Under constant barrage from chemical, pathogenic, and mechanical stresses, the intestinal epithelium is homeostatically replenished by a pool of Lgr5⁺ intestinal stem cells (ISCs), residing at the bottom of submucosal invaginations termed crypts. Decorated with the RSPO-receptor LGR5, which potentiates canonical Wnt/β-catenin signalling, these actively cycling cells can both self-renew and give rise to short-lived transit-amplifying cells. In turn, transit-amplifying cells undergo successive rounds of cell division and differentiation to generate the full gamut of terminally differentiated intestinal cell types tasked with performing pleiotropic absorptive, secretory, immune, and barrier functions. The self-renewal capabilities and multipotency of Lgr5⁺ ISCs are tightly controlled by instructive cues emanating from epithelial and stromal components of the ISC niche in the vicinity of the lower crypt.
The intestinal epithelium displays a remarkable ability to regenerate following demise of homeostatic Lgr5⁺ ISCs post injury. Plasticity—the ability of lineage-restricted cells to regain self-renewal capacity and multi-lineage differentiation potential in response to environmental cues—is pervasive among multiple intestinal cell populations. Reserve stem-like cells, lineage-committed progenitors, and/or fully differentiated cell types can all contribute to regeneration and repair through dedifferentiation and reversion to an Lgr5⁺ stem-like state. In line with the pervasive plasticity of the intestinal epithelium, accumulating evidence supports both “bottom-up” and “top-down” histogenesis of colorectal tumours whereby the cells-of-origin comprise either ISCs at the crypt base or differentiated cells at the crypt apex, respectively.
- intestinal stem cells (ISCs)
- plasticity
- LGR5
- intestinal epithelium
- colorectal cancer
- regeneration
- YAP
- cancer stem cells
1. Introduction
The single-layer epithelium lining the intestinal tract is integral to its functions in water and nutrient absorption, waste elimination, and immune surveillance while also forming a barrier against luminal toxins and gut-resident microbiota. To weather the barrage of chemical, pathogenic, and mechanical stresses posed by the digestive process, and counterbalance cell attrition, the intestine must continuously replenish its epithelial lining and regenerate the full gamut of specialized cell types that underpin its diverse functions. The homeostatic renewal of this epithelium is critically dependent on the sustained activity of multipotent intestinal stem cells (ISCs), residing within submucosal invaginations, called crypts. ISCs can self-renew while also giving rise to short-lived transit-amplifying (TA) cells, which in turn undergo successive rounds of cell division to generate multiple mature intestinal cell types (
). Broadly, differentiated intestinal cell lineages are specialized to perform either absorptive or secretory functions [1]. Absorptive enterocytes retrieve nutrients and water from luminal contents, whereas rare microfold (M) cells function in immune surveillance, delivering luminal antigens to underlying lymphoid structures (Peyer’s patches). Secretory lineages include multiple hormone- and neurotransmitter-secreting enteroendocrine cell types that regulate physiological responses to food intake and interface with the enteric nervous system, mucus-secreting goblet cells that fortify the host epithelium against mechanical stresses and luminal microorganisms, and chemosensory tuft cells that orchestrate type-2 immunity responses to helminth parasites and allergens [1].
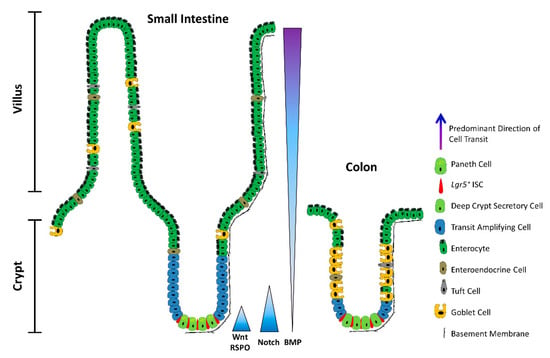
The architecture of the small intestine and the colon. Schematic depicting a longitudinal section of the intestinal mucosa. The mucosa of the small intestine extends finger-like projections (villi) into the gut lumen, which provide an increased surface area for optimal nutrient absorption. The villi are populated by mature, differentiated absorptive and secretory cell types, including absorptive enterocytes, hormone- and neurotransmitter-secreting enteroendocrine cells, mucus-secreting goblet cells, tuft cells, and microfold (M) cells (not shown). The mucosa surrounding the villi forms tubular invaginations into the lamina propria, called crypts, which serve as a protected reservoir of stem and progenitor cell populations. Notably, the epithelium of the colon is devoid of villi, with the crypts opening onto a flat mucosal surface, reflecting its role in waste compaction. To support homeostatic turnover, ISCs self-renew and give rise to short-lived transit-amplifying (TA) cells, which in turn beget lineage-restricted progenitors that differentiate into the mature cell types lining the villi. During their limited lifespan, intestinal epithelial cells migrate from the base of the crypt to the tip of the villus or the colonic surface, from where they are shed into the gut lumen and replaced by neighbouring cells. In contrast, Paneth cells are relatively long-lived, and migrate to the base of the crypt, where they secrete antimicrobial peptides and form a vital component of the ISC niche. Paneth cells are absent from the colon, but deep crypt secretory (DCS) cells may fulfil an equivalent role. Opposing gradients of morphogens specify intestinal-cell fate and differentiation along the vertical crypt axis: Wnt and Notch signalling prevail at the crypt base, whereas BMP transduction is highest near the lumen.
Since aberrant ISC proliferation or, conversely, the failure to mobilize ISCs in response to injury is invariably detrimental, ISC activity is kept in check by the local milieu: the ISC niche. Set within the confines of the crypt base, the ISC niche is comprised of either Paneth cells in the small intestine or deep crypt secretory (DCS) cells in the colon, in addition to pericryptal fibroblasts, immune cells, endothelial cells, enteric neurocytes, extracellular matrix (ECM) components, and soluble cytokines and growth factors. Multiple converging niche-signalling pathways—primarily Wnt, Notch, and BMP—maintain ISCs in a multipotent state and fine-tune the balance between self-renewal and differentiation.
2. ISCs in a Nutshell
Daily homeostatic turnover of the intestinal epithelium is orchestrated by crypt-base columnar cells, nestled between either Paneth or DCS cells at the crypt base in the small intestine and colon, respectively [2][3]. Decorated with the RSPO-receptor LGR5, which potentiates canonical Wnt/β-catenin signalling [4][5][6], these highly proliferative cells (hereafter
ISCs) exhibit the ability to self-renew and differentiate into all intestinal lineages in vitro and in vivo [7][8] and are tasked with the homeostatic renewal of the epithelium in both the small intestine and the colon [7] (
a). Yet, remarkably, the adult intestinal epithelium can fully recover following acute ablation of
-expressing cells [9], and conditional deletion of the
gene does not visibly perturb crypt architecture [4]. Together, these findings bring forth the redundancy of
ISCs for homeostasis and suggest that other cell types can compensate for their deficiency in this setting. Nevertheless, sustained depletion of
ISCs severely compromises the regenerative response to radiation-induced damage [10], suggesting that any compensatory cell types, deployed post injury, must first repopulate the
compartment prior to reconstituting the lost epithelium. Indeed, multiple putative reserve ISC pools have been proposed to reside just above the crypt base—around the so-called +4 position—based on DNA-label retention [11] and lineage tracing with eGFP−IRES−Cre
reporters inserted into the loci encoding BMI1 [12], mTERT [13], HOPX [14], or LRIG1 [15]. These slow-cycling reserve populations can be mobilized post injury to replenish lost or damaged
ISCs [9][12][13][14][15][16][17][18][19]. In addition, multiple lineage-committed progenitors and fully differentiated cell types can dedifferentiate and regain stem-like traits. Notably,
enterocyte precursors [20],
enteroendocrine-lineage cells co-expressing tuft-cell markers [21] and other subsets of enteroendocrine cells [22][23][24], CD69
CD274
goblet cell precursors [25], secretory progenitors expressing
[26] or
(also known as
) [27][28][29][30], differentiated KRT20
surface enterocytes in the colon [31], as well as post-mitotic tuft [32], enterochromaffin [33], and Paneth cells [34][35][36] can contribute to varying degrees to crypt homeostasis and injury-induced regeneration (
b).
