Cellulose-based materials are a viable alternative to synthetic polymers due to their favorable physico-chemical and biological characteristics. They are also an appropriate organic matrix for the incorporation of hydroxyapatite particles, inter and intramolecular hydrogen bonds, as well as electrostatic interactions being formed between the functional groups on the polymeric chains surface and the inorganic filler. Considering the versatility of hydroxyapatite particles, the hybrid materials offer favorable prospects for applications in water purification, tissue engineering, drug delivery, and hemodialysis. The preparation technique and the chemical composition have a big influence on the final membrane properties. The well-established membrane fabrication methods such as phase inversion, electrospinning, or gradual electrostatic assembly are discussed, together with the various strategies employed to obtain a homogenous dispersion of the inorganic particles in the polymeric matrix. Finally, the main conclusions and the future directions regarding the preparation and applications of cellulose derivatives/hydroxyapatite composite membranes are presented.
- cellulose
- membrane
- hydroxyapatite
- water purification
- tissue engineering
1. Introduction
Among the functional materials currently known, membranes possess a unique characteristic and that is selectivity [1]. Another aspect is represented by the fact that they were the first functional materials known on earth—the membrane of the first unicellular organism [2]. Polymeric membranes were manufactured on a wide scale as filtering materials after the Second World War from the practical necessity of obtaining drinkable water from affected or contaminated natural sources. The first polymer applied industrially to obtain these membranes was cellulose nitrate, an explosive powder used to fabricate bombs and as a consequence, available in large quantities. Membrane technology was constituted in an individual scientific field starting with the research conducted by Loeb and Sourirajan that explained the formation mechanism of asymmetric membranes and the coagulation phenomenon of a polymer from concentrated solution in the presence of a non-solvent [3]. Once the formation mechanism of polymeric membranes was established and understood, more and more materials were developed for different practical applications such as gas separation [4][5][6], protein concentration [7][8][9], heavy metals separation [10], and removal of environmental pollutants [11][12][13]. As time passed, beyond their primary role as filtering materials used to obtain drinking water, membranes were employed in more and more advanced applications, the field with the most advanced performance demands being biomedical engineering. To fulfill the requests of such applications, composite membranes were developed by incorporating nanostructured inorganic fillers in the polymeric matrix thus resulting in a synergistic performance by combining the advantages of organic and inorganic materials [14]. One of the first niche domains targeted the obtainment of hemodialysis membranes [15]. Hemodialysis is the membrane process designed for patients with chronic kidney disease [16] and it is used to substitute kidney functions once every two days. The main chemical species that are separated during this process are urea, uric acid, and excess of creatinine and salts from the human body [17]. Other biomedical applications based on the extracorporeal blood circulation is the artificial lung used especially during open-heart surgeries [18][19] or experimental studies for an artificial liver based on composite membranes functionalized with porcine hepatocytes [20].
