The mammalian genome comprehends a small minority of genes that encode for proteins (barely 2% of the total genome in humans) and an immense majority of genes that are transcribed into RNA but not encoded for proteins (ncRNAs). These non-coding genes are intimately related to the expression regulation of protein-coding genes. The ncRNAs subtypes differ in their size, so there are long non-coding genes (lncRNAs) and other smaller ones, like microRNAs (miRNAs) and piwi-interacting RNAs (piRNAs).
Due to their important role in the maintenance of cellular functioning, any deregulation of the expression profiles of these ncRNAs can dissemble in the development of different types of diseases, such as cancer, neurodegenerative, or cardiovascular disorders.
- ncRNAs
- miRNAs
- circRNAs
- lncRNAs
- piRNAs
- diseases
- cancer
- cardiovascular disease
- neurodegenerative
1. Introduction
ncRNAs are usually classified in accordance with their size. For instance, short RNAs are those smaller than 200 nucleotides in length. In this group there are included small interfering RNAs (siRNAs), piwi-interacting RNAs (piRNAs), and microRNAs (miRNAs) [1][2]. On the other hand, we also find long noncoding RNAs (lncRNAs) which are longer than 200 nucleotides [3]. It is accepted that most ncRNAs are associated to gene expression regulation, but in addition, it has recently been discovered that ncRNAs are also involved in guide DNA synthesis or genome rearrangement [4]. This fact implies that these RNAs are capable of modulating both, gene production and genome reorganization [5]. To perform their role, some ncRNAs need their secondary RNA structure (such as ribozymes and riboswitches), although most of them require a complementary sequence (snRNP, snoRNP, miRNA, piRNA, and lncRNA) to accomplish its cellular role [6][7]. Please see
.
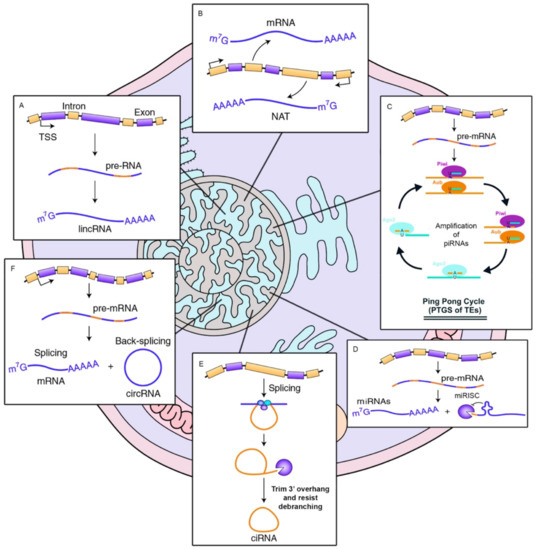
Biogenesis of ncRNAs. (
) long intergenic non-coding RNAs are transcribed by Pol II in a pre-RNA immature form. Once matured, they are poly-adenylated. (
) Natural antisense transcripts transcription from the opposed strand of protein-coding genes. (
) piRNAs recognize their targets and recruits piwi proteins. It results in the cleavage of the primary piRNA transcript, producing the secondary piRNA in an amplification mechanism called ping-pong. (
) miRNAs exported are bound with AGO proteins to form the miRISC complex to inhibit mRNA expression. (
,
) circular RNAs, (derived from lariats introns: ciRNAs, or from back-splicing events of exonic pre-mRNAs: circRNAs) are covalently closed and usually contains miRNA binding sites.
2. Involvement in Human Diseases
There is an obvious relationship between all the ncRNAs we have described, so they should not be seen as RNAs that exert their function in isolation. As it has been aforementioned, miRNAs are negative modulators of gene expression since they bind to mRNAs of different genes (a miRNA can regulate several different mRNAs and therefore a gene can be regulated by more than one different miRNA); thus, these ncRNAs are linked to gene regulatory networks. Moreover, miRNAs functionally cooperate with ncRNA molecules, such as circRNAs and lncRNAs, to control their cellular presence. In turn, lncRNAs and circRNAs module the expression of miRNAs by a sponge-like process sharing common MRE (miRNAs responses elements), inhibiting normal miRNA targeting activity on mRNA. The alterations of this equilibrium have a very important outcome that affects the cellular fate, since important imbalances can propitiate the development of diseases. (Please see
). In consequence, this competing endogenous RNA (ceRNA) network has been found to be distorted in many diseases such as cancer [8] or cardiovascular diseases [9], among others.
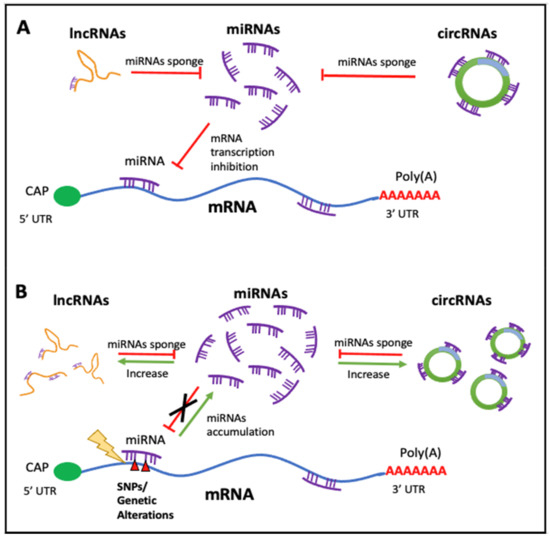
Regulatory feedback between ncRNAs. The interconnection existent among the main ncRNAs species involved in human diseases. (
) Regular interaction in a normal scenario (healthy cells). (
) When an alteration occurs (i.e., SNPs, mutation, expression alteration) there is an imbalance in the competing endogenous RNA (ceRNA) characteristic of many diseases.
Another relevant processes that affect the expression and function of ncRNAs, and thus the network of their targets, are genetic variants. These sequence changes are associated with diseases by performing genome-wide association studies (GWAS). The majority of GWAS-associated variants fall in ncRNAs loci, suggesting that they affect complex traits and diseases by altering expression of nearby genes, through regulatory mechanisms [10]. Among the examples that will be mentioned, we will see how the presence of SNPs in the sequence of a ncRNA can inhibit its maturation and therefore its cellular function. The presence of these variants in promoter regions also affects the expression levels of ncRNAs.This fact confers a higher probability of suffering from a disease. Finally, since ncRNAs exert their function by means of complementary sequence binding to their target RNAs, sequence changes in these regions (in the ncRNA itself or in its targets) can alter the binding efficiency. ln line with this, Gong et al. predicted that 52% of SNPs in the dbSNP database (release 132) would be able to create novel miRNA binding sites [11].
This review will be focused on explaining how the most described non-coding RNA species (miRNAs, lncRNAs, piRNAs, and circRNAs) play an important role in the development of human diseases. A summary of different types of ncRNAs, along with their chromosome location and their implication in various human diseases, can be found in
. Among them, we highlight the following ones due to their high incidence in the population:
2.1. Non-Coding RNAs in Cancer
A large number of laboratories have published and experimentally validated that alteration in the expression profiles of certain non-coding RNAs are linked to development of cancer [12]. In addition, single nucleotide polymorphisms (SNPs) are the genetic variations that are most frequently associated with the onset of cancer. Remarkably, 85% of SNPs are located in non-coding regions and linked to the development of this disease [13].
