The management of large volumes of organic residues generated in different livestock, urban, agricultural and industrial activities is a topic of environmental and social interest. The high organic matter content of these residues means that their application as soil organic amendments in agriculture is considered one of the more sustainable options, as it could solve the problem of the accumulation of uncontrolled wastes while improving soil quality and avoiding its irreversible degradation. However, the behavior of pesticides applied to increase crop yields could be modified in the presence of these amendments in the soil.
- soil amendment
- organic matter
1. Introduction
The use of large quantities of pesticides in today’s intensive agricultural systems is a widespread practice for controlling pests, diseases and weeds. This increases the yield per hectare, ensuring the food supply for the world’s ever-growing population [1][2], which currently stands at over 7.7 billion people, and is estimated to rise above 9.6 billion by 2050, and reach nearly 11 billion around 2100 [3]. The application of a wide range of pesticides is considered a regular and required practice in agriculture, as almost 45% of annual food production is lost due to pest infestation or the competition between crops and weeds for soil nutrients [4]. In fact, 3.5 million tons of pesticides are being used, of which 47.5% are herbicides, 29.5% are insecticides, 17.5% are fungicides, and 5.5% are other pesticides [5]. The global pesticide market recorded a value of nearly USD 84.5 billion in 2019, increasing at an annual growth rate of 4.2% since 2015, and it is likely to reach 11.5% with a value of nearly USD 130.7 billion by 2023 [6]. The ten countries consuming the most pesticide in the world are China, USA, Argentina, Thailand, Brazil, Italy, France, Canada, Japan, and India [7].
However, this extensive use of pesticides over recent decades is now of considerable environmental concern because of the release of mobile and/or persistent pollutants into the environment, and the potential accumulation of these toxic substances in soils and/or waters [8][9][10]. The fate of pesticides and their degradation products determines the contamination of the soil, water and air ecosystems over time. Moreover, if agrochemicals remain in the crops, they could finally enter the food chain, posing a threat to human, animal, and plant welfare [11][12][13][14].
The contamination of agricultural soils with pesticides could lead to changes in their chemical and biological properties, affecting their quality and causing a negative impact on crop yields [15]. They may impair soil microbial biodiversity and enzymatic activity (a vital indicator of soil tolerance to pollutants), and the associated degradation of soil organic matter (OM) [16][17]. Many reports are available on these negative effects on soil microbial communities [17][18], and on the processes associated with microbial activities [19].
A recent study involving 317 agricultural topsoil samples from the European Union and 76 pesticide residues as target compounds has revealed that 83% of the soils have been contaminated by one or more residues [9]. The contamination of surface and ground waters by pesticides has also been detected in recent years, probably due to deficient pesticide management, and increased by precipitation and/or irrigation that give rise to the runoff or leaching process of these compounds through the soil [20][21][22][23]. In fact, the contamination of water by pesticides is increasing in agricultural areas across different countries, and a broad range of pesticide concentrations has been found, in some cases exceeding the limit established for drinking water by European Union (EU) legislation (0.1 µg·L
These environmental contamination data highlight the need to roll out strategies to optimize agricultural sustainability by maximizing crop productivity and reducing or preventing soil and water contamination by pesticides. This has been widely addressed in recent years due to the requirement to meet European Community regulations [27]. One of these strategies is based on the in-situ application of organic residues as organic amendments [28]. This method is a common agricultural practice which allows increasing soil OM content, and it can be used to control soil and water contamination by pesticides: (i) promoting the immobilization of pesticides in soil OM, enhancing their subsequent biodegradation, and preventing or reducing their potential mobility into water resources [28][29][30], and (ii) delivering nutrients to the soil by increasing OM content to promote soil fertility and plant growth and stimulate ecological restoration with concomitant benefits for the health of the soil ecosystem [11]. In addition, organic materials require minimal pre-treatment before their application to the soil because of their biological origin [31].
