Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Bruce Ren and Version 1 by Carmen Lidia Chitescu.
Phenolic compounds are plants’ bioactive metabolites that have been studied for their ability to confer extensive benefits to human health. As currently there is an increased interest in natural compounds identification and characterization, new analytical methods based on advanced technologies have been developed.
- extraction
- high-resolution mass spectrometry
- fragmentation pathway
- non-targeted analysis
- chemical profiling
Note: The following contents are extract from your paper. The entry will be online only after author check and submit it.
1. Introduction
Plants have been used for centuries as remedies in several forms: unprocessed, as complex mixtures of different species as in Traditional Chinese Medicine [1], or, more recently, in commercial products as phyto-pharmaceuticals and dietary supplements [2,3,4][2][3][4]. The bioactivity of those natural products is generally high [4,5[4][5][6],6], supporting the high use of those products as a primary form of healthcare for a large part of the population [7]. Furthermore, about 80% of all synthetic drugs are directly or indirectly derived from them [8,9][8][9]. The pharmaceutical industry is currently showing an increasing interest in the development of new formulations with integrated vegetal extracts as a source of bioactive compounds [9]. Therefore, the assessment of the chemical components of herbs, spices or functional foods has become an essential part of our understanding.
Polyphenols are secondary plant metabolites, and comprise a wide range of compounds that strongly differ in their structure, physicochemical and nutritional properties. Dietary polyphenols are one of the most important groups of natural antioxidants and chemopreventive agents in human diets, playing a vital role in supporting the functioning of biological systems [6,10][6][10]. Epidemiological, clinical and nutritional studies strongly support the suggestion that polyphenolic compounds enhance human health by lowering risk and preventing several diseases and disorders [5,10][5][10]. It has been reported that polyphenolic compounds exhibit anticarcinogenic, anti-inflammatory, antimicrobial, antiviral, antidiabetic and hepatoprotective activities, as well as estrogen-like activities [11,12,13][11][12][13].
In the last few decades, different studies have intensely investigated the antioxidative, antimicrobial, antiproliferative or enzyme-inhibition effects of polyphenols [6,12,14][6][12][14]. Studies using in vitro and in vivo approaches together with LC-MS analytical techniques have led to a better understanding of the bioavailability and bioactivity of polyphenols. However, the theory that the beneficial effects of polyphenols are direct consequences of antioxidant activity in vivo is obsolete. Their protective activity was firstly attributed to their antioxidant, free radical-scavenging, and metal chelator properties, and then to their ability to inhibit different enzymes [12]. New research supports the hypothesis according to which polyphenols’ interaction with signal transduction pathways and cell receptors induce adaptive responses that drive antioxidant, antiplatelet, vasodilatory or anti-inflammatory effects [6,11,15][6][11][15]. Thus, the current research places more emphasis on the individual identification/quantification of each compound rather than on the in vitro assay of bioactive properties [16].
2. Extract Hydrolysis and Purification
The hydrolysis can be performed before, during or after extraction, using different procedures—acidic, alkaline or enzymatic hydrolysis—in order release bound polyphenols and increase the extraction yield of their aglycone form [46,56,57][17][18][19]. While acidic hydrolysis breaks glycosidic bonds, alkaline hydrolysis breaks ester bonds and removes acetyl- or malonyl- groups from glucosides, allowing only β-glucosides and native aglycone forms to remain in the extract [56][18]. Enzymatic hydrolysis produces aglycone forms, similar to acid hydrolysis [56][18]. The official AOAC method 2001.10 for isoflavones in soy and in various foods containing soy uses alkaline hydrolysis [57][19]. For enzymatic hydrolysis, β-glucosidase, β-galactosidase, β-glucuronidase or mixtures of these are usually used [45][20].
The purification, fractionation and concentration of the extract are of great importance, both for the analysis of polyphenols and for the subsequent use of the extracts in various fields, including pharmaceuticals.
The extract, hydrolyzed or not, may be subjected to clean-up techniques allowing the accurate identification and quantification of the target analytes. The extraction in the solid phase (SPE) is the most-employed technique in clean-up procedures [58][21]. In SPE, the target compounds are retained in a specific sorbent and then eluted with an adequate solvent, such as methanol, ethanol, and ethyl acetate. The SPE process allows for the purification and concentration of polyphenols at the same time. SPE columns type C18 [23][22], HLB [58][21] or Oasis MCX [59][23] were used as stationary phases. An alternative to traditional SPE is the matrix solid-phase dispersion extraction (MSPD) method, with the advantage of less solvent consumption [60][24]. Dispersive solid-phase extraction (d-SPE), another alternative to SPE, was recently evaluated for the determination of phenolic compounds in Myrciaria cauliflora peel [61][25], proving its efficiency in removing the interfering compounds without significant retention of polyphenols.
QuEChERS (quick, easy, cheap, effective, rugged, and safe) is an alternative method of polyphenol extraction and purification that reduces solvent amount and procedure duration. Initially developed for the determination of pesticide residues in food matrices, due to its versatility, QuEChERS has been progressively applied for the extraction of other compounds in different matrices, resulting in the good recovery of the target analytes and lower interference [62][26]. The technique involves liquid–liquid partitioning with organic solvents and purification of the extract using solid-phase dispersive extraction (d-SPE) with sorbents and buffers [62,63][26][27].
QuEChERS assisted by ultrasound extraction was recently optimized for the isolation of polyphenols from several fruit and vegetable samples [62][26]. Acetonitrile, methanol, ethanol, and a combination of them were tested, methanol being selected in the subsequent experiments. An ultra-sonification time of 5 min was also selected as optimum. A mixture of buffered salts, including disodium hydrogen citrate sesquihydrate, trisodium citrate dihydrate sodium chloride, and MgSO4, was used [62][26].
However, due to the development of advanced, sensitive spectrometric methods, sample preparation has become simplified, often consisting of the filtration and convenient dilution of the extract.
3. Analytical Detection Techniques
Due to polyphenols’ structural diversity and low concentrations, and the plant matrix complexity, their analysis remains challenging. Currently, there is a requirement for sensitive and accurate methods for the analysis of polyphenols, as knowledge of their identity and dosage are prerequisites in evaluating health benefits [64][28]. Novel techniques have been employed in the past few decades, ranging from high-pressure liquid chromatography (HPLC) to mass spectrometry (MS) and spectroscopic methods. Recent (2015–2020) developments, and the application of analytical methods in qualitative and quantitative studies of polyphenols following extraction, were reviewed in the present work. High-resolution mass spectrometry is particularly addressed due to its applicability in the targeted/untargeted metabolomic analysis of polyphenols.
3.1. Liquid Chromatography with Ultraviolet/Visible (UV/Vis)-Based Detection: HPLC Fingerprint with Chemometric Analysis
High-performance liquid chromatography (HPLC) is still one of the most widely used analytical tools for the identification and quantification of polyphenols [65][29]. The quality consistency of herbal medicines reflects variations in their chemical composition from batch to batch, depending on several factors, such as botanical species, chemotypes, morphological parts of the plant, geographical area, time of harvest, and storage conditions. With the continuous development of HPLC technology, chromatographic fingerprint analysis has been recognized as an innovative, rapid, and comprehensive method for the identification and qualification of herbal medicines [66][30]. The fingerprint profiles show variations in a given herb in an integrated manner, and can identify a particular herb, distinguishing it from closely related species [66,67][30][31]. The chromatographic fingerprint of the herbal profile can be defined as the characteristic signal of selected plant that allows unambiguous identification via the evaluation of the chemical similarities and differences in the obtained chromatograms of studied samples [66][30]. Fingerprint analysis has been internationally accepted as a method for the evaluation and quality control of herbal medicines and preparations, and is currently applied in combination with other chemometric modeling methods, namely, similarity analysis (SA), hierarchical clustering analysis (HCA), principal component analysis (PCA), and partial least square regression [68,69,70][32][33][34].
For a reliable investigation of a matrix as complex as is found vegetal extracts, the fingerprint method should display good precision, repeatability, and stability, evaluated based on the relative standard deviations (RSD) of the relative retention times (RRT < 3%), and the relative peak areas (RPA < 3%) of the characteristic peaks compared with the reference peak. Similarity values above 0.98 are accepted [69][33].
The chromatographic fingerprint method can also distinguish authentic materials from substitutes and adulterants, suggesting new applications for food products and pharmaceuticals. Recently, several methods were developed for the fingerprint analysis of different species, such as Flos Carthami (Carthamus tinctorius L.), [71][35], Aurantii Fructus (Citrus aurantium L.) [66][30], chamomile (Matricaria chamomilla L.) [72][36], licorice (Glycyrrhiza glabra L.) [67][31], and selected lavender species (Lavandulae spp.) [73][37] (Table 1). An HPLC-UV polyphenolic fingerprint method was applied on pure cranberry extracts and cranberry-based extracts adulterated with grape at different percentages [2].
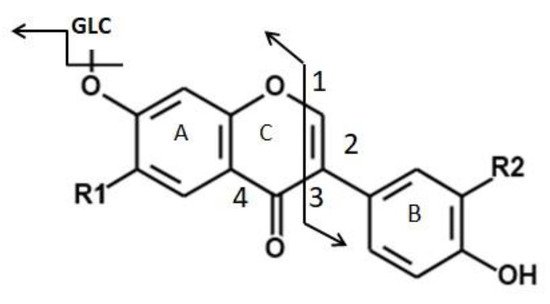
Table 1. Some examples of the recent applications of HPLC fingerprinting methods in natural polyphenols analysis.
| Plant Material | Extraction | HPLC Condition | Characteristic Fingerprint Peaks | Chemometric Analysis Approach | Ref. | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cranberry ( | Vaccinium macrocarpon | )-based products (fresh and dried fruits, juice) | Lyophilized samples were extracted with an acetone:water:hydrochloric acid (70:29.9:0.1 | v | / | v | / | v | ) solution by UAE for 10 min | Kinetex C18 (100 4.6 mm i.d., 2.6 µm particle size) column; mobile phase: 0.1% formic acid in water ( | v | / | v | ) and MeOH; flow rate of 1 mL/min; monitoring wavelength range: 190–550 nm |
gallic acid, homogentisic acid, protocatechuic acid, protocatechualdehyde, (+) catechin hydrate, gentilic acid, | p | -salicilic acid, chlorogenic acid, vanillic acid, (−) epicatechin, syringic acid, syringaldehyde, ethyl gallate, | p | -coumaric acid, ferulic acid, resveratrol and quercitrin | Partial least square regression and PCA | [2] |
| 27 |

On the other hand, small radical losses such as CO and CO2, H2O, CH2-CO, and CH4O were observed [91,92,95][56][57][60]. The fragments resulting from the rDA reaction are particularly useful for elucidating the structure, because they allow not only the positioning of the OH- group, but also for the identification of the position of glycosidic bonds. March et al. [94][59] proposed an intermediate structure of the C ring, which successfully explained the mechanism of CO2 removal at the C ring in the negative ionization of isoflavone-glycoside. Fabreet al. [95][60] found a loss in ketene moiety (C2H2O) in flavones and flavanones at the C ring following fragmentation due to the low probability of rearrangement, whereas Kang et al. [86][51] showed that the loss of ketene occurs at ring A for isoflavones.