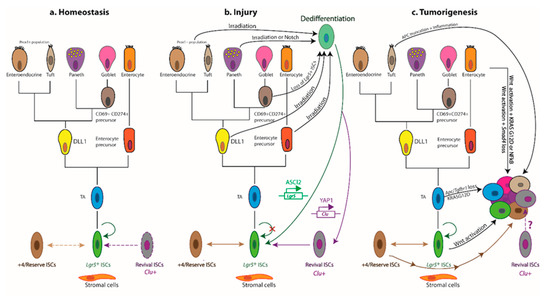
Cellular hierarchies and phenotypic plasticity in the intestinal epithelium during homeostasis, post-injury regeneration, and tumorigenesis. The dendrograms summarize key lineage relationships between
ISCs, transit-amplifying (TA) cells, lineage-committed precursors, terminally differentiated intestinal cell types, +4/reserve ISCs, and revival stem cells in different settings. (
) The homeostatic remodelling of the intestinal epithelium is orchestrated by niche signals that fine-tune the balance between
ISC self-renewal and differentiation. Revival stem cells are rare under homeostatic conditions (dashed boundary), and +4/reserve ISCs contribute only weakly to daily turnover during homeostasis (double-headed dashed arrow). (
) Following ablation of
ISCs post injury, +4/reserve ISCs can mobilize to replenish lost
ISCs and repopulate damaged crypts. Differentiated
progeny upregulate
in a YAP1-dependent manner, transiently serving as revival stem cells that can generate
ISCs de novo. Lineage-committed precursors and/or fully differentiated cells can dedifferentiate, revert to an
state, and regain stem-like traits. The transcription factor ASCL2 is critical for the ability of recent
ISC progeny to undergo dedifferentiation to an
state. Of note,
cells are distinct from the
and
populations. (
) Aberrant activation of Wnt signalling drives unbridled proliferation of
ISCs leading to intestinal hyperplasia. TA and differentiated cells, with hyperactive Wnt signalling and mutant KRAS
, may progress to malignancy in the context of TGFβ-receptor loss or inflammation (i.e., NFκB activation). Multiple +4/reserve ISCs can also initiate tumorigenesis, and tuft cells are readily transformed by inflammation following
loss. Whether revival stem cells can serve as tumour-initiating cells remains unclear. Solid arrows indicate the ability to dedifferentiate and revert to a stem-like state, or the susceptibility to transformation and hyperplastic progression. Reflexive arrows indicate the ability to self-renew. Double-headed solid arrows denote dynamic interconversion between indicated cell types. Note that, to date, goblet cell progenitors have not been lineage-traced.
Classification of ISCs is further confounded by the expression of markers of +4/reserve ISCs (
,
,
,
) in
cells, suggesting considerable overlap between these populations [37][38][39]. Indeed,
and +4/reserve ISCs can dynamically interconvert during both homeostatic and radiation-induced regeneration, although +4/reserve ISCs contribute only weakly to daily turnover under non-pathological conditions [9][14][16][17]. Nevertheless, most +4/reserve ISC populations (including
secretory precursors) differ profoundly from
ISCs as they are relatively refractory to Wnt stimulation, lack expression of canonical Wnt-target genes, and exhibit resistance to high-dose irradiation [14][16][17][40][41].
In addition to
—itself a Wnt-target gene—
ISCs express other Wnt targets, including
,
,
, and
[39][42][43], underscoring the importance of Wnt signalling for safeguarding the stem cell state at the crypt base [43]. ASCL2—a master regulator of the
ISC gene expression program—perpetuates its own expression in a positive feedback loop, controlled by WNT/RSPO levels. Thus, while an “
-on” state imparts stemness, “
-off” cells are destined to differentiate, with the corollary that TA cells can regain stemness upon encountering increased WNT/RSPO levels sufficient to drive
expression [44].
Recent insights have further highlighted heterogeneity within the
compartment itself. Addressing a long-standing controversy in the field, Buczacki and colleagues identified label-retaining cells as
eGFP
secretory precursors of Paneth and enteroendocrine cells that do not contribute to homeostasis [30] and are discrete from the +4/reserve ISCs marked by Cre
knock-in reporters [45]. Two additional slow-cycling
ISC subpopulations, expressing
[46] or
[47], were found to survive genotoxic stress and contribute to radiation-induced regeneration. In this respect, these slow-cycling
ISC subsets exhibit purported traits of +4/reserve ISCs and markedly contrast with the highly proliferative, radio-sensitive
ISC population. Furthermore, a rapidly-cycling, DNA damage-resistant subpopulation of
cells, that expresses little-to-no
and resides at the +4 position, has recently been shown to repopulate the intestinal epithelium post irradiation [48]. Crucially,
cells are mobilized before the reappearance of
cells, challenging the widely held contention that +4/reserve ISCs must regain
expression prior to instigating repair [49]. Although able to repopulate all major intestinal lineages,
cells preferentially differentiate into Paneth cells, suggesting that they may first replenish the ISC niche to help restore
ISC functionality in the newly remodelled crypt [48]. An additional distinct—but transient—population comprises the immediate progeny of
ISCs, expressing modestly reduced levels of ISC-associated transcripts alongside markers of mature secretory cells and enterocytes [50]. An example of multilineage gene priming, this transient bipotential progenitor population is poised to lose
expression entirely as cells move further from the crypt base along their ultimate cell-fate trajectory [50][51]. Collectively, these findings suggest considerable overlap and dynamic interconversions between crypt ISC populations and implicate the local niche as the main “influencer” of stem-like behavioural and phenotypic traits.
Bringing a long-standing debate to an apparent close [52], recent studies have attributed the bulk of intestinal epithelial regeneration to the dedifferentiation of recent progeny of
ISCs. Both absorptive and secretory cell lineages are recruited to replenish the stem-cell pool post injury, with the underlying kinetics precluding the mobilization and expansion of dedicated reserve ISC populations [53]. In fact, the induction of
is critical for the ability of recent
ISC progeny to undergo dedifferentiation to an
state prior to regenerating the injured intestinal epithelium [53]. Mechanistically, such pervasive dedifferentiation is underpinned by a permissive open chromatin configuration in progenitor cells undergoing differentiation [54], with only incremental chromatin remodelling of lineage-restricted genes required to interconvert between homeostatic
ISCs and their secretory and absorptive eventual progeny during differentiation, and vice versa during crypt regeneration [25][54].
While at times confounding, these studies collectively converge on the fact that most, if not all, crypt-resident cell types display phenomenal plasticity and retain (dormant) stemness potential, calling into question the existence of a “dedicated” reserve ISC pool. Importantly, they underscore the notion that stemness is not a cell-intrinsic trait and refocus attention on the role of the niche in governing ISC function during homeostasis and the de novo acquisition of stemness in times of stress.
3. The YAP-Driven Foetal-Like Stem Cell State
Additional studies have interrogated the molecular mechanisms underlying the response of the mouse intestinal epithelium to helminth infection [55] and treatment with dextran sodium sulphate (DSS) [56], both of which breach the mucosal barrier. An emergent theme is that the regenerating intestinal epithelium is transiently reprogrammed into a highly plastic foetal-like state, orchestrated by changes in the inflammatory milieu [55] and ECM [56], respectively. The extensive tissue remodelling that ensues entails the deployment of highly proliferative SCA1
progenitors [55][56], lacking markers of secretory lineages as well as of adult ISCs—most notably,
and LRIG1 [39][56]. Instead, these regenerating cells express foetal epithelial markers, such as
and
/
, alongside the multipotent progenitor marker SCA1 (also known as LY6A) [56][57]. Notably, although
is absent from the human genome,
is highly expressed in the regenerating epithelium of inflamed ulcerative colitis, compared with non-inflamed regions in matched patient specimens [56]. Moreover, the transcriptional signatures of the mouse repairing epithelium and the foetal-like state are enriched in patients with active inflammation, compared with healthy counterparts, lending relevance of this foetal-like program to human disease [56]. Similarly, crypts overlying helminth larvae-associated granulomas become devoid of
expression, with a discrete subset of SCA1
crypt cells activating an IFNγ-dependent foetal-like transcriptional program [55]. Indeed, similar injury-response programs are deployed following irradiation and
ISC ablation, suggesting that the transient “revival” of latent foetal-like traits is likely a common denominator of the intestinal epithelial response regardless of the mode of injury [55].
