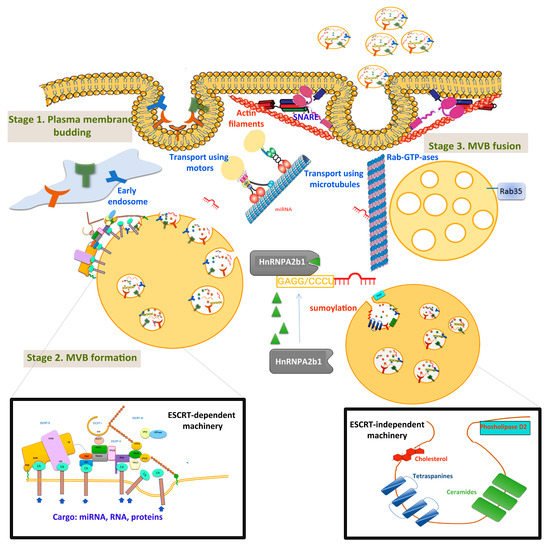
3.1. Correlation of Exosomal miRNAs with the Clinicopathological Features and Prognosis of HCC
The current incidence and mortality rates of HCC tend to be high. The five-year survival rate for HCC-diagnosed patients is 6%. This unfavourable prognosis stems from the aggressive progression of tumours and high recurrence rates
[56,57][56][57]. Therefore, early diagnosis is the most important prerequisite for the success of HCC treatment. Exosomes from various sources have different molecular expression profiles, important when comparing the exosome content of patients with those extracted from healthy individuals.
The population of exosomes is relatively simple and stable, transporting different functional molecules—such as proteins, miRNAs, mRNAs, and DNA—to the targeted cells through circulation. Out of all of the above, miRNA species are currently the most investigated molecules as putative biomarkers in HCC therapy, because of their specific signatures and correlation with the presence of disease or with clinical features such as tumour size, disease staging, overall survival, and disease recurrence. Analysis of exosomal miRNA species (mainly from serum) is useful for early detection and assessment of disease progression, without the need for tumour biopsy, which is a major advantage in the diagnosis of liver cancer.
Based on the published data, the role of miRNAs specific to exosomes can be divided into (1) miRNAs specific to HCC patients, (2) miRNAs that significantly correlate with the early stages of the disease, and (3) miRNAs that correlate with tumour staging. Changes in the expression levels of all of these miRNA species derived from exosomes have implications for the diagnosis of hepatic disorders.
illustrates different miRNA species extracted from the serum of HCC patients and identified as being clinically significant. miRNA quantification was performed with qRT-PCR. As illustrated, species of miRNA are either over-expressed—as indicated for miR-224, miR-21, miR-210-3p, miR-93, miR-92b, miR-155, and miR-665—or under-expressed—as for miR-718, miR-744, miR-9-3p, and miR-125b—in HCC patients. To this end, we lack explanations about the high variation of miRNA species in HCC patients, which downsizes their specificity for this disease. The crucial message is that exosomal miRNAs are different or better expressed than those extracted from serum, and that some of them have expression correlated with tumour stage or clinical parameters.
Table 1. Exosomal miRNAs with clinical significance in HCC.
| miR Species |
Expression Profile in HCC Down- or Upregulated |
Exosome Isolation Methods |
Groups and Sample Size |
Normalized (Reference miRs/Internal Control) |
Clinical Significance |
References |
| miR-224 |
Up |
Total Exosome Isolation Kit |
HCC (n = 89) and healthy controls (n |
The main endpoints in HCC investigation are overall survival (OS) and disease-free survival (DFS); therefore, a direct connection between exosomal miRNAs and patients’ outcomes would be a valuable asset for clinical research.
summarizes statistical correlation between the expression of different miRNAs and clinical data. We selected the reports in which the expression levels of miRNAs were obtained following ROC curve analysis, and the diagnostic values were investigated using the area under the curve (AUC) and p-values.