Table 2. Examples of the target screening methods used for the identification/quantification of the selected polyphenols.
| Plant Material | Extraction | Hyphenated Technique Used | Instrumental Methodology (HPLC Condition, Ionization, Acquisition Mode) | Selected Polyphenols | Ref. | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Flowers, leaves, and stalks of alfalfa ( | Medicago sativa | L.) and goldenrod ( | Solidago virgaurea | L.); flowers, leaves, stalks, and roots of phacelia ( | Phacelia tanacetifolia Benth | .); buckwheat ( | Fagopyrum esculentum | ); licorice root ( | Glycyrrhiza glabra | ); and lavender flower ( | Lavandula spica | L.) | solid–liquid extraction (SLE) consecutively using H | 2 | O; mixture H | 2 | O/EtOH (1:1; | v | / | v | ), mixture H | 2 | O/MeOH (1:1; | v | / | v | ), and finally NeOH shaking for 5 h; SPE purification using a C18 column | UHPLC-MS/MS | Zorbax Eclipse XDB-C18 column (50 × 2.1 mm, 1.8 μm); mobile phase: 0.1% | v | / | v | formic acid in water and ACN; ESI ionization source in negative mode, acquisition in selected reactions monitoring mode, SRM |
3-(4-hydroxyphenyl) propionic acid, 4-hydroxybenzoic acid, and 3,4-dihydroxybenzoic acid, quercetin, rutin, glabridin, and naringenin |
[23] | [22] |
| Salvia | L. Species, leaf and root | Maceration in MeOH (2 × 10 mL for 24 h) followed by solvents removal on rotary evaporator under vacuum at 40 °C to dryness | ||||||||||||||||||||||||||||||||||
| Edible lotus ( | Nelumbo nucifera | ) rhizome knotRP C18 Eurospher-100 column, (5 µm particle, 125 mm × 4 mm); mobile phase: 0.2% ( | v | / | Enzymatic hydrolysis with cellulose and pectinase, at 62 °C, pH 4, 90 min followed by | v | ) glacial acetic acid in water and ACN; flow rate of 1 mL/min; monitoring wavelength: 280 nm |
ultrafiltrationrosmarinic acid, carnosic acid, caffeic acid, salvianolic acids A and B |
PCA | [65] | HPLC–QqQ-MS/MS | ZORBAX Eclipse XDB-C18 column (150 mm × 4.6 mm, 5 μm; mobile phase: aqueous 0.4% acetic acid, and acetonitrile; ESI in negative ion mode Acquisition in MRM mode[29] |
||||||||||||||||||||||||
| chlorogenic acid, B-type procyanidin | dimer, (+)-catechin, B-type procyanidin dimer, (−)-epicatechin, propyl | gallate, caffeic acid, (−)-epicatechin-3-gallate, and rutin | [43] | [61] | Aurantii Fructus | , dried mature and imature fruits of | Citrus aurantium | L. (medicinal herbs in TCM); | UAE (200 W) with MEOH for 45 min |
Symmetry C18 column (250 × 4.6 mm, 5 μm); mobile phase: ACN and 0.1% aqueous phosphoric acid; flow rate of 1 mL/min; monitoring wavelength range: 285–324 nm |
eriocitrin, neoeriocitrin, narirutin, naringin, hesperidin, neohesperidin, meranzin, poncirin, naringenin, nobiletin, tangeretin and auraptene | Quantitative analysis of multiple components by single marker (QAMS); similarity analysis; standard method difference; HCA | ||||||||||||||||||||||||
| Trifolium pratense | L. (Red Clover), dried leaves | MAE, 300 W, with MeOH at 70 °C | HPLC-ESI-MS/MS | Zorbax Eclipse XDB-C18 50 mm × 4.6 mm × 1.8 μm column; mobile phase: 0.05% aqueous formic acid and MeOH; ESI ionisation source in negative mode, acquisition in SRM |
43 phenolic including: genistein, daidzein, p-hydroxy-benzoic acid, caffeic acids, kaempferol 3- | O | -glucoside, quercetin 3- | O[66] | [30] | |||||||||||||||||||||||||||
| -glucoside, hyperoside | [ | 87 | ] | [ | 52] | Licorice root ( | Liquiritiae radix | ) - | Glycyrrhiza glabra | L. | UAE with 80% MeOH-water, 120 W, 40 KHZ, 20 min | Cosmosil column (5C18-MS-II, 5 µm, 4.6 × 250 mm), at 35 °C; mobile phase: 5 mmol/L sodium heptane sulfonate solution phosphoric acid (499:1, | ||||||||||||||||||||||||
| Heartwood samples of | Dalbergia odorifera | (medicinal herbs in TCM); | UAE with 70% methanol ( | v | / | v | ) for 45 min | v | / | v | ) and ACN-MeOH (9:1, | v | / | v | ); flow rate of 1 mL/min; monitoring wavelengths: 203 nm, 220 nm, 250 nm, 280 nm and 344 nm |
glycyrrhizic acid, liquiritigenin, isoliquiritigenin, isoliquiritin, liquiritin apioside, isoliquiritin apioside and glabridin | Geometric linear quantified fingerprint method (GLQFM) and PCA | UHPLC–QqQ-MS/MS and UHPLC-Q/TOF-MS/MS |
Acquity HSS T3 column (100 mm × 2.1 mm, 1.8 µm); mobile phase: ACNand water containing 0.05% formic acid ESI negative ion mode. Acquisition in MRM mode for QqQ analysis. | [67] | 17 flavonoids including: daidzein, dalbergin, 30-hydroxydaidein, liquiritigenin, isoliquiritigenin, alpinetin, butein, naringenin, butin, prunetin, eriodictyol, tectorigenin, pinocembrin, formononetin, genistein, sativanone[31] |
|||||||||||||||
| [ | 88 | ] | [ | 53 | ] | Flos Carthami, the dried flower of | Carthamus tinctorius | L.; (medicinal herbs in TCM) | UAE with 50% MeOH, 270 W, 30 min |
|||||||||||||||||||||||||||
| Green, black and oolong tea ( | Camellia sinensis | C18 column (4.6 mm × 250 mm, 5 μm); mobile phase: 0.1% formic acid in water and ACN | ) | Extraction at high temperature (80 °C) for 3 min with mild stirringguanosine, hydroxysafflor yellow A, |
HPLC–QqQ-MS/MSanhydrosafflor yellow B, kaempferol 3- | O | -β-rutinoside, rutin, quercetin, kaempferol | Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine (Version 2004 A) | Capcell Pak C18 MGIII (2.0 mm × 100 mm, 3 m) column; ESI in positive and negative ion mode. Acquisition in MRM mode. | [71] | [35] | |||||||||||||||||||||||||
| gallocatechin, epigallocatechin, catechin, epigallocatechin gallate, epicatechin, gallocatechin gallate, epicatechin gallate, catechin gallate, theaflavin, theaflavin-3-gallate, theaflavin-3′-gallate; theaflavin-3,3′-digallate. | [ | 89 | ] | [ | 54] | Chamomile— | Matricaria chamomilla | L., commercial samples | UAE with MeOH:water mixture (80:20, | |||||||||||||||||||||||||||
| Sour Guava ( | Psidium friedrichsthalianum Nied | ) lyophilized whole fruits | v | / | v | ) at 35 °C for 30 min, three times repeated | UAE of lyophilized fruits with acetone:water (7:3). The extract was submitted to successive partitions with ethyl ether, ethyl acetate, and | N | -butanol.Hypersil Gold C18 column (250 × 4.6 mm; 5 μm) at 35 °C; mobile phase: 0.05% TFA in ACN and 0.05% TFA in water; flow rate of 1 mL/min; monitoring wavelength: 254 nm |
UPLC-ESI/QqQ-MS/MS | UPLC BEH C18-column (2.1 × 100 mm, 1.7 µm); mobile phase water/formic acid (99.9:0.1, | v | / | v | ), and ACN/formic acid (99.9:0.1, | v | / | v | ) ESI ionization source in negative mode. Acquisition in MRM |
22 phenolic compounds including several hydroxybenzoic, phenylacetic, and hydroxycinnamic acid derivatives | [90] | [55] | ||||||||||||||
| Barks of Connarus var. angustifolius, and leaves of Cecropia obtusa | , | Cecropia palmata | and | Mansoa alliacea | ; | dried samples | UAE in 70% hydroethanolic, butanol/ethyl acetate, 4 h Butanol and ethyl acetate were evaporated at 40 °C |
HPLC-ESI/QqQ-MS/MS | SB-C18 Rapid Resolution HD column (2.1 × 50 mm, 1.8 μm; mobile phase 0.1% acetic acid in water and ACN; ESI ionization source in negative mode Acquisition in MRM |
gallic acid, catechin, caffeic acid, rutin, ferulic acid, quercitrin and resveratrol | [96] | [62] |
In addition, the ion [MH-C3O2]− has shown a relatively high abundance in flavonones fragmentation (luteolin, apigenin, genistein), it being one of the key ions for the differentiation of isobaric compounds apigenin and genistein. The subsequent fragmentation MS3 of this ion from the apigenin showed the ion [MH-2C3O2] resulting from the loss of another C3O2 fragment, and in genistein the loss of CO from [MH-C3O2] was displayed [97][63].
For methoxylated flavonoids (biochanin, formononetin, prunetin, calicosin, glycitein), the [M-H-CH3]− ion is characteristic of negative ionization [94][59]. The ionic fragments CH3, CHO, and CO2, resulting from successive or simultaneous losses, are attributed to the type of isoflavone [4′-OCH3], [86,97][51][63]. In addition, for isoflavones containing a hydroxyl group or a methoxy- group at the B ring, a relatively highly abundant fragment ion at m/z 132 was detected and assigned to [0.3B-2H]- for prunetin and glycitein, and [0.3B-CH3-H]- for formononetin and biochanin A [86,97][51][63]. The fragment ion at m/z 117 indicates the presence of a hydroxyl group at ring B or in position 3. On the other hand, the fragments [MH−CH3−CO] are characteristic of methoxilates isoflavones, CO loss being subsequent to radical loss [CH3]. The fragment [MH-CH3-CO-B-ring] was found to be characteristic of the distinction between glycitein and other isomers as collision energy increases [98][64]. Thus, the mass spectral decomposition of polyphenols under ESI-HRMSn allows a structural characterization of the corresponding compounds by assigning specific key ions.