Following DSS treatment, the regenerating mouse intestinal epithelium is also characterized by upregulation of several ECM components and the accumulation of collagen type I fibres around newly formed crypts. These dynamic changes in the ECM composition of the niche are propagated via FAK/SRC-mediated mechanotransduction, culminating in the activation and nuclear translocation of YAP and TAZ [56], two paralogous transcriptional coactivators inhibited by the Hippo tumour-suppressor pathway [58]. YAP has similarly been shown to transiently reprogram
ISCs into a regenerative state post irradiation. Here, YAP suppresses homeostatic Wnt signalling and Paneth cell differentiation while concomitantly activating expression of the EGF-family member EREG to drive proliferation and promote cell survival [59]. Indeed, several studies concur that YAP/TAZ can inhibit Wnt signalling during intestinal regeneration and tumorigenesis [59][60][61][62], consistent with the suppression of
and the ISC signature during the foetal-like regenerative response [56].
A critical role for YAP has also been ascribed in the damage-induced mobilization of “revival stem cells”, recently identified in the regenerating intestinal epithelium using a single-cell transcriptomics approach [63]. The revival stem cell pool is a rare, quiescent population in homeostasis, characterized by elevated expression of clusterin (
),
,
, and
. While these
revival stem cells do not contribute to daily homeostatic renewal, they are mobilized and expanded following ablation of
ISCs, irradiation, or DSS-induced inflammation and colitis. Their transient expansion post damage regenerates the full gamut of intestinal cell types, including
ISCs, in a YAP1-dependent manner [63]. Interestingly,
revival stem cells express elevated levels of
post irradiation [63], raising the possibility that this damage-induced, expanded revival stem cell population overlaps with
foetal-like crypt cells, which also rely on YAP for their regenerative potential [56].
Whereas the YAP-driven regenerative response is a transient, reversible process [56][63], persistent tissue injury and repair set up a vicious cycle of chronic inflammation—a known risk factor for colorectal cancer (CRC) [64]. Indeed, the YAP-mediated regenerative response can be hijacked to facilitate the progression of APC-deficient foci to adenomas to the extent that
Yapdeletion abrogates adenoma formation in
ApcMin/+ mice [152,158]. Moreover, the YAP transcriptional program correlates with the gene expression signatures of earlymice [59][65]. Moreover, the YAP transcriptional program correlates with the gene expression signatures of early
ApcMin/+ tumours as well as of revival stem cells [156,159]. Accordingly, YAP decorates the nuclei of tubular adenomas from patients afflicted with familial adenomatous polyposis [158]. Yet, the role of YAP in intestinal tumorigenesis remains controversial as both tumour-suppressive [153,155] and oncogenic functions [152,158,160,161] have been ascribed in different contexts. It further remains to be seen whether the foetal-like, YAP/TAZ-dependent,tumours as well as of revival stem cells [63][66]. Accordingly, YAP decorates the nuclei of tubular adenomas from patients afflicted with familial adenomatous polyposis [65]. Yet, the role of YAP in intestinal tumorigenesis remains controversial as both tumour-suppressive [60][62] and oncogenic functions [59][65][67][68] have been ascribed in different contexts. It further remains to be seen whether the foetal-like, YAP/TAZ-dependent,
Lgr5−regenerative state plays a role in the development of colonic tumours lacking overt Wnt-pathway mutations. In support of this notion, BRAF
V600E-driven colonic organoids exhibit a foetal-like dedifferentiation program enriched for Hippo-pathway targets, which recapitulates the transcriptional profiles of human BRAF
V600E-driven CRCs [162]. In addition, the acquisition of a YAP/TAZ-dependent foetal-like signature is associated with resistance to Wnt-targeted therapy [163], and-driven CRCs [69]. In addition, the acquisition of a YAP/TAZ-dependent foetal-like signature is associated with resistance to Wnt-targeted therapy [70], and
Sca1+reserve-like stem cells, with a regenerative/tumorigenic YAP transcriptional signature and concomitant suppression of β-catenin signalling, fuel adenoma initiation in
ApcMin/+mice as well as an azoxymethane-induced tumour model. Crucially, the tumorigenic capacity of these
Sca1+ reserve-like stem cells depends on the druggable PGE2–PTGER4 axis, which in turn controls the nuclear localization/activity of YAP [159]. Indeed, PGE2-induced YAP signalling is also implicated in colitis-associated regeneration and spontaneous tumorigenesis [164], suggesting that the pro-oncogenic, YAP-dependent, foetal-like regenerative program may serve as a therapeutically actionable target.reserve-like stem cells depends on the druggable PGE2–PTGER4 axis, which in turn controls the nuclear localization/activity of YAP [66]. Indeed, PGE2-induced YAP signalling is also implicated in colitis-associated regeneration and spontaneous tumorigenesis [71], suggesting that the pro-oncogenic, YAP-dependent, foetal-like regenerative program may serve as a therapeutically actionable target.
Overall, multiple cell types can mobilize to regenerate the injured intestinal epithelium by adopting a highly plastic foetal-like state, although the degree to which each population contributes to the repair warrants further study. It also remains unclear whether regenerative cues can mobilize different crypt progenitors and/or mature cell types to dedifferentiate into a foetal-like state, and/or whether pre-existing homeostatic crypt cell types, such as the
Lgr5−Clu+Sca1+ revival stem cells [156], expand in an attempt to restore the epithelium independently of Wnt signalling. Furthermore, whether revival stem cells can serve as tumour-initiating cells remains untested at present. For example, it is conceivable that PGE2-dependentrevival stem cells [63], expand in an attempt to restore the epithelium independently of Wnt signalling. Furthermore, whether revival stem cells can serve as tumour-initiating cells remains untested at present. For example, it is conceivable that PGE2-dependent
Sca1+ reserve-like tumour-initiating cells [159] derive from transformation ofreserve-like tumour-initiating cells [66] derive from transformation of
Lgr5−Clu+Sca1+ revival stem cells [156]. Consistent with this notion, the revival stem cell signature correlates with resistance to 5-fluorouracil chemotherapy in patient-derived CRC organoids, and elevatedrevival stem cells [63]. Consistent with this notion, the revival stem cell signature correlates with resistance to 5-fluorouracil chemotherapy in patient-derived CRC organoids, and elevated
CLU expression is associated with poor patient survival and disease recurrence [165]. In addition, the revival stem cell signature is reportedly enriched in L1CAM-positive metastasis-initiating CRC cells [166].expression is associated with poor patient survival and disease recurrence [72]. In addition, the revival stem cell signature is reportedly enriched in L1CAM-positive metastasis-initiating CRC cells [73].
4. Cells-of-Origin and Plasticity in Colorectal Cancer
Analogous to normal crypts, current dogma posits that only a subset of intestinal tumour cells—termed cancer stem cells (CSCs)—are endowed with tumour-initiating potential, i.e., the capacity to self-renew and generate the differentiated non-CSCs that constitute the tumour bulk. CSCs are also thought to underpin metastatic competence, drug resistance, disease recurrence and, ultimately, poor therapeutic outcome. In line with the pervasive plasticity of the intestinal epithelium, accumulating evidence supports both “bottom-up” [74] and “top-down” [75] histogenesis of colorectal tumours whereby the cells-of-origin comprise either ISCs at the crypt base or differentiated cells at the crypt apex, respectively (
c).