Large amounts of organic residues are generated from livestock, urban, agricultural and industrial activities, and their management is a topic of environmental and social interest in many countries today due to the problems surrounding their disposal [32][33]. In general, these wastes have a high OM content, and they could be used as organic amendments in agriculture, with this being one of the most sustainable options and with greater environmental advantages. Moreover, numerous organic residues could perform as possible sorbents for pesticides [34][35][36]. These studies have assessed the effects that organic carbon (OC) from exogenous sources have on the behavior and environmental fate of pesticides in soils due to the affinity of pesticides, which are generally hydrophobic substances, by these organic materials. The OC of the amendments, depending on their nature, composition and content, can modify the main physicochemical processes of pesticides (adsorption–desorption, dissipation and leaching) in soils. These processes determine their efficiency as well as the dissipation or persistence of these compounds in the soil and their effects as potential environmental contaminants of the soil and surface or ground waters [37].
2. Organic Residues as Soil Amendments
2.1. Origin, Characteristics and Impact on Soil Properties
2.2. European Legislation on the Use of Organic Residues as Soil Amendments
2.1. Origin, Characteristics and Impact on Soil Properties
2.2. European Legislation on the Use of Organic Residues as Soil Amendments

3. Effect of Organic Residues on the Fate of Pesticides in Soil
3.1. Effect of Organic Residues on the Adsorption-Desorption of Pesticides
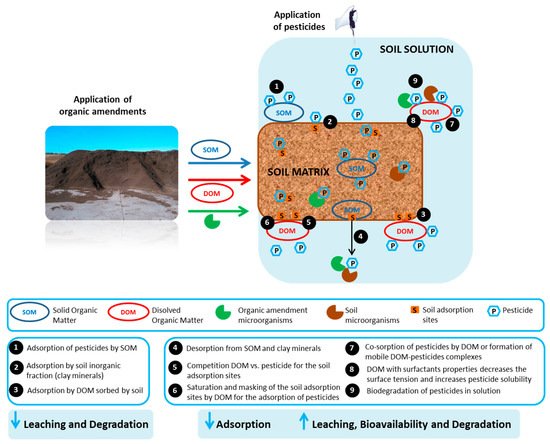
The combined application of pesticides and organic residues in soils modifies the former’s physicochemical behavior, mainly through their adsorption-desorption by the amended soils [78][79]. Organic amendments increase soil OC content, and this parameter is the most relevant factor influencing the adsorption process and the affinity of hydrophobic pesticides by soils [80][81]. Adsorption-desorption determines the environmental fate of any organic pollutants in the soil-water environment [82]; it directly or indirectly controls the availability of pesticides to be transported to surface waters by runoff or to groundwaters by leaching, to the air by volatilization, to be degraded/transformed by microbial attack, or be taken up by plants [83]. Thus, the weak adsorption and/or strong desorption of pesticides promotes leaching, run-off, volatilization, biodegradation and even ecotoxicological impacts on non-target organisms, including human beings, while strong adsorption prevents losses of pesticides by such processes [84]. Accordingly, the addition of organic amendments to soil could lead to a greater or lesser degree of pesticide immobilization in the amended soil. This effect has consequences for pesticide degradation, persistence or mobility, enhancing a pesticide’s subsequent chemical, physical, and biological transformation or degradation, decreasing its transport through the soil profile, and consequently reducing groundwater pollution in some cases [28][78]. However, it could also affect the final concentration bioavailable for absorption by the targeted weeds [37]. Therefore, adsorption and desorption processes help to understand how to predict the mobility and availability of pesticides in unamended and amended soils. Numerous references report the ability that organic amendments have to adsorb pesticides [29][85][86][87]. Adsorption is a physicochemical process in which pesticide molecules are retained on a solid surface (especially by the soil colloidal fraction) within a solution through hydrophobic interactions, van der Waals