Analytical approaches based on HRMS have been successfully applied for polyphenols profiling in herbal remedies from traditional Chinese medicine, such as Dingkun Dan [91][56] or Aster tartaricus risoma [99][65], rare plants such as Ophryosporus triangularis (native to the Acatama Desert, Chile) [100][66], or common plants, e.g., culinary herbs [22][67], green tea products [101][68], leaves of green perilla [102][69], pomegranates [103][70] and goji [104][71] (Table 3).
Table 3. Examples of the non-target screening methods used for chemical profiling of the plant extracts.
| Plant Material | Extraction | Hyphenated Technique Used |
Instrumental Methodology (LC Condition, Ionization, Acquisition Mode) | Data Processing Approach Used for Tentatively Identification | Ref. | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ground dried culinary herbs and spices: dill ( | Anethum graveolens | ), marjoram ( | Origanum majorana | ), turmeric ( | Curcuma longa | ), caraway ( | Carum carvi | ), and nutmeg ( | Myristica fragans | ) | UAE with 5 mL of 50% ethanol in 0.1% formic acid in ultrapure water; 5 min | UHPLC- LTQ Orbitrap MS | Atlantis T3 C18 (100 × 2.1 mm, 3 µm) column; mobile phase; 1% formic acid in water and 0.1% formic acid in ACN. Full scan MS mode at 60,000 FWHM and MS/MS mode with the Orbitrap at 30,000 FWHM DDA scan. |
In-house database Comparison with HR-MS data found in literature PCA, HCA |
[22] | [67] | |
| Dingkun Dan (traditional Chinese medicine prescription) |
Ultrasonic extraction with MeOH, 30 min | UHPLC-Q/Orbitrap –HRMS/MS | Acquity T3 (2.1 × 100 mm, 1.8 μm) column; mobile phase: 0.1% formic acid in water (A) and 0.1% formic acid in MeOH. HESI II ionization source in positive and negative mode; collision energy: 25–60 V. Data-dependent acquisition (DDA). |
In-house database Searching in Chemspider; Pubchem. |
[91] | [56] | |||||||||||
| Aster tataricus | rhizoma | UAE with MeOH for 30 min | UHPLC-Q-TOF-MS | Poroshell 120 EC-C18 column (100 mm × 2.1 mm, 2.6 µm); mobile phase: water + 0.1% formic acid and ACN + 0.1% formic acid. ESI source in both positive and negative ion mode. DDA and DIA acquisition methods. Multiple mass defect filter (MMDF) and dynamic background subtraction (DBS) by AB Sciex software |
Searching for reported metabolites; in-house database. Searching in Chemspider database using MasterView | TM | 1.0. Product ions strategy (KPIs). Clog P (calculated by Chemdraw Ultra 12.0 software)—used for distinguishing isomers. |
[99] | [65] | ||||||||
| Ophryosporus triangularis | Meyen, dried aerial parts (leaves and stems) and flowers | UAE with MeOH, 30 min | UHPLC-Q- Orbitrap HRMS/MS |
Acclaim UHPLC C18 column, (150 mm × 4.6 mm ID, 2.5 µm); mobile phases: 1% formic aqueous solution and ACN; HESI negative ionization mode; full scan at 70,000 FWHM followed by targeted MS/MS at 17,500 FWHM; collision energy (HCD cell)–30 kv |
In-house database comparison with HR-MS data found in literature |
[100] | [66] | ||||||||||
| Dried leaves of green perilla ( | Perilla frutescens | ) | Extraction with MeOH by shaking for 8 h at ambient temperature | LC- TOF-MS/MS | Poroshell 120 EC-C18, 150 × 2.1 mm, I.D., 4 mm; mobile phase: 0.1% formic acid in water and MeOH; ESI source in negative ion mode; resolution of 2700; collision energy 10 eV. |
The analyst TF software (version 1.7); identification of the compounds by comparison with previous studies. Tentatively characterized by fragmentation pathway identification. |
[102] | [69] | |||||||||
| gallic acid, caffeic acid, syringic acid, | p | -coumaric acid, ferulic acid, rutin, myricetin, quercetin and kaempferol | ANOVA, PCA, hierarchical cluster analysis (HCA) | [72] | [36] | ||||||||||||
| Seven species of | Lavandulae flos | UAE with xylen (1:30) under reflux for 4 h followed by solvents removal on a rotary evaporator under vacuum to dryness | Kinetex RP18 column (5 μm, 150 × 4.6 mm); mobile phase: MeOHwater–0.1% formic acid (gradient 5–100% ( | v | / | v | ) of MEOH) at 30 °C; flow rate of 1 mL/min; monitoring wavelength: 280 nm | apigenin, myricetin, luteolin, luteolin 7-glucoside, chlorogenic acid, caffeic acid, ferulic acid | Cluster analysis using SpecAlign program (Pearson correlation coefficient, r, and Euclidean) and PCA | [73] | [37] | ||||||
| Pomegranate ( | Punica granatum | L.), dried peel | UAE of the dried samples with 60% ethanol, 26 min | Zorbax SB-C18 column (5 μm, 4.6 mm × 250 mm); mobile phase: glacial acetic acid (99:1, | v | / | v | ; pH 3.0) and MeOH flow rate of 1 mL/min; monitoring wavelength: 280 nm |
gallic acid, punicalagin, catechin, chlorogenic acid, caffeic acid, epicatechin, rutin, and ellagic acid | Similarity Evaluation System for Chromatographic; Fingerprint of Traditional Chinese Medicine (Version 2004A) software | [75] | [38] | |||||
| Sedi linearis Herba | , dried whole herb | UAE of the dried samples with 70% methanol, 60 min | BDS Hypersil C18 column (4.6 m × 250 mm, 5 μm), 30 °C; Mobile phase: ACN and 0.1% acetic acid solution; flow rate of 1 mL/min; monitoring wavelength: 265 nm |
hyperoside, isoquercetin and astragalin |
- | [76] | [39] | ||||||||||
| Black tea samples | Enzymatic extraction using immobilized polyphenol oxidase followed by fractionating using a Mitsubishi SP-207 resin chromatography with an elution gradient of 20%, 30%, 40%, 50% and 70% aqueous ethanol |
C18 column; mobile phase: water: ACN: glacial acetic acid (73.5/26/0.5, | v | / | v | / | v | ); flow rate of 5 mL/min | caffeine, (−) epigallocatechin gallate, (−) epigallocatechin, (−) epicatechin gallate, (−) epicatechin, (+) gallocatechin gallate, (+) gallocatechin, (+) catechin gallate, (+) catechin, theaflavin, theaflavin 3-monogallate, theaflavin 3′-monogallate and theaflavin 3,3′-digallate | - | [77] | [40] | |||||
| Raw elderberry ( | Sambucus nigra | L.) | UAE of the dried samples with 80% methanol, 45 min | C18 column, 35 °C, mobile phase: MeOH and acetic acid in water (1.0%, | v | / | v | ); flow rate of 1 mL/min; monitoring wavelength: 285 nm |
gallic acid, chlorogenic acid, caffeic acid, syringic acid, pcoumaric acid, ferulic acid, rutin, myricetin, quercetin kaempferol and quercetin 3-glucoside | HCA and PCA | [78] | [41] | |||||
| Phyllanthus emblica | , dried fruits | Fruit powder was extracted in 70% ethanol (1:8) at 50 °C using a magnetic stirrer | DiKMA C18 column (250 mm × 4.6 mm × 5 μm); mobile phase: 0.2% formic acid in water and methanol; flow rate of 1 mL/min; Monitoring wavelength: 273 nm |
gallic acid, corilagin, ellagic acid, quercetin | Similarity Evaluation System for Chromatographic Fingerprints of Traditional Chinese Medicine (Version, 2004A) and HCA | [79] | [42] | ||||||||||
| Medicago | spp. in different phenologic stages: vegetative elongation, late bud and late flower; dried leaves | MSPD extraction using C18 column. Elution with methanol: H | 2 | O (9:1, | v | / | v | ) | Luna 5 U C18 column (5 μm, 150 × 4.60 mm) at 40 °C; mobile phase: acetonitrile and acetic acid in water (1.0%, | v | / | v | ); flow rate of 1 mL/min; monitoring wavelength: 254 nm | puerarin, daidzin, genistin, daidzein, glycitein, genistein, pratensein, formononetin, irilone, prunetin and biochanin A |
generalized linear model (GLM) and linear discriminant analysis (LDA) | [80] | [43] |
| Tithonia diversifolia | , dried leaves | Maceration in 80% acetone for 72 h; evaporate to dryness at 40 °C |
C18 column (5 µm, 4.6 mm × 250 mm); mobile phase: water/acetic acid, 98:2% | v | / | v | and methanol/water/acetic acid, 70:28:2% | v | / | v | ; flow rate 0.6 mL/min; monitoring wavelengths: 254, 327, 366 |
gallic acid, chlorogenic acid, caffeic acid and | p | -coumaric acid, and apigenin | analysis of variance model and Tukey’s test | [81] | [44] |
| lyophilized leaves of ten mango varieties | UAE with 70% ethanol, 320 W for 30 min; evaporate to dryness by vacuum rotary evaporator at 30 °C | C18 column (250 mm × 4.6 mm, 5 μm); mobile phase: 0.1% ( | v | / | v | ) formic acid in water and 0.1% ( | v | / | v | ) formic acid in acetonitrile; flow rate: 0.8 mL/min; all wavelengths scanning detection from 200 to 600 nm |
neomangiferin, galic acid, 5-caffeoylquinic acid, 3-chlorogenic acid, mangiferin, 4-hydroxybenzoic acid, sinpic acid, isoquercitrin, quercetin | similarity analysis, PCA, HCA, discriminant analysis | [82] | [45] |
For assessing the chromatographic condition, the C18 or C8 reversed-phase LC (packed with particles of silica bonded with alkyl chains) columns are preferred for the separation. C12 columns have also been investigated in herbal drug standardization [74][46].
A mixture of water with organic solvents (i.e., methanol, acetonitrile) is usually used as the mobile phase. The development of LC methods for polyphenols analysis is almost always based on a slow solvent gradient (duration between 45 and 80 min) with the advantage of sharper peaks because of the compression effects induced by the gradient, as well as minimizing the LC column contamination due to the increased solvent strength. A gradient based on a combination of acetonitrile/water/methanol is often applied on a C18 column for the separation polyphenols [66,74][30][46]. Acetic acid, trimethylamine, phosphoric acid or trifuloroacetic acid (TFA) are usually used as mobile phase additives for enhancing the chromatographic separation [66,73,74,75,78][30][37][46][38][41] (Table 1).
However, conventional HPLC suffers the disadvantages of long analysis time, low resolution and large solvent consumption. UPLC and multi-dimensional separation techniques have emerged as alternatives to HPLC, presenting superior separation capabilities and high levels of system stability, providing technical support for further pharmacological and pharmaceutical research [77,83][40][47].
3.2. Liquid Chromatography–Mass Spectrometry Analytical Methods
Although many studies had been published concerning the phenolic profiles of various plant species through RP-HPLC, mass spectrometric techniques have recently gained a considerable advancement in the analysis of complex biological matrices, and thus in evaluating the bioactivity and nutraceutical potential of plants [20,23,84,85][48][22][49][50].