ISCs have been amply demonstrated to serve as tumour-initiating cells [76][77]. Indeed, targeted deletion of
in
ISCs drives aberrant Wnt signalling and hyperproliferation, leading to rapid adenoma formation in mice [76]. Moreover, overexpression of
in
cells drives hyperplastic bottom-up lesions, containing mislocalized Paneth cells and expanded
and
populations, in keeping with the fact that RSPO3 is a secreted protein that nurtures both
ISCs and their supportive epithelial niche. Besides
cells, however, lineage tracing implicates
populations as putative cells-of-origin of the resulting hyperplastic adenomas and adenocarcinomas in this model [77]. Similarly, constitutive activation of Wnt signalling in cells expressing
[12],
[78], or
[15][79] drives bottom-up intestinal neoplasia in mice. Collectively, these findings suggest the existence of multiple possible cells-of-origin within the crypt.
Other crypt cell types can also assume the mantle of tumour-initiating cell, contributing to bottom-up tumorigenesis. Hence, loss of
in
cells—a heterogeneous population, encompassing
and
cells, spanning the crypt base as well as the TA zone—leads to adenomas that occasionally progress to invasive adenocarcinomas [47]. Such lesions are not typically observed upon sole deletion of
in other putative tumour-initiating cell populations, including
ISCs [15][18][76][79]. It remains to be determined whether the coexistence of adenomas and adenocarcinomas—frequently a feature of human polyposis syndromes—reflects tumour initiation from distinct differentially localized subsets of
cells [47]. Remarkably, while the majority of
subsets are exquisitely sensitive to DNA damage,
cells are radioresistant and may thus survive to spawn tumours post injury.
Conversely, top-down lesions likely derive from cells located in the TA zone or the villus, induced to undergo dedifferentiation. Notably, sole deletion of
in TA cells yields only microscopic lesions, which rarely progress to adenoma [76]. Additional TGFβ dysfunction is not sufficient to drive dedifferentiation in this compartment or the formation of top-down lesions [80]. However, following exposure to inflammation and/or upon accumulating cooperating mutations, differentiated villus cells can re-express
and ISC markers, and initiate tumours [81][82]. Thus, constitutive activation of β-catenin and NFκB signalling [81] or dual
/
mutations [82] can drive tumour formation both from crypt ISCs and villus epithelial cells in the small intestine [81] and colon [82], respectively. Deletion of
further augments the dedifferentiation potential of
villus epithelial cells, exacerbating top-down tumorigenesis [80]. This suggests that, during early tumour progression, the elevated stromal-derived TGFβ levels that prevail further up the crypt–villus axis restrain dedifferentiation, whereas cells in lower regions or the crypt base can escape to form tumours. Consequently, mutations enabling differentiated cells to evade TGFβ-mediated tumour suppression will extend the pool of tumour-initiating cells.
Additional examples whereby dedifferentiation bestows tumorigenic potential have been reported. Long-lived differentiated
tuft cells, which remain quiescent following
loss, are readily transformed by inflammation, forming poorly differentiated colonic adenocarcinomas [32]. APC truncation or post-irradiation depletion of
ISCs induces radioresistant
upper-crypt progenitors to dedifferentiate, via an
state, spawning tumours both in the small intestine and colon [18].
secretory cell precursors are another candidate cell-of-origin located just above the ISC zone. In the small intestine, these cells can dedifferentiate to form tumours with serrated features upon sustained activation of Notch signalling, combined with
loss. In the colon, the counterpart
cell population is mobilized upon DSS treatment via the activation of SRC and YAP [83]. Thus,
secretory cell precursors respond differently to tumorigenic insult in distinct niches, although the clinical relevance of these findings remains unclear [83].
Aberrant expression of the BMP inhibitor
in the intestinal epithelium disrupts homeostatic morphogen gradients, prompting the proliferative expansion of
progenitor cells that spur the formation of ectopic crypt foci perpendicular to the villus axis. Cells, within these structures, accumulate multiple somatic mutations, with concomitant suppression of cytostatic and differentiation programs, eventually progressing to polyps that recapitulate features of hereditary mixed polyposis syndrome and traditional serrated adenomas [84]. These findings further confirm that
cells, outwith the ISC niche, can undergo malignant transformation.
Together, the above findings reinforce the links between deregulated niche signalling, prolonged inflammation, and CRC risk/progression [85] (
). Crucially, they underscore that the initiation of tumours from differentiated cells requires cooperating mutations or exacerbating stimuli, such as an inflammatory drive, alongside Wnt deregulation.
5. All Roads Lead through LGR5
Notwithstanding the existence of multiple putative cells-of-origin within the crypt base or more differentiated luminal regions, compelling evidence supports the contention that LGR5 marks a subset of mouse and human intestinal CSCs endowed with tumorigenic potential and multi-lineage differentiation capacity [76][86][87][88][89][90][91][92]. Perhaps unsurprisingly, considering the pervasive plasticity of the intestinal epithelium, diphtheria toxin-mediated ablation of cells engineered to express the diphtheria-toxin-receptor, DTR, under the control of the
promoter (
), failed to achieve regression of non-metastatic
subcutaneous organoid allografts. Instead, tumours remained in a state of stasis while
populations mobilized to sustain growth, albeit less efficiently than
counterparts [91]. Notably, tumour growth resumed unabated following treatment withdrawal, underpinned by dynamic conversion of
non-CSCs into
cells. Intriguingly, comparable growth dynamics were observed in cultured organoids, suggesting that the repopulation of
cells may partly rely on intrinsic
cell properties and proceed independently of tumour-activated stroma [91]. The mechanisms whereby non-CSCs, or distinct subsets thereof, sense the depletion of
CSCs within a tumour, and the intrinsic and extrinsic cues that trigger their mobilization remain an important avenue for investigation to better understand therapy resistance and tumour recurrence.
Similarly, xenografted patient-derived organoids contain differentiated
cells that can re-express
and fuel tumour regrowth [93]. In this model, short-term ablation of
cells, in combination with anti-EGFR therapy, elicited a more pronounced inhibition of tumour growth than either treatment alone [93]. Consistent with this, residual drug-resistant
cells that can reconstitute tumour growth, following
cell depletion, express the EGF-family member EREG [94]. Interestingly, oxaliplatin did not synergize with anti-EGFR, owing to the failure of chemotherapy to induce
expression in
cells [93]. Since
and
cells appear to reside within distinct tumour niches [92], it is plausible that depletion of the
population exposes differentiated
cells to aberrant instructive signals that incite their dedifferentiation and acquisition of CSC traits, analogous to the reversion of multiple intestinal cell types to an
state during injury-induced regeneration [9][20][26].
Until recently, little was known about the identity of metastasis-initiating cells in CRC and their relationship to primary tumour CSCs. Ablation of
CSCs in orthotopically implanted
organoids demonstrated an indispensable role for
CSCs in the formation and maintenance of metastatic outgrowths, even though their ablation proved inefficacious in the primary tumour setting [91]. Most notably, treatment cessation was not accompanied by regrowth of liver metastases, highlighting the potential therapeutic benefits of targeting
CSCs in the metastatic setting [91]—the ultimate cause of patient demise. In addition, these findings suggest that distinct tumour cell subsets may harbour differential abilities to drive primary tumour growth and initiate metastases, and underscore the importance of a permissive microenvironment as a prelude for colonization at the distant site [91].
Unexpectedly, ablation of
CSCs did not impair primary tumour invasiveness per se, yet still reduced liver metastatic burden, raising the possibility of LGR5-independent mechanisms of productive invasion [91]. Indeed, using intravital multiphoton microscopy to observe spontaneous metastatic progression from orthotopically implanted, genome-edited CRC organoids [95], van Rheenen and colleagues made the striking observation that the majority of circulating tumour cells lacks
. In vitro,
cells were intrinsically competent to form organoids and spawn functional
progeny, independently of niche signals, although the emergence of
cells was increased in the presence of HGF and FGF [96]. Importantly, targeted ablation of
cells prevented the progression of micrometastases, similar to the earlier findings of de Sousa e Melo et al. [91], with colonization and outgrowth of seeded
cells dependent on the de novo expression of
[96]. While
CSCs were detected in the migratory population, they were not typically recovered from the circulation, raising the intriguing possibility that, upon escaping the confines of the primary tumour niche,
cells enter an
non-CSC state that likely confers the ability to navigate and survive the perils of the metastatic cascade. Following seeding of
cells at the distant site, their reversion to an
state allows the outgrowth and progression of micrometastases. Although organoid cultures suggest that
non-CSCs can spontaneously revert to an
state in a niche-independent manner, signals emanating from the tumour microenvironment may nevertheless influence this transition in vivo. Deciphering the signals that instruct the plasticity transitions between
and
states, and the underlying molecular mechanisms, may yield important insights into critical determinants of disease progression and therapy resistance, and inform new strategies to target metastatic plasticity.