In the case of miRNA and more specifically in cancer, there are the so-called OncomiRs, which appear overexpressed in tumor samples and linked to tumor development [14]. It has been reported that OncomiRs affect the proliferation and signaling of cancer cells, prevent apoptosis, favors invasion, angiogenesis and metastasis [15]. (Please see
.) For example, the miR-17/92 cluster was the first OncomiR described. This is a polycistronic RNA that encodes for 6 miRNAs. Its overexpression is related to the inhibition of E2F1 [16] and AIB1 genes [17] triggering tumor initiation by stimulating cell progression and proliferation. On the contrary, anti-OncomiRs are a group of miRNAs that behaves as tumor suppressors, such as the miR-143/145 cluster. Both miRNAs control the expression of dozens of genes and its deregulation is believed to propitiate the appearance of the initial events in cancer. In comparison with cells from non-tumor tissues, these miRNAs are expressed in a smaller amount in colon, head and neck, breast, bladder, and lung cancers [18].
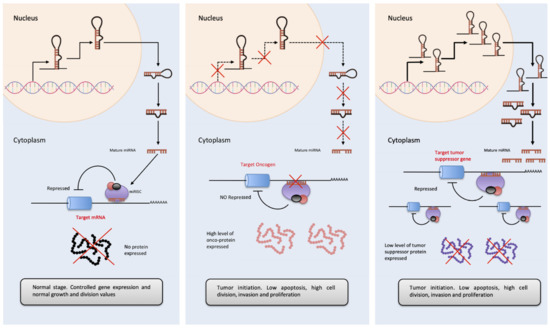
OncomiRs involvement in tumor initiation process.
. The diagram represents basal expression of miRNAs. These are released to the cytoplasm in a highly controlled amount, to exert their function by binding to the 3′-UTR of their target mRNA, inhibiting the formation of protein.
. In the case of cancer, the expression of multiple genes is altered, if this variation causes an inhibition of OncomiRs (miRNAs that regulate oncogene expressions), these are not able to inhibit the amount of oncogenic protein, favoring proliferation, cell cycle progression as well as a decrease in apoptosis and in the tumor initiation.
. On the contrary, in cancer, the overexpression of miRNAs acting as tumor suppressor regulators has also been observed (essentially, they are miRNAs that inhibit the expression of tumor suppressor genes). A considerable increase in the cytoplasm of OncomiRs, causes the almost disappearance of tumor suppressor proteins, favoring the development of cancer, its progression and tumor invasion.
This indicates that a recurrent oncogenic process is implicated. Well known is the TP53 gene loss-of-function, as the activation of the p53 pathway increases the miR-143 and miR-145 levels through a transcriptional mechanism [19]. Another mechanism that affects miR-143 and miR-145 levels depends on the mitogen-activated protein kinase (MAPK) cascade activity, with RREB1 being the effector for the repression of the cluster [20]. In addition, in the case of colorectal cancer, the variant rs353292 in the flanking region of miR-143/145 increases the risk of developing this disease [21], as rs353292 CT/TT individuals presented a lower expression of miR-143.
Furthermore, there is a group of miRNAs involved in an extensive range of diseases, those related to hypoxia (low oxygen levels) and called hypoxamirs. In the case of cancer, there are a high number of tumors with hypoxic tendency (being large masses of tissue with a high proliferation rate and little vascular development, the development of hypoxic regions is favored), such as breast cancer. Among this group of microRNAs, we must highlight miR-210, recognized as a mediator of cellular responses to stress related to hypoxia. Recent studies have postulated that hypoxamirs regulate hypoxic transcriptional cascades, angiogenesis, and endothelial growth [22] and consequently, involved in metastasis. miRNAs that suffer an alteration in their expression levels, have been related to various functions in tumoral events, including the maintenance of proliferation, resistance to cell death, invasion, and metastasis and very recently with resistance to drugs [23]. These anomalous expression patterns have been endorsed to DNA modifications (deletions, amplifications, or mutations) [24], epigenetic alterations [25], miss-regulated transcription factors [26], and deregulation of RNA-binding proteins (RBP) that contribute to miRNA synthesis [27].
In addition, single nucleotide polymorphisms (SNPs) are the genetic variations that are most frequently associated with the onset of cancer. Remarkably, 85% of SNPs are located in non-coding regions and linked to the development of this disease [13]. For example, the SNP rs11671784 is localized within the miR-27a sequence affecting its processing efficiency. This variant has been implicated in gastric cancer reduction risk by impairing the maturation of pre-miR-27a to mature miR-27a [28].
According to the atlas of human long non-coding RNAs in FANTOM5 [29], there are about 28,000 known human lncRNA genes, making them the most abundant transcribed RNAs from DNA. They can work as enhancers, scaffolds or decoys by binding with RNAs or even with RNA binding proteins, which alters all cellular signaling regulation networks [5]. For instance, the lncRNA MEG3 (maternally expressed gene 3) binds the p53 DNA binding domain, favoring the activation of P53 [30]. Interestingly, in cervical cancer, MEG3 expression is inversely associated to tumor volume and metastasis, which indicates its strong role as a tumor suppressor through P53 and pointing out as a possible therapeutic target [31]. Likewise, low levels of MEG3 have been linked with an increment in cell cycle progression and autophagy in bladder cancer [32]. Moreover, another example of the role of the long non-coding RNAs in gene regulation is the lncRNA-PVT1 that usually acts as miRNA sponge towards the miR-200 family in normal breast tissue and loses its functionality in breast cancer cells, altering the genetic regulation in this tissue [33].
A recent study proved that LINC00883 could act as miRNA “sponge” for miR-150 [34] which promotes tumor cell proliferation by negatively regulating tumor suppressor gene SRCIN1 [35] and Notch3 [36]. In fact, Notch3 played an important role in oncogenesis and resistance to chemotherapy in lung cancer [37]. Qi Sun et al. demonstrated that T allele of rs793544 was associated with the low expression level of LINC00883 in lung tumor tissues [38]. Taken together, it seems that SNP rs793544 is related with the diminished expression level of LNC00883, increasing the risk of lung cancer by altering the expression of miR-150.
In the case of piRNAs, it was initially thought that their only function was related to the suppression of the activity of mobile elements (or transposons). In fact, the inhibition of piRNAs that normally suppresses the function of these elements can facilitate mutagenic retro-transpositions and DNA instability, thus favoring cancer initiation. Nevertheless, it is not an exclusive mechanism, because it has been proposed that the piRNA/PIWI complex can cause aberrant DNA methylation, which results in genomic silencing, stimulating the cell to enter in a “stem-like” stage [39]. In detail, piRNAs can stimulate
methylation, in human lymphoma and breast cancer cell lines, single copy piRNAs favor methylation of gene-specific DNA through imperfect binding to genomic DNA [40]. Moreover, piRNAs have been associated to cell cycle regulation, in fact, it has been discovered that alteration in the expression of piRNA/PIWI complex correlates with clinical variables in tumorigenic samples indicating a new role for piRNA in cancer. For instance, in breast cancer, four significantly regulated piRNAs have been discovered: piR-20365, piR-4987, piR-20582, and piR-20485 [41]. In addition, genetic association studies have identified variants associated with cancer in piRNA loci. As an example, the SNP rs147061479 in piR-598 increases glioma risk by affecting the tumor-suppressive function of piR-598 [42].