forces, π–π interaction, and covalent, ionic or hydrogen bonds [88][89]. Soil OM and its more active components, humic acids (HAs) and fulvic acid (FAs), are the principal adsorbents for pesticides, followed by clay colloids and oxyhydroxides of iron and manganese, which interact with pollutants when they reach the soil [90][91]. HA is the OM fraction with the highest reactivity (determined by the number and type of functional groups) and largest surface area. OM’s highly variable composition means it can interact with neutral or ionizable molecules [75]. The pesticide adsorption capacity varies, in general, according to the physicochemical characteristics of the adsorbent and pesticide properties, mainly its water solubility and its hydrophobic, polar, or ionic character [37][92]. The nature and composition of the amendment’s OM vary, with the consequent difficulty in predicting its efficiency for adsorbing pesticides [34][76]. The addition of organic amendments to soil introduces not just SOM but also DOM (Figure 1). The influence of the SOM and DOM content of organic residues on the adsorption of pesticides with different characteristics by amended soils has been frequently studied [34][76][77]. The DOM content in unamended soil is usually very low, but it could become relevant if the organic amendment has a high content in this fraction [93]. DOM is a diverse mixture of complex compounds with different chemical structures and molecular weights that might enhance the formation of multiple interactions with organic pesticides, controlling their distribution in the soil [94]. This is why DOM may modify the movement of pesticides, generally decreasing their adsorption by SOM and increasing their leaching, leading to groundwater contamination [78][95][96], although other authors have indicated that DOM could also be adsorbed by the soil, increasing the adsorption of pesticides and decreasing their leaching [97]. Different processes have been proposed to explain the decreased adsorption of pesticides in the presence of DOM [94][98][99][100] (Figure 1). These include the competition between DOM and pesticide molecules for the adsorption sites in soil, the saturation of soil adsorption sites by DOM, masking these sites for the adsorption of pesticides, the co-sorption of pesticides by DOM, and the formation of mobile DOM-pesticides complexes. Some authors have also indicated that DOM has characteristics similar to surfactants with the capacity to decrease surface tension and increase the solubility of pesticides, reducing their adsorption [101]. The extent and nature of DOM-pesticide interactions depend on factors such as pesticide molecular weight and polarity [102]. These relative effects of DOM will be greater for more hydrophobic chemicals, and will be influenced by the concentration, source, size, polarity, and molecular configuration of the organic colloids [95]. Table 1 includes a summary of the main results obtained from the recent literature on the adsorption and/or desorption of pesticides by some organic residues and by soils amended with different organic residues used as amendments. An organic material widely studied for these purposes is biochar (BC). It is an efficient adsorbent and a potential material for soil amendment [30]. It is a carbonaceous and porous product generated from the partial combustion of biomass, and its effects as soil amendment in the adsorption–desorption of pesticides has been assessed from different points of view. The effects of different types of BC, treatments or aging periods, and their different doses or application forms as organic amendments in soils have been reported for pesticides with different characteristics. Parlavecchiaetal. [103] have investigated the effect of two types of BC from grape vine