Nanobiotechnology has grown rapidly and become an integral part of modern disease diagnosis and treatment. Biosynthesized silver nanoparticles (AgNPs) are a class of eco-friendly, cost-effective and biocompatible agents that have attracted attention for their possible biomedical and bioengineering applications. Like many other inorganic and organic nanoparticles, such as AuNPs, iron oxide and quantum dots, AgNPs have also been widely studied as components of advanced anticancer agents in order to better manage cancer in the clinic. AgNPs are typically produced by the action of reducing reagents on silver ions. In addition to numerous laboratory-based methods for reduction of silver ions, living organisms and natural products can be effective and superior source for synthesis of AgNPs precursors. Currently, plants, bacteria and fungi can afford biogenic AgNPs precursors with diverse geometries and surface properties.
- silver nanoparticles
- green chemistry
- cancer
- anti-cancer effect
Note: The following contents are extract from your paper. The entry will be online only after author check and submit it.
1. Introduction
2. Properties of AgNPs
2.1. Shape and Size
2.2. Optical Properties
2.3. Electrical Properties
3. Biologic Synthesis of AgNPs
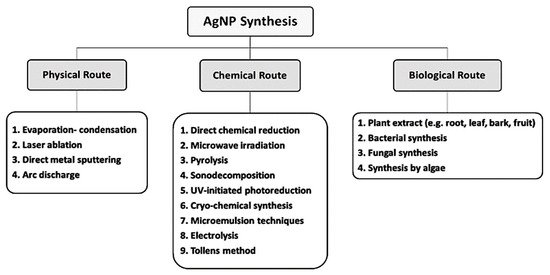
3.1. Bacterial Synthesis of AgNPs
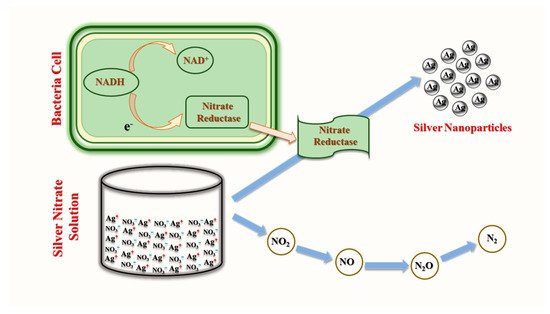
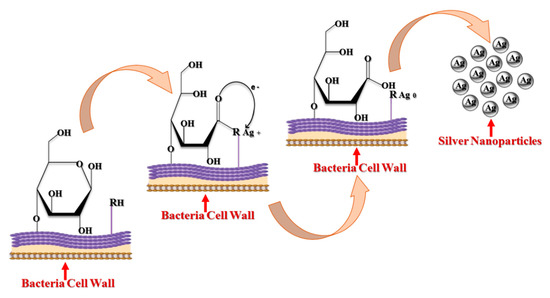
3.2. Fungal Synthesis of AgNPs
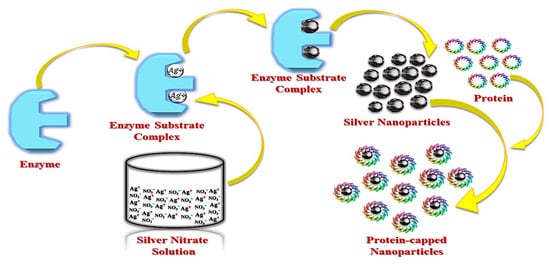
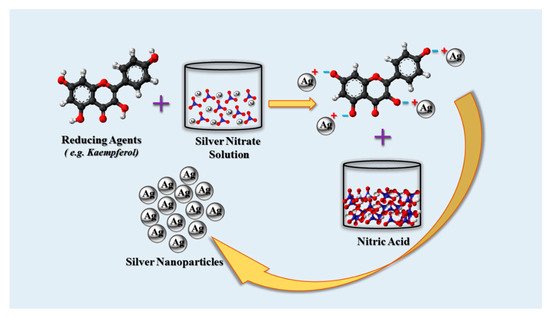
3.3. Plant Synthesis of AgNPs
| Plant | Phytochemical | Reference |
|---|---|---|
| Alternanthera tenella | Flavonoids | [65] |
| Cocos nucifera | Carbohydrates, alkaloids, terpenoids, tannins, saponins, phenolics and reducing sugars | [68] |
| Lemongrass | Reducing sugars | [69] |
| Ocimum sanctum | Caffeine | [70] |
| Chrysanthemum indicum | Tannins, flavonoids and glycosides | [71] |
| Dalbergia spinose | Reducing sugars and flavonoids | [72] |
| Cinnamomum camphora | Phenolics, terpenoids, polysaccharides and flavones | [73] |
| Eucalyptus hybrid | Flavonoids and terpenoids | [74] |