Finally, as we mentioned, miRNAs are crucial key actors in the pathogenesis of most human tumors [43]. Therefore, because circRNAs function as miRNA modulators, these in turn are also linked to cancer. Currently, a small number of circRNAs have been discovered with several targets’ sites for a particular miRNA, but it has also been observed that most of the circRNAs have, in fact, other functions besides the regulation of miRNA [44]. For example, it has been revealed that circRNAs are highly expressed in several tumor cell lines from the ENCODE consortium data [45]. These studies have demonstrated the involvement of circRNAs in cancer through unexpected functions. This is the case of colorectal carcinoma (RCC) in which Bachmayr-Heyda et al. confirmed a general decrease in circRNAs expression levels in tumor tissue compared to adjacent non-cancerous tissues [46]. This reduction has been negatively correlated with proliferation. The authors affirmed that circRNAs are accumulated in stable-arrested cells, while they are disseminated among the proliferating ones.
2.2. Neurodegenerative Diseases
Although ncRNAs are expressed in all cell types, the Central Nervous System (CNS) is especially enriched [47]. Thus, approximately 40% of the genes encoding lncRNA are specifically expressed in brain tissue. Other types of non-coding RNAs, such as circRNA and certain miRNAs, are also specifically abundant in the CNS (and some of them specifically involved in synapses) [48][49]. miRNAs such as miR-124 and miR-132 have a regulatory impact on neurogenesis [50], while other lncRNAs, as rhab-domyosarcoma 2 associated transcript (RMST) and Tcl1 upstream neuron-associated long intergenic ncRNA (TUNA), stimulates neuronal differentiation [51]. In addition, ncRNAs enriched in synaptic connections are known, for example certain miRNA precursors and several miRNAs (e.g., miR-9, miR-132, miR-134, and miR-138) [52]. Moreover, most circRNAs that are expressed in the brain act as synaptic regulators to regulate local protein expression.
In the case of Parkinson Disease (PD), it has been found that there is a negative correlation between the levels of expression of certain miRNAs and two of the genes involved in this disease: α-synuclein (SNCA) and leucine-rich repeat kinase2 (LRRK2). For instance, miR-7 and miR-153 [53] are natural regulators of SNCA and LRRK2. Both miRNAs are highly expressed in the brain [54]. In addition, a specific circRNA related to this disease has also been found through the inhibition of miR-7. Recent research has revealed that circRNA CDR1 acts as a negative regulator of miR-7 causing increased expression of SNCA, which is involved in the development of PD and contributes to oxidative stress. Therefore, circRNA CDR1AS as a miR-7 sponge plays a crucial role in PD by repressing miR-7 [55].
GWAS studies in PD cohorts have revealed that polymorphisms such as rs2070535 and rs10849446 are located in genes highly related to PD, as PDCK and SCNN1A [56][57]. Further studies on these polymorphisms have determined that rs2070535 is located in the 3′-UTR region of PDCK, and differential expression of miRNAs may influence the changes in expression of this gene as seen in this disease. Similarly, rs10849446 is located in a intronic region of SCNN1A related to lncRNAs [58].
LncRNAs involved in PD have also been discovered, for example the antisense transcript of the UCHL1 gene (UCHL1-AS) is also linked to the prognosis of this disease. The Ubiquitin carboxyl-terminal hydrolase isozyme L1 (UCHL1] is highly expressed in the substantia nigra and related to neuron differentiation. Several authors had pointed out that UCHL1 is a PD risk gene [59]. In that sense, UCHL1-AS targets Uchl1 mRNA to induce its translation and increasing UCHL1 levels [60].
Alzheimer’s disease (AD) is another type of neurodegenerative disorder caused by many different factors. Among them, we can highlight the following ones related to ncRNAs. Elevated levels of amyloid-β protein (Aβ), BACE1 proteins, as well as the NAT generated in the strand opposite to this gene, BACE1-AS, have been detected in AD. Aβ is resulting from the cleavage of the amyloid precursor protein (APP) by the APP enzyme of the beta 1 site (BACE1) and the γ-secretase complex. The aberrant expression of these molecules has been related with numerous neurological syndromes and mainly in subjects with Alzheimer’s disease, which emphasizes the importance of regulating the catalytic activity of BACE1 in this disease. In that sense, it was discovered that the CIRS-7 circRNA, downregulated in AD brain tissue, has the main function of reducing APP and BACE1 protein levels [61]. In addition, other set of circRNAs has been implicated in AD through the alteration of myelin function [62].
Another example is 17A, this is a ncRNA of a size of 159 nt from the third intron of the G 51 gene receptor coupled to the G protein (GPR51). This ncRNA is involved in increasing expression of the GABA B receptor affecting all signaling cascades dependent on GABA B. ncRNA 17A is overexpressed in AD patients versus healthy tissues, proposing that it might be involved somehow in the initiation of AD [63][64].
Furthermore, miRNAs are also associated in the deposition of Aβ, the formation of neurofibrillar nodes (NFT) and extensive neuronal degeneration in the brain. It has been found that miRNAs are able to control APP expression in several ways: for example, miR-106a, miR-520c, members of the miR-20a family (miR-20a, miR-17), miR-16, miR-101, miR-147, miR-655, miR-323-3p, miR-644, and miR-153 are able to bind to a specific sequence in the 3′UTR of APP. Through experimental in vitro studies, the reduction in the expression level of APP when co-expressed with these miRNAs has been verified [65][66].
Single nucleotide polymorphisms in ncRNAs have been also associated with AD. It is important to mention that rs7232 and rs12453 in lncRNA NONHSAT160355.1 significantly downregulate the expression of a known AD pathogenic gene, TCN1 in Temporal Cortex, which participates in the regulation of homocysteine in brain to increase risk of AD [67].
2.2.1. Cardiovascular Diseases
Cardiovascular diseases (CVD) are considered the first cause of mortality all over the world. This concept involves not only heart diseases but also blood vessel associated diseases. The main risk factors classically associated with these diseases are hypertension, diabetes, and smoking, and can be used for stratification and prediction factors for prognosis of the patients. The rest of the risk factors can vary depending on the pathology examined.
Due to the high number of diseases associated to this term, in this section we are going to examine the main ncRNAs historically associated with the development of CVD, and two different cardiovascular pathologies such as atherosclerosis and myocardial infarction.
The lncRNA Braveheart (lncRNA-Bvhrt) was the first one described with a functional implication in cardiac function in mouse heart development. This lncRNA has been associated to the cardiac linage establishment during cardiac cells differentiation [68]. This role was performed through the interactions with other gene implicated in cell differentiation, SUZ12 (part of the polycomb-repressive complex 2, PRC2), pointing out to the possible role of lncRNA-Bvht in the epigenetic regulation of cardiac commitment [69]. Furthermore, it suggested the implication of lncRNA-Bvht in directing the neonatal Cardiomyocytes (CM) to a cardiac differentiation. It has been described its involvement in mouse stem cells differentiation to CMs. Other lncRNA called lncRNA-fendrr was described as a proper modulator of heart differentiation at chromatin level [70].
The implication of other ncRNA, lncRNA-HBL1 (Heart Break LncRNA1) was recently unveil in human induced pluripotent stem cells (hiPSCs) acting as a regulator of CM development. The overexpression of this lncRNA inhibit the CM differentiation from hiPSCs. This is not a direct procedure due to the mechanism of sequestration of hsa-miR-1 during the process. The deregulation of the expression of lncRNA-HBL1 has been described in ischemic heart failure and several cardiovascular diseases in mouse models and human [71]. Moreover, every cardiac pathophysiology has some lncRNAs which are different for the particular cell type related to each condition.