2. Cellulose Derivatives-Based Composite Membranes with Hydroxyapatite
Among the functional materials currently known, membranes possess a unique characteristic and that is selectivity [1]. Another aspect is represented by the fact that they were the first functional materials known on earth—the membrane of the first unicellular organism [2]. Polymeric membranes were manufactured on a wide scale as filtering materials after the Second World War from the practical necessity of obtaining drinkable water from affected or contaminated natural sources. The first polymer applied industrially to obtain these membranes was cellulose nitrate, an explosive powder used to fabricate bombs and as a consequence, available in large quantities. Membrane technology was constituted in an individual scientific field starting with the research conducted by Loeb and Sourirajan that explained the formation mechanism of asymmetric membranes and the coagulation phenomenon of a polymer from concentrated solution in the presence of a non-solvent [3]. Once the formation mechanism of polymeric membranes was established and understood, more and more materials were developed for different practical applications such as gas separation [4,5,6], protein concentration [7,8,9], heavy metals separation [10], and removal of environmental pollutants [11,12,13]. As time passed, beyond their primary role as filtering materials used to obtain drinking water, membranes were employed in more and more advanced applications, the field with the most advanced performance demands being biomedical engineering. To fulfill the requests of such applications, composite membranes were developed by incorporating nanostructured inorganic fillers in the polymeric matrix thus resulting in a synergistic performance by combining the advantages of organic and inorganic materials [14]. One of the first niche domains targeted the obtainment of hemodialysis membranes [15]. Hemodialysis is the membrane process designed for patients with chronic kidney disease [16] and it is used to substitute kidney functions once every two days. The main chemical species that are separated during this process are urea, uric acid, and excess of creatinine and salts from the human body [17]. Other biomedical applications based on the extracorporeal blood circulation is the artificial lung used especially during open-heart surgeries [18,19] or experimental studies for an artificial liver based on composite membranes functionalized with porcine hepatocytes [20].Due to the porosity and semi-permeable properties, polymeric membranes are also used in the development of controlled drug delivery devices [21], in an attempt to avoid the toxic effects of high quantities of pharmaceutically active substances on the human body. Another increasingly researched field of application is the one of osseointegration membranes that are placed at the interface between a metallic implant and the bone, with the purpose of favoring the pre-osteoblasts growth and spreading, in order to integrate the implant into the host bone tissue [22][23][24]. These latter membranes are based especially on composites with hydroxyapatite. Hydroxyapatite is one of the natural bone components, the so called “soft component”. It can be used as such, to obtain composites [25][26] or with the addition of other elements, like silver, to obtain hydroxyapatite with antibacterial properties [27]. Hydroxyapatite also has a high synthesis versatility because it can be of animal [28][29] or synthetic origin [30]. The present review desires to offer the reader an overview on the recent progress made in the domain of composite membranes based on cellulose derivatives and hydroxyapatite. Why these two components? They both present the great advantage of originating from natural sources, which is associated with biocompatibility of the resulting composites. More than that, once inserted into the human body, in the case of tissue engineering scaffolds, cellulose presents the remarkable property of bioresorbability, with only glucose molecules resulting after its hydrolysis and degradation. Cellulose derivatives-based membranes for various applications such as water purification, tissue engineering, osseointegration, drug delivery, and hemodialysis will be presented, all having in common hydroxyapatite as a filler agent.
Due to the porosity and semi-permeable properties, polymeric membranes are also used in the development of controlled drug delivery devices [21], in an attempt to avoid the toxic effects of high quantities of pharmaceutically active substances on the human body. Another increasingly researched field of application is the one of osseointegration membranes that are placed at the interface between a metallic implant and the bone, with the purpose of favoring the pre-osteoblasts growth and spreading, in order to integrate the implant into the host bone tissue [22,23,24]. These latter membranes are based especially on composites with hydroxyapatite. Hydroxyapatite is one of the natural bone components, the so called “soft component”. It can be used as such, to obtain composites [25,26] or with the addition of other elements, like silver, to obtain hydroxyapatite with antibacterial properties [27]. Hydroxyapatite also has a high synthesis versatility because it can be of animal [28,29] or synthetic origin [30]. The present review desires to offer the reader an overview on the recent progress made in the domain of composite membranes based on cellulose derivatives and hydroxyapatite. Why these two components? They both present the great advantage of originating from natural sources, which is associated with biocompatibility of the resulting composites. More than that, once inserted into the human body, in the case of tissue engineering scaffolds, cellulose presents the remarkable property of bioresorbability, with only glucose molecules resulting after its hydrolysis and degradation. Cellulose derivatives-based membranes for various applications such as water purification, tissue engineering, osseointegration, drug delivery, and hemodialysis will be presented, all having in common hydroxyapatite as a filler agent.Pure cellulose is frequently turned into cellulose derivatives to overcome the drawbacks related to its poor solubility in common organic solvents [31]. Due to the low cost and widespread availability of cellulose [32], cellulose acetate (CA) and carboxymethylcellulose (CMC) particularly are among the most used cellulose-based matrices for the incorporation of inorganic hydroxyapatite particles in order to obtain hybrid composite membranes with improved physico-chemical and biological characteristics.