Overall, the above findings attest to the plasticity of
tumour-bulk cells, suggesting it may underpin failed treatment outcomes and metastatic competence. Which differentiated/non-CSC
subsets are mobilized to replenish primary tumour growth, when the
CSC pool is compromised, and whether these are the same cell subsets that exhibit metastatic competence remains to be seen. The ability of differentiated/non-CSC
cells to activate a dormant plasticity program at the distant site, seed metastases, and re-establish a cellular hierarchy de novo highlights the need to target intrinsic plasticity mechanisms as well as extrinsic niche pathways in order to ablate metastatic potential.
References
- Beumer, J.; Clevers, H. Cell fate specification and differentiation in the adult mammalian intestine. Nat. Rev. Mol. Cell Biol. 2021, 22, 39–53.
- Cheng, H.; Leblond, C.P. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. V. Unitarian Theory of the origin of the four epithelial cell types. Am. J. Anat. 1974, 141, 537–561.
- Sasaki, N.; Sachs, N.; Wiebrands, K.; Ellenbroek, S.I.; Fumagalli, A.; Lyubimova, A.; Begthel, H.; van den Born, M.; van Es, J.H.; Karthaus, W.R.; et al. Reg4+ deep crypt secretory cells function as epithelial niche for Lgr5+ stem cells in colon. Proc. Natl. Acad. Sci. USA 2016, 113, E5399–E5407.
- De Lau, W.; Barker, N.; Low, T.Y.; Koo, B.K.; Li, V.S.; Teunissen, H.; Kujala, P.; Haegebarth, A.; Peters, P.J.; van de Wetering, M.; et al. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature 2011, 476, 293–297.
- Carmon, K.S.; Gong, X.; Lin, Q.; Thomas, A.; Liu, Q. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/beta-catenin signaling. Proc. Natl. Acad. Sci. USA 2011, 108, 11452–11457.
- De Lau, W.; Peng, W.C.; Gros, P.; Clevers, H. The R-spondin/Lgr5/Rnf43 module: Regulator of Wnt signal strength. Genes Dev. 2014, 28, 305–316.
- Barker, N.; van Es, J.H.; Kuipers, J.; Kujala, P.; van den Born, M.; Cozijnsen, M.; Haegebarth, A.; Korving, J.; Begthel, H.; Peters, P.J.; et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 2007, 449, 1003–1007.
- Sato, T.; Vries, R.G.; Snippert, H.J.; van de Wetering, M.; Barker, N.; Stange, D.E.; van Es, J.H.; Abo, A.; Kujala, P.; Peters, P.J.; et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 2009, 459, 262–265.
- Tian, H.; Biehs, B.; Warming, S.; Leong, K.G.; Rangell, L.; Klein, O.D.; de Sauvage, F.J. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 2011, 478, 255–259.
- Metcalfe, C.; Kljavin, N.M.; Ybarra, R.; de Sauvage, F.J. Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration. Cell Stem Cell 2014, 14, 149–159.
- Potten, C.S.; Hume, W.J.; Reid, P.; Cairns, J. The segregation of DNA in epithelial stem cells. Cell 1978, 15, 899–906.
- Sangiorgi, E.; Capecchi, M.R. Bmi1 is expressed in vivo in intestinal stem cells. Nat. Genet. 2008, 40, 915–920.
- Montgomery, R.K.; Carlone, D.L.; Richmond, C.A.; Farilla, L.; Kranendonk, M.E.; Henderson, D.E.; Baffour-Awuah, N.Y.; Ambruzs, D.M.; Fogli, L.K.; Algra, S.; et al. Mouse telomerase reverse transcriptase (mTert) expression marks slowly cycling intestinal stem cells. Proc. Natl. Acad Sci. USA 2011, 108, 179–184.
- Takeda, N.; Jain, R.; LeBoeuf, M.R.; Wang, Q.; Lu, M.M.; Epstein, J.A. Interconversion between intestinal stem cell populations in distinct niches. Science 2011, 334, 1420–1424.
- Powell, A.E.; Wang, Y.; Li, Y.; Poulin, E.J.; Means, A.L.; Washington, M.K.; Higginbotham, J.N.; Juchheim, A.; Prasad, N.; Levy, S.E.; et al. The pan-ErbB negative regulator Lrig1 is an intestinal stem cell marker that functions as a tumor suppressor. Cell 2012, 149, 146–158.
- Li, N.; Yousefi, M.; Nakauka-Ddamba, A.; Jain, R.; Tobias, J.; Epstein, J.A.; Jensen, S.T.; Lengner, C.J. Single-cell analysis of proxy reporter allele-marked epithelial cells establishes intestinal stem cell hierarchy. Stem Cell Rep. 2014, 3, 876–891.
- Yan, K.S.; Chia, L.A.; Li, X.; Ootani, A.; Su, J.; Lee, J.Y.; Su, N.; Luo, Y.; Heilshorn, S.C.; Amieva, M.R.; et al. The intestinal stem cell markers Bmi1 and Lgr5 identify two functionally distinct populations. Proc. Natl. Acad. Sci. USA 2012, 109, 466–471.
- Asfaha, S.; Hayakawa, Y.; Muley, A.; Stokes, S.; Graham, T.A.; Ericksen, R.E.; Westphalen, C.B.; von Burstin, J.; Mastracci, T.L.; Worthley, D.L.; et al. Krt19(+)/Lgr5(-) cells are radioresistant cancer-initiating stem cells in the colon and intestine. Cell Stem Cell 2015, 16, 627–638.
- Bankaitis, E.D.; Ha, A.; Kuo, C.J.; Magness, S.T. Reserve stem cells in intestinal homeostasis and injury. Gastroenterology 2018, 155, 1348–1361.
- Tetteh, P.W.; Basak, O.; Farin, H.F.; Wiebrands, K.; Kretzschmar, K.; Begthel, H.; van den Born, M.; Korving, J.; de Sauvage, F.; van Es, J.H.; et al. Replacement of lost Lgr5-positive stem cells through plasticity of their enterocyte-lineage daughters. Cell Stem Cell 2016, 18, 203–213.
- Yan, K.S.; Gevaert, O.; Zheng, G.X.Y.; Anchang, B.; Probert, C.S.; Larkin, K.A.; Davies, P.S.; Cheng, Z.F.; Kaddis, J.S.; Han, A.; et al. Intestinal enteroendocrine lineage cells possess homeostatic and injury-inducible stem cell activity. Cell Stem Cell 2017, 21, 78–90.
- Schonhoff, S.E.; Giel-Moloney, M.; Leiter, A.B. Neurogenin 3-expressing progenitor cells in the gastrointestinal tract differentiate into both endocrine and non-endocrine cell types. Dev. Biol. 2004, 270, 443–454.
- Gross, S.; Balderes, D.; Liu, J.; Asfaha, S.; Gu, G.; Wang, T.C.; Sussel, L. Nkx2.2 is expressed in a subset of enteroendocrine cells with expanded lineage potential. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G975–G987.
- Sei, Y.; Lu, X.; Liou, A.; Zhao, X.; Wank, S.A. A stem cell marker-expressing subset of enteroendocrine cells resides at the crypt base in the small intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 300, G345–G356.
- Jadhav, U.; Saxena, M.; O’Neill, N.K.; Saadatpour, A.; Yuan, G.C.; Herbert, Z.; Murata, K.; Shivdasani, R.A. Dynamic reorganization of chromatin accessibility signatures during dedifferentiation of secretory precursors into Lgr5+ intestinal stem cells. Cell Stem Cell 2017, 21, 65–77.
