Suprachoroidal drug delivery technology has advanced rapidly and emerged as a promising administration route for a variety of therapeutic candidates, targeting multiple ocular diseases, ranging from neovascular age-related macular degeneration to choroidal melanoma. This entry summarizes the latest preclinical and clinical progress in suprachoroidal delivery of therapeutic agents, focusing on small molecule suspensions.
- suprachoroidal
- microinjector
- SCS
1. Current Treatment Landscape and Unmet Clinical Need
Highly active research and development efforts are ongoing to optimize ocular drug delivery for potential improvement in efficacy, safety, and durability benefits. The most common ocular drug delivery method for treating posterior eye diseases is intravitreal (IVT) injections [1][2][3][4]. It is estimated that 24.4 million IVT injections were performed globally in 2019 with 6.9 million of these injections performed in the United States, most often for neovascular age-related macular degeneration (nAMD) and diabetic macular edema (DME) [5]. While IVT injections of anti-vascular endothelial growth factor (anti-VEGF) therapies are commonly performed in an office setting, and have demonstrated remarkable therapeutic benefits in controlled clinical trials [6][7][8][9][10][11][12][13][14][15][16][17], real world data on patients receiving IVT injections of anti-VEGF therapies show a much more modest improvement in patients’ vision [18][19][20][21][22]. This disparity may relate to treatment burden, with patients undertreated due to the inability to maintain the frequent treatments in fixed regimens utilized in clinical trials. Furthermore, a subset of patients responds incompletely to anti-VEGF treatments regardless of the frequency of administration [23]. Other pharmacological agents such as corticosteroids, that are commonly injected intravitreally for posterior eye diseases, may have undesirable side effects, including ocular hypertension and/or cataracts, due to their anterior chamber exposure [24][25][26][27][28][29][30][31].
In addition to posterior eye diseases that are currently treated with IVT injections, there remain significant unmet clinical needs in other ocular disorders, such as glaucoma, that are currently treated topically with eye drops and/or surgery. Glaucoma patients face challenges including compliance with proper administration of topical eye drops of beta blockers (e.g., timolol), alpha agonists (e.g., brimonidine), Rho kinase inhibitors (e.g., netarsudil), and prostaglandin analogs (e.g., latanoprost) [32][33][34]. These drugs act by decreasing the production and/or increasing the outflow of the aqueous humor. In some instances, combination treatments may also be required to provide adequate treatment. Specifically related to topical eye drops, in addition to patient compliance, there are challenges maintaining drug concentrations above therapeutic levels due to the high clearance rate via nasolacrimal drainage and/or dilution from production of reflex tears [35][36]. Taken together, there remains an unmet clinical need for improved delivery to targeted ocular tissues, compared to current topical or intravitreal delivery methods.
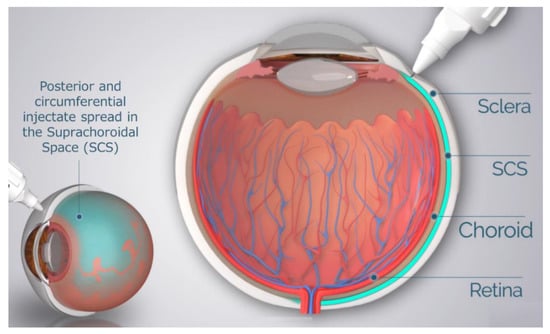
One possible solution to address this unmet need is the delivery of therapeutic agents into the suprachoroidal space (SCS). The SCS is the ‘potential space’ between the sclera and choroid that circumferentially spans the entire posterior segment of the eye from the ciliary body rearwards. Potential spaces in the body are areas between directly apposed organs or tissue layers. These potential spaces, such as the pleural space, the pericardial cavity, and the SCS, expand when fluid enters and flows within the space and collapse upon fluid egress. These potential spaces can serve as “druggable” targets for the delivery of therapeutic agents.
Drug delivery to the SCS has unique potential advantages in that, (1) it specifically targets affected chorioretinal tissues with posterior and circumferential spread of the drug administered, (2) it may provide sustained drug kinetic release profiles, depending on the physicochemical formulation properties, and (3) it may spare the unaffected anterior segment of the eye and the vitreous chamber, thus minimizing risks associated with off-target effects to potentiate safety [37][38]. To reliably access the SCS, microneedles have been designed to be long enough to penetrate through the sclera, delivering therapeutic agents into the SCS, without penetrating into the vitreous. The therapeutic agent flows both circumferentially and posteriorly towards the back of the eye, targeting chorioretinal tissues (
). In recent years, multiple clinical trials have evaluated this technique of suprachoroidal (SC) delivery with various therapeutic agents ranging from small molecule suspensions to viral vector therapies.
Schematic of Microneedle Injection into the Suprachoroidal Space (SCS).