Due to noise reduction, and improved detection sensitivities and method specificities, these techniques require minimal sample preparation by dilution. Thus, the limitations of LC-UV methods, such as the presence of interferences in complex samples and the high levels of detection and quantification limits, have been overcome [23,84][22][49]. Mass spectrometry is currently the most sensitive method of structural analysis, consisting in the ionization of the investigated chemical substances followed by the separation of the ions according to the mass to charge ratio. The mass spectrum represents the plot of the ions’ relative abundances versus their mass to charge ratio, and is a characteristic of each compound [20][48].
3.2.1. Liquid Chromatography (LC) Tandem Low-Resolution Mass Spectrometry
Liquid chromatography (LC) coupled with single–quadruple low-resolution mass spectrometry, or more often, with tandem mass spectrometry (MS-MS) using ion trap spectrometers (IT) [84][49] or triple quadrupole (QQQ) [23,86,87,88][22][51][52][53], is common in targeted screening and quantification methods for polyphenols. The comparative study of the theoretical mass and fragmentation patterns of the reference standards vs. target compounds is used to unequivocally confirm the identity of polyphenolic compounds in ‘‘targeted’’ analysis. However, due to the limited availability of analytical standards, a limited number of compounds can be identified and quantified using this approach.
Four MS scan approaches are possible, and all can generate valuable information: (i) Full scan (FS) and (ii) selective ion monitoring (SIM) represent the most common data acquisition modes in methods without fragmentation. Confirmatory analysis uses the monitoring of the fragment ions through (iii) product reaction monitoring (PRM) or (iv) multiple reactions monitoring (MRM), which monitor all ion fragments resulting from a single selected precursor ion (MS2 spectra) [86][51].
The most common ionization source in the LC-MS analysis of polyphenols is electrospray ionization (ESI) in the negative mode, providing the deprotonated molecule [M-H]− [86][51]. ESI in the positive ionization mode has also been proposed in various applications [89][54].
For the chromatographic separation of polyphenols, prior to spectrometric analysis, reverse phase chromatography (RP-LC) using the stationary phases C18 and C8 is the most commonly used in LC-MS. Short columns (e.g., 150 mm for HPLC and 100 mm for UHPLC) with small particle diameters (e.g., 5 μm for HPLC or between 1.8 and 2.6 μm for UHPLC) are generally preferred [23,87,90][22][52][55]. Mixtures of ultrapure water acidified with formic acid or acetic acid 0.05–0.5% and methanol or acetonitrile as organic solvents (often acidified with formic acid or acetic acid) are usually used as mobile phases [20,23][48][22]. The addition of 0.1% trifluoroacetic acid can result in improved peak separation, tailing reduction and superior peak symmetry [20][48].
For adequate detection of the analytes, the parameters of the ESI ion source (nebulizing gas flow, their temperature, voltage and source temperature, declustering potential, etc.) as well as the different parameters of the MS instrument (e.g., collision energy) are optimized by injection of analytical standards.
Liquid chromatography (LC) tandem low-resolution mass spectrometry has recently been used in various targeted analyses of polyphenols in alfalfa (Medicago sativa L.), lavender (Lavandula spica L.), buckwheat (Fagopyrum esculentum), phacelia (Phacelia tanacetifolia Benth.) and licorice (Glycyrrhiza glabra) root extract [23][22], Chinese rose (Dalbergia odorifera) used in traditional Chinese medicine [88][53], red clover (Trifolium pratense L.) extract [87][52], and green, black and oolong tea (Camellia sinensis) [89][54] (Table 2).
3.2.2. Non-Targeted High-Resolution Mass Spectrometry Use in “Chemical Profiling”
Although low-resolution mass spectrometry is still used for the identification and quantification of polyphenols in different vegetal matrices, particularly food, high-resolution mass spectrometry (HRMS), represented by quadrupole–flight time spectrometry (Q-TOF) and quadrupole–Orbitrap technologies, have recently gained popularity due to their ability to provide complete information on the exact molecular mass, elemental composition and chemical structure of a given compound [91,92][56][57].
The exact mass provided by high-resolution mass spectrometry with a mass error ≤ 2 ppm is currently the ideal tool for the structural characterization of the compounds in various applications, including polyphenols identification [91][56]. In the MS/MS tandem analysis, the quasi-molecular ion [M-H]− or [M-H]+ fragmentation model can be studied by the retro-Diels–Alder reaction (rDA) or other fragmentation models [91,92][56][57].
For polyphenols’ analysis, the negative ionization mode provides more characteristic fragments than positive ionization [93,94,95][58][59][60]. A fragmentation pattern characterized by loss of the carboxylic fraction (CO2, 44 Da) is observed for phenolic acids. The remaining skeleton showed the ion [MH]− at m/z 137.02 (C7H5O3), and in the MS/MS spectra, the fragment [MH-COOH]− at m/z 93.03 (C6H5O) is generated [91][56]. The loss of a hydroxyl radical leads to the fragment m/z 107.01, visible in several such compounds [91][56].
For O-glycosylated flavonoids, the cleavage to the neutral glycosidic residue is characteristic in both positive and negative ionization modes, resulting in fragments such as 162 Da (glucose), 132 Da (pentose), 146 Da (deoxyhexose), 146 Da (rhamnosis), 180 Da (glucopyranose) and 308 Da (rutinoside) [92][57]. In aglycones flavonoids, two characteristic fragmentation pathways can be distinguished: ring heterocyclization and gradual degradation of the molecule [92][57].
Thus, for negatively ionized flavones, isoflavones, flavonols and flavanones, the MS-MS ion spectra show fragmentation following the retro-Diels–Alder reaction path to the C ring, leading to = molecule cleavage at bonds 1 and 3 (Figure 1). Two product ions containing intact A and B rings result [91,92,93][56][57][58].
| Goji berries ( | ||||||||||||||||||||||||
| Lycium barbarum | ||||||||||||||||||||||||
| L.) | ||||||||||||||||||||||||
| Extraction in 1% formic acid in 80:20 methanol/water solution by centrifugation at | 25,000 rpm/3 min |
UHPLC-ESI-QTOF-MS | Zorbax eclipse plus C18 column (50 × 2.1 mm, 1.8 μm); mobile phase: water and MeOH ESI ionisation source in positive; full scan MS | 2 | (30,000 FWHM); mass accuracy ≤ 5 |
Profinder B.07 software. Phenol-Explorer 3.6 database |
[104] | [71] | ||||||||||||||||
| Stellera chamaejasme | extracts | reflux with MeOH for 30 min at 50 °C. | UHPLC–LTQ-Orbitrap MS | n | Agilent Zorbax Ecipse Plus C18 column (100 × 3.0 mm, 1.8 µm) at 50 °C; mobile phase water with 0.5% FA and ACN; HESI in positive and negative mode DDA, MS | n | scan (four ions for MS | 2 | and one ion for MS | 3 | ); resolution of MS | 2 | –60,000 and MS | 3 | –30,000; collision energy: MS | 2 | –30 V and MS | 3 | –35 V. | SciFinder database ( | https://scifinder.cas.org | ) for chemical formula annotation; comparison with HR-MS data found in literature |
[105] | [72] |
| The male flowers of date palm ( | Phoenix dactylifera | ) | Soxhlet extraction with 80% MeOH for 6 h at 50 °C, fractionation in a C18 column | HPLC-ESI-ITMS | Symmetry C18 column (5 µm, 2.1 mm × 150 mm; mobile phase: ACN containing 0.03% ( | v | / | v | ) formic acid and water containing 0.03% ( | v | / | v | ) formic acid; ESI ion source in negative ion mode full-scan followed by MS-MS |
Density functional theory (DFT) study | [106] | [73] | ||||||||
| Mentha rotundifolia | (L.) Huds, aerial parts | maceration in MeOH for 24 h at room temperature | UHPLC-ESI-Q- Orbitrap- HRMS/MS | Kromasil RP-18 column (250 mm 10 mm); mobile phase: water/ACN 75:25, 0.25% FA and ACN, 0.25% FA; HESI ionization source in negative mode; full MS followed by DDA scan |
In-house database, confirmation by NMR approaches | [107] | [74] | |||||||||||||||||
| Baoyuan decoction (traditional Chinese medicine formula) | reflux with water for 1.5 h | UHPLC–Q-TOF-MS/MS | Cortecs UPLC C18 column (1.6 μm, 2.1 × 100 mm; mobile phase: 0.05% aqueous formic acid and ACN containing 0.05% formic acid; ESI ionization in negative mode; DIA |
UNIFI software. In-house database |
[108] | [75] | ||||||||||||||||||
| The bark, twigs, leaves, and fruits of | Alnus japonica, Alnus hirsuta | and | Alnus hirsuta var. sibirica | UAE with MeOH (1:10 g DW/g), 60 °C, 60 min, at 60 kHz | UHPLC–Q-TOF-MS/MS | Acquity BEH C18 (100 × 2.1 mm 1.7 μm) column; mobile phase: H | 2 | O and MeCN, both of which were acidified with 0.1% formic acid; ESI ionization mode in negative ion; DDA (full MS followed by MS/MS scans for the three most intense ion). |
Spectral preprocessing using MZmine; in silico annotation with network; annotation propagation GNPS molecular networking; integration of annotation data using MolNetEnhancer |
[109] | [76] | |||||||||||||
| Medicago sativa | L. and | Trifolium pratense | L. dried sprouts | UAE with 70% ethanol (1:10 g DW/g), 60 °C, 60 min, at 60kHz | UHPLC-Q- Orbitrap HRMS/MS |
Accucore U-HPLC Column C18 (150 × 2.1 mm, 2.6 μm); mobile phase: water containing 500 μL/L | −1 | formic acid (pH 2.5) and MeOH; HESI ionization in negative mode; DIA (Full scan 70,000 FWHM, MS/MS 35,000 FWHM). Normalized collision energy: 30, 60 and 80 NCE |
Chemspider database; NORMAN MassBank, mzCloude | TM | Advanced Mass Spectral Database; in silico fragmentation with ACDLabs MS Fragmenter 2019.2.1 software | [110] | [77] | |||||||||||
| Blackcurrant ( | Ribes nigrum | L.) leaves | UAE with ethanol/water (1:1), 20 °C, 60 min | ESI-LTQ-Orbitrap MS | Kinetex Evo C18 5 μm column; mobile phase: water + 0.1% formic acid and ACN + 0.1% formic acid. ESI source in negative ion mode. DDA |
MZmine; PCA. | [111] | [78] | ||||||||||||||||
| Persimmon leaves (Chinese traditional medicine) | UAE with methanol/water (80:20, | v | / | v | ) | LC-ESI-LTQ- Orbitrap-MS | Atlantis T3 column 2.1 × 100 mm, 3 m; mobile phase water/0.1% formic acid and ACN; ESI source in both positive and negative ion mode; DDA approach-full scan at 30,000 FWHM and MS/MS at 15,000 FWHM; Collision energy (HCD cell)–35 kv |
Identification by generating the molecular formula using accurate mass (C = 30, H = 100, O = 15), and matching with the isotopic pattern. Searching in polyphenol database: ( | http://phenol-explorer.eu/ | ). Confirmation by comparison with HR-MS data in literature and databases |
[112] | [79] | ||||||||||||
| Flowers and leaves of Chilean Mistletoe (Quintral, | Tristerix tetrandus | ) | Lyophilized flowers and leaves were defatted thrice with | N | -hexane (1:10) and then extracted by UAE with 0.1% HCl in MeOH (1:10) for 60 min. Purification with XAD-7 column. | UHPLC–Q/Orbitrap/HRMS/MS | UHPLC C18 Column, Acclaim, 150 mm × 4.6 mm ID, 5 µm. Mobile phase: 0.1% aqueous formic acid and ACN 0.1% formic acid. HESI II ionization source in positive and negative mode. Full scan MS (70,000 FWHM) acquisition followed by targeted MS/MS analysis (17,500 FWHM). Collision energy: 30 kv. Mass accuracy ≤ 5 |
Trace Finder 3.2 software | [113] | [80] |
Hybrid mass spectrometers using linear ion capture technology, such as LTQ-Orbitrap, have also become common in this field. LTQ-Orbitrap provides the possibility of the screening, identification and structural characterization of unknown compounds using MSn fragmentation [111,112][78][79]. For example, the UHPLC technique coupled with LTQ-Orbitrap has recently been used for the characterization of components in Stellera chamaejasme [105][72] and in blackcurrant leaves [111][78].
