1. Introduction
Ionizing radiation (radiation) is commonly used for medical diagnosis and cancer treatment. Amongst these uses, radiation therapy is known to be one of the most effective treatments for cancer. It is difficult to control radiation-induced damage with conventional radiation therapy; therefore, intensity-modulated radiation therapy (IMRT) has recently been used
[1]. However, various radiation damages can also occur with IMRT. The harmful effects of radiation on the living body can be classified into direct and indirect effects. Direct effects are caused by the direct absorption of radiation energy into nucleic acids (DNA), proteins, and lipids
[2][3][4][5][2,3,4,5]. Indirect effects are caused by free radicals, such as hydroxyl radicals (•OH), and molecular products generated in the process of water radiolysis
[2][3][4][5][2,3,4,5]. In addition to the direct damage on DNA, secondary damages to non-DNA targets cannot be ignored because low-dose radiation damage is mainly caused by these indirect effects. Secondary damages include oxidation, inflammation, apoptosis, and effects on gene expression related to intracellular responses.
Medical applications of H
2 were first reported by Dole et al. in 1975
[6]. They reported that the inhalation of hyperbaric H
2 caused a marked regression in squamous cell carcinoma in mice induced by UV radiation. With the exception of a few studies, however, H
2 has not been extensively studied for medical applications. In 2007, Ohsawa et al. reported that the inhalation of H
2 gas ameliorated ischemia-reperfusion injury in a rat model with cerebral infarction
[7]. In this paper, they showed that H
2 is an antioxidant that selectively reduces highly oxidative reactive oxygen species (ROS) and reactive nitrogen species (RNS), such as •OH and peroxynitrite (ONOO
−), respectively, but does not react with other ROS such as superoxide anions (O
2−) and hydrogen peroxide (H
2O
2). However, we need to reacquaint ourselves with the pioneering paper on the antioxidant effects of H
2 by Yanagihara et al.
[8]in 2005,
published two years before the study by Ohsawa et al.
[7][8]. They reported that the ingestion of neutral H
2-rich water produced by water electrolysis alleviated liver damage in rats induced by chemical oxidants. These papers have led to global research on the medical applications of H
2. We recently showed that although H
2 is an inactive substance, compared to other antioxidants, it is the only molecule with mitochondrial permeability and an ability to reduce •OH, which is promising for future medical applications
[9][10][9,10]. Selective •OH scavengers may have potential medical applications as radioprotective agents. The efficacy of H
2 against various diseases and disease models have been reported, and there are now more than 1000 papers on the medical applications of H
2, including 80 clinical trials.
The use of a safer and more effective radioprotective agent in clinical practice is of great importance. Many drugs have been evaluated in a variety of ways. For instance, the radioprotective effects of many synthetic and natural compounds have been investigated. Cytokines such as granulocyte-macrophage colony-stimulating factor (GM-CSF), interleukin (IL)-1, IL-12, and natural compounds such as vitamin C, vitamin D, vitamin E, melatonin, succinate, alpha lipoic acid, and N-acetyl cysteine (NAC) have been reported to exhibit radioprotective effects in animal studies
[11][12][13][14][15][16][11,12,13,14,15,16]. Many drugs are in various stages of evaluation, but many are far from being ideal radioprotective agents. However, amifostine (WR2721) has been developed as a radioprotective agent with free radical scavenging properties, such as against •OH, and is the only radioprotective agent approved by the U.S. FDA for clinical use
[17][18][19][20][21][22][17,18,19,20,21,22]. However, this drug has not been widely considered as a useful radioprotective agent of choice because of its dose-dependent side effects such as hypotension, nausea, and vomiting
[20]. Therefore, it is not an exaggeration to say that there are no clinically usable radioprotective agents with high efficacy and few side effects.
On the other hand, H
2 has been reported to show radioprotective effects in many animal studies, and because H
2 has also shown to have no side effects in clinical studies, it may be a clinically reliable radioprotective agent. As for its radioprotective effects in clinical trials, Kang et al. reported that H
2-rich water improved the quality of life (QOL) of liver cancer patients receiving radiotherapy
[23]. We recently reported that the inhalation of H
2 gas reduced bone marrow damage in end-stage cancer patients receiving IMRT without compromising the antitumor effects
[24][25][24,25].
2. Biological Effects of Radiation
Exposure to radiation induces many detrimental effects, including genetic mutation, cell death, and carcinogenesis. The most radiation-sensitive organs are in the hematopoietic, digestive, reproductive, and skin systems, consisting of those with high cell proliferation
[26][27][26,27]. Radiation damage occurs at the cellular level, either directly or indirectly. Thus, harmful effects of radiation on living organisms can be divided into direct and indirect effects
[2][3][4][5][2,3,4,5].