In contrast with other diseases, in cardiovascular diseases several lncRNAs have been described associated to their development and because of their potential use as a biomarker. In recent studies, some specific lncRNAs were associated and experimentally validated with CVDs: lncRNA ANRIL (antisense noncoding RNA located in INK4 locus), MALAT1 (metastasis associated lung adenocarcinomas transcript 1), KCNQ1OT1 (KCNQ1 overlapping transcript 1), aHIF (natural antisense transcript derived from HIF1alpha), and MIAT (myocardial infarction associated transcript). In that sense, the 9p21.3 risk locus, identified in several genome-wide association studies (GWAS) for coronary artery disease (CAD) susceptibility, is adjacent to the last exons of ANRIL which encompasses multiple SNPs [72].
The potential therapeutic application of these lncRNAs in atherosclerosis was pointed out since the expression levels of MIAT and ANRIL were higher compared with a non-atherosclerotic tissue control, as well as the expression levels of MALAT were lower than control [73].
Other classes of ncRNAs, as miRNAs, have been also linked to the development of cardiovascular diseases. Several miRNAs currently related with cardiovascular disorders were previously described as onco-miRs, in association with different cancer types, or related with other cellular functions that are not directly associated with CADs. Some miRNAs like miR-126-3p or miR-21-5p play a key role in cardiovascular diseases such as the response to ischemia process, regulating myocardial fibrosis, ventricular remodeling, arrythmia, or heart failure. In a similar manner, rs687289-A and rs532436-A are intronic variants to the ABO gene relevant to thrombosis and atherosclerosis risk. Nikpay M. et al. stated that rs532436-A and rs687289-A are associated to an increase level of miR-10b-5p, and higher circulating level of miR-10b-5p is associated with increased risk of CAD [74].
Recently, it was published the updated version of HMDD2.0 database [75]. This database included experimentally validated associations between miRNAs and different diseases, and included 165 cardiovascular-related miRNAs, spread across the human genome.
Within the definition of Cardiovascular diseases, atherosclerosis is one of the main concerns in the field. It has been defined as a multifactorial disease that involves multiple associated mechanisms, traditionally described as a chronic inflammatory and lipid disorder, and implicating diverse cell types [76]. During the study of atherosclerosis and its vascular-associated complications, different evidences have shown the involvement of ncRNAs in the development of this disease, and it is considered as other epigenetic mechanism implicated. Even more, the epigenome-wide associated studies allowed to observe the underlying implication of the epigenetic regulation of several related functions with the development of atherosclerosis, such as inflammation, lipid metabolism, and redox cellular status.
Maegdefesset and colleagues were investigating the advantages of a local delivery of some mimic miRs to increase the expression of miR-21 and miR-210 [77][78]. Replacing the lack of these miRNAs, conferred stability to the atherosclerotic plaque, decreasing the risk of atherothrombotic vascular complications. Recent studies found a useful signature of five plasmatic miRNAs (circulating miRNAs) for predicting Myocardial Infarction (MI) with a percentage of accuracy of the 81.8% in women and the 74.1% in men. This panel of circulating miRNAs was generated using the data extracted from the HUNT study [79] and included the following miRNAs: let-7g-5p, miR-106a-5p, miR-424-5p, miR-144-3p, and miR-660-5p. Other circulating miRNAs, which expression is p53-dependent, were described as a novel predictor of heart failure after suffered from MI: miR-192-5p, miR-194-5p, and miR-34a-5p [80]. General speaking, there is more than 60 miRNAs already associated with CVDs that could be used with diagnostic and prognostic purposes.
The association of another circulating lncRNAs, Zinc finger antisense 1 (ZFAS1) and CDR1 antisense (CDR1AS), has been described as differentially expressed in acute MI patients in comparison with healthy individuals [81]. The urothelial carcinoma-associated 1 (UCA1) is a lncRNA that was previously associated to bladder and lung cancer, and it was known as a predictive biomarker for its expression in this kind of cancers. Furthermore, it was described its expression in heart tissue of healthy individuals. Recent findings have shown its deregulation from early stage of acute MI until three days after the MI. Moreover, there is an inverse correlating among circulating UCA1 levels and miR-1 expression levels [82].
The presence of circRNAs in human heart tissue is directly related with the abundance of their related mRNAs. Some of the most important cardiac expressed transcript, as RYR2, DMD, and TTN genes, generate the most abundant circRNAs in heart tissue. In atherosclerosis, CDKN2B-AS1 is perhaps one of the molecularly best-studied circRNAs. According to other studies, this circular RNA from the 9p21 locus, contains a vast number of single nucleotide polymorphisms that have been linked to atherosclerotic vascular disease, as well as to type 2 diabetes mellitus (T2DM) [83].
2.2.2. Other Diseases
The purpose of this section is to include some of the remaining diseases that could obtain a benefit from the research in ncRNAs as a potential therapeutic target, as well as biomarkers for their use in diagnosis and prognosis of the different pathologies.
Type 1 diabetes is a chronic disease in which very low or absent insulin production by the pancreas has two main consequences: insulitis and pancreatic beta cells destruction. In the organism response to this kind of stress, the pro-inflammatory cytokines interferon gamma (IFNG) and interleukin 1 beta (IL1B) are produced during insulitis and enhance endoplasmic reticulum (ER) stress response and trigger the expression of BCL2-related proteins in beta cells, participating in their cell death. Grieco et al., has recently unveil the implication of the downregulation of two miRNAs associated to these facts from the same miRNA family, miR-204-5p, and miR-211-5p [84]. These miRNAs have been involved in the regulation of the transcriptional expression of some ER stress genes downstream PERK, specifically DDIT3 (CHOP, pro-apoptotic protein). These novel findings related with early type 1 diabetes point out a relationship between miRNAs, ER stress and beta cell apoptosis.
Type 2 diabetes is a form of the disease characterized by three different conditions in the human body: high levels of glucose in blood, a decrease in the amount of insulin, and an insulin resistance. This is another kind of defect of the pancreas and it is caused by an impairment between the insulin sensitivity and the insulin secretion.
The involvement of miRNAs in the production of insulin has been extensively studied. miRNAs play an important role in the regulation of the gene coding for the insulin. The expression of several miRNAs has been associated with this process. For instance, the upregulation of miR-30d directly correlated with high glucose levels. This fact has been previously described by Tang et al., enhancing the transcription of the insulin gene, and the opposite effect was observed during its inhibition [85]. Another important miRNA in the pancreas is miR-375. Its overexpression is able to inhibit the glucose-stimulate insulin expression. This miRNA achieves this effect by means of the regulation of 3′-phosphoinositide-dependent protein kinase-1 (PDK1), unveiling its important role within the pancreas [86][87][88].
The main organs depending on the glucose consumption in the organism are the liver and the skeletal muscle. Recent studies have also described the implication of other miRNAs in mechanisms associated to insulin resistance. For example, miR-29 has been described by Zhou et al., in the insulin resistance by targeting PPARδ in skeletal muscle [89]. In mice, the upregulation of miR-29 was also related with the inhibition of the gluconeogenesis. In mice liver, miR-33 regulates the expression of insulin receptor substrate 2 (IRS2), that is related with insulin resistance [90].