Table 2. Exosomal miRNAs with statistically significant expression with respect to clinical data.
| No. |
miR Species |
Diagnostic Values (AUC Values, Area under the Curve) |
Correlation with Prognosis and/or with Clinicopathological Features (p Values) |
References |
| AUC Values (>0.7) |
p Values |
Large Tumour Size (>3 cm or 5 cm) |
Advanced Tumour Stage (III/IV) |
Kaplan–Meier Curve Analysis: OS and DFS |
| = 50) |
let-7i, let-7g, and let-7d |
Detection, prognosis, recurrence, and therapuetic target |
| 1 |
miR-224 |
0.910 (95% CI: 0.84–0.98) |
p < 0.001 |
p < 0.001 | [ | 58 |
p < 0.001 | ] |
| p | < 0.01 |
[ | 58 | ] |
miR-21 |
Total Exosome Isolation Reagent (serum) |
HCC (n = 30), CHB (n = 30), and healthy controls (n = 30) |
U6 |
Detection and diagnosis |
[59] |
| 2 |
miR-93 |
0.825 (95% CI: 0.730–0.919) |
p < 0.0001 |
p = 0.047 |
p = 0.006 |
p = 0.046 |
[60] |
miR-93 |
Total Exosome Isolation kit |
HCC (n = 85) and healthy controls (n = 23) |
miR-16 |
| 3 |
miR-92b | Detection, prognosis, and recurrence |
0.702 (95% CI: 0.576–0.828) | [ |
p = 0.004 |
ND | 60] |
| ND |
ND |
[ | 62 | ] |
miR-1247-5p |
Ultracentrifugation |
HCC patients without lung metastasis (n = 90), HCC patients suffering lung metastasis (n = 20), and healthy controls (n = 25) |
18S |
Detection, diagnosis, and therapuetic target |
[61 |
| 4 | ] |
| miR-665 |
ND |
p | = 0.0042 (<5 cm) |
p = 0.0276 |
p < 0.05 |
[65] |
miR-92b |
ExoQuick Exosome Precipitation Solution |
Non-HCC (n = 26), HCC no recurrence (n = 28), early HCC recurrence (n = 43), and late HCC recurrence (n = 22) |
Synthetic C. elegans miR-39 |
Detection, prognosis, and recurrence |
| 5 |
miR-718 | [ | 62 | ] |
| ND |
p | = 0.04 |
p | = 0.026 |
p = 0.0002 (DFS) |
[66] |
miR-210-3p |
Ultracentrifugation |
HCC (n = 104) and healthy controls (n = 60) |
cel-miR-67 (NC67) |
Detection, diagnosis, and therapuetic target |
[63] |
| 6 |
miR-125b |
0.739 (95% CI: 0.648–0.830) |
p = 0.048 |
p = 0.11 |
p = 0.011 |
p < 0.001 (DFS and OS) |
[69] |
miR-155 |
| 5 |
miR-21, miR-10b |
up |
Ultracentrifugation |
Rat serum |
Male fisher 344 rats (n = 108) (HCC models) |
miRNA-484 |
Biomarkers for early-stage HCC combined with exosomal miRNAs and AFP. |
[84] |
ExoQuick Exosome Precipitation Solution (System Biosciences) |
HCC (n = 40): high level miR-155 of HCC (n = 20) and low level miR-155 of HCC (n = 20) |
Not mentioned |
Prognosis and recurrence |
[64] |
| miR-665 |
| 7 |
miR-194 |
0.738 (95% CI: 0.638–0.838) |
p = 0.0001 |
p |
| miR-122, miR-200a |
down | = 0.013 |
p | > 0.05 |
ND |
[ | 72] |
ExoQuick TM Kit (System Biosciences) |
HCC (n = 30) and healthy controls (n = 10) |
U6 |
Detection and prognosis |
[65] |
| miR-17-5p |
0.850 (95% CI: 0.764–0.936) |
p = 0.0001 |
p = 0.047 |
p > 0.05 |
ND |
miR-718 |
Down |
Ultracentrifugation |
Patients with HCC who underwent living donor liver transplantation (LDLT) (n = 59) |
Synthetic C. elegans miR-39 |
Detection, prognosis, recurrence, and therapeutic target |
[66] |
| miR-122 |
| miR-106a |
0.704 (95% CI: 0.534–0.873) |
p = 0.016 |
p = 0.035 |
p > 0.05 |
p = 0.041 |
ExoQuick |
Samples collected before and after TACE treatment of HCC patients (n = 75). |
cel-miR-39 |
Detection and prognosis |
[67] |
| 8 |
miR-10b-5p |
0.968 (95% CI: 0.85–0.99) |
p < 0.0001 |
ND |
not statistically significant |
[73] |
miR-638 |
Total Exosome Isolation kit |
HCC (n = 126) and healthy controls (n = 21) |
| miR-215-5p |
0.936 (95% CI: 0.80–0.99) |
p | miR-16 |
< 0.0001 |
ND | Detection, prognosis, and recurrence |
p < 0.01 | [68] |
| p | = 0.02 (DFS) |
mi-125b |
ExoQuick Exosome Precipitation Solution |
Group 1: CHB (n = 30), LC (liver cirrhosis) (n = 30), and HCC (n = 30). Group 2: HCC (n = 128) |
cel-miR-39 |
Detection, prognosis, and recurrence |
[69] |
| 9 |
miR-595 |
0.92 (95% CI: 0.86–0.97) |
p < 0.0001 |
ND |
p = 0.007 |
ND |
[74] |
miR-9-3p |
Three miRNAs—miR-125b, miR-665, and miR-638—are associated with improved OS rates
[65[65][68][69],
68,69], whereas exosomal miR-10b-5p overexpression significantly correlates with disease-free survival in patients with HCC
[73]. Despite improvements in therapies, the recurrence rates of HCC after surgical resection remain ≥10%. Thus, exploiting exosomal miRNAs (expressed pre- and post-surgery) as predictive markers for recurrence pursues the unmet clinical need.