pruning residues (BC-G) and spruce wood (BC-S) and two vermicomposts (VC) involving digestates from a mixture of manure and olive mill wastewater (VC-M) and buffalo manure (VC-B) in the sorption-desorption capacity of the fungicide metalaxyl-M. Both types of amendments (BC and VC) have a significant capacity to adsorb the high-water-soluble fungicide. However, BC has recorded a much higher sorption efficiency than VC and lower desorption, which is explained by the composition and structural differences in OM between the two (VC has less aromatic carbon and a higher content of hydrophilic functional groups interacting with polar compounds and solvents than BC). Metalaxyl-M is adsorbed to a similar extent on the two VCs, while a different sorption behavior is observed in BC-G and BC-S due to their different porous structures. Likewise, Wu et al. [89] have assessed the effects of different types of BC from peanuts (BCP), chestnuts (BCC), bamboo (BCB), maize straw (BCM), and rice husk (BCR), and the effects of BCR aging on the sorption, degradation and bioavailability of the herbicide oxyfluorfen in various amended soils. The sorption capacity of the five BC differs significantly due to their physicochemical properties. The sorption capacity of BC for oxyfluorfen is significantly correlated with the specific surface area and elemental composition, but it decreases with longer aging time. BC reduces the bioavailability of oxyfluorfen in amended soils, but a higher bioavailability is recorded with an increase in the aging period of BC. Nevertheless, the sorption capacity of amended soil for oxyfluorfen after six months is still better than the unamended soil, highlighting that BCR is an effective way of reducing the risk of contaminating soil with oxyfluorfen, although it could also diminish the herbicide’s bioavailability and efficacy. Deng et al. [104] have studies the effect that BC obtained from cassava residues at 750 °C (MS750) applied at different rates between 0% and 5% has on the sorption-desorption and mobility of atrazine. The MS750 application significantly enhances the sorption capacity and decreases the sorption reversibility of atrazine in the amended soil compared to the unamended soil, due to the larger surface area and greater aromaticity of MS750 (with favorable sorption domains for organic compounds). Moreover, sorption affinity increases with higher BC application rates, although it is also influenced by solution pH, ambient temperature, and contact time between soil and BC (equilibrium time). The entrapment of atrazine in micropore or pore deformation could lead to desorption hysteresis in BC-amended soils.3.1. Effect of Organic Residues on the Adsorption-Desorption of Pesticides
| Pesticide | Soil Characteristics | Organic Amendment/Dose | Experimental Design | Results | Reference | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Metalaxyl-M | Silt loam soil (pH 6.70, OC 2.90%) |
Biochar from grape vine pruning residues (BC–G) (pH 9.9, OC 75.1%) and spruce wood (BC–S) (pH 9.1, OC 83.8%). Vermicomposts (VC) from manure and olive mill wastewater (VC–M) (pH 7.9, OC 31.6%) and buffalo manure (VC–B) (pH 7.8, OC 36.6%) Biochar/soil: 2% (w w | –1 | ) | Sorbent/Solution: 25 mg biochar/5 mL or 3 g soil/8 mL water solution Herbicide concentration: 1–20 mg L | −1 | Shaken: 24 h, T: 20 °C Analytical determination: HPLC |
Metalaxyl sorption order: non–amended soil < soil–VC–M ≤ soil–VC–B < soil–BC–S < soil–BC–G Much higher sorption efficiency by BC than by VC and a lower extent of metalaxyl desorption due to composition and structural differences of the organic matter of BC. |
Parlavecchia et al. [103] | |||||
| Oxyfluorfen | Loamy clay soil (pH 4.85, OC 0.84%) Sandy loam soil (pH 7.55, OC 0.98%) Clay loam soil (pH 6.59, OC 2.23%) |