Regarding the methods’ optimization, in the LC-HRMS/MS non-targeted screening analysis, resolution, scan rate and mass acquisition interval are the most critical parameters [99][65]. In addition to data acquisition modes also common in low-resolution spectrometry (SIM and MRM, PRM), new approaches such as data-dependent acquisition (DDA) and independent data acquisition (DIA) strategies, used so far only in proteomics, have recently been transposed into the analysis of small molecules in HRMS [110,114][77][81].
The limitation of the DDA strategy comes from the selection of precursors for MS/MS analysis [99][65]. Instead, in the recently developed independent data acquisition (DIA) strategy, all molecules within consecutive preselected m/z windows are subject to fragmentation, leading to higher specificity compared to the AIF (all ions fragmentation) approach [99,110][65][77]. As a disadvantage, the lack of precursor preselection can lead to impure mass spectra and low sensitivity. In this particular situation, the scan speed is the critical parameter [99,114][65][81].
In a recent study, Sun et al. [99][65] compared the DDA and DIA acquisition mode approaches using UHPLC-Q-TOF-MS technology for chemical characterization of the Aster tataricus rhizome, a traditional Chinese medicinal remedy. The key product ions strategy (KPIs) was used for the first time for searching for and identifying bioactive compounds. A total of 131 compounds, of which 31 were flavonoids, were identified or provisionally characterized. For the DDA setting, a complete scan and the ten most intense ion fragments from each analyte were applied to perform a TOF scan. For flavonoids, the ionic fragment m/z 153.0180 of kaempferol was used as the key ion in positive ionization. Based on the metabolic patterns of apigenin, isorhamnetin and kaempferol, several flavonoids were provisionally characterized: asapigenin-5-ramnoside, isorhamnetin-3-O-neohespeidoside, isorhamnetin-3-O-glucoside and biorobine (kaempferios 3-robin). The DDA strategy allowed the identification of 120 compounds, while 131 was identified by the DIA approach. However, certain glycosides identified in the DDA could not be detected by DIA [99][65].
The use of the DIA-MS strategy using an HRMS Q-Exactive Orbitrap instrument has recently been reported for the identification of polyphenols in extracts of red clover and alfalfa sprouts [110][77].
A workflow strategy in chemical profiling and metabolomics using HRMS technologies is shown in Figure 2.
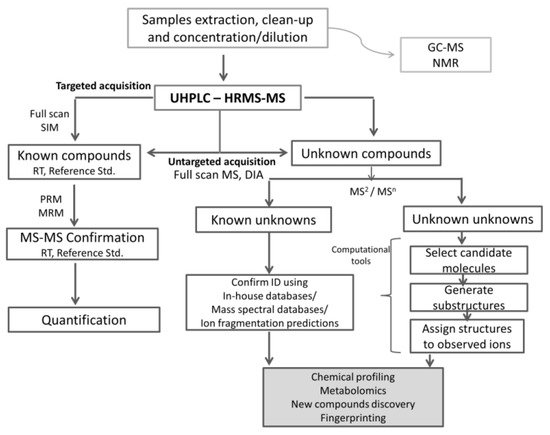

Figure 2. Workflow strategy in chemical profiling and metabolomics using HRMS technologies.
Although HRMS non-target screening offers clear advantages, investigating the enormous amount of data produced by such techniques remains a challenge [110][77]. The identification of unknown compounds in complex samples always requires MS and MS/MS databases, and the evaluation of the exact mass of the ions obtained by HRMS (molecular ion, ionic fragments, isotopic models) for provisionally assigning the chemical formula based on the rules defined in organic chemistry or using designed software.
Practically, considering polyphenolic compounds, the potential elementary molecular compositions of the compounds are established as C, H and O, then molecular formulas calculated based on mass accuracy are generated with a reasonable degree of measurement error (≤2 ppm). Based on molecular fragments and a ‘’match’’ factor of 70%, presumptive compounds are selected from a database, such as the Chemspider, (http://www.chemspider.com/), Pub-chem, (https://pubchem.ncbi.nlm.nih.gov/) or SciFinder database (https://scifinder.cas.org) or the polyphenol database (http://phenol-explorer.eu/) [105,110,111][72][77][78].
The comparison of the fragmentation patterns in MS-MS databases in the literature led to structure confirmation. There are currently various collections of public and private MS/MS mass spectra, such as NBS/EPA/NIH Mass Spectral Date Base, G.W.A. National Bureau of Standards Washinton; “eight peck index’’ at the Mass Spectrometry Data Center of Royal Society, Cambrige; Norman MassBank (https://massbank.eu/MassBank/); network clusters such as as mzCloudeTM (https://www.mycloud.com/#/); ReSpect, NIST (The National Institute of Standards and Technology; http://www.nist.gov/srd/nist1a.cfm) and Global Molecular Social Networking (GNPS). These allow users to compare the obtained MS-MS spectra with reference spectra for chemical structure annotation.
Spectrum processing and comparative analysis can be done manually, requiring extensive work and time, or automatically using software such as Compound DiscovererTM, Trace FinderTM, MassHunter, MasterViewTM, MZmine, MAGMa software, or MetabolitePilotTM.
In addition, predictors of silico fragmentation, such as as software tools like ACD Labs MS Fragmenter or Mass FrontierTM, are used to extend the primary annotation of presumptively identified molecules to the confirmation of the chemical structure [110][77].
Peak selection is one of the key steps in any non-target screening, and it can be done using a software algorithm (e.g., UNIFI platform by Waters Corporation). In the ‘‘suspect’’ compounds screening, the selection is made based on theoretical/predicted reference data. In non-target screening, presumptive peak identification can be performed when the signal strength for both the precursor ion and the fragments is sufficient to elucidate the molecular formula. Recently, Xu et al. [115][82] applied a UPLCQ-TOF-MSE method using data processing in the UNIFI computer platform for the determination of the chemical composition of Gandou decoction, a traditional Chinese medicine (TCM) formula. In total, 96 compounds, including flavonoids and phenolic acid, were identified or provisionally characterized based on retention time, exact mass (mass separation window of 5 ppm) and MS-MS fragmentation patterns [115][82].
In addition to MS databases, information on the environmental context (samples source, literature) may also be essential for the compounds’ identification. As well, combined statistical approaches, including chemometrics and bioinformatics, are currently being used to identify unknown or new compounds [116][83].
Guo et al. [116][83] performed a chemical profiling of several Keemun black tea classes via LC-Orbitrap-MS/MS. Data were subsequently analyzed using multivariate metabolomics statistics (PCA and HCA), leading to the identification of tea class markers (theasinensin A, afzelechin galate and kaempferol-glucoside).
The reported studies have highlighted that the digitized analysis of MS/MS data significantly improves the phytochemical analysis. Moreover, it demonstrates that molecules with high complexity can be identified and chemically characterized only with specialized software tools and require highly qualified personnel.
4.3. Analysis of Phenolic Compounds in Plants by GC-MS Using Derivatization Techniques
Although the use of GC coupled with MS and tandem MS-MS has been proven a powerful analytical tool for natural products’ characterization [117][84], there is a limited range of polyphenolic compounds that can be analyzed by GC-MS methods. Phenolic acids, phenolic aldehydes, ketones and phenolic alcohols were recently analyzed by GC-MS in wild plant fruits [117][84], fruit samples [36[85][86],118], Curcuma caesia Roxb [119][87], Kodo millet (Paspalum scrobiculatum) [120][88], and Scambiosa Columbabria L. [121][89]. Although newer two-dimensional GC x GC systems improved the separation performance and resolution [122][90], currently, the GC-MS technique is less commonly employed mainly because complex sample pre-treatments are required to increase the volatility and stability of the phenolic compounds. In addition to sample extraction and clean-up, a previous chemical derivatization step is needed to convert the hydroxyl groups to ethers, or make esters more volatile and thermostable.
Among the variety of derivation reagents, the most used is the trialkylsilyl, substituting the active hydrogen in alcohols and other polar organic compounds by the trimethylsilyl (TMS) group, –Si(CH3)3. N,O-bis-(trimethylsilyl)trifluoroacetamide (BSTFA), N,O-bis-(trimethylsilyl)acetamide (BSA), N-methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA) and N-[dimethyl-(2-methyl-2-propyl)silyl]-2,2,2-trifluoro-N-methylacetamide (MTBSTFA) are the most commonly used reagents [118,122][86][90]. Regarding the chromatographic conditions, fused silica capillary columns with lengths ranging from 25 to 30 m and inner dimensions from 0.25 to 0.5 mm, coated with 5% phenyl-95% dimethylpolysiloxane, are commonly used. The temperature gradients use initial column temperatures ranging from 40 to 80 °C, and final temperatures between 200 and 370 °C, with increasing rates ranging from 2 to 50 °C/min. High-purity helium is commonly used as a carrier gas at a flow-rate ranging from 0.4 to 3 mL/min, although high-purity hydrogen has also been used. Usually, GC-MS systems use the electron ionization (EI) mode prior to quadrupole MS quadrupole ion trap (QIT) and time-of-flight (TOF) analyzers [118,122][86][90].