Direct damages occur when radiation energy is directly absorbed by the target molecule, DNA. This direct action excites or ionizes the DNA, making it unstable because of the extra energy that is accumulated. In the process of releasing this extra energy, the ionization of DNA directly breaks chemical bonds in the DNA
[2][3][4][5][2,3,4,5]. On the other hand, there are also indirect effects, which occur when molecules other than the target absorb radiation energy and produce active bodies, such as radicals, which eventually react with the target molecule. In aqueous solutions, radiation is first absorbed by water molecules to produce radicals and molecular products such as •OH, hydrogen radicals (H•), hydration electrons (e
−aq), H
2, and H
2O
2 [4] (). These active substances then move through the water and induce chemical reactions with DNA.
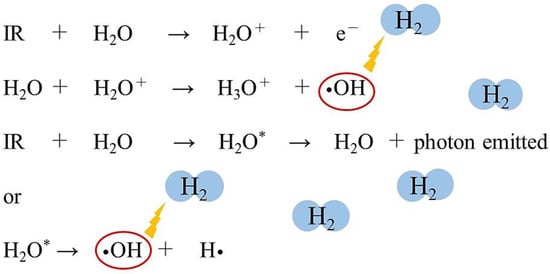
Figure 1. Ionizing radiation (IR) acts on water, a component of living organisms, ionizing and exciting the water molecules. Short-lived radical-cations (H2O+) are very unstable and decompose to produce hydroxyl radicals (•OH) and hydronium (H3O+). Electronically excited water molecules (H2O*) cleave to produce •OH and hydrogen radicals (H•). Molecular hydrogen (H2) can selectively eliminate the •OH by the following chemical reaction: •OH + H2 → H• + H2O.
In other words, radiation acts on water, which is a constituent of cells, and causes the ionization and excitation of water molecules. The water molecule ion (H
2O
+) is highly unstable and produces •OH and hydronium (H
3O
+). Excited water molecules (H
2O*) cleave to produce •OH and H•. The electrons from the water molecules are trapped between other water molecules and produce e
−aq [4] (). Approximately 60–70% of DNA damage is induced by the indirect action of free radicals
[3].
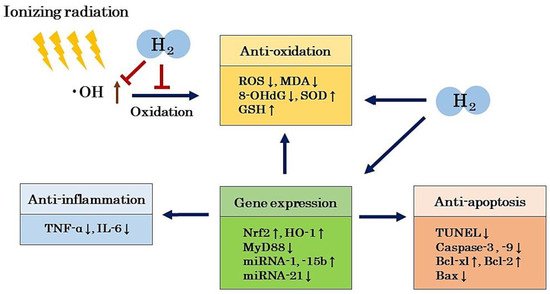
The •OH produced during water radiolysis causes the oxidation of DNA, lipids, amino acids, and saccharides, and the oxidation of these biological materials leads to the formation of various secondary free radicals
[26][27][26,27]. DNA is one of the major targets of free radicals. The compound 8-hydroxydeoxyguanosine (8-OHdG) is produced by •OH from deoxyguanosine in DNA and is considered to be one of the biomarkers of DNA damage and carcinogenesis
[28][29][28,29]. Structural changes in proteins are induced by •OH and other free radicals, leading to functional changes in proteins
[30]. Lipids in cell membranes are one of the major targets of •OH and other free radicals. Lipid peroxides such as malondialdehyde (MDA) and 2-thiobarbituric acid reactive substances (TBARS) are indicators of lipid damage
[31]. These lipid peroxides induce changes in cell membrane permeability
[32].
On the other hand, as an indirect effect of radiation, the molecular products generated by water radiolysis, such as e
−aq, H
2 and H
2O
2, also cause chemical reactions in biomolecules
[4]. In particular, low doses of radiation induce modifications of intracellular molecules, leading to effects on oxidation, inflammation, apoptosis, and gene expression. It has been reported that there is also a bystander effect, in which information can be transmitted from exposed cells to unexposed cells, transferring radiation damage to these unexposed cells
[33], as well as an abscopal effect, in which the local radiation therapy of a tumor can also shrink distant untreated tumors
[34]. The involvement of radiation in cellular responses and the immune system has also been considered. Furthermore, the effects of radiation on epigenetic effects, i.e., changes in gene expression or cellular phenotypes that are inherited after cell division without changes in DNA sequence, have also been pointed out
[35].