There are many renal diseases associated with the dysregulation of ncRNAs, specifically miRNAs, such as acute renal damage, renal cell carcinoma, diabetic nephropathy, polycystic kidney disease (PKD), and others. In the development of these diseases, it has been described an important involvement of miRNAs. Different studies were coincident in the importance of miRNAs and their use as biomarkers for the diagnosis of these renal diseases. The miR-200 family has been described as abundant in renal tissue and highly expressed [91]. Some of the components of this family has been found in a very low expression in patients suffering from IgA neuropathy, being the level of decrease of the expression considered as a prognostic factor in this disease. Moreover, the increase on the expression levels of miR-155 and miR-146a has been also correlated with a poor prognosis of the disease, at the clinical and pathophysiology level [92].
The deregulation of the expression of miRNAs can be used as a diagnostic or prognostic factor for several diseases, such as cancer. In this specific case, they could be useful in the detection of diseases related with the presence of cysts, like PKD, or associated with a process of cystogenesis in the organs with like liver, pancreas, and ovary. Thus, these miRNAs could be used as a novel biomarker for detecting renal diseases.
In the case of autoimmune diseases, there are also ncRNAs implicated at different levels. As an example, rs57095329 is located in the miR-146a promoter which confers risk of systemic lupus erythematosus (SLE). It turns out that individuals carrying the risk allele in the promoter showed lower expression levels of miR-146a [93]. Further, in SLE, the over expression of linc00513 plays a role in lupus pathogenesis by promoting IFN signalling pathway. SNP variants in the linc00513 promoter are functionally significant in regulating linc00513 expression and conferring predisposition to SLE [94].
References
- Melton, C.; Reuter, J.A.; Spacek, D.V.; Snyder, M. Recurrent somatic mutations in regulatory regions of human cancer genomes. Nat. Genet. 2015, 47, 710–716.
- Bartel, D.P. Metazoan MicroRNAs. Cell 2018, 173, 20–51.
- Batista, P.J.; Chang, H.Y. Long noncoding RNAs: Cellular address codes in development and disease. Cell 2013, 152, 1298–1307.
- Hanly, D.J.; Esteller, M.; Berdasco, M. Interplay between long non-coding RNAs and epigenetic machinery: Emerging targets in cancer? Philos. Trans. R. Soc. Lond. B Biol. Sci. 2018, 373, 20170074.
- Cech, T.R.; Steitz, J.A. The noncoding RNA revolution-trashing old rules to forge new ones. Cell 2014, 157, 77–94.
- Ishizu, H.; Siomi, H.; Siomi, M.C. Biology of PIWI-interacting RNAs: New insights into biogenesis and function inside and outside of germlines. Genes Dev. 2012, 26, 2361–2373.
- Sivagurunathan, S.; Arunachalam, J.P.; Chidambaram, S. PIWI-like protein, HIWI2 is aberrantly expressed in retinoblastoma cells and affects cell-cycle potentially through OTX2. Cell Mol. Biol. Lett. 2017, 22, 17.
- Karreth, F.A.; Tay, Y.; Perna, D.; Ala, U.; Tan, S.M.; Rust, A.G.; DeNicola, G.; Webster, K.A.; Weiss, D.; Perez-Mancera, P.A.; et al. In Vivo Identification of Tumor-Suppressive PTEN ceRNAs in an Oncogenic BRAF-Induced Mouse Model of Melanoma. Cell 2011, 147, 382–395.
- Song, C.; Zhang, J.; Qi, H.P.; Feng, C.C.; Chen, Y.P.; Cao, Y.G.; Ba, L.N.; Ai, B.; Wang, Q.Y.; Huang, W.; et al. The global view of mRNA-related ceRNA cross-talks across cardiovascular diseases. Sci. Rep. 2017, 7.
- Ghoussaini, M.; Mountjoy, E.; Carmona, M.; Peat, G.; Schmidt, E.M.; Hercules, A.; Fumis, L.; Miranda, A.; Carvalho-Silva, D.; Buniello, A.; et al. Open Targets Genetics: Systematic identification of trait-associated genes using large-scale genetics and functional genomics. Nucleic Acids Res. 2021, 49, D1311–D1320.
- Gong, J.; Tong, Y.; Zhang, H.M.; Wang, K.; Hu, T.; Shan, G.; Sun, J.; Guo, A.Y. Genome-Wide Identification of SNPs in MicroRNA Genes and the SNP Effects on MicroRNA Target Binding and Biogenesis. Hum. Mutat. 2012, 33, 254–263.
- Tanic, M.; Andres, E.; Rodriguez-Pinilla, S.M.; Marquez-Rodas, I.; Cebollero-Presmanes, M.; Fernandez, V.; Osorio, A.; Benitez, J.; Martinez-Delgado, B. MicroRNA-based molecular classification of non-BRCA1/2 hereditary breast tumours. Br. J. Cancer 2013, 109, 2724–2734.
- Freedman, M.L.; Monteiro, A.N.; Gayther, S.A.; Coetzee, G.A.; Risch, A.; Plass, C.; Casey, G.; De Biasi, M.; Carlson, C.; Duggan, D.; et al. Principles for the post-GWAS functional characterization of cancer risk loci. Nat. Genet. 2011, 43, 513–518.
- Andres-Leon, E.; Cases, I.; Alonso, S.; Rojas, A.M. Novel miRNA-mRNA interactions conserved in essential cancer pathways. Sci. Rep. 2017, 7, 46101.
- Esquela-Kerscher, A.; Slack, F.J. Oncomirs—microRNAs with a role in cancer. Nat. Rev. Cancer 2006, 6, 259–269.
- Hammond, S.M. MicroRNAs as oncogenes. Curr. Opin. Genet. Dev. 2006, 16, 4–9.
- Hossain, A.; Kuo, M.T.; Saunders, G.F. Mir-17-5p regulates breast cancer cell proliferation by inhibiting translation of AIB1 mRNA. Mol. Cell Biol. 2006, 26, 8191–8201.
- Akao, Y.; Nakagawa, Y.; Naoe, T. MicroRNAs 143 and 145 are possible common onco-microRNAs in human cancers. Oncol. Rep. 2006, 16, 845–850.
- Suzuki, H.I.; Yamagata, K.; Sugimoto, K.; Iwamoto, T.; Kato, S.; Miyazono, K. Modulation of microRNA processing by p53. Nature 2009, 460, 529–533.
- Kent, O.A.; Chivukula, R.R.; Mullendore, M.; Wentzel, E.A.; Feldmann, G.; Lee, K.H.; Liu, S.; Leach, S.D.; Maitra, A.; Mendell, J.T. Repression of the miR-143/145 cluster by oncogenic Ras initiates a tumor-promoting feed-forward pathway. Genes Dev. 2010, 24, 2754–2759.
- Yuan, F.; Sun, R.F.; Li, L.J.; Jin, B.; Wang, Y.Y.; Liang, Y.D.; Che, G.L.; Gao, L.B.; Zhang, L. A functional variant rs353292 in the flanking region of miR-143/145 contributes to the risk of colorectal cancer. Sci. Rep. 2016, 6.
- Janaszak-Jasiecka, A.; Bartoszewska, S.; Kochan, K.; Piotrowski, A.; Kalinowski, L.; Kamysz, W.; Ochocka, R.J.; Bartoszewski, R.; Collawn, J.F. miR-429 regulates the transition between Hypoxia-Inducible Factor (HIF)1A and HIF3A expression in human endothelial cells. Sci. Rep. 2016, 6, 22775.