4.4. Metabolomic Analysis of Polyphenolc Using Nuclear Magnetic Resonance Spectroscopy
Nuclear magnetic resonance (NMR) spectroscopy and high-resolution mass spectrometry (HRMS) are currently the main analytical methods applied in metabolomics studies [123][91]. Both techniques can enable two approaches for “profiling” studies: a non-targeted approach using chemometric analysis; and a targeted approach for the identification and quantification of known compounds in the plant extract [123,124][91][92]. Both techniques have met the requirements of metabolomics by being able to provide information that can lead to molecular structure characterization.
One of the disadvantages of HRMS compared to NMR is the complexity of operations, including elaborate sample preparation, chromatographic separation, molecule fragmentation, and extensive data processing, all of which requiring highly qualified personnel. In addition, because of the complexity and variability of the factors that influence the analysis, analytical protocols are difficult to standardize. Although less sensitive than mass spectrometry, NMR is highly reproducible, allowing the operations’ standardization [124][92]. Due to the distinct advantages of each method, NMR and HRMS are considered complementary techniques for the characterization of plant extracts [123][91].
Various open databases for the metabolites’ identification are currently being developed, such as the Human Metabolism Database (http://www.hmdb.ca), and the Biological Magnetic Resonance Databank (BMB) (http://www.bmrb.wisc.edu/). However, most of these databases contain information on human metabolites and very few resources for the identification of secondary metabolites of plants [74][46].
In NMR spectroscopy, the intensity of the peaks is directly proportional to the number of atomic nuclei, and hence, in combination with chemometric analysis, NMR is currently used to classify plant samples according to species, origin, processing, age or other quality parameters, based on the NMR profiles [74][46]. The main approach of metabolomics is to compare two data sets (for example, in the case of plant matrices several species of the same genus) and, through data filtering and multivariate analysis (PCA) techniques, a discrimination between these two data sets is performed. This process highlights any differences between the data and aims to identify the compounds responsible for this differentiation [74][46].
A typical NMR workflow for the identification of unknown compounds in natural extracts involves the collection of fractions containing the metabolites of interest after chromatographic separation followed by off-line NMR analysis. RMB databases and computational methods, such as the prediction and simulation software PERCH, or StrucEluc from ACD Labs, in combination with complete 1 H NMR iterative full-spin analysis (HiFSA approach), provide an accurate distinction between the natural compounds with almost identical NMR spectra. In addition, computer-assisted structural elucidation (CASE) is a methodology that allows users to enter NMR data and identify clusters by comparison algorithms [123,124,125][91][92][93].
Using an integrated approach (HPLC-DAD-MS/MS) and nuclear magnetic resonance (NMR) techniques, four derivatives of ellagic acid (ellagic hexoside, ellagic pentoside, methyl ellagic deoxiexoside acid, 4- (alpha-Ramnopyranosyl) ellagic acid (eschweilenol C)) were characterized for the first time in the biomass of Brazil nuts, Bertholletia excelsa Bonpl. (Lecythidaceae) [123][91]. A triple-quadrupole negative ionization was used for MS analysis, and NMR analysis was performed after an SPE fractionation of the plant extract.
References
- Sun, Z.; Zuo, L.; Sun, T.; Tang, J.; Ding, D.; Zhou, L.; Kang, J.; Zhang, X. Chemical Profiling and Quantification of XueBiJing Injection, a Systematic Quality Control Strategy Using UHPLC-Q Exactive Hybrid Quadrupole-Orbitrap High-Resolution Mass Spectrometry. Sci. Rep. 2017, 7, 16921.
- Pardo-Mates, N.; Vera, A.; Barbosa, S.; Hidalgo-Serrano, M.; Núñez, O.; Saurina, J.; Hernández-Cassou, S.; Puignou, L. Characterization, Classification and Authentication of Fruit-Based Extracts by Means of HPLC-UV Chromatographic Fingerprints, Polyphenolic Profiles and Chemometric Methods. Food Chem. 2017, 221, 29–38.
- Baranowska, I.; Bajkacz, S. A New UHPLC-MS/MS Method for the Determination of Flavonoids in Supplements and DPPH-UHPLC-UV Method for the Evaluation of the Radical Scavenging Activity of Flavonoids. Food Chem. 2018, 256, 333–341.
- Chen, L.-R.; Ko, N.-Y.; Chen, K.-H. Isoflavone Supplements for Menopausal Women: A Systematic Review. Nutrients 2019, 11, 2649.
- Minatel, I.O.; Borges, C.V.; Ferreira, M.I.; Gomez, H.A.G.; Chen, C.-Y.O.; Lima, G.P.P. Phenolic Compounds: Functional Properties, Impact of Processing and Bioavailability. In Phenolic Compounds: Biological Activity; IntechOpen: London, UK, 2017; pp. 1–24. ISBN 978-953-51-2960-8.
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The Effects of Polyphenols and Other Bioactives on Human Health. Food Funct. 2019, 10, 514–528.
- Šatínský, D.; Jägerová, K.; Havlíková, L.; Solich, P. A New and Fast HPLC Method for Determination of Rutin, Troxerutin, Diosmin and Hesperidin in Food Supplements Using Fused-Core Column Technology. Food Anal. Methods 2013, 6, 1353–1360.
- Bidlack, W.R.; Omaye, S.T.; Meskin, M.S.; Topham, D.K.W. Phytochemicals as Bioactive Agents; CRC Press: Boca Raton, FL, USA, 2000; ISBN 978-1-56676-788-0.
- Veeresham, C. Natural Products Derived from Plants as a Source of Drugs. J. Adv. Pharm. Technol. Res. 2012, 3, 200–201. Available online: (accessed on 5 April 2021).
- Tomás-Barberán, F.A.; González-Sarrías, A.; García-Villalba, R. Dietary Polyphenols: Metabolism and Health Effects; John Wiley & Sons: Hoboken, NJ, USA, 2020; ISBN 978-1-119-56371-6.
- Kim, H.-S.; Quon, M.J.; Kim, J. New Insights into the Mechanisms of Polyphenols beyond Antioxidant Properties; Lessons from the Green Tea Polyphenol, Epigallocatechin 3-Gallate. Redox Biol. 2014, 2, 187–195.
- Li, A.-N.; Li, S.; Zhang, Y.-J.; Xu, X.-R.; Chen, Y.-M.; Li, H.-B. Resources and Biological Activities of Natural Polyphenols. Nutrients 2014, 6, 6020–6047.
- Granado-Lorencio, F.; Hernández-Alvarez, E. Functional Foods and Health Effects: A Nutritional Biochemistry Perspective. Curr. Med. Chem. 2016, 23, 2929–2957.
- Sarian, M.N.; Ahmed, Q.U.; Mat So’ad, S.Z.; Alhassan, A.M.; Murugesu, S.; Perumal, V.; Syed Mohamad, S.N.A.; Khatib, A.; Latip, J. Antioxidant and Antidiabetic Effects of Flavonoids: A Structure-Activity Relationship Based Study. BioMed Res. Int. 2017, 2017, 8386065.
- Goszcz, K.; Duthie, G.G.; Stewart, D.; Leslie, S.J.; Megson, I.L. Bioactive Polyphenols and Cardiovascular Disease: Chemical Antagonists, Pharmacological Agents or Xenobiotics That Drive an Adaptive Response? Br. J. Pharmacol. 2017, 174, 1209–1225.
- Plaza, M.; Domínguez-Rodríguez, G.; Castro-Puyana, M.; Marina, M.L. 6—Polyphenols analysis and related challenges. In Polyphenols: Properties, Recovery, and Applications; Galanakis, C.M., Ed.; Woodhead Publishing: Cambridge, UK, 2018; pp. 177–232. ISBN 978-0-12-813572-3.
- Domínguez-Rodríguez, G.; Marina, M.L.; Plaza, M. Strategies for the Extraction and Analysis of Non-Extractable Polyphenols from Plants. J. Chromatogr. A 2017, 1514, 1–15.
- Rodrigues, S.; Fernandes, F.A.N.; de Brito, E.S.; Sousa, A.D.; Narain, N. Ultrasound Extraction of Phenolics and Anthocyanins from Jabuticaba Peel. Ind. Crops Prod. 2015, 69, 400–407.
- Collison, M.W. Determination of Total Soy Isoflavones in Dietary Supplements, Supplement Ingredients, and Soy Foods by High-Performance Liquid Chromatography with Ultraviolet Detection: Collaborative Study. J. AOAC Int. 2008, 91, 489–500.
- Gligor, O.; Mocan, A.; Moldovan, C.; Locatelli, M.; Crișan, G.; Ferreira, I.C.F.R. Enzyme-Assisted Extractions of Polyphenols—A Comprehensive Review. Trends Food Sci. Technol. 2019, 88, 302–315.
- Köseoğlu Yılmaz, P.; Kolak, U. SPE-HPLC Determination of Chlorogenic and Phenolic Acids in Coffee. J. Chromatogr. Sci. 2017, 55, 712–718.
- Bajkacz, S.; Baranowska, I.; Buszewski, B.; Kowalski, B.; Ligor, M. Determination of Flavonoids and Phenolic Acids in Plant Materials Using SLE-SPE-UHPLC-MS/MS Method. Food Anal. Methods 2018, 11, 3563–3575.
- Ruiz, A.; Sanhueza, M.; Gómez, F.; Tereucán, G.; Valenzuela, T.; García, S.; Cornejo, P.; Hermosín-Gutiérrez, I. Changes in the Content of Anthocyanins, Flavonols, and Antioxidant Activity in Fragaria Ananassa Var. Camarosa Fruits under Traditional and Organic Fertilization. J. Sci. Food Agric. 2019, 99, 2404–2410.
- Ding, M.; Bai, Y.; Li, J.; Yang, X.; Wang, H.; Gao, X.; Chang, Y. A Diol-Based-Matrix Solid-Phase Dispersion Method for the Simultaneous Extraction and Determination of 13 Compounds From Angelicae Pubescentis Radix by Ultra High-Performance Liquid Chromatography. Front. Pharmacol. 2019, 10, 227.
- Senes, C.E.R.; Nicácio, A.E.; Rodrigues, C.A.; Manin, L.P.; Maldaner, L.; Visentainer, J.V. Evaluation of Dispersive Solid-Phase Extraction (d-SPE) as a Clean-up Step for Phenolic Compound Determination of Myrciaria Cauliflora Peel. Food Anal. Methods 2020, 13, 155–165.
- Aguiar, J.; Gonçalves, J.L.; Alves, V.L.; Câmara, J.S. Chemical Fingerprint of Free Polyphenols and Antioxidant Activity in Dietary Fruits and Vegetables Using a Non-Targeted Approach Based on QuEChERS Ultrasound-Assisted Extraction Combined with UHPLC-PDA. Antioxidants 2020, 9, 305.
- Nicácio, A.E.; Rodrigues, C.A.; Jardim, I.C.S.F.; Visentainer, J.V.; Maldaner, L. Modified QuEChERS Method for Phenolic Compounds Determination in Mustard Greens (Brassica juncea) Using UHPLC-MS/MS. Arab. J. Chem. 2020, 13, 4681–4690.