Biochar from peanut (BCP) (pH, 7.05, C 49.17%), chestnut (BCC) (pH 6.08, C 58.07%), bamboo (BCB) (pH 7.45, C 63.25%), maize straw (BCM) (pH 6.83, C 43.36%), rice hull (BCR) (pH 6.96, C 33.60%) BCR/soil: 0.5%, 1%, or 2% (w w | –1 | ) | Sorbent/Solution: 0.1 g biochar/40 mL or 2 g soil/200 mL 0.01 M CaCl2 Herbicide concentration: 0.05–10 mg L−1 Shaken: 6 days, T: 25 °C Aging time of BCR-soil: 1, 3, 6 months Analytical determination: GC/MS |
BC sorption capacities followed the order: BCR > BCB > BCM > BCC > BCP owing to differences in physicochemical properties. BCR sorption capacity decreased with aging time. |
Wu et al. [89] | |||||||
| Atrazine | Krasnozem soil (pH 7.05, OC 0.89%, clay 28.2%, silt 37.8%) |
Biochar from cassava wastes (pH 9.55, C 62.38%) obtained at 750 °C (MS750). SSA: 430.4 m2/g, MP: 0.144 m3/g Biochar/soil: 0%, 0.1%, 0.5%, 1%, 3% and 5% (w w–1) |
Sorbent/Solution: 0.2–2 g/10 mL 0.01 M CaCl2 Herbicide concentration: 0.5–20 mg L−1 Shaken: 24 h T: 15, 25, 35 °C, pH: 3,5, 7, 9 Analytical determination: HPLC |
Great sorption capacity for atrazine of MS750 in soil due to high surface area and micropore volume. High degrees of aromaticity and hydrophobicity (H/C: 0.02, N + O/C: 0.09) of MS750 supplied numerous sorption sites. | Deng et al. [104] | |||||||||
| Hexazinone Metribuzin Quinclorac |
Sandy loam soil (pH 6.9, OC 0.52%, clay 15.1%, silt 3.3%) |
Bone char (BC) (pH 9.72, C 11%) BC/soil: 5% (w w | –1 | ) or 60 t ha | −1 | Sorbent/Solution: 10 g/10 mL 0.01 M CaCl2 Herbicide concentration: 0.63–3.13 mgL−1, 1.60–8 mgL | Sorbent/Solution: 10 g/10 mL 0.01 M CaCl2 Herbicide concentration: 0.63–3.13 mgL | −1, 0.31–1.56 mgL | , 1.60–8 mgL | −1 Shaken: 24 h, T: 20 °C Analytical determination: Liquid scintillation | , 0.31–1.56 mgL−1 Shaken: 24 h, T: 20 °C Analytical determination: Liquid scintillation |
High sorption of herbicides by BC, regardless of the application form of the material (topsoil or incorporated in the surface layer in leaching columns). | Mendes et al. [105] | |
| Aminocyclopyrachlor Mesotrione |
Clay soil (pH 6.44, OC 2.73%), clay 50.9%, silt 19.6%) |
Bone Char (BC) (pH 9.72, C 11%) BC/soil: 0%, 1%, 5%, 10%, and 100% (w w | −1 | ) or 0, 12, 60, 120, and 1200 t ha | −1 | BC particle size groups: 0.3–0.6 and 0.15–0.3 mm |
Sorbent/Solution: 10 g/10 mL 0.01 M CaCl2 Herbicide concentration: 0.051 mg L−1 (0.32 Bq L | Sorbent/Solution: 10 g/10 mL 0.01 M CaCl2 Herbicide concentration: 0.051 mg L | −1) aminocyclopyrachlor 5.0 mg L−1 (1.13 Bq L | (0.32 Bq L−1) aminocyclopyrachlor 5.0 mg L | −1) mesotrione Shaken: 24 h, T: 20 °C Analytical determination: Liquid scintillation | (1.13 Bq L−1) mesotrione Shaken: 24 h, T: 20 °C Analytical determination: Liquid scintillation |
Higher BC rates (regardless of the particle size) increased both herbicides adsorption and decreased their desorption. | Mendes et al. [106] |
| Linuron Alachlor Metalaxyl |
Sandy loam soil (pH 6.3, OC 0.51%, clay 11.8, silt 13.6%), Sandy clay soil (pH 6.9, OC 1.04%, clay 38.1%, silt 5.8%) |
Pine Wood (OC 41.6%, DOM 1.62%, lignin 24.4%), oak wood (OC 38.5%, DOM 6.86%, lignin 18.2%) Wood/soil: 5% and 50% (w w | –1 | ) (40 and 400 t C ha | –1 | ) | Sorbent/Solution: 5 g/10 mL water solution Herbicide concentration: 1–25 mg L | −1 | (100 kBq L | −1 | ) Shaken: 24 h, T: 20 °C Incubation times: 0, 5 and 12 months Analytical determination: Liquid scintillation |
Pesticide adsorption increased with high wood dose but OC nature was not relevant. Adsorption did not change after incubation times. The adsorption irreversibility decreased in presence of wood for alachlor and increased that of linuron and metalaxyl. | Marín–Benito et al. [107] | |
| Aminocyclopyrachlor Mesotrione |
Clay soil (pH 6.0, OC 2.21%, clay 60.5%, silt 11.3%) |