Pure cellulose is frequently turned into cellulose derivatives to overcome the drawbacks related to its poor solubility in common organic solvents [31]. Due to the low cost and widespread availability of cellulose [32], cellulose acetate (CA) and carboxymethylcellulose (CMC) particularly are among the most used cellulose-based matrices for the incorporation of inorganic hydroxyapatite particles in order to obtain hybrid composite membranes with improved physico-chemical and biological characteristics.Cellulose acetate is a cellulose ester formed by partial or full acetylation of the free hydroxyl groups in the anhydroglucose unit. Depending on the acetyl content that usually ranges between 29% and 48%, mono-, di-, and triacetate can be differentiated [[33]. The ability of facile processing by various techniques and its broad range of applications make cellulose acetate the most commonly synthesized cellulose derivative worldwide, the global production of CA from biomass being projected by Global Industry Analysis to be 751.1 thousand metric tons until 2024 [34]. The classic cellulose acetylation process is based on the reaction between wood or cotton pulp with acetic anhydride as the acetylation agent and sulfuric acid as catalyst in an acetic acid reaction media [35]. Currently, this approach is used industrially but more and more research is being conducted on the use of agro-industrial residues and environmentally friendly synthesis routes that involve replacing the sulfuric and acetic acids by eco-friendly reagents [36]. Cellulose acetate is employed for the production of a variety of consumer goods including textiles, photographic films, personal hygiene products, and cigarette filters [37] but its resistance to the action of chemical agents, good thermal stability, flexibility, and mechanical strength [38], coupled with low fouling susceptibility and a hydrophilic nature [39], recommend this polymer especially for the production of membranes, with applications in industrial and biomedical processes (e.g., adsorption, separation, catalysis, biosensing, drug delivery, or tissue regeneration). Cellulose acetate was used in the purification process of contaminated resources of natural gas starting with the mid 1980s, when several companies applied dried CA membranes for CO
Cellulose acetate is a cellulose ester formed by partial or full acetylation of the free hydroxyl groups in the anhydroglucose unit. Depending on the acetyl content that usually ranges between 29% and 48%, mono-, di-, and triacetate can be differentiated [33]. The ability of facile processing by various techniques and its broad range of applications make cellulose acetate the most commonly synthesized cellulose derivative worldwide, the global production of CA from biomass being projected by Global Industry Analysis to be 751.1 thousand metric tons until 2024 [34]. The classic cellulose acetylation process is based on the reaction between wood or cotton pulp with acetic anhydride as the acetylation agent and sulfuric acid as catalyst in an acetic acid reaction media [35]. Currently, this approach is used industrially but more and more research is being conducted on the use of agro-industrial residues and environmentally friendly synthesis routes that involve replacing the sulfuric and acetic acids by eco-friendly reagents [36]. Cellulose acetate is employed for the production of a variety of consumer goods including textiles, photographic films, personal hygiene products, and cigarette filters [37] but its resistance to the action of chemical agents, good thermal stability, flexibility, and mechanical strength [38], coupled with low fouling susceptibility and a hydrophilic nature [39], recommend this polymer especially for the production of membranes, with applications in industrial and biomedical processes (e.g., adsorption, separation, catalysis, biosensing, drug delivery, or tissue regeneration). Cellulose acetate was used in the purification process of contaminated resources of natural gas starting with the mid 1980s, when several companies applied dried CA membranes for CO2
/CH4