The expression level of exosomal miR-92b in the serum of HCC patients was significantly higher than that of the control group (non-HCC)
[62]. If the levels of exosomal miR-92b continue to be upregulated, premature recurrence is induced. This result identifies exosomal miR-92b as a prognostic biomarker of post-transplant HCC recurrence. Sugimachi et al. showed using microarray analysis that miR-718 is a potential biomarker for predicting HCC recurrence after surgery. The authors reported different expression levels in patients with and without HCC recurrence
[66].
Many studies reported low expression of miR-155 in the plasma of HCC patients compared with that of healthy individuals
[75,76,77][75][76][77]. Others detected overexpressed miR-155 in preoperative plasma, and found it to be significantly correlated with early recurrence in patients with HCC
[64].
A different approach correlates exosomal miRNA expression with tumour staging, which allows for the selection of the most suitable biomarkers for early diagnosis. Overexpression of miR-665 in plasma exosomes correlates with tumour size, invasion, and staging
[65], whereas miR-21 expression strongly correlates with tumour stage and cirrhosis. Because exosomal miR-519d and miR-494 are upregulated in HCC compared with liver cirrhosis patients, they are considered to be independent diagnostic biomarkers
[70]. In addition, the level of exosomal miR-224 was significantly higher in patients with large tumours (>3 cm) and advanced tumour stages (III/IV)
[58]. Similarly, Shi et al. showed that low levels of exosomal miR-638 in HCC were significantly correlated with tumour size (>5 cm) and advanced staging (III/IV)
[68].
To gain a better understanding of intercellular communication, and ultimately improve therapeutic strategies for HCC, researchers have recently performed in vitro studies using HCC cell cultures treated with exosomes derived from other cells. HCC cells (Huh7 and SMMC 7721) proliferate significantly less when co-cultured with high concentrations of miR-638 extracted from serum exosomes
[68]. Clinical data support these results: low levels of miR-638 extracted from serum exosomes correlate with a lower survival rate of HCC patients. The authors imply that serum exosomal miR-638 affects liver carcinogenesis by inhibiting cancer cell proliferation. In contrast, the expression level of serum-derived exosomal miR-638 did not correlate with viral hepatitis B or C, tumour grade, or liver cirrhosis.
More complex studies were conducted to correlate the type of therapy and development of HCC with the expression of different exosomal miRNAs, based on the putative association of exosomal miRNA species with hepatitis and cirrhosis in HCC patients. The relative expression of exosomal miR-122 (calculated as relative expression of miR-122 after/before TACE) is significantly decreased after TACE
[67]. One study was conducted on 57 patients with liver cirrhosis and 18 chronic hepatitis patients with HCC; miR-122 relative expression was higher in patients with liver cirrhosis and longer disease-specific survival. The authors point toward the applicability of miR-122 in the therapeutic guidance of TACE-treated patients.
Other studies suggest that exosomal miR-21 is a potential biomarker for the diagnosis of CHB (chronic hepatitis B) patients. Different reference genes (miRNAs and small RNAs) were used to normalize expression levels of exosomal serum-derived miRs—including miR-221, let-7a, miR-191, miR-26a, and miR-181a—in CHB patients, HCC patients, and healthy individuals. The expression level of exosomal miR-21 was significantly increased in the CHB group compared to the other two groups
[78].