Drug delivery into the SCS has been achieved in multiple ways, in addition to the microneedle method, the predominant technique clinically utilized. Small case series have also been performed by creating a scleral flap and a suprachoroidal (SC) pocket to insert autologous tissue. Preclinically, SC injections have also been achieved using either the scleral flap technique, standard hypodermic needles or catheters (
).
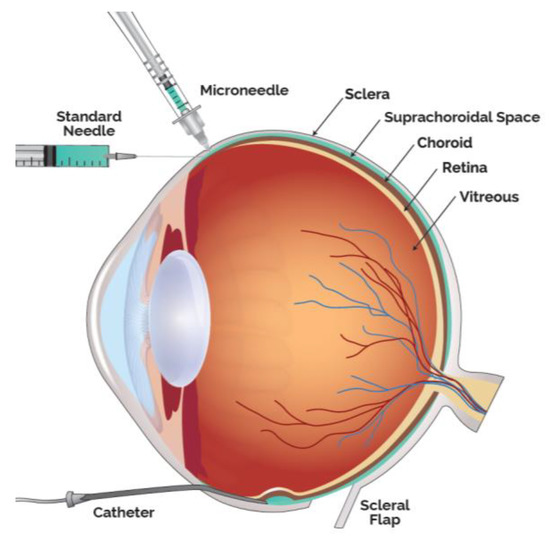
Modalities to Administer Therapeutic Agents into the Suprachoroidal Space (SCS).
The objective of this review article is to summarize the current status of preclinical and clinical therapeutic agents that have been administered into the SCS. As described in this review article, SC administration may offer unique advantages, such as compartmentalization away from the anterior segment and prolonged durability with appropriate formulations. Clinical trials with suprachoroidally administered triamcinolone acetonide, a small molecule suspension, have already demonstrated safety and efficacy. This route of administration may further play a significant role in ocular drug delivery of other novel therapeutic agents for a wide range of ocular diseases, spanning from glaucoma and ocular melanoma to several common chorioretinal diseases, such as age-related macular degeneration (AMD) and diabetic retinopathy (DR).
2. Small Molecule Suspensions
Introduction of low-solubility therapeutic agents, such as small molecules in suspension, into the SCS is one strategy employed to promote both improved pharmacokinetic (PK) profiles and targeted drug delivery. Several well-characterized therapeutic agents, compounded as small molecule suspensions, have been administered suprachoroidally.
2.1. Corticosteroid
Safety and efficacy of an SC-delivered investigational formulation of triamcinolone acetonide (TA) has been evaluated in multiple controlled clinical trials [39][40][41][42][43] and its pharmacokinetic and pharmacodynamic characteristics have also evaluated in multiple animal studies.
Preclinically, favorable pharmacokinetics of SC TA has been demonstrated. This commonly used corticosteroid is a crystalline small molecule with low aqueous solubility (ranging between 0.02 mg/mL at 28 °C and 0.03 mg/mL at 50 °C [44]) and has historically been administered via topical, periocular or intravitreal routes, for the treatment of ocular inflammation. At a particle size distribution in the low micrometer range after micronization, it has been demonstrated that the concentration of TA in the SCS in various preclinical models can be maintained above therapeutic levels for an extended period of time [45].
With respect to ocular distribution, when TA was injected into the SCS in rabbits, TA concentrations in the retinal pigment epithelium (RPE)-choroid-sclera (RCS) and the retina were significantly higher than those in both the aqueous humor and the vitreous through the duration of the study (91 days) [46]. This study illustrated that drug delivery into the SCS can be compartmentalized, away from the anterior segment and the vitreous humor, preferentially targeting chorioretinal tissues and provide sustained PK profile.
Pharmacodynamically, efficacy of SC TA was supported in a porcine model of uveitis. In this model, lipopolysaccharides (LPS) were introduced into pigs to induce uveitis [47]. Suprachoroidal administration of TA meaningfully reduced inflammation over time. In an in vivo model, Chen et al. showed that TA delivered via the SC route provided excellent targeting to the posterior retina; resulting in improved efficacy with significantly fewer aqueous humor cells and lower vitreous opacity scores compared to TA administered via the sub-Tenon route in rabbits [48]. Similar studies have also shown that TA administered via the SC or IVT routes in a model of endotoxin-induced panuveitis in albino rabbits led to comparable efficacy in the reduction of ocular inflammation [49]. Furthermore, an in vivo porcine model of acute uveitis demonstrated that 1/10th the dose of TA administered suprachoroidally was as effective as the full dose administered intravitreally, with no adverse effects [47]. Using a similar porcine model, another study showed that TA administered suprachoroidally was more effective in reducing ocular inflammation than low dose oral prednisone (0.1 mg/kg/day) administered for three days, and resulted in an improved rate of reduction in inflammation when compared to high