natural gas separations. The hydrophilic nature of cellulose acetate makes the membranes suitable for assisting chemical and biochemical reactions as well as for the removal of polar compounds or specific organic–organic separations using pervaporation. The separation of helium particularly is of great interest in the natural gas purification process, due to its high added value. Asymmetric CA membranes presented an acceptable permeability for He and a good He/N2
and He/CH4 selectivity, being considered economically feasible for usage in three stage membrane processes with recycle streams [40]. Carboxymethyl cellulose, one of the most important cellulose ethers, is synthesized by treating alkali cellulose with monochloroacetic acid or sodium monochloro-acetate in an aqueous sodium hydroxide (NaOH) medium [41]. Wood residues, cotton linters, paper sludge, and agricultural waste biomass such as orange peels, corncobs, sugarcane bagasse, rice, or corn husks were used so far as cellulose sources for the preparation of carboxymethylcellulose [42]. As the reaction takes place, the hydroxyl groups in the cellulose backbone are replaced with carboxymethyl groups in the C6 > C3 > C2 order [43]. The chemical structure obtained following etherification is responsible for the unique properties of carboxymethylcellulose, such as water solubility, non-toxicity, biodegradability, transparency, and good film forming ability [44]. Owing to these characteristics, carboxymethylcellulose is already applied in the food, pharmaceutical, and daily-use chemical industries as an emulsifier, thickener, and a flocculating or chelating agent [45][46]. Currently, carboxymethylcellulose based materials are investigated for biomedical applications such as tissue engineering [47] and drug delivery [48] mainly in the shape of hydrogels, membranes, and nanoparticles [49].
selectivity, being considered economically feasible for usage in three stage membrane processes with recycle streams [40]. Carboxymethyl cellulose, one of the most important cellulose ethers, is synthesized by treating alkali cellulose with monochloroacetic acid or sodium monochloro-acetate in an aqueous sodium hydroxide (NaOH) medium [41]. Wood residues, cotton linters, paper sludge, and agricultural waste biomass such as orange peels, corncobs, sugarcane bagasse, rice, or corn husks were used so far as cellulose sources for the preparation of carboxymethylcellulose [42]. As the reaction takes place, the hydroxyl groups in the cellulose backbone are replaced with carboxymethyl groups in the C6 > C3 > C2 order [43]. The chemical structure obtained following etherification is responsible for the unique properties of carboxymethylcellulose, such as water solubility, non-toxicity, biodegradability, transparency, and good film forming ability [44]. Owing to these characteristics, carboxymethylcellulose is already applied in the food, pharmaceutical, and daily-use chemical industries as an emulsifier, thickener, and a flocculating or chelating agent [45,46]. Currently, carboxymethylcellulose based materials are investigated for biomedical applications such as tissue engineering [47] and drug delivery [48] mainly in the shape of hydrogels, membranes, and nanoparticles [49].Some of the most popular membrane manufacturing techniques include electrospinning, gradual electrostatic assembly, and phase inversion by immersion precipitation or solvent evaporation.
Some of the most popular membrane manufacturing techniques include electrospinning, gradual electrostatic assembly, and phase inversion by immersion precipitation or solvent evaporation.Phase inversion is a popular method for the preparation of cellulose acetate membranes. The first step of this process consists in the dissolution of the polymer in an appropriate solvent, such as hexafluoro-2-propanol [50], formic acid [51], N, N-dimethylformamide [52], acetone [53][54][55], or a mixed solvent system of the latter two [56], to obtain a homogenous polymeric solution. Afterwards, the obtained solution is cast on a glass plate and submerged in a coagulation bath containing a non-solvent, usually distilled water. The penetration of the solvent into the non-solvent and non-solvent into the polymeric solution cause demixing and polymer precipitation with the formation of a membrane with an asymmetric structure, composed in most cases of a thin film top layer also called “skin”, a support porous substructure, and a bottom layer [54][57].
