Light is the primary regulator of various biological processes during the plant life cycle. Although plants utilize photosynthetically active radiation to generate chemical energy, they possess several photoreceptors that perceive light of specific wavelengths and then induce wavelength-specific responses. Light is also one of the key determinants of the initiation of leaf senescence, the last stage of leaf development. As the leaf photosynthetic activity decreases during the senescence phase, chloroplasts generate a variety of light-mediated retrograde signals to alter the expression of nuclear genes. On the other hand, phytochrome B (phyB)-mediated red-light signaling inhibits the initiation of leaf senescence by repressing the phytochrome interacting factor (PIF)-mediated transcriptional regulatory network involved in leaf senescence.
1. Introduction
Note: The following contents are extract from your paper. The entry will be online only after author check and submit it.
1. Introduction
Leaf senescence, the final stage of leaf development, is a highly controlled developmental process accompanied by massive transcriptional and metabolic changes that destabilize intracellular organelles and macromolecules and translocate nutrients into developing tissues and storage organs. Numerous studies conducted in the past two decades have greatly expanded our knowledge of the molecular mechanisms underlying the regulation of leaf senescence (reviewed in [1][2][3][4][5]). The initiation of leaf senescence is tightly controlled by endogenous factors, such as the state of phytohormones and other metabolites [5][6] and external stimuli such as drought, high salinity, high temperature, and pathogens [7][8][9][10]. Furthermore, a large number of transcription factors (TFs) involved in the regulation of leaf senescence has been identified in the model plant Arabidopsis (
Leaf senescence, the final stage of leaf development, is a highly controlled developmental process accompanied by massive transcriptional and metabolic changes that destabilize intracellular organelles and macromolecules and translocate nutrients into developing tissues and storage organs. Numerous studies conducted in the past two decades have greatly expanded our knowledge of the molecular mechanisms underlying the regulation of leaf senescence (reviewed in [1,2,3,4,5]). The initiation of leaf senescence is tightly controlled by endogenous factors, such as the state of phytohormones and other metabolites [5,6] and external stimuli such as drought, high salinity, high temperature, and pathogens [7,8,9,10]. Furthermore, a large number of transcription factors (TFs) involved in the regulation of leaf senescence has been identified in the model plant Arabidopsis ( Arabidopsis thaliana) [5][11] and in other plant species [12], uncovering transcriptional regulatory networks that regulate leaf senescence.
) [5,11] and in other plant species [12], uncovering transcriptional regulatory networks that regulate leaf senescence.
Light is the foremost regulator of various biological processes in the plant life cycle. Light characteristics, such as wavelength, fluence rate, and photoperiod, greatly affect plant traits, including growth habit, floral induction, and plant productivity [13]. Plants transform light energy into chemical energy through the process of photosynthesis and also capture light energy using distinct sets of photoreceptors that perceive specific wavelengths of light through the process of light signaling [14]. Light is also a key determinant of the initiation of leaf senescence. In recent years, molecular mechanisms underlying photosynthesis- and light-signaling-mediated regulation of leaf senescence have been uncovered.
2. Regulation of Leaf Senescence by Light via Photosynthesis
Photosynthesis is the process that transforms light energy into chemical energy and thus forms the basis of all life on Earth, ranging from photosynthetic bacteria to higher plants. In plants, photosynthesis takes place in chloroplasts. Since chloroplasts contain approximately 80% of the total leaf nitrogen (N) [15], translocation of N compounds from chloroplasts in older leaves (source organs) to new developing tissues (sink organs) is one of the most crucial events during the process of leaf senescence. In addition, increased expression of senescence-associated genes (SAGs) is promoted by the decline in photosynthetic activity and photosynthesis-related gene expression [16]. Thus, the expression of SAGs during senescence is probably greatly affected by the status of photosynthesis, and the nucleus may obtain the information about the status of photosynthesis through signals produced in chloroplasts. The transport of such retrograde signals from chloroplasts to the nucleus to increase the expression of SAGs is still largely unknown, but several important clues have been found in recent studies.