Similarly, Murakami et al. analysed miRNA expression profiles from the serum of 64 CHC (chronic hepatitis C) patients and 24 controls with normal liver (NL). The specific expression patterns of exosomal miRNAs expressed in chronic liver disease and inflammation correlated with the types and grades of liver disease. The expression patterns of nine miRNA species (miR-1225-5p, miR-1275, miR-638, miR-762, miR-320c, miR-451, miR-1974, miR-1207-5p, and miR-1246) identified CHC and NL with 96.59% accuracy
[79].
Exosomes isolated from HCV J6/JFH-1-infected HuH-7.5 cells, and from the sera of chronic HCV-infected patients, contain the replication enhancers VHC-Ago2, miR-122, and HSP90. Recent studies have consistently shown that miR-122, Ago2, and HSP90 enhance HCV replication. Moreover, miR-122 is a host factor used by HCV for replication, and is present in exosomes isolated from HCV patients
[80].
To date, we have identified many miRNA species isolated from exosomes, considering their potential roles as specific biomarkers for HCC. Therefore, scientists have used a more insightful approach of late: studies of miRNA profiling panels that match specific pathological conditions. Panels of miRNAs readily applicable as biomarkers or prognostic tools are yet to be discovered, but studies consider them to be sufficiently accurate to guide therapy in patients with advanced HCC disease. reviews in detail these exosomal miRNA panels and their clinical relevance. The exosomes’ sources are indicated.
Table 3. Exosome panels and their clinical relevance.
| No. |
miR Analysed in Panels |
Expression Profile in HCC (Up- or Downregulated) |
Exosome Isolation Methods |
Source of Exosomes |
Sample Size and Groups |
Quantification Methods |
Normalized (Reference miRs/Internal Control) |
Clinical Relevance |
References |
| 1 |
miR-122, miR-125b, miR-145, miR-192, miR-194, miR-29a, miR-17-5p, and miR-106a. |
up |
Total Exosome Isolation Kit (GenePharma) |
Human serum |
HCC (n = 80) and healthy controls (n = 30) |
qRT-PCR |
miR-16 |
Distinguish HCC patients from healthy controls. |
[72] |
| 2 |
miR-18a, miR-221, miR-222, miR-224 |
up |
ExoQuick Exosome Precipitation Solution |
HCC (n = 20), CHB (n = 20), and LC (n = 20) |
miR-16 |
Distinguish HCC from LC and CHB. |
[81] |
| miR-101, miR-106b, miR-122, miR-195 |
down |
| 3 |
miR-26a, miR-29c, miR-21 |
down |
ExoQuick reagent |
Human serum |
HCC (n = 20), CHB (n = 20), and LC (n = 20) |
let-7a |
Lower levels in HCC patients than in cirrhotic and HBV patients. |
[82] |
| 4 |
miR-122, miR-148a and miR-1246 |
up |
Polyethylene glycol (PEG) 6000 (Sigma-Aldrich, St Louis, MO) of 8% concentration |
Discovery group: HCC (n = 5) with LC (n = 5) for deep sequencing. Validation group: NC (n = 64), CHB(n = 50), LC (n = 53), and HCC(n = 68) for further study 85 HCC patients |
cel-miR-39 |
Distinguish early-stage HCC from liver cirrhosis. |
[83] |
| 6 |
| miR-10b-5p, miR-18a-5p, miR-215-5p, and miR-940 |
up |
SeraMir Exosome RNA Amplification Kit (System Biosciences) |
Human serum |
HCC ( | n = 90), CHB (n = 27), LC (n = 33), and healthy controls (n = 28) |
qRT-PCR and miR sequencing for discovery set |
miR-1228-3p |
Biomarker for early-stage HCC and poor disease-free survival |
[73] |
| 7 |
miR-519d, miR-21, miR-221 and miR-1228 |
up |
Ultracentrifugation |
30 patients (10 patients with liver cirrhosis without nodular liver lesions, 13 patients with early HCC, and 7 patients with advanced HCC) and HCC-derived cell lines (n = 7). HepG2, Hep3B, Huh-7, SNU449, SNU398, SNU182, and SNU475 |