Phase inversion is a popular method for the preparation of cellulose acetate membranes. The first step of this process consists in the dissolution of the polymer in an appropriate solvent, such as hexafluoro-2-propanol [50], formic acid [51], N, N-dimethylformamide [52], acetone [53,54,55], or a mixed solvent system of the latter two [56], to obtain a homogenous polymeric solution. Afterwards, the obtained solution is cast on a glass plate and submerged in a coagulation bath containing a non-solvent, usually distilled water. The penetration of the solvent into the non-solvent and non-solvent into the polymeric solution cause demixing and polymer precipitation with the formation of a membrane with an asymmetric structure, composed in most cases of a thin film top layer also called “skin”, a support porous substructure, and a bottom layer [54,57].Gradual electrostatic assembly is based on the spontaneous interactions between two oppositely charged polysaccharides, mixed in an aqueous solution. The electrostatic interactions are followed by polymeric chain entanglement and hydrogen bond formation, this resulting in a polyelectrolyte complex membrane. Due to its anionic nature, carboxymethylcellulose was used in combination with cationic polymers such as chitosan (CS) to prepare such composites [58].
Gradual electrostatic assembly is based on the spontaneous interactions between two oppositely charged polysaccharides, mixed in an aqueous solution. The electrostatic interactions are followed by polymeric chain entanglement and hydrogen bond formation, this resulting in a polyelectrolyte complex membrane. Due to its anionic nature, carboxymethylcellulose was used in combination with cationic polymers such as chitosan (CS) to prepare such composites [58].Cellulose acetate was among the first electrospun polymers [59]; it is considered suitable for electrospinning because it can be easily dissolved in common organic solvents and maintains high mechanical strength during membrane fabrication [60]. To date, there are no studies that report the effective electrospinning of carboxymethylcellulose without the addition of another polymer; nonetheless, blends of CMC with polyvinylpyrrolidone (PVP) [61], polyvinyl alcohol (PVA) [62], or polyethylene oxide (PEO) [63][64] were successfully electrospun into nanofibrous membranes with applications especially in the biomedical area. The electrospinning experiments usually take place at room temperature under normal atmospheric conditions. The device consists of three major parts: high voltage power supply, feeding nozzle or spinneret, and a grounded collecting plate (metal plate or rotating drum). The polymeric solution is inserted into a capillary tube connected to a feeding nozzle. A high voltage source is used to inject a certain polarity charge into the polymeric solution or melt. When the electric field reaches a critical value the repulsive electrical forces surpass the surface tension forces at the tip of the nozzle and the solution is accelerated towards the opposite polarity collector. The solvent is evaporated and polymeric fibers are formed [65]. Solution viscosity, polymer concentration, and molecular weight are important factors that influence the electrospinning process and an optimal balance between them must be achieved in order to generate uniform fibers. Due to their unique properties, electrospun fibers have been successfully applied in various domains such as environmental engineering, pharmaceutics, optoelectronics, biomedicine, and biotechnology [66].
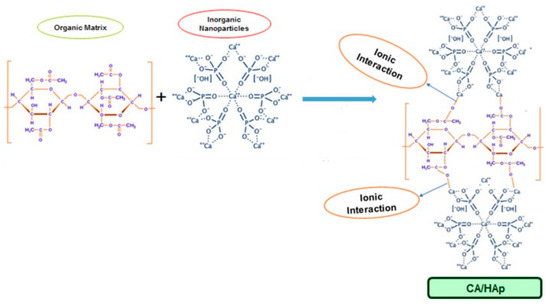
Cellulose acetate was among the first electrospun polymers [59]; it is considered suitable for electrospinning because it can be easily dissolved in common organic solvents and maintains high mechanical strength during membrane fabrication [60]. To date, there are no studies that report the effective electrospinning of carboxymethylcellulose without the addition of another polymer; nonetheless, blends of CMC with polyvinylpyrrolidone (PVP) [61], polyvinyl alcohol (PVA) [62], or polyethylene oxide (PEO) [63,64] were successfully electrospun into nanofibrous membranes with applications especially in the biomedical area. The electrospinning experiments usually take place at room temperature under normal atmospheric conditions. The device consists of three major parts: high voltage power supply, feeding nozzle or spinneret, and a grounded collecting plate (metal plate or rotating drum). The polymeric solution is inserted into a capillary tube connected