- Van Es, J.H.; Sato, T.; van de Wetering, M.; Lyubimova, A.; Yee Nee, A.N.; Gregorieff, A.; Sasaki, N.; Zeinstra, L.; van den Born, M.; Korving, J.; et al. Dll1+ secretory progenitor cells revert to stem cells upon crypt damage. Nat. Cell Biol. 2012, 14, 1099–1104.
- Castillo-Azofeifa, D.; Fazio, E.N.; Nattiv, R.; Good, H.J.; Wald, T.; Pest, M.A.; de Sauvage, F.J.; Klein, O.D.; Asfaha, S. Atoh1(+) secretory progenitors possess renewal capacity independent of Lgr5(+) cells during colonic regeneration. EMBO J. 2019, 38, e99984.
- Ishibashi, F.; Shimizu, H.; Nakata, T.; Fujii, S.; Suzuki, K.; Kawamoto, A.; Anzai, S.; Kuno, R.; Nagata, S.; Ito, G.; et al. Contribution of ATOH1(+) cells to the homeostasis, repair, and tumorigenesis of the colonic epithelium. Stem Cell Rep. 2018, 10, 27–42.
- Tomic, G.; Morrissey, E.; Kozar, S.; Ben-Moshe, S.; Hoyle, A.; Azzarelli, R.; Kemp, R.; Chilamakuri, C.S.R.; Itzkovitz, S.; Philpott, A.; et al. Phospho-regulation of ATOH1 is required for plasticity of secretory progenitors and tissue regeneration. Cell Stem Cell 2018, 23, 436–443.
- Buczacki, S.J.; Zecchini, H.I.; Nicholson, A.M.; Russell, R.; Vermeulen, L.; Kemp, R.; Winton, D.J. Intestinal label-retaining cells are secretory precursors expressing Lgr5. Nature 2013, 495, 65–69.
- Harnack, C.; Berger, H.; Antanaviciute, A.; Vidal, R.; Sauer, S.; Simmons, A.; Meyer, T.F.; Sigal, M. R-spondin 3 promotes stem cell recovery and epithelial regeneration in the colon. Nat. Commun. 2019, 10, 4368.
- Westphalen, C.B.; Asfaha, S.; Hayakawa, Y.; Takemoto, Y.; Lukin, D.J.; Nuber, A.H.; Brandtner, A.; Setlik, W.; Remotti, H.; Muley, A.; et al. Long-lived intestinal tuft cells serve as colon cancer-initiating cells. J. Clin. Investig. 2014, 124, 1283–1295.
- Sei, Y.; Feng, J.; Samsel, L.; White, A.; Zhao, X.; Yun, S.; Citrin, D.; McCoy, J.P.; Sundaresan, S.; Hayes, M.M.; et al. Mature enteroendocrine cells contribute to basal and pathological stem cell dynamics in the small intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2018, 315, G495–G510.
- Schmitt, M.; Schewe, M.; Sacchetti, A.; Feijtel, D.; van de Geer, W.S.; Teeuwssen, M.; Sleddens, H.F.; Joosten, R.; van Royen, M.E.; van de Werken, H.J.G.; et al. Paneth cells respond to inflammation and contribute to tissue regeneration by acquiring stem-like features through SCF/c-Kit signaling. Cell Rep. 2018, 24, 2312–2328.
- Yu, S.; Tong, K.; Zhao, Y.; Balasubramanian, I.; Yap, G.S.; Ferraris, R.P.; Bonder, E.M.; Verzi, M.P.; Gao, N. Paneth Cell Multipotency Induced by Notch Activation following Injury. Cell Stem Cell 2018, 23, 46–59.
- Jones, J.C.; Brindley, C.D.; Elder, N.H.; Myers, M.G., Jr.; Rajala, M.W.; Dekaney, C.M.; McNamee, E.N.; Frey, M.R.; Shroyer, N.F.; Dempsey, P.J. Cellular plasticity of Defa4(Cre)-expressing Paneth cells in response to Notch activation and intestinal injury. Cell. Mol. Gastroenterol. Hepatol. 2019, 7, 533–554.
- Snippert, H.J.; van Es, J.H.; van den Born, M.; Begthel, H.; Stange, D.E.; Barker, N.; Clevers, H. Prominin-1/CD133 marks stem cells and early progenitors in mouse small intestine. Gastroenterology 2009, 136, 2187–2194.
- Itzkovitz, S.; Lyubimova, A.; Blat, I.C.; Maynard, M.; van Es, J.; Lees, J.; Jacks, T.; Clevers, H.; van Oudenaarden, A. Single-molecule transcript counting of stem-cell markers in the mouse intestine. Nat. Cell Biol. 2011, 14, 106–114.
- Munoz, J.; Stange, D.E.; Schepers, A.G.; van de Wetering, M.; Koo, B.K.; Itzkovitz, S.; Volckmann, R.; Kung, K.S.; Koster, J.; Radulescu, S.; et al. The Lgr5 intestinal stem cell signature: Robust expression of proposed quiescent ‘+4’ cell markers. EMBO J. 2012, 31, 3079–3091.
- Tao, S.; Tang, D.; Morita, Y.; Sperka, T.; Omrani, O.; Lechel, A.; Sakk, V.; Kraus, J.; Kestler, H.A.; Kuhl, M.; et al. Wnt activity and basal niche position sensitize intestinal stem and progenitor cells to DNA damage. EMBO J. 2015, 34, 624–640.
- Li, N.; Yousefi, M.; Nakauka-Ddamba, A.; Tobias, J.W.; Jensen, S.T.; Morrisey, E.E.; Lengner, C.J. Heterogeneity in readouts of canonical wnt pathway activity within intestinal crypts. Dev. Dyn. 2016, 245, 822–833.
- Van der Flier, L.G.; Sabates-Bellver, J.; Oving, I.; Haegebarth, A.; De Palo, M.; Anti, M.; Van Gijn, M.E.; Suijkerbuijk, S.; Van de Wetering, M.; Marra, G.; et al. The intestinal Wnt/TCF signature. Gastroenterology 2007, 132, 628–632.
- van der Flier, L.G.; van Gijn, M.E.; Hatzis, P.; Kujala, P.; Haegebarth, A.; Stange, D.E.; Begthel, H.; van den Born, M.; Guryev, V.; Oving, I.; et al. Transcription factor achaete scute-like 2 controls intestinal stem cell fate. Cell 2009, 136, 903–912.
- Schuijers, J.; Junker, J.P.; Mokry, M.; Hatzis, P.; Koo, B.K.; Sasselli, V.; van der Flier, L.G.; Cuppen, E.; van Oudenaarden, A.; Clevers, H. Ascl2 acts as an R-spondin/Wnt-responsive switch to control stemness in intestinal crypts. Cell Stem Cell 2015, 16, 158–170.
- Li, N.; Nakauka-Ddamba, A.; Tobias, J.; Jensen, S.T.; Lengner, C.J. Mouse Label-Retaining Cells Are Molecularly and Functionally Distinct from Reserve Intestinal Stem Cells. Gastroenterology 2016, 151, 298–310.
- Barriga, F.M.; Montagni, E.; Mana, M.; Mendez-Lago, M.; Hernando-Momblona, X.; Sevillano, M.; Guillaumet-Adkins, A.; Rodriguez-Esteban, G.; Buczacki, S.J.A.; Gut, M.; et al. Mex3a marks a slowly dividing subpopulation of Lgr5+ intestinal stem cells. Cell Stem Cell 2017, 20, 801–816.
- Giroux, V.; Stephan, J.; Chatterji, P.; Rhoades, B.; Wileyto, E.P.; Klein-Szanto, A.J.; Lengner, C.J.; Hamilton, K.E.; Rustgi, A.K. Mouse intestinal Krt15+ crypt cells are radio-resistant and tumor initiating. Stem Cell Rep. 2018, 10, 1947–1958.
- Sheng, X.; Lin, Z.; Lv, C.; Shao, C.; Bi, X.; Deng, M.; Xu, J.; Guerrero-Juarez, C.F.; Li, M.; Wu, X.; et al. Cycling Stem Cells Are Radioresistant and Regenerate the Intestine. Cell Rep. 2020, 32, 107952.