qRT-PCR |
cel-miR-39 |
More efficient diagnostic role than AFP for HCC patients. |
[74] |
| 8 |
miR-140-3p, miR-30d-5p, miR-29b-3p, miR-130b-3p and miR-330-5p |
up |
3D Medicine exosome isolation kit (CFDA license no. Hu min xie bei 20170019) |
10 fast-migrated and 10 slow-migrated PDC cultures from 36 HCC samples |
Small RNA library construction and sequencing |
Normalization |
Migratory abilities of tumour cells |
[85] |
| Ultracentrifugation |
| miR-296-3p |
down |
| HCC ( | n | = 30) and healthy controls (n = 10) |
not mentioned |
Potential treatment/therapeutic target |
[70] |
| miR-744 |
Ultracentrifugation |
Group 1 (serum):HCC patients (n = 10) and healthy controls (n = 10). Group 2 (tissue): HCC patients (n = 68) and normal liver tissue (n = 52) |
U6 |
Detection, diagnosis, and therapeutic target |
[71] |
3.2. Effects of Exosomal microRNAs on Cell Survival, Proliferation, and Angiogenesis
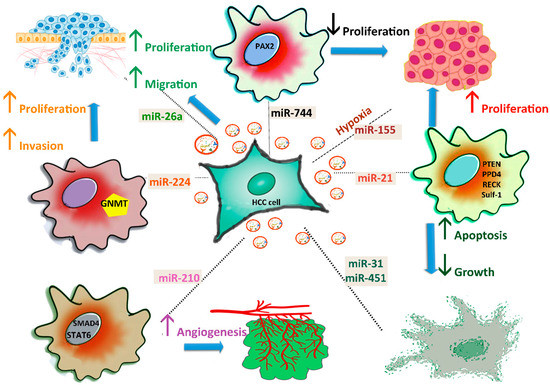
HCC cells can release exosomes to promote tumour cell proliferation, angiogenesis, migration, or metastasis ().
Figure 2. Roles of exosomal miRNAs in HCC progression. HCC cells secrete exosomes and affect proliferation by transferring miR-744, miR-21, and miR-26a into neighbouring cells. Angiogenesis is stimulated by miR-210, which blocks the SMAD4 and STAT6 pathways, while apoptosis is activated by miR-31 and miR-451. Inhibition of GNMT by miR-224 promotes the proliferation and invasion of HCC cells. miR-155 secretion increases under hypoxic conditions.
The two-way signalling pathways impact the drug resistance of HCC. For instance, high levels of miR-744 promote proliferation, and induce resistance of HepG2 cells to sorafenib, targeting paired box gene 2 (
PAX2), which is overexpressed in HCC tissue. Following treatment with exosomes, miR-744 significantly decreases HCC cell proliferation and resistance to therapeutic drugs
[71]. miR-21 induces cell proliferation and metastasis by inhibiting the expression of PTEN
[86], PDD4, RECK, and SULF-1 (human sulphatase-1)
[87], and confers resistance to chemotherapeutic drugs
[88].
In vitro studies indicate that miR-224 decreases the expression of
GNMT (glycine
N-methyltransferase) by directly targeting the 3’-UTR mRNA and promoting the proliferation and invasion of HCC cells
[58].
Exosomal miR-210 derived from HCC cells is internalized by endothelial cells, and promotes tumour angiogenesis through direct inhibition of the
SMAD4 (SMAD family member 4) and
STAT6 (signal transducer and activator of transcription 6) genes
[63]. Matsuura et al. showed that both cellular and exosomal miR-155 expression levels were significantly increased under hypoxic conditions in HCC cells
[64]. Of a group of six miRNAs, only miR-155 was upregulated under hypoxic conditions. Another study demonstrated that miR-31 and miR-451 species in exosomes derived from adult human liver stem cells inhibit HCC growth and stimulate apoptosis
[89].
The release of miR-26a species in HepG2 cells treated with exosomes led to ectopic overexpression of miR-26a and decreased migration and tumour proliferation. Exosomes loaded with miR-26a blocked cell growth, pointing to the possibility of using engineered exosomes as therapeutic agents
[90]. The next subsection of this review notes some results deciphering the mechanism of communication through exosomal miRNAs between HCC cells and cells from the tumour microenvironment.