to a feeding nozzle. A high voltage source is used to inject a certain polarity charge into the polymeric solution or melt. When the electric field reaches a critical value the repulsive electrical forces surpass the surface tension forces at the tip of the nozzle and the solution is accelerated towards the opposite polarity collector. The solvent is evaporated and polymeric fibers are formed [65]. Solution viscosity, polymer concentration, and molecular weight are important factors that influence the electrospinning process and an optimal balance between them must be achieved in order to generate uniform fibers. Due to their unique properties, electrospun fibers have been successfully applied in various domains such as environmental engineering, pharmaceutics, optoelectronics, biomedicine, and biotechnology [66].Lately, an increased research interest was directed towards the utilization of cellulose derivatives/hydroxyapatite composite membranes in environmental or biomedical engineering (e.g., water purification, bone tissue regeneration, wound healing, controlled drug delivery). It was found that there is a good compatibility between cellulose-based matrices and hydroxyapatite, the inorganic particles interacting with the organic component by inter and intramolecular hydrogen bonds, and also by ion-dipole forces formed between the calcium ions of HA and the functional groups on the cellulose derivatives surface [60]. For example, in the case of CA, the positively charged calcium ions bind with the negatively charged carboxylate groups and hydrogen bonds are formed between the hydroxyl groups of cellulose acetate and hydroxyapatite (
Lately, an increased research interest was directed towards the utilization of cellulose derivatives/hydroxyapatite composite membranes in environmental or biomedical engineering (e.g., water purification, bone tissue regeneration, wound healing, controlled drug delivery). It was found that there is a good compatibility between cellulose-based matrices and hydroxyapatite, the inorganic particles interacting with the organic component by inter and intramolecular hydrogen bonds, and also by ion-dipole forces formed between the calcium ions of HA and the functional groups on the cellulose derivatives surface [60]. For example, in the case of CA, the positively charged calcium ions bind with the negatively charged carboxylate groups and hydrogen bonds are formed between the hydroxyl groups of cellulose acetate and hydroxyapatite (Figure 1).
).
It is demonstrated that hydroxyapatite (HA) particles are good mineral adsorbers and have the capacity to bind divalent heavy metal ions, hence they were researched for the removal of harmful substances in drinking water [53]. Hydroxyapatite particles can also be used as additives for cellulose acetate to improve the morphology and properties of the membranes, this resulting in a better separation performance and higher water flux values [54]. More than that they could act as a catalyst for the precipitation process involved in phase inversion, by increasing the viscosity of the solution [39][57]. The most common synthesis methods for membranes are phase inversion with two possible variants—precipitation in a non-solvent and solvent evaporation. The most versatile is the precipitation of the membrane in a non-solvent. The non-solvent flow through solution polymer film will determine the porosity and morphology depending on several parameters—viscosity, temperature, and miscibility with the polymer solvent. These properties directly influence the speed of membrane formation—the higher the formation speed, the smaller the pore diameter with a high distribution of pores at the surface. A slow process of membrane coagulation will lead to pores with a large diameter and a lower distribution of pores on the surface. Each possibility is preferred depending on the application of the synthesized membrane. The presence of hydroxyapatite in a polymer solution acts in two ways—firstly it will increase the viscosity of the solution and second it will influence the speed of non-solvent through the polymer solution film. The first influence is always higher, so from a more viscous initial solution, membranes with a decreased diameter of pores will be obtained. This can be translated into a more efficient separation. More than that, hydroxyapatite particles itself, in the structure of the membrane, participate in the separation process due to their porosity and ability to retain small chemical species like cations, pesticides, and dyes.