2.1. Chloroplast-Produced Reactive Oxygen Species (ROS) Affect Leaf Senescence
Reactive oxygen species (ROS) are highly reactive molecules generated by the reduction of ground-state molecular oxygen (3
O2
) through the acceptance of an electron pair. In plants, the major forms of ROS include superoxide anion (O2−
), hydroxyl radical (·OH), hydrogen peroxide (H2
O2
), and singlet oxygen (1
O2
). Among these, O2−
, ·OH, and H2
O2
are produced in most subcellular compartments, while the highly reactive 1
O2
is uniquely produced in chloroplasts, which are the primary source of ROS under light conditions [17]. ROS production in chloroplasts is tightly associated with light-dependent photosynthetic reactions. During photosynthesis, 1
O2
is produced mainly within photosystem II (PSII) in thylakoid membranes because of the reaction between the triplet state of chlorophyll molecule and 3
O2
[17]. On the other hand, the reduction of 3
O2
by PSI generates O2−
, which is immediately converted into H2
O2
on the stromal side of the thylakoid membranes either spontaneously or through the action of superoxide dismutases (SODs) [18]. The production of ROS is drastically increased under unfavorable environmental conditions, such as excess light, drought, and high temperature [19]. In addition, it is well known that the production of ROS, especially H2
O2
, is significantly increased in leaves during senescence [20], which is accompanied by a significant reduction in the abundance of light harvesting complex II (LHCII) and other photosystem proteins. Consequently, the captured light energy is in excess for the remaining photosynthetic units, leading to the production of a large amount of ROS even under moderate light conditions. In addition, the dismantling of LHCII subunits releases a large number of chlorophyll molecules and their degradation products. It is well known that several chlorophyll biosynthesis intermediates, including protoporphyrin IX and protochlorophyllide, act as photosensitizers that produce high amounts of 1
O2 under light conditions [21][22]. Similarly, several chlorophyll degradation products, such as 7-hydroxymethyl chlorophyll
under light conditions [21,22]. Similarly, several chlorophyll degradation products, such as 7-hydroxymethyl chlorophyll a
, pheophorbide a, and red chlorophyll catabolite, also act as photosensitizers [23][24][25] (
, and red chlorophyll catabolite, also act as photosensitizers [23,24,25] ().
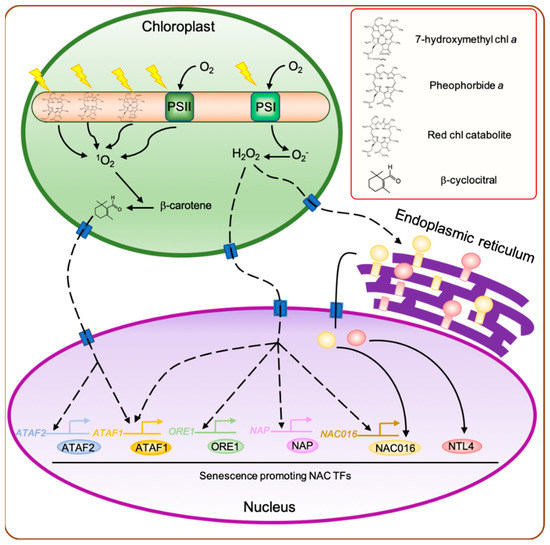
Figure 1.
Regulatory network of leaf senescence induced by chloroplast-derived retrograde signaling. During the senescence phase, the abundance of photosystem proteins decreases significantly. Consequently, the captured light energy exceeds the amount needed by the remaining photosynthetic units, leading to the production of a large amount of O
2−
by photosystem I (PSI) and
1
O
2
by PSII. In addition, the dismantling of light harvesting complex II (LHCII) proteins releases a large number of chlorophyll molecules and their degradation products, such as 7-hydroxymethyl chlorophyll
a
, pheophorbide
a
, and red chlorophyll catabolite, which act as photosensitizers, producing high amounts of
1
O
2
in the presence of light. The
1
O
2
generated in chloroplasts promotes the cleavage of β-carotene to produce β-cyclocitral, which then acts as a retrograde signal between the chloroplast and nucleus to activate the expression of
1
O
2
-responsive genes, including senescence-associated
Arabidopsis thaliana
activating factor (ATAF) subfamily no apical meristem/
ATAF1,2
/cup-shaped cotyledon (NAC) genes,
ATAF1
and
ATAF2
. O
2−
generated by PSI is rapidly dismutated into H
2
O
2
, which acts as a signaling molecule to alter the expression of H
2
O
2
-responsive genes, including
ATAF1
,
ORESARA1
(
ORE1
),
NAC-
like, activated by
AP3/PI
(
NAP
), and
ANAC016
. In addition, H
2
O
2
generated in the chloroplasts also triggers the translocation of membrane-bound NAC transcription factors (TFs), such as ANAC016 and NAC with transmebrane motif 1-LIKE4 (NTL4), from the endoplasmic reticulum to the nucleus. Dashed arrowheads indicate the indirect activation of genes. chl, chlorophyll.