- Li, L.; Clevers, H. Coexistence of quiescent and active adult stem cells in mammals. Science 2010, 327, 542–545.
- Kim, T.H.; Saadatpour, A.; Guo, G.; Saxena, M.; Cavazza, A.; Desai, N.; Jadhav, U.; Jiang, L.; Rivera, M.N.; Orkin, S.H.; et al. Single-cell transcript profiles reveal multilineage priming in early progenitors derived from Lgr5(+) intestinal stem cells. Cell Rep. 2016, 16, 2053–2060.
- Baulies, A.; Angelis, N.; Foglizzo, V.; Danielsen, E.T.; Patel, H.; Novellasdemunt, L.; Kucharska, A.; Carvalho, J.; Nye, E.; De Coppi, P.; et al. The transcription co-repressors MTG8 and MTG16 regulate exit of intestinal stem cells from their niche and differentiation into enterocyte vs secretory lineages. Gastroenterology 2020, 159, 1328–1341.
- Leedham, S.J. Reserving the right to change the intestinal stem cell model. Cell Stem Cell 2020, 26, 301–302.
- Murata, K.; Jadhav, U.; Madha, S.; van Es, J.; Dean, J.; Cavazza, A.; Wucherpfennig, K.; Michor, F.; Clevers, H.; Shivdasani, R.A. Ascl2-dependent cell dedifferentiation drives regeneration of ablated intestinal stem cells. Cell Stem Cell 2020, 26, 377–390.
- Kim, T.H.; Li, F.; Ferreiro-Neira, I.; Ho, L.L.; Luyten, A.; Nalapareddy, K.; Long, H.; Verzi, M.; Shivdasani, R.A. Broadly permissive intestinal chromatin underlies lateral inhibition and cell plasticity. Nature 2014, 506, 511–515.
- Nusse, Y.M.; Savage, A.K.; Marangoni, P.; Rosendahl-Huber, A.K.M.; Landman, T.A.; de Sauvage, F.J.; Locksley, R.M.; Klein, O.D. Parasitic helminths induce fetal-like reversion in the intestinal stem cell niche. Nature 2018, 559, 109–113.
- Yui, S.; Azzolin, L.; Maimets, M.; Pedersen, M.T.; Fordham, R.P.; Hansen, S.L.; Larsen, H.L.; Guiu, J.; Alves, M.R.P.; Rundsten, C.F.; et al. YAP/TAZ-dependent reprogramming of colonic epithelium links ECM remodeling to tissue regeneration. Cell Stem Cell 2018, 22, 35–49.
- Mustata, R.C.; Vasile, G.; Fernandez-Vallone, V.; Strollo, S.; Lefort, A.; Libert, F.; Monteyne, D.; Perez-Morga, D.; Vassart, G.; Garcia, M.I. Identification of Lgr5-independent spheroid-generating progenitors of the mouse fetal intestinal epithelium. Cell Rep. 2013, 5, 421–432.
- Gregorieff, A.; Wrana, J.L. Hippo signalling in intestinal regeneration and cancer. Curr. Opin. Cell Biol. 2017, 48, 17–25.
- Gregorieff, A.; Liu, Y.; Inanlou, M.R.; Khomchuk, Y.; Wrana, J.L. Yap-dependent reprogramming of Lgr5(+) stem cells drives intestinal regeneration and cancer. Nature 2015, 526, 715–718.
- Barry, E.R.; Morikawa, T.; Butler, B.L.; Shrestha, K.; de la Rosa, R.; Yan, K.S.; Fuchs, C.S.; Magness, S.T.; Smits, R.; Ogino, S.; et al. Restriction of intestinal stem cell expansion and the regenerative response by YAP. Nature 2013, 493, 106–110.
- Li, Q.; Sun, Y.; Jarugumilli, G.K.; Liu, S.; Dang, K.; Cotton, J.L.; Xiol, J.; Chan, P.Y.; DeRan, M.; Ma, L.; et al. Lats1/2 sustain intestinal stem cells and Wnt activation through TEAD-dependent and independent transcription. Cell Stem Cell 2020, 26, 675–692.
- Cheung, P.; Xiol, J.; Dill, M.T.; Yuan, W.C.; Panero, R.; Roper, J.; Osorio, F.G.; Maglic, D.; Li, Q.; Gurung, B.; et al. Regenerative Reprogramming of the intestinal stem cell state via Hippo signaling suppresses metastatic colorectal cancer. Cell Stem Cell 2020, 27, 590–604.
- Ayyaz, A.; Kumar, S.; Sangiorgi, B.; Ghoshal, B.; Gosio, J.; Ouladan, S.; Fink, M.; Barutcu, S.; Trcka, D.; Shen, J.; et al. Single-cell transcriptomes of the regenerating intestine reveal a revival stem cell. Nature 2019, 569, 121–125.
- Bigaeva, E.; Uniken Venema, W.T.C.; Weersma, R.K.; Festen, E.A.M. Understanding human gut diseases at single-cell resolution. Hum. Mol. Genet. 2020, 29, R51–R58.
- Cai, J.; Maitra, A.; Anders, R.A.; Taketo, M.M.; Pan, D. β-Catenin destruction complex-independent regulation of Hippo-YAP signaling by APC in intestinal tumorigenesis. Genes Dev. 2015, 29, 1493–1506.
- Roulis, M.; Kaklamanos, A.; Schernthanner, M.; Bielecki, P.; Zhao, J.; Kaffe, E.; Frommelt, L.S.; Qu, R.; Knapp, M.S.; Henriques, A.; et al. Paracrine orchestration of intestinal tumorigenesis by a mesenchymal niche. Nature 2020, 580, 524–529.
- Zhou, D.; Zhang, Y.; Wu, H.; Barry, E.; Yin, Y.; Lawrence, E.; Dawson, D.; Willis, J.E.; Markowitz, S.D.; Camargo, F.D.; et al. Mst1 and Mst2 protein kinases restrain intestinal stem cell proliferation and colonic tumorigenesis by inhibition of Yes-associated protein (Yap) overabundance. Proc. Natl. Acad. Sci. USA 2011, 108, E1312–E1320.
- Rosenbluh, J.; Nijhawan, D.; Cox, A.G.; Li, X.; Neal, J.T.; Schafer, E.J.; Zack, T.I.; Wang, X.; Tsherniak, A.; Schinzel, A.C.; et al. β-Catenin-driven cancers require a YAP1 transcriptional complex for survival and tumorigenesis. Cell 2012, 151, 1457–1473.
- Reischmann, N.; Andrieux, G.; Griffin, R.; Reinheckel, T.; Boerries, M.; Brummer, T. BRAF(V600E) drives dedifferentiation in small intestinal and colonic organoids and cooperates with mutant p53 and Apc loss in transformation. Oncogene 2020, 39, 6053–6070.
- Han, T.; Goswami, S.; Hu, Y.; Tang, F.; Zafra, M.P.; Murphy, C.; Cao, Z.; Poirier, J.T.; Khurana, E.; Elemento, O.; et al. Lineage Reversion Drives WNT Independence in Intestinal Cancer. Cancer Discov. 2020, 10, 1590–1609.
- Kim, H.B.; Kim, M.; Park, Y.S.; Park, I.; Kim, T.; Yang, S.Y.; Cho, C.J.; Hwang, D.; Jung, J.H.; Markowitz, S.D.; et al. Prostaglandin E(2) activates YAP and a positive-signaling loop to promote colon regeneration after colitis but also carcinogenesis in mice. Gastroenterology 2017, 152, 616–630.
- Koulis, C.; Yap, R.; Engel, R.; Jardé, T.; Wilkins, S.; Solon, G.; Shapiro, J.D.; Abud, H.; McMurrick, P. Personalized medicine-current and emerging predictive and prognostic biomarkers in colorectal cancer. Cancers 2020, 12, 812.
- Ganesh, K.; Basnet, H.; Kaygusuz, Y.; Laughney, A.M.; He, L.; Sharma, R.; O’Rourke, K.P.; Reuter, V.P.; Huang, Y.H.; Turkekul, M.; et al. L1CAM defines the regenerative origin of metastasis-initiating cells in colorectal cancer. Nat. Cancer 2020, 1, 28–45.