It is demonstrated that hydroxyapatite (HA) particles are good mineral adsorbers and have the capacity to bind divalent heavy metal ions, hence they were researched for the removal of harmful substances in drinking water [53]. Hydroxyapatite particles can also be used as additives for cellulose acetate to improve the morphology and properties of the membranes, this resulting in a better separation performance and higher water flux values [54]. More than that they could act as a catalyst for the precipitation process involved in phase inversion, by increasing the viscosity of the solution [39,57]. The most common synthesis methods for membranes are phase inversion with two possible variants—precipitation in a non-solvent and solvent evaporation. The most versatile is the precipitation of the membrane in a non-solvent. The non-solvent flow through solution polymer film will determine the porosity and morphology depending on several parameters—viscosity, temperature, and miscibility with the polymer solvent. These properties directly influence the speed of membrane formation—the higher the formation speed, the smaller the pore diameter with a high distribution of pores at the surface. A slow process of membrane coagulation will lead to pores with a large diameter and a lower distribution of pores on the surface. Each possibility is preferred depending on the application of the synthesized membrane. The presence of hydroxyapatite in a polymer solution acts in two ways—firstly it will increase the viscosity of the solution and second it will influence the speed of non-solvent through the polymer solution film. The first influence is always higher, so from a more viscous initial solution, membranes with a decreased diameter of pores will be obtained. This can be translated into a more efficient separation. More than that, hydroxyapatite particles itself, in the structure of the membrane, participate in the separation process due to their porosity and ability to retain small chemical species like cations, pesticides, and dyes.An arising issue is represented by the aggregation tendency of the inorganic particles but various strategies such as ultrasound-assisted mixing [52], surfactants addition [54], or modification of the hydroxyapatite surface [56] are investigated for an improved dispersion. The aggregation tendency of hydroxyapatite particles in aqueous media was successfully overcome by using electrospinning as membrane fabrication technique. Electrospun membranes have some advantages over phase inversion membranes in terms of volume and aspect ratio, specific area, and porosity, but, according to a recent study, HA concentrations higher than 3 wt/v% generated a “beads on a string” morphology in the case of CA solutions (15 wt/v%) in a mixed solvent system of acetone and N,N-dimethylformamide (DMF) [[60]. Therefore, the inorganic filler concentration must be carefully chosen in order to obtain smooth nanofibers. Due to its biocompatibility, bioactivity, and osteoconductive properties, hydroxyapatite distinguished itself among materials used for bone tissue regeneration. However, the direct use of hydroxyapatite in such applications was associated with poor mechanical and chemical stability especially in the case of synthetic particles [67]. Therefore hybrid polymer/hydroxyapatite composites were developed, natural polymers such as collagen [68][69], polylactic acid [24][70], chitosan [71][72], and cellulose [73] being preferred, instead of synthetic ones [74], for the preparation of novel composite materials with superior bioactivity compared to pure components.
An arising issue is represented by the aggregation tendency of the inorganic particles but various strategies such as ultrasound-assisted mixing [52], surfactants addition [54], or modification of the hydroxyapatite surface [56] are investigated for an improved dispersion. The aggregation tendency of hydroxyapatite particles in aqueous media was successfully overcome by using electrospinning as membrane fabrication technique. Electrospun membranes have some advantages over phase inversion membranes in terms of volume and aspect ratio, specific area, and porosity, but, according to a recent study, HA concentrations higher than 3 wt/v% generated a “beads on a string” morphology in the case of CA solutions (15 wt/v%) in a mixed solvent system of acetone and N,N-dimethylformamide (DMF) [60]. Therefore, the inorganic filler concentration must be carefully chosen in order to obtain smooth nanofibers. Due to its biocompatibility, bioactivity, and osteoconductive properties, hydroxyapatite distinguished itself among materials used for bone tissue regeneration. However, the direct use of hydroxyapatite in such applications was associated with poor mechanical and chemical stability especially in the case of synthetic particles [67]. Therefore hybrid polymer/hydroxyapatite composites were developed, natural polymers such as collagen [68,69], polylactic acid [24,70], chitosan [71,72], and cellulose [73] being preferred, instead of synthetic ones [74], for the preparation of novel composite materials with superior bioactivity compared to pure components.