- Lu, Y.; Zhao, X.; Liu, Q.; Li, C.; Graves-Deal, R.; Cao, Z.; Singh, B.; Franklin, J.L.; Wang, J.; Hu, H.; et al. lncRNA MIR100HG-derived miR-100 and miR-125b mediate cetuximab resistance via Wnt/beta-catenin signaling. Nat. Med. 2017, 23, 1331–1341.
- Sevignani, C.; Calin, G.A.; Nnadi, S.C.; Shimizu, M.; Davuluri, R.V.; Hyslop, T.; Demant, P.; Croce, C.M.; Siracusa, L.D. MicroRNA genes are frequently located near mouse cancer susceptibility loci. Proc. Natl. Acad. Sci. USA 2007, 104, 8017–8022.
- Suzuki, H.; Maruyama, R.; Yamamoto, E.; Kai, M. Epigenetic alteration and microRNA dysregulation in cancer. Front. Genet. 2013, 4, 258.
- O’Donnell, K.A.; Wentzel, E.A.; Zeller, K.I.; Dang, C.V.; Mendell, J.T. c-Myc-regulated microRNAs modulate E2F1 expression. Nature 2005, 435, 839–843.
- Viswanathan, S.R.; Powers, J.T.; Einhorn, W.; Hoshida, Y.; Ng, T.L.; Toffanin, S.; O’Sullivan, M.; Lu, J.; Phillips, L.A.; Lockhart, V.L.; et al. Lin28 promotes transformation and is associated with advanced human malignancies. Nat. Genet. 2009, 41, 843–848.
- Yang, Q.; Jie, Z.; Ye, S.; Li, Z.; Han, Z.; Wu, J.; Yang, C.; Jiang, Y. Genetic variations in miR-27a gene decrease mature miR-27a level and reduce gastric cancer susceptibility. Oncogene 2014, 33, 193–202.
- Hon, C.C.; Ramilowski, J.A.; Harshbarger, J.; Bertin, N.; Rackham, O.J.L.; Gough, J.; Denisenko, E.; Schmeier, S.; Poulsen, T.M.; Severin, J.; et al. An atlas of human long non-coding RNAs with accurate 5 ‘ ends. Nature 2017, 543, 199–204.
- Zhu, J.; Liu, S.; Ye, F.; Shen, Y.; Tie, Y.; Zhu, J.; Wei, L.; Jin, Y.; Fu, H.; Wu, Y.; et al. Long Noncoding RNA MEG3 Interacts with p53 Protein and Regulates Partial p53 Target Genes in Hepatoma Cells. PLoS ONE 2015, 10, e0139790.
- Zhang, J.; Yao, T.; Wang, Y.; Yu, J.; Liu, Y.; Lin, Z. Long noncoding RNA MEG3 is downregulated in cervical cancer and affects cell proliferation and apoptosis by regulating miR-21. Cancer Biol. Ther. 2016, 17, 104–113.
- Ying, L.; Huang, Y.; Chen, H.; Wang, Y.; Xia, L.; Chen, Y.; Liu, Y.; Qiu, F. Downregulated MEG3 activates autophagy and increases cell proliferation in bladder cancer. Mol. Biosyst. 2013, 9, 407–411.
- Conte, F.; Fiscon, G.; Chiara, M.; Colombo, T.; Farina, L.; Paci, P. Role of the long non-coding RNA PVT1 in the dysregulation of the ceRNA-ceRNA network in human breast cancer. PLoS ONE 2017, 12, e0171661.
- Perry, M.M.; Tsitsiou, E.; Austin, P.J.; Lindsay, M.A.; Gibeon, D.S.; Adcock, I.M.; Chung, K.F. Role of non-coding RNAs in maintaining primary airway smooth muscle cells. Respir. Res. 2014, 15, 58.
- Zhang, L.R.; Lin, J.; Ye, Y.Q.; Oba, T.; Gentile, E.; Lian, J.; Wang, J.; Zhao, Y.; Gu, J.; Wistuba, I.I.; et al. Serum MicroRNA-150 Predicts Prognosis for Early-Stage Non-Small Cell Lung Cancer and Promotes Tumor Cell Proliferation by Targeting Tumor Suppressor Gene SRCIN1. Clin. Pharmacol. Ther. 2018, 103, 1061–1073.
- Ghisi, M.; Corradin, A.; Basso, K.; Frasson, C.; Serafin, V.; Mukherjee, S.; Mussolin, L.; Ruggero, K.; Bonanno, L.; Guffanti, A.; et al. Modulation of microRNA expression in human T-cell development: Targeting of NOTCH3 by miR-150. Blood 2011, 117, 7053–7062.
- George, J.; Lim, J.S.; Jang, S.J.; Cun, Y.P.; Ozretic, L.; Kong, G.; Leenders, F.; Lu, X.; Fernandez-Cuesta, L.; Bosco, G.; et al. Comprehensive genomic profiles of small cell lung cancer. Nature 2015, 524, 47–53.
- Sun, Q.; Wang, Y.Z.; Fan, J.Y.; Li, Z.H.; Zhang, J.H.; Wang, L.J.; Fan, X.K.; Ji, M.M.; Zhu, M.; Dai, J.C.; et al. Association of expression quantitative trait loci for long noncoding RNAs with lung cancer risk in Asians. Mol. Carcinog. 2019, 58, 1303–1313.
- Siddiqi, S.; Matushansky, I. Piwis and piwi-interacting RNAs in the epigenetics of cancer. J. Cell. Biochem. 2012, 113, 373–380.
- Fu, A.; Jacobs, D.I.; Zhu, Y. Epigenome-wide analysis of piRNAs in gene-specific DNA methylation. RNA Biol. 2014, 11, 1301–1312.
- Huang, G.; Hu, H.; Xue, X.; Shen, S.; Gao, E.; Guo, G.; Shen, X.; Zhang, X. Altered expression of piRNAs and their relation with clinicopathologic features of breast cancer. Clin. Transl. Oncol. 2013, 15, 563–568.
- Jacobs, D.I.; Qin, Q.; Lerro, M.C.; Fu, A.; Dubrow, R.; Claus, E.B.; DeWan, A.T.; Wang, G.L.; Lin, H.F.; Zhu, Y. PIWI-Interacting RNAs in Gliomagenesis: Evidence from Post-GWAS and Functional Analyses. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1073–1080.
- Rupaimoole, R.; Calin, G.A.; Lopez-Berestein, G.; Sood, A.K. miRNA Deregulation in Cancer Cells and the Tumor Microenvironment. Cancer Discov. 2016, 6, 235–246.
- Guo, J.U.; Agarwal, V.; Guo, H.; Bartel, D.P. Expanded identification and characterization of mammalian circular RNAs. Genome Biol. 2014, 15, 409.
- Salzman, J.; Chen, R.E.; Olsen, M.N.; Wang, P.L.; Brown, P.O. Cell-type specific features of circular RNA expression. PLoS Genet. 2013, 9, e1003777.
- Bachmayr-Heyda, A.; Reiner, A.T.; Auer, K.; Sukhbaatar, N.; Aust, S.; Bachleitner-Hofmann, T.; Mesteri, I.; Grunt, T.W.; Zeillinger, R.; Pils, D. Correlation of circular RNA abundance with proliferation--exemplified with colorectal and ovarian cancer, idiopathic lung fibrosis, and normal human tissues. Sci. Rep. 2015, 5, 8057.