- Gleichenhagen, M.; Schieber, A. Current Challenges in Polyphenol Analytical Chemistry. Curr. Opin. Food Sci. 2016, 7, 43–49.
- Fotovvat, M.; Radjabian, T.; Saboora, A. HPLC Fingerprint of Important Phenolic Compounds in Some Salvia L. Species from Iran. Rec. Nat. Prod. 2018, 13.
- Lei, Y.; Wang, Y.; Sun, Z.; Lin, M.; Cai, X.; Huang, D.; Luo, K.; Tan, S.; Zhang, Y.; Yan, J.; et al. Quantitative Analysis of Multicomponents by Single Marker Combined with HPLC Fingerprint Qualitative Analyses for Comprehensive Evaluation of Aurantii Fructus. J. Sep. Sci. 2020, 43, 1382–1392.
- Yang, F.; Chu, T.; Zhang, Y.; Liu, X.; Sun, G.; Chen, Z. Quality Assessment of Licorice (Glycyrrhiza glabra L.) from Different Sources by Multiple Fingerprint Profiles Combined with Quantitative Analysis, Antioxidant Activity and Chemometric Methods. Food Chem. 2020, 324, 126854.
- FDA. Guidance for Industry Botanical Drug Products; Center for Drug Evaluation and Research (CDER); Food and Drug Administration: Silver Spring, MD, USA, 2016; p. 34.
- SFDA State Drug Administration of China. Requirements for Studying Fingerprint of Traditional Chinese Medicine Injection (Draft). Chin. Tradit. Pat. Med. 2000, 22, 671–675.
- WHO. WHO Traditional Medicine Strategy 2002–2005; WHO: Geneva, Switzerland, 2002.
- Yu, S.; Zhu, L.; Xiao, Z.; Shen, J.; Li, J.; Lai, H.; Li, J.; Chen, H.; Zhao, Z.; Yi, T. Rapid Fingerprint Analysis of Flos Carthami by Ultra-Performance Liquid Chromatography and Similarity Evaluation. J. Chromatogr. Sci. 2016, 54, 1619–1624.
- Viapiana, A.; Struck-Lewicka, W.; Konieczynski, P.; Wesolowski, M.; Kaliszan, R. An Approach Based on HPLC-Fingerprint and Chemometrics to Quality Consistency Evaluation of Matricaria chamomilla L. Commercial Samples. Front. Plant Sci. 2016, 7, 1561.
- Hawrył, A.; Hawrył, M.; Waksmundzka-Hajnos, M. Liquid Chromatography Fingerprint Analysis and Antioxidant Activity of Selected Lavender Species with Chemometric Calculations. PLoS ONE 2019, 14, e0218974.
- Li, J.; He, X.; Li, M.; Zhao, W.; Liu, L.; Kong, X. Chemical Fingerprint and Quantitative Analysis for Quality Control of Polyphenols Extracted from Pomegranate Peel by HPLC. Food Chem. 2015, 176, 7–11.
- Liu, D.; Mei, Q.; Long, W.; Wan, X.; Wan, D.; Wang, L. HPLC Fingerprint Analysis and Content Determination of Extract with Anticancer Activities of Sedi Linearis Herba. Pharmacogn. J. 2017, 9, 128–134.
- Pan, H.-B.; Zhang, D.; Li, B.; Wu, Y.-Y.; Tu, Y.-Y. A Rapid UPLC Method for Simultaneous Analysis of Caffeine and 13 Index Polyphenols in Black Tea. J. Chromatogr. Sci. 2017, 55, 495–496.
- Viapiana, A.; Wesolowski, M. HPLC Fingerprint Combined with Quantitation of Phenolic Compounds and Chemometrics as an Efficient Strategy for Quality Consistency Evaluation of Sambucus Nigra Berries. Nat. Prod. Commun. 2016, 11, 1449–1454.
- Li, W.; Zhang, X.; Chen, R.; Li, Y.; Miao, J.; Liu, G.; Lan, Y.; Chen, Y.; Cao, Y. HPLC Fingerprint Analysis of Phyllanthus Emblica Ethanol Extract and Their Antioxidant and Anti-Inflammatory Properties. J. Ethnopharmacol. 2020, 254, 112740.
- Barreira, J.C.M.; Visnevschi-Necrasov, T.; Nunes, E.; Cunha, S.C.; Pereira, G.; Oliveira, M.B.P.P. Medicago Spp. as Potential Sources of Bioactive Isoflavones: Characterization According to Phylogenetic and Phenologic Factors. Phytochemistry 2015, 116, 230–238.
- Ojo, O.A.; Ojo, A.B.; Ajiboye, B.O.; Olaiya, O.; Okesola, M.A.; Boligon, A.A.; de Campos, M.M.A.; Oyinloye, B.E.; Kappo, A.P. HPLC-DAD Fingerprinting Analysis, Antioxidant Activities of Tithonia diversifolia (Hemsl.) A. Gray Leaves and Its Inhibition of Key Enzymes Linked to Alzheimer’s Disease. Toxicol. Rep. 2018, 5, 585–592.
- Wu, L.; Wu, W.; Cai, Y.; Li, C.; Wang, L. HPLC Fingerprinting-Based Multivariate Analysis of Phenolic Compounds in Mango Leaves Varieties: Correlation to Their Antioxidant Activity and in Silico α-Glucoidase Inhibitory Ability. J. Pharm. Biomed. Anal. 2020, 191, 113616.
- Bansal, A.; Chhabra, V.; Rawal, R.K.; Sharma, S. Chemometrics: A New Scenario in Herbal Drug Standardization. J. Pharm. Anal. 2014, 4, 223–233.
- Cacciola, F.; Farnetti, S.; Dugo, P.; Marriott, P.J.; Mondello, L. Comprehensive Two-Dimensional Liquid Chromatography for Polyphenol Analysis in Foodstuffs. J. Sep. Sci. 2017, 40, 7–24.
- López-Fernández, O.; Domínguez, R.; Pateiro, M.; Munekata, P.E.S.; Rocchetti, G.; Lorenzo, J.M. Determination of Polyphenols Using Liquid Chromatography–Tandem Mass Spectrometry Technique (LC–MS/MS): A Review. Antioxidants 2020, 9, 479.
- Llorent-Martínez, E.J.; Molina-García, L.; Ruiz-Medina, A.; Ortega-Barrales, P. Quantitation of Selected Polyphenols in Plant-Based Food Supplements by Liquid Chromatography–Ion Trap Mass Spectrometry. Food Anal. Methods 2014, 7, 2177–2183.
- Huang, F.-Q.; Dong, X.; Yin, X.; Fan, Y.; Fan, Y.; Mao, C.; Zhou, W. A Mass Spectrometry Database for Identification of Saponins in Plants. J. Chromatogr. A 2020, 1625, 461296.
- Kang, J.; Hick, L.A.; Price, W.E. A Fragmentation Study of Isoflavones in Negative Electrospray Ionization by MSn Ion Trap Mass Spectrometry and Triple Quadrupole Mass Spectrometry. Rapid Commun. Mass Spectrom. 2007, 21, 857–868.
- Vlaisavljević, S.; Kaurinović, B.; Popović, M.; Vasiljević, S. Profile of Phenolic Compounds in Trifolium Pratense L. Extracts at Different Growth Stages and Their Biological Activities. Int. J. Food Prop. 2017, 20, 3090–3101.
- Zhao, X.; Zhang, S.; Liu, D.; Yang, M.; Wei, J. Analysis of Flavonoids in Dalbergia Odorifera by Ultra-Performance Liquid Chromatography with Tandem Mass Spectrometry. Molecules 2020, 25, 389.
- Tao, W.; Zhou, Z.; Zhao, B.; Wei, T. Simultaneous Determination of Eight Catechins and Four Theaflavins in Green, Black and Oolong Tea Using New HPLC-MS-MS Method. J. Pharm. Biomed. Anal. 2016, 131, 140–145.
- Cuadrado-Silva, C.T.; Pozo-Bayón, M.Á.; Osorio, C. Targeted Metabolomic Analysis of Polyphenols with Antioxidant Activity in Sour Guava (Psidium friedrichsthalianum Nied.) Fruit. Molecules 2016, 22, 11.
- Gao, X.; Wang, N.; Jia, J.; Wang, P.; Zhang, A.; Qin, X. Chemical Profliling of Dingkun Dan by Ultra High Performance Liquid Chromatography Q Exactive Orbitrap High Resolution Mass Spectrometry. J. Pharm. Biomed. Anal. 2020, 177, 112732.
- Demarque, D.P.; Crotti, A.E.M.; Vessecchi, R.; Lopes, J.L.C.; Lopes, N.P. Fragmentation Reactions Using Electrospray Ionization Mass Spectrometry: An Important Tool for the Structural Elucidation and Characterization of Synthetic and Natural Products. Nat. Prod. Rep. 2016, 33, 432–455.
- Schmidt, J. Negative Ion Electrospray High-Resolution Tandem Mass Spectrometry of Polyphenols. J. Mass Spectrom. 2016, 51, 33–43.
- March, R.E.; Miao, X.-S.; Metcalfe, C.D.; Stobiecki, M.; Marczak, L. A Fragmentation Study of an Isoflavone Glycoside, Genistein-7-O-Glucoside, Using Electrospray Quadrupole Time-of-Flight Mass Spectrometry at High Mass Resolution. Int. J. Mass Spectrom. 2004, 232, 171–183.
- Fabre, N.; Rustan, I.; de Hoffmann, E.; Quetin-Leclercq, J. Determination of Flavone, Flavonol, and Flavanone Aglycones by Negative Ion Liquid Chromatography Electrospray Ion Trap Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 707–715.
- Zhu, Z.; Li, S.; He, J.; Thirumdas, R.; Montesano, D.; Barba, F.J. Enzyme-Assisted Extraction of Polyphenol from Edible Lotus (Nelumbo nucifera) Rhizome Knot: Ultra-Filtration Performance and HPLC-MS2 Profile. Food Res. Int. 2018, 111, 291–298.
- Pires, F.B.; Dolwitsch, C.B.; Dal Prá, V.; Faccin, H.; Monego, D.L.; de Carvalho, L.M.; Viana, C.; Lameira, O.; Lima, F.O.; Bressan, L.; et al. Qualitative and Quantitative Analysis of the Phenolic Content of Connarus Var. Angustifolius, Cecropia Obtusa, Cecropia Palmata and Mansoa Alliacea Based on HPLC-DAD and UHPLC-ESI-MS/MS. Rev. Bras. Farmacogn. 2017, 27, 426–433.
- Marczak, Ł.; Znajdek-Awiżeń, P.; Bylka, W. The Use of Mass Spectrometric Techniques to Differentiate Isobaric and Isomeric Flavonoid Conjugates from Axyris Amaranthoides. Molecules 2016, 21, 1229.