Sewage sludge (SS) (pH 6.8, OC 16.64%) SS/soil: 0.1%, 1%, and 10% (w∙w | –1 | ) or 1.2, 12, and 120 t∙ha | –1 | Sorbent/Solution: 10 g/10 mL 0.01 M CaCl2 Herbicide concentration: 0.08–0.64 Bq·L−1 (aminocyclopyrachlor) 0.28–2.27 Bq·L−1 (mesotrione) Shaken: 24 h, T: 20 °C Analytical determination: Liquid scintillation |
SS slightly affected sorption–desorption of both herbicides (lowest Kd at soil-SS1%). Kd for mesotrione was ~3.5–fold higher than for aminocyclopyrachlor (higher water solubility). | Mendes et al. [108] | ||||||
| Imazapic Atrazine Hexazinone Diuron Metribuzin |
Red Ferrusol (pH 7.1, OC 2.1%, clay 41%, silt 23%), Grey Dermosol (pH 5.7, OC 0.9%, clay 30%, silt 22%), Red Kandosol (pH 6.5, OC 3.5%, clay 22%, silt 8%) |
Eleven mill muds/ash from different sugar mills (pH 6.04–7.26, OC 27.7–37.8%) Mill muds/soil: 5–25% (w w | –1 | ) | Sorbent/Solution: 1 g/5 mL 0.01 M CaCl2 Herbicide concentration: 0.5 mg L−1 Shaken: 24 h, T: 25 °C Analytical determination: Q-TOF |
Sorption order: diuron > atrazine = metribuzin > hexazinone = imazapic (consistent with herbicide properties). Mill muds at 5% dose increased herbicide retention up to tenfold. Amendments reduced desorption of mobile herbicides in low OC soils. | Duhan et al. [109] | |||||||
| MCPA Diuron Clomazone Terbuthylazine |
Sandy loam soil (pH 7.93, OC 0. 54%, clay 6.7%, silt 16.8%) Loam soil (pH 6.77, OC 1.77%, clay 22.1%, silt 34.2%) Clay loam soil (pH 8.14, OC 1.38%, clay 31.1%, silt 26.8%) |
Mucilage extracted from chia seeds (Salvia hispanica L.) Organic residue/soil: 10% (w w–1) |
Sorbent/Solution: 0.5 g unamended or amended soil/8 mL water solution Herbicide concentration: 1 mg L | −1 | Shaken: 24 h, T: 20 °C Analytical determination: HPLC |
Soil porosity decreased by mucilage amendment. Sorption of herbicides increased in amended soils (sandy–loam < loam < clay–loam). Diuron recorded the highest Kd value and desorption was observed only for terbuthylazine. | Marsico et al. [110] | |||||||
| Dichlorvos Chlorpyrifos |
Sandy soil (pH 8.52, OC 0.7%, clay + silt 9.3%) | Compost (C) from mixed wastes (pH 6.61, OC 29.5%, DOM 354 mg L | −1 | ), and dried goat organic manure (OM) (pH 8.67, OC 14.4%, DOM 620 mg L | −1 | ) Organic residues/soil: 2.5 and 5% (w w | –1 | ) | Sorbent/Solution: 5 g soil/100 mL in C-DOM or 0.01 M CaCl2 Herbicide concentration: 0.1–10 mg L−1 (chlorpyrifos) 0.25–100 mg L−1 (dichlorvos) Shaken: 24 h, T: 25 °C Analytical determination: GC |
C–and OM–DOM increased dichlorvos sorption (S < S–OM–DOM< S–C–DOM) and decreased chlorpyrifos sorption (S > S–C–DOM> S–OM–DOM). Humified and aromatic nature of DOM determines the interactions with pesticides with different hydrophobic character. | Gaonkar et al. [111] | Gaonkar et al. [112] | ||
| Triasulfuron Prosulfocarb Chlorotoluron Flufenacet |
Sandy loam soil (pH 7.36, OC 1.20%, clay 17%, silt 25%) Loamy sand soil (pH 7.61, OC 0.9%, clay 13%, silt 6%) |
Spent mushroom substrate (pH 7.9, C 26.4%, DOM 1.29%), green compost (pH 7.2, C 23.6%, DOM 0.69%), manure (C 18.5%, DOM 1.32%), sewage sludge (pH 7.6, C 28.9%, DOM 1.18%) Organic residues/soils: 10% (w w | –1 | ) | Sorbent/Solution: 5 g soil or 0.1 g organic residues/10 mL 0.01 M CaCl2 Herbicide concentration: 1–25 mg L−1 (TSF, CTL, FNC) 0.25–10 mg L−1 (100 Bq mL | Sorbent/Solution: 5 g soil or 0.1 g organic residues/10 mL 0.01 M CaCl2 Herbicide concentration: 1–25 mg L−1 (TSF, CTL, FNC) 0.25–10 mg L | −1) (PSC) Shaken: 24 h, T: 20 °C Analytical determination: HPLC/MS and Liquid scintillation | (100 Bq mL−1) (PSC) Shaken: 24 h, T: 20 °C Analytical determination: HPLC/MS and Liquid scintillation |
Highest adsorption for prosulfocarb (lowest water solubility and highest Kow) in all materials. Aliphatic and aromatic structures optimize adsorption and O-alkyl and N-alkyl groups enhance desorption hysteresis. |