- Kadakkuzha, B.M.; Liu, X.A.; McCrate, J.; Shankar, G.; Rizzo, V.; Afinogenova, A.; Young, B.; Fallahi, M.; Carvalloza, A.C.; Raveendra, B.; et al. Transcriptome analyses of adult mouse brain reveal enrichment of lncRNAs in specific brain regions and neuronal populations. Front. Cell. Neurosci. 2015, 9, 63.
- Rybak-Wolf, A.; Stottmeister, C.; Glazar, P.; Jens, M.; Pino, N.; Giusti, S.; Hanan, M.; Behm, M.; Bartok, O.; Ashwal-Fluss, R.; et al. Circular RNAs in the Mammalian Brain Are Highly Abundant, Conserved, and Dynamically Expressed. Mol. Cell 2015, 58, 870–885.
- You, X.; Vlatkovic, I.; Babic, A.; Will, T.; Epstein, I.; Tushev, G.; Akbalik, G.; Wang, M.; Glock, C.; Quedenau, C.; et al. Neural circular RNAs are derived from synaptic genes and regulated by development and plasticity. Nat. Neurosci. 2015, 18, 603–610.
- Davis, G.M.; Haas, M.A.; Pocock, R. MicroRNAs: Not “Fine-Tuners” but Key Regulators of Neuronal Development and Function. Front. Neurol. 2015, 6, 245.
- Ng, S.Y.; Johnson, R.; Stanton, L.W. Human long non-coding RNAs promote pluripotency and neuronal differentiation by association with chromatin modifiers and transcription factors. EMBO J. 2012, 31, 522–533.
- Schratt, G. microRNAs at the synapse. Nat. Rev. Neurosci. 2009, 10, 842–849.
- Junn, E.; Lee, K.W.; Jeong, B.S.; Chan, T.W.; Im, J.Y.; Mouradian, M.M. Repression of alpha-synuclein expression and toxicity by microRNA-7. Proc. Natl. Acad. Sci. USA 2009, 106, 13052–13057.
- Bak, M.; Silahtaroglu, A.; Moller, M.; Christensen, M.; Rath, M.F.; Skryabin, B.; Tommerup, N.; Kauppinen, S. MicroRNA expression in the adult mouse central nervous system. RNA 2008, 14, 432–444.
- Ashwal-Fluss, R.; Meyer, M.; Pamudurti, N.R.; Ivanov, A.; Bartok, O.; Hanan, M.; Evantal, N.; Memczak, S.; Rajewsky, N.; Kadener, S. circRNA biogenesis competes with pre-mRNA splicing. Mol. Cell 2014, 56, 55–66.
- Elstner, M.; Morris, C.M.; Heim, K.; Lichtner, P.; Bender, A.; Mehta, D.; Schulte, C.; Sharma, M.; Hudson, G.; Goldwurm, S.; et al. Single-Cell Expression Profiling of Dopaminergic Neurons Combined with Association Analysis Identifies Pyridoxal Kinase as Parkinson’s Disease Gene. Ann. Neurol. 2009, 66, 792–798.
- Giraldez, T.; Dominguez, J.; Alvarez de la Rosa, D. ENaC in the brain--future perspectives and pharmacological implications. Curr. Mol. Pharmacol. 2013, 6, 44–49.
- Mortezaei, Z.; Lanjanian, H.; Masoudi-Nejad, A. Candidate novel long noncoding RNAs, MicroRNAs and putative drugs for Parkinson’s disease using a robust and efficient genome-wide association study. Genomics 2017, 109, 158–164.
- Liu, Y.; Chen, Y.Y.; Liu, H.; Yao, C.J.; Zhu, X.X.; Chen, D.J.; Yang, J.; Lu, Y.J.; Cao, J.Y. Association between ubiquitin carboxy-terminal hydrolase-L1 S18Y variant and risk of Parkinson’s disease: The impact of ethnicity and onset age. Neurol. Sci. 2015, 36, 179–188.
- Carrieri, C.; Cimatti, L.; Biagioli, M.; Beugnet, A.; Zucchelli, S.; Fedele, S.; Pesce, E.; Ferrer, I.; Collavin, L.; Santoro, C.; et al. Long non-coding antisense RNA controls Uchl1 translation through an embedded SINEB2 repeat. Nature 2012, 491, 454–457.
- Lukiw, W.J. Circular RNA (circRNA) in Alzheimer’s disease (AD). Front. Genet. 2013, 4, 307.
- Zhang, S.; Zhu, D.; Li, H.; Li, H.; Feng, C.; Zhang, W. Characterization of circRNA-Associated-ceRNA Networks in a Senescence-Accelerated Mouse Prone 8 Brain. Mol. Ther. 2017, 25, 2053–2061.
- Wei, C.W.; Luo, T.; Zou, S.S.; Wu, A.S. The Role of Long Noncoding RNAs in Central Nervous System and Neurodegenerative Diseases. Front. Behav. Neurosci. 2018, 12, 175.
- Massone, S.; Vassallo, I.; Fiorino, G.; Castelnuovo, M.; Barbieri, F.; Borghi, R.; Tabaton, M.; Robello, M.; Gatta, E.; Russo, C.; et al. 17A, a novel non-coding RNA, regulates GABA B alternative splicing and signaling in response to inflammatory stimuli and in Alzheimer disease. Neurobiol. Dis. 2011, 41, 308–317.
- Hebert, S.S.; De Strooper, B. Alterations of the microRNA network cause neurodegenerative disease. Trends Neurosci. 2009, 32, 199–206.
- Vilardo, E.; Barbato, C.; Ciotti, M.; Cogoni, C.; Ruberti, F. MicroRNA-101 regulates amyloid precursor protein expression in hippocampal neurons. J. Biol. Chem. 2010, 285, 18344–18351.
- Han, Z.J.; Xue, W.W.; Tao, L.; Zhu, F. Identification of Key Long Non-Coding RNAs in the Pathology of Alzheimer’s Disease and their Functions Based on Genome-Wide Associations Study, Microarray, and RNA-seq Data. J. Alzheimers Dis. 2019, 68, 339–355.
- Kataoka, M.; Huang, Z.P.; Wang, D.Z. Build a braveheart: The missing linc (RNA). Circ. Res. 2013, 112, 1532–1534.
- Hou, J.; Long, H.; Zhou, C.; Zheng, S.; Wu, H.; Guo, T.; Wu, Q.; Zhong, T.; Wang, T. Long noncoding RNA Braveheart promotes cardiogenic differentiation of mesenchymal stem cells in vitro. Stem Cell Res. Ther. 2017, 8, 4.
- Viereck, J.; Kumarswamy, R.; Foinquinos, A.; Xiao, K.; Avramopoulos, P.; Kunz, M.; Dittrich, M.; Maetzig, T.; Zimmer, K.; Remke, J.; et al. Long noncoding RNA Chast promotes cardiac remodeling. Sci. Transl. Med. 2016, 8, 326ra322.
- Stępień, E.; Costa, M.C.; Kurc, S.; Drozdz, A.; Cortez-Dias, N.; Enguita, F.J. The circulating non-coding RNA landscape for biomarker research: Lessons and prospects from cardiovascular diseases. Acta Pharmacol. Sin. 2018, 39, 1085–1099.