3.3. Role of Exosomal microRNAs in Intra- and Intercellular Communication and Therapies
Exosomes secreted by HCC cells provide autocrine and paracrine signals to surrounding cells but also deliver products to distant cells, fine-tuning diverse biological responses. The process is two-way: blood cells or other cells from the tumoural microenvironment secrete exosomes, which mediate carcinogenesis. All of these processes have been intensively analysed, and many communications pathways have been depicted in recent years.
Of note, even during biogenesis, two miR species (miR-27b-3p and miR-92a-3p) inhibit VPS4A expression in HCC tissues
[20]. In turn, dysregulation of VPS4A facilitates the secretion of oncogenic miRNAs in exosomes. The expression of VPS4A in HCC tissue is linked to tumour growth and metastasis.
Exosomal miR-21 derived from HCC cell lines converts normal hepatic stellate cells (HSCs) into cancer-associated fibroblasts (CAFs) through directly targeting the
PTEN gene, which activates the PDK1/AKT signalling pathway. Finally, angiogenic cytokines (VEGF, MMP-2, MMP-9, FGF2, and TGF) are secreted, which promote cancer progression. These results concur with the clinical data: patients with HCC have high levels of serum exosomal miR-21—correlated with CAF activation, higher vessel density, and lower survival rates
[91].
A newly described mechanism shows that miRNAs secreted by HCC cells inhibit T cell function, escaping the immune system. Next-generation sequencing analysis (NGS) reveals that miR-23a-3p has a high expression level in exosomes derived from tunicamycin-treated HCC cells (Exo-TM). Co-cultivation of T cells with macrophages treated with Exo-TM leads to decreased cell ratio of CD8 T and decreased interleukin-2 expression, as a result of the activation of the PTEN/AKT pathway
[92].
Drug-resistant HCC cells (Bel/5-FU) secrete high levels of exosomal miR-32-5p, and low levels of the tumour suppressor gene
PTEN. This discrepancy results from miR-32-5p overexpression, which inhibits PTEN and, subsequently, activates the PI3K/AKT signalling pathway. Inhibited PTEN induces drug resistance, and promotes angiogenesis and EMT. Clinically, miR-32-5p overexpression and low PTEN expression are positively associated with poor prognosis in HCC patients
[93].
Exosome-derived miRNAs are mediators of the carcinogenesis induced by environmental chemicals. Exosomes derived from arsenite-transformed L-02 cells transfer miR-155 to normal L-02 and THLE-3 cells (grown in co-culture) and induce pro-inflammatory activity in normal liver cells. Moreover, exosome-derived miR-155 from serum was abundant in the patient group exposed to arsenite
[94].
In hepatocytes, miR-155 binds to Toll receptor ligands, triggering Toll receptor-mediated inflammation and promoting liver injury and inflammation
[95].
One study identifies new pathways of functional miRNA transfer from circulating tumour cells (CTCs) to the tumoural cells of origin, with the potential to inhibit tumour recurrence and metastatic spread. Exosomal miR-25-5p derived from HCC contributes to “tumour self-seeding”, defined as colonization by CTCs. This mechanism was linked to the expression of the
LRRC7 (Leucine-rich repeat-containing 7) gene involved in CTC reattachment and the reformation of tumour cell seeding. The uptake of exosomes secreted by CTC cells greatly improved the migratory and invasive abilities of HCC cells, with miR-25-5p declared as the main target of
LRRC7. Inhibition of exosomal miR-25-5p reversed these effects
[96].
Exosomal miR-103 produced by hepatic cells and delivered to endothelial cells (HUVECs, LSECs) and normal liver sinusoidal endothelial cells (Sk-Hep-1) inhibited protein junctions, zonula occludens-1, VE cadherin, and p120-catenin. MiR-103 reduced the integrity of endothelial junctions, increased the grade of vascular permeability and, finally, facilitated tumour metastasis
[97]. To analyse metastasis and proliferation of target cells, in vitro co-cultivation studies used the transfer of long non-coding RNA (lncRNA FAL1 (Focally amplified lncRNA on chromosome 1)) molecules to HepG2 and HuH-7 cell cultures. The competitive binding to miR-1236 in target cells leads to the overexpression of ZEB1 and AFP, and the subsequent promotion of metastasis and proliferation of the targeted cells
[98].