For many years, it has been proposed that ROS generated in chloroplasts are involved in the promotion of leaf senescence (reviewed in [26][27]). Among these ROS, H
For many years, it has been proposed that ROS generated in chloroplasts are involved in the promotion of leaf senescence (reviewed in [26,27]). Among these ROS, H2
O2
plays a crucial role in the regulation of leaf senescence. Since H2
O2
can easily cross cellular membranes [28], it can be transmitted as a signaling molecule from the chloroplast to the nucleus. Transcriptome analyses of Arabidopsis revealed that several genes upregulated during leaf senescence are also upregulated by H2
O2 treatment [29][30]; for example, no apical meristem/ATAF1,2/cup-shaped cotyledon (NAC) TF-encoding genes, such as
treatment [29,30]; for example, no apical meristem/ATAF1,2/cup-shaped cotyledon (NAC) TF-encoding genes, such as Arabidopsis thaliana activating factor1 (ATAF1)/ANAC002
[31], ANAC016
[32], NAC-
like, activated by AP3/PI (NAP)/ANAC029
[33], JUNGBRUNNEN1 (JUB1)/ANAC042
[34], and ORESARA1
(ORE1
)/ANAC092
[35], which have been characterized as key regulators of leaf senescence (). Among these, JUB1
acts as a negative regulator of leaf senescence, while the other four NAC
genes promote leaf senescence.
H2
O2
is also one of the triggers that induce the translocation of membrane-bound NAC TFs from the endoplasmic reticulum (ER) membrane to the nucleus. Several membrane-bound NAC TFs are activated by H2
O2 generated in mitochondria under environmental stresses [36][37]. The induction of nuclear gene expression by treatment with methyl viologen, which accepts electrons from PSI via ferredoxin and leads to the production of a large amount of O
generated in mitochondria under environmental stresses [36,37]. The induction of nuclear gene expression by treatment with methyl viologen, which accepts electrons from PSI via ferredoxin and leads to the production of a large amount of O2−
in chloroplasts, is strongly inhibited in the knockout mutant of ANAC017
, an Arabidopsis membrane-bound NAC TF gene [38], indicating that H2
O2
generated in chloroplasts is also important for the activation of membrane-bound NAC TFs. In Arabidopsis and rice (Oryza sativa
L.), several membrane-bound NAC TFs including ANAC016 [32], NAC WITH TRANSMEMBRANE MOTIF 1-LIKE4 (NTL4)/ANAC053 [39], and ONAC054 [40] have been shown to promote the initiation of leaf senescence. Thus, H2
O2
generated in chloroplasts during the senescence phase may modulate the gene expression partially via the activation of senescence-associated membrane-bound NAC TFs ().
O2−
also modulates the expression of SAGs. In Arabidopsis seedlings treated with methyl viologen, an O2−
-specific propagator, several genes encoding senescence-associated TFs including ATAF1
, WRKY6
[41], and WRKY22
[42] were strongly upregulated [43]. WRKY6 and WRKY22 antagonistically regulate leaf senescence; WRKY6 acts as a negative regulator, while WRKY22 acts as an enhancer of leaf senescence. In contrast with H2
O2
, O2−
is a short-lived ROS and cannot cross the chloroplast membrane [44]. Thus, O2−
probably generated in chloroplasts cannot serve as a signaling molecule.
Compared with H2
O2
and O2−
, 1
O2
is a highly reactive molecule and consequently a more potent oxidizing agent than the other ROS [45]. In leaf tissues, in particular, 1
O2
is required for most of the lipid peroxidation reactions. Lipid peroxidation promotes the generation of free radicals, which accelerate senescence [46]. 1
O2
also gives rise to a signal that affects the expression of nuclear genes, similar to the other two ROS. Op den Camp et al. (2003) investigated the effect of