- Jarinova, O.; Stewart, A.F.R.; Roberts, R.; Wells, G.; Lau, P.; Naing, T.; Buerki, C.; McLean, B.W.; Cook, R.C.; Parker, J.S.; et al. Functional Analysis of the Chromosome 9p21.3 Coronary Artery Disease Risk Locus. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 1671–U1670.
- Arslan, S.; Berkan, O.; Lalem, T.; Ozbilum, N.; Goksel, S.; Korkmaz, O.; Cetin, N.; Devaux, Y.; Cardiolinc, N. Long non-coding RNAs in the atherosclerotic plaque. Atherosclerosis 2017, 266, 176–181.
- Nikpay, M.; Beehler, K.; Valsesia, A.; Hager, J.; Harper, M.E.; Dent, R.; McPherson, R. Genome-wide identification of circulating-miRNA expression quantitative trait loci reveals the role of severalmiRNAs in the regulation of cardiometabolic phenotypes. Cardiovasc. Res. 2019, 115, 1629–1645.
- Li, Y.; Qiu, C.; Tu, J.; Geng, B.; Yang, J.; Jiang, T.; Cui, Q. HMDD v2.0: A database for experimentally supported human microRNA and disease associations. Nucleic Acids Res. 2014, 42, D1070–D1074.
- Xu, S.; Kamato, D.; Little, P.J.; Nakagawa, S.; Pelisek, J.; Jin, Z.G. Targeting epigenetics and non-coding RNAs in atherosclerosis: from mechanisms to therapeutics. Pharmacol. Ther. 2019, 196, 15–43.
- Eken, S.M.; Jin, H.; Chernogubova, E.; Li, Y.; Simon, N.; Sun, C.; Korzunowicz, G.; Busch, A.; Backlund, A.; Osterholm, C.; et al. MicroRNA-210 Enhances Fibrous Cap Stability in Advanced Atherosclerotic Lesions. Circ. Res. 2017, 120, 633–644.
- Jin, H.; Li, D.Y.; Chernogubova, E.; Sun, C.; Busch, A.; Eken, S.M.; Saliba-Gustafsson, P.; Winter, H.; Winski, G.; Raaz, U.; et al. Local Delivery of miR-21 Stabilizes Fibrous Caps in Vulnerable Atherosclerotic Lesions. Mol. Ther. 2018, 26, 1040–1055.
- Bye, A.; Rosjo, H.; Nauman, J.; Silva, G.J.; Follestad, T.; Omland, T.; Wisloff, U. Circulating microRNAs predict future fatal myocardial infarction in healthy individuals—The HUNT study. J. Mol. Cell. Cardiol. 2016, 97, 162–168.
- Matsumoto, S.; Sakata, Y.; Suna, S.; Nakatani, D.; Usami, M.; Hara, M.; Kitamura, T.; Hamasaki, T.; Nanto, S.; Kawahara, Y.; et al. Circulating p53-responsive microRNAs are predictive indicators of heart failure after acute myocardial infarction. Circ. Res. 2013, 113, 322–326.
- Zhang, X.O.; Dong, R.; Zhang, Y.; Zhang, J.L.; Luo, Z.; Zhang, J.; Chen, L.L.; Yang, L. Diverse alternative back-splicing and alternative splicing landscape of circular RNAs. Genome Res. 2016, 26, 1277–1287.
- Yan, Y.; Zhang, B.; Liu, N.; Qi, C.; Xiao, Y.; Tian, X.; Li, T.; Liu, B. Circulating Long Noncoding RNA UCA1 as a Novel Biomarker of Acute Myocardial Infarction. Biomed. Res. Int. 2016, 2016, 8079372.
- Du, W.W.; Zhang, C.; Yang, W.N.; Yong, T.Q.; Awan, F.M.; Yang, B.B. Identifying and Characterizing circRNA-Protein Interaction. Theranostics 2017, 7, 4183–4191.
- Grieco, F.A.; Schiavo, A.A.; Brozzi, F.; Juan-Mateu, J.; Bugliani, M.; Marchetti, P.; Eizirik, D. The microRNAs miR-211-5p and miR-204-5p modulate ER stress in human beta cells. J. Mol. Endocrinol. 2019, 63, 139–149.
- Zhao, X.; Mohan, R.; Ozcan, S.; Tang, X. MicroRNA-30d induces insulin transcription factor MafA and insulin production by targeting mitogen-activated protein 4 kinase 4 (MAP4K4) in pancreatic beta-cells. J. Biol. Chem. 2012, 287, 31155–31164.
- El Ouaamari, A.; Baroukh, N.; Martens, G.A.; Lebrun, P.; Pipeleers, D.; van Obberghen, E. miR-375 targets 3′-phosphoinositide-dependent protein kinase-1 and regulates glucose-induced biological responses in pancreatic beta-cells. Diabetes 2008, 57, 2708–2717.
- Hu, S.; Zhang, M.; Sun, F.; Ren, L.; He, X.; Hua, J.; Peng, S. miR-375 controls porcine pancreatic stem cell fate by targeting 3-phosphoinositide-dependent protein kinase-1 (Pdk1). Cell. Prolif. 2016, 49, 395–406.
- Poy, M.N.; Eliasson, L.; Krutzfeldt, J.; Kuwajima, S.; Ma, X.; Macdonald, P.E.; Pfeffer, S.; Tuschl, T.; Rajewsky, N.; Rorsman, P.; et al. A pancreatic islet-specific microRNA regulates insulin secretion. Nature 2004, 432, 226–230.
- Zhou, Y.; Gu, P.; Shi, W.; Li, J.; Hao, Q.; Cao, X.; Lu, Q.; Zeng, Y. MicroRNA-29a induces insulin resistance by targeting PPARdelta in skeletal muscle cells. Int. J. Mol. Med. 2016, 37, 931–938.
- Yang, W.M.; Jeong, H.J.; Park, S.Y.; Lee, W. Induction of miR-29a by saturated fatty acids impairs insulin signaling and glucose uptake through translational repression of IRS-1 in myocytes. FEBS Lett. 2014, 588, 2170–2176.
- Patel, V.; Hajarnis, S.; Williams, D.; Hunter, R.; Huynh, D.; Igarashi, P. MicroRNAs regulate renal tubule maturation through modulation of Pkd1. J. Am. Soc. Nephrol. 2012, 23, 1941–1948.
- Wang, G.; Kwan, B.C.; Lai, F.M.; Chow, K.M.; Li, P.K.; Szeto, C.C. Elevated levels of miR-146a and miR-155 in kidney biopsy and urine from patients with IgA nephropathy. Dis. Markers 2011, 30, 171–179.
- Luo, X.B.; Yang, W.L.; Ye, D.Q.; Cui, H.J.; Zhang, Y.; Hirankarn, N.; Qian, X.X.; Tang, Y.J.; Lau, Y.L.; de Vries, N.; et al. A Functional Variant in MicroRNA-146a Promoter Modulates Its Expression and Confers Disease Risk for Systemic Lupus Erythematosus. Plos Genet. 2011, 7.
- Xue, Z.X.; Cui, C.J.; Liao, Z.J.; Xia, S.W.; Zhang, P.J.; Qin, J.L.; Guo, Q.; Chen, S.; Fu, Q.; Yin, Z.H.; et al. Identification of LncRNA Linc00513 Containing Lupus-Associated Genetic Variants as a Novel Regulator of Interferon Signaling Pathway. Front. Immunol. 2018, 9.