The development of HCC is mainly based on LC disorders, as the tumour microenvironment is enriched with activated fibroblasts. Highly metastatic HCC cells release exosomal miR-1247-3p to convert fibroblasts into CAFs by targeting the
B4GALT3 (β-1,4-galactosyltransferase 3) gene, which activates the β1-integrin/NF-κB signalling pathway. Consequently, the activated pathway decreases the secretion of the IL-6 and IL-8 cytokines. The same study showed that increased expression levels of exosomal miR-1247-3p significantly correlate with lung metastases in patients with HCC
[61]. CAFs induce tumourigenic processes, tumour niches, EMT (epithelial–mesenchymal transition), and chemoresistance. All of these processes are important in the development of the inflammatory microenvironment, and promote lung metastases derived from liver cancer. A different group performed sequencing analysis of CAFs and corresponding para-cancer fibroblasts (PAFs), and found a significant reduction in exosomal miR-320a levels in CAF-derived exosomes. The transfer of stromal-cell-derived miR-320a inhibits the tumour progression of HCC cells by binding to its direct target
PBX3. These results qualify the method of miRNA transfer as a potential treatment option for HCC progression
[99].
Human macrophages transfer miRNA species to HCC cells in a manner that requires cell contact through gap junctions. Two specific miRNAs—miR-142 and miR-223—are efficiently transferred; they block the stathmin-1 and IGFR-1 receptors, and finally HCC cell proliferation
[100].
Exosomal miR-21, miR-192, and miR-221 derived from colorectal cancer cell lines and transferred to HepG2 and A549 cells promoted the invasion and metastasis of recipient cells
[101]. Hep3B-derived exosomes containing specific miRNAs—such as miR-584, miR-517c, miR-378, miR-520f, miR-142-5p, miR-451, miR-518d, miR-215, miR-376a, miR-133b, and miR-367—were internalized in another HCC cell line, HepG2. Consecutively, these miRNAs modulated the TAK1 (TGF-β-activated kinase 1) inhibition process. Loss of TAK1 has been associated with hepatocarcinogenesis
[102].
Exosomes released from umbilical mesenchymal stem cells (UMSCs) inhibit HCV infection, especially viral replication. Qian et al. found that the anti-hepatitis C virus infection effect was mediated by various miRNAs (let-7f, miR-145, miR-199a, and miR-221) specifically transported by exosomes derived from UMSCs
[103].
The E2 envelope glycoprotein of the hepatitis C virus (HCV-E2) stimulated mast cells to secrete exosomes with high levels of miR-490. The exosome shuttled into HCC cells inhibited the ERK1/2 signalling pathway and, in the end, suppressed HCC cells’ metastasis
[104].
Using exosomes to mediate the transfer of miRNAs between different cells encourages clinical application, but for the moment, studies are limited to experiments using cell culture models. Exosomes operate on hepatocytes even when they derive from the cells of other tissues or organs, and contribute to the physiological or pathological processes of the liver
[105,106,107,108][105][106][107][108].
Adipose tissue-derived mesenchymal stem cells (AMSCs) transfected with a plasmid-encoding miR-122 (122-Exo) effectively loaded miR-122 in secreted exosomes. Then, 122-Exo was transported to HCC cells, and altered the expression profiles of cyclin G1 (
CCNG1),
ADAM10 (a disintegrin and metalloprotease domain-containing protein 10), and
IGFR-1 (insulin-like growth factor receptor 1). These changes occurred after HepG2 cells were exposed to 122-Exo for 24 h
[109]. AMSCs infected with pre-miR-199a-3p (LV-199a) plasmid abundantly expressed miR-199, and transferred the species via exosomes after puromycin selection. This process increased the sensitivity of HCC cells to doxorubicin by inhibiting the mTOR signalling pathway. Intravenous injection of an orthotopic HCC murine model with the AMSC-Exo-199a produces a rapid increase in the efficiency of doxorubicin therapy in HCC tumours
[110].
Other groups report the use of exosomes as nanotransporters in therapies, paving the way for new applications in HCC treatment. Exosomes transfected with antineoplastic and antifibrotic miR-335-5p were administered in vitro and in vivo to mice with developed HCC tumours; this treatment inhibited cell proliferation, invasion, and tumour growth. Previous reports demonstrated that HSC-derived exosomes can encapsulate and transfer miR-335-5p to HCC cells in vitro or in vivo, inhibiting tumour growth
[111]. In a recent study, exosomes were used as vehicles for the transport and targeted internalization of small RNA molecules in HCC cells. HepG2 cells were conditioned to bind (through the Apo-A1 receptor) and internalize exosomes designed by genetic engineering
[90].