- Preston, S.L.; Wong, W.M.; Chan, A.O.; Poulsom, R.; Jeffery, R.; Goodlad, R.A.; Mandir, N.; Elia, G.; Novelli, M.; Bodmer, W.F.; et al. Bottom-up histogenesis of colorectal adenomas: Origin in the monocryptal adenoma and initial expansion by crypt fission. Cancer Res. 2003, 63, 3819–3825.
- Shih, I.M.; Wang, T.L.; Traverso, G.; Romans, K.; Hamilton, S.R.; Ben-Sasson, S.; Kinzler, K.W.; Vogelstein, B. Top-down morphogenesis of colorectal tumors. Proc. Natl. Acad. Sci. USA 2001, 98, 2640–2645.
- Barker, N.; Ridgway, R.A.; van Es, J.H.; van de Wetering, M.; Begthel, H.; van den Born, M.; Danenberg, E.; Clarke, A.R.; Sansom, O.J.; Clevers, H. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 2009, 457, 608–611.
- Hilkens, J.; Timmer, N.C.; Boer, M.; Ikink, G.J.; Schewe, M.; Sacchetti, A.; Koppens, M.A.J.; Song, J.Y.; Bakker, E.R.M. RSPO3 expands intestinal stem cell and niche compartments and drives tumorigenesis. Gut 2017, 66, 1095–1105.
- Zhu, L.; Gibson, P.; Currle, D.S.; Tong, Y.; Richardson, R.J.; Bayazitov, I.T.; Poppleton, H.; Zakharenko, S.; Ellison, D.W.; Gilbertson, R.J. Prominin 1 marks intestinal stem cells that are susceptible to neoplastic transformation. Nature 2009, 457, 603–607.
- Powell, A.E.; Vlacich, G.; Zhao, Z.Y.; McKinley, E.T.; Washington, M.K.; Manning, H.C.; Coffey, R.J. Inducible loss of one Apc allele in Lrig1-expressing progenitor cells results in multiple distal colonic tumors with features of familial adenomatous polyposis. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 307, G16–G23.
- Cammareri, P.; Vincent, D.F.; Hodder, M.C.; Ridgway, R.A.; Murgia, C.; Nobis, M.; Campbell, A.D.; Varga, J.; Huels, D.J.; Subramani, C.; et al. TGFbeta pathway limits dedifferentiation following WNT and MAPK pathway activation to suppress intestinal tumourigenesis. Cell Death Differ. 2017, 24, 1681–1693.
- Schwitalla, S.; Fingerle, A.A.; Cammareri, P.; Nebelsiek, T.; Goktuna, S.I.; Ziegler, P.K.; Canli, O.; Heijmans, J.; Huels, D.J.; Moreaux, G.; et al. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell 2013, 152, 25–38.
- Tetteh, P.W.; Kretzschmar, K.; Begthel, H.; van den Born, M.; Korving, J.; Morsink, F.; Farin, H.; van Es, J.H.; Offerhaus, G.J.; Clevers, H. Generation of an inducible colon-specific Cre enzyme mouse line for colon cancer research. Proc. Natl. Acad. Sci. USA 2016, 113, 11859–11864.
- Hayakawa, Y.; Tsuboi, M.; Asfaha, S.; Kinoshita, H.; Niikura, R.; Konishi, M.; Hata, M.; Oya, Y.; Kim, W.; Middelhoff, M.; et al. BHLHA15-positive secretory precursor cells can give rise to tumors in intestine and colon in mice. Gastroenterology 2019, 156, 1066–1081.
- Davis, H.; Irshad, S.; Bansal, M.; Rafferty, H.; Boitsova, T.; Bardella, C.; Jaeger, E.; Lewis, A.; Freeman-Mills, L.; Giner, F.C.; et al. Aberrant epithelial GREM1 expression initiates colonic tumorigenesis from cells outside the stem cell niche. Nat. Med. 2015, 21, 62–70.
- Herrinton, L.J.; Liu, L.; Levin, T.R.; Allison, J.E.; Lewis, J.D.; Velayos, F. Incidence and mortality of colorectal adenocarcinoma in persons with inflammatory bowel disease from 1998 to 2010. Gastroenterology 2012, 143, 382–389.
- Vermeulen, L.; Todaro, M.; de Sousa Mello, F.; Sprick, M.R.; Kemper, K.; Perez Alea, M.; Richel, D.J.; Stassi, G.; Medema, J.P. Single-cell cloning of colon cancer stem cells reveals a multi-lineage differentiation capacity. Proc. Natl. Acad. Sci. USA 2008, 105, 13427–13432.
- Junttila, M.R.; Mao, W.; Wang, X.; Wang, B.E.; Pham, T.; Flygare, J.; Yu, S.F.; Yee, S.; Goldenberg, D.; Fields, C.; et al. Targeting LGR5+ cells with an antibody-drug conjugate for the treatment of colon cancer. Sci. Transl. Med. 2015, 7, 314ra186.
- Kemper, K.; Prasetyanti, P.R.; De Lau, W.; Rodermond, H.; Clevers, H.; Medema, J.P. Monoclonal antibodies against Lgr5 identify human colorectal cancer stem cells. Stem Cells 2012, 30, 2378–2386.
- Schepers, A.G.; Snippert, H.J.; Stange, D.E.; van den Born, M.; van Es, J.H.; van de Wetering, M.; Clevers, H. Lineage tracing reveals Lgr5+ stem cell activity in mouse intestinal adenomas. Science 2012, 337, 730–735.
- Roper, J.; Tammela, T.; Cetinbas, N.M.; Akkad, A.; Roghanian, A.; Rickelt, S.; Almeqdadi, M.; Wu, K.; Oberli, M.A.; Sanchez-Rivera, F.J.; et al. In vivo genome editing and organoid transplantation models of colorectal cancer and metastasis. Nat. Biotechnol. 2017, 35, 569–576.
- de Sousa e Melo, F.; Kurtova, A.V.; Harnoss, J.M.; Kljavin, N.; Hoeck, J.D.; Hung, J.; Anderson, J.E.; Storm, E.E.; Modrusan, Z.; Koeppen, H.; et al. A distinct role for Lgr5(+) stem cells in primary and metastatic colon cancer. Nature 2017, 543, 676–680.
- Cortina, C.; Turon, G.; Stork, D.; Hernando-Momblona, X.; Sevillano, M.; Aguilera, M.; Tosi, S.; Merlos-Suarez, A.; Stephan-Otto Attolini, C.; Sancho, E.; et al. A genome editing approach to study cancer stem cells in human tumors. EMBO Mol. Med. 2017, 9, 869–879.
- Shimokawa, M.; Ohta, Y.; Nishikori, S.; Matano, M.; Takano, A.; Fujii, M.; Date, S.; Sugimoto, S.; Kanai, T.; Sato, T. Visualization and targeting of LGR5(+) human colon cancer stem cells. Nature 2017, 545, 187–192.
- Kobayashi, S.; Yamada-Okabe, H.; Suzuki, M.; Natori, O.; Kato, A.; Matsubara, K.; Jau Chen, Y.; Yamazaki, M.; Funahashi, S.; Yoshida, K.; et al. LGR5-positive colon cancer stem cells interconvert with drug-resistant LGR5-negative cells and are capable of tumor reconstitution. Stem Cells 2012, 30, 2631–2644.
- Fumagalli, A.; Suijkerbuijk, S.J.E.; Begthel, H.; Beerling, E.; Oost, K.C.; Snippert, H.J.; van Rheenen, J.; Drost, J. A surgical orthotopic organoid transplantation approach in mice to visualize and study colorectal cancer progression. Nat. Protoc. 2018, 13, 235–247.
- Fumagalli, A.; Oost, K.C.; Kester, L.; Morgner, J.; Bornes, L.; Bruens, L.; Spaargaren, L.; Azkanaz, M.; Schelfhorst, T.; Beerling, E.; et al. Plasticity of Lgr5-negative cancer cells drives metastasis in colorectal cancer. Cell Stem Cell 2020, 26, 569–578.