- Frański, R.; Gierczyk, B.; Kozik, T.; Popenda, Ł.; Beszterda, M. Signals of Diagnostic Ions in the Product Ion Spectra of [M − H]− Ions of Methoxylated Flavonoids. Rapid Commun. Mass Spectrom. 2019, 33, 125–132.
- Sun, Y.; Li, L.; Liao, M.; Su, M.; Wan, C.; Zhang, L.; Zhang, H. A Systematic Data Acquisition and Mining Strategy for Chemical Profiling of Aster tataricus Rhizoma (Ziwan) by UHPLC-Q-TOF-MS and the Corresponding Anti-Depressive Activity Screening. J. Pharm. Biomed. Anal. 2018, 154, 216–226.
- Simirgiotis, M.J.; Quispe, C.; Mocan, A.; Villatoro, J.M.; Areche, C.; Bórquez, J.; Sepúlveda, B.; Echiburu-Chau, C. UHPLC High Resolution Orbitrap Metabolomic Fingerprinting of the Unique Species Ophryosporus Triangularis Meyen from the Atacama Desert, Northern Chile. Rev. Bras. Farmacogn. 2017, 27, 179–187.
- Vallverdú-Queralt, A.; Regueiro, J.; Alvarenga, J.F.R.; Martinez-Huelamo, M.; Leal, L.N.; Lamuela-Raventos, R.M.; Vallverdú-Queralt, A.; Regueiro, J.; Alvarenga, J.F.R.; Martinez-Huelamo, M.; et al. Characterization of the Phenolic and Antioxidant Profiles of Selected Culinary Herbs and Spices: Caraway, Turmeric, Dill, Marjoram and Nutmeg. Food Sci. Technol. 2015, 35, 189–195.
- López-Gutiérrez, N.; Romero-González, R.; Plaza-Bolaños, P.; Martínez Vidal, J.L.; Garrido Frenich, A. Identification and Quantification of Phytochemicals in Nutraceutical Products from Green Tea by UHPLC-Orbitrap-MS. Food Chem. 2015, 173, 607–618.
- Lee, Y.H.; Kim, B.; Kim, S.; Kim, M.-S.; Kim, H.; Hwang, S.-R.; Kim, K.; Lee, J.H. Characterization of Metabolite Profiles from the Leaves of Green Perilla (Perilla frutescens) by Ultra High Performance Liquid Chromatography Coupled with Electrospray Ionization Quadrupole Time-of-Flight Mass Spectrometry and Screening for Their Antioxidant Properties. J. Food Drug Anal. 2017, 25, 776–788.
- Abdulla, R.; Mansur, S.; Lai, H.; Ubul, A.; Sun, G.; Huang, G.; Aisa, H.A. Qualitative Analysis of Polyphenols in Macroporous Resin Pretreated Pomegranate Husk Extract by HPLC-QTOF-MS. Phytochem. Anal. PCA 2017, 28, 465–473.
- Rocchetti, G.; Chiodelli, G.; Giuberti, G.; Ghisoni, S.; Baccolo, G.; Blasi, F.; Montesano, D.; Trevisan, M.; Lucini, L. UHPLC-ESI-QTOF-MS Profile of Polyphenols in Goji Berries (Lycium barbarum L.) and Its Dynamics during in Vitro Gastrointestinal Digestion and Fermentation. J. Funct. Foods 2018, 40, 564–572.
- Wang, Z.; Qu, Y.; Wang, L.; Zhang, X.; Xiao, H. Ultra-High Performance Liquid Chromatography with Linear Ion Trap-Orbitrap Hybrid Mass Spectrometry Combined with a Systematic Strategy Based on Fragment Ions for the Rapid Separation and Characterization of Components in Stellera Chamaejasme Extracts. J. Sep. Sci. 2016, 39, 1379–1388.
- Ben Said, R.; Hamed, A.I.; Mahalel, U.A.; Al-Ayed, A.S.; Kowalczyk, M.; Moldoch, J.; Oleszek, W.; Stochmal, A. Tentative Characterization of Polyphenolic Compounds in the Male Flowers of Phoenix Dactylifera by Liquid Chromatography Coupled with Mass Spectrometry and DFT. Int. J. Mol. Sci. 2017, 18, 512.
- Ben Haj Yahia, I.; Zaouali, Y.; Ciavatta, M.L.; Ligresti, A.; Jaouadi, R.; Boussaid, M.; Cutignano, A. Polyphenolic Profiling, Quantitative Assessment and Biological Activities of Tunisian Native Mentha rotundifolia (L.) Huds. Molecules 2019, 24, 2351.
- Ma, X.; Guo, X.; Song, Y.; Qiao, L.; Wang, W.; Zhao, M.; Tu, P.; Jiang, Y. An Integrated Strategy for Global Qualitative and Quantitative Profiling of Traditional Chinese Medicine Formulas: Baoyuan Decoction as a Case. Sci. Rep. 2016, 6, 38379.
- Kang, K.B.; Woo, S.; Ernst, M.; van der Hooft, J.J.J.; Nothias, L.-F.; da Silva, R.R.; Dorrestein, P.C.; Sung, S.H.; Lee, M. Assessing Specialized Metabolite Diversity of Alnus Species by a Digitized LC–MS/MS Data Analysis Workflow. Phytochemistry 2020, 173, 112292.
- Chiriac, E.R.; Chiţescu, C.L.; Borda, D.; Lupoae, M.; Gird, C.E.; Geană, E.-I.; Blaga, G.-V.; Boscencu, R. Comparison of the Polyphenolic Profile of Medicago Sativa L. and Trifolium Pratense L. Sprouts in Different Germination Stages Using the UHPLC-Q Exactive Hybrid Quadrupole Orbitrap High-Resolution Mass Spectrometry. Molecules 2020, 25, 2321.
- D’Urso, G.; Montoro, P.; Piacente, S. Detection and Comparison of Phenolic Compounds in Different Extracts of Black Currant Leaves by Liquid Chromatography Coupled with High-Resolution ESI-LTQ-Orbitrap MS and High-Sensitivity ESI-Qtrap MS. J. Pharm. Biomed. Anal. 2020, 179, 112926.
- Heras, R.M.-L.; Quifer-Rada, P.; Andrés, A.; Lamuela-Raventós, R. Polyphenolic Profile of Persimmon Leaves by High Resolution Mass Spectrometry (LC-ESI-LTQ-Orbitrap-MS). J. Funct. Foods 2016, 23, 370–377.
- Simirgiotis, M.J.; Quispe, C.; Areche, C.; Sepúlveda, B. Phenolic Compounds in Chilean Mistletoe (Quintral, Tristerix Tetrandus) Analyzed by UHPLC-Q/Orbitrap/MS/MS and Its Antioxidant Properties. Molecules 2016, 21, 245.
- Zhou, J.; Li, Y.; Chen, X.; Zhong, L.; Yin, Y. Development of Data-Independent Acquisition Workflows for Metabolomic Analysis on a Quadrupole-Orbitrap Platform. Talanta 2017, 164, 128–136.
- Xu, L.; Liu, Y.; Wu, H.; Wu, H.; Liu, X.; Zhou, A. Rapid Identification of Chemical Profile in Gandou Decoction by UPLC-Q-TOF-MSE Coupled with Novel Informatics UNIFI Platform. J. Pharm. Anal. 2020, 10, 35–48.
- Guo, X.; Long, P.; Meng, Q.; Ho, C.-T.; Zhang, L. An Emerging Strategy for Evaluating the Grades of Keemun Black Tea by Combinatory Liquid Chromatography-Orbitrap Mass Spectrometry-Based Untargeted Metabolomics and Inhibition Effects on α-Glucosidase and α-Amylase. Food Chem. 2018, 246, 74–81.
- Ahmad, N.; Zuo, Y.; Lu, X.; Anwar, F.; Hameed, S. Characterization of Free and Conjugated Phenolic Compounds in Fruits of Selected Wild Plants. Food Chem. 2016, 190, 80–89.
- Barrales, F.M.; Silveira, P.; Barbosa, P.D.P.M.; Ruviaro, A.R.; Paulino, B.N.; Pastore, G.M.; Macedo, G.A.; Martinez, J. Recovery of Phenolic Compounds from Citrus By-Products Using Pressurized Liquids—An Application to Orange Peel. Food Bioprod. Process. 2018, 112, 9–21.
- Marsol-Vall, A.; Balcells, M.; Eras, J.; Canela-Garayoa, R. Injection-Port Derivatization Coupled to GC-MS/MS for the Analysis of Glycosylated and Non-Glycosylated Polyphenols in Fruit Samples. Food Chem. 2016, 204, 210–217.
- Pakkirisamy, M.; Kalakandan, S.; Ravichandran, K.; Ravichandran, K. Phytochemical Screening, GC-MS, FT-IR Analysis of Methanolic Extract of Curcuma Caesia Roxb (Black Turmeric). Pharmacogn. J. 2017, 9, 952–956.
- Sharma, S.; Saxena, D.C.; Riar, C.S. Using Combined Optimization, GC-MS and Analytical Technique to Analyze the Germination Effect on Phenolics, Dietary Fibers, Minerals and GABA Contents of Kodo Millet (Paspalum scrobiculatum). Food Chem. 2017, 233, 20–28.
- Sagbo, I.J.; Orock, A.E.; Kola, E.; Otang-Mbeng, W. Phytochemical Screening and Gas Chromatography-Mass Spectrometry Analysis of Ethanol Extract of Scambiosa columbabria L. Pharmacogn. Res. 2020, 12, 35.
- Rohloff, J. Analysis of Phenolic and Cyclic Compounds in Plants Using Derivatization Techniques in Combination with GC-MS-Based Metabolite Profiling. Molecules 2015, 20, 3431–3462.
- Da Silva, F.M.A.; Hanna, A.C.S.; de Souza, A.A.; da Silva Filho, F.A.; Canhoto, O.M.F.; Magalhães, A.; Benevides, P.J.C.; de Azevedo, M.B.M.; Siani, A.C.; Pohlit, A.M.; et al. Integrative Analysis Based on HPLC-DAD-MS/MS and NMR of Bertholletia Excelsa Bark Biomass Residues:Determination of Ellagic Acid Derivatives. J. Braz. Chem. Soc. 2019, 30, 830–836.
- Der Kooy, F.V.; Venkataya, B.; Pearson, J.L.; Torres, A.; Li, C.G.; Chang, D. Sensitivity of NMR-Based Metabolomics in Drug Discovery from Medicinal Plants. Eur. J. Med. Plants 2015, 191–203.
- Valli, M.; Russo, H.M.; Pilon, A.C.; Pinto, M.E.F.; Dias, N.B.; Freire, R.T.; Castro-Gamboa, I.; Bolzani, V.D.S. Computational Methods for NMR and MS for Structure Elucidation II: Database Resources and Advanced Methods. Phys. Sci. Rev. 2019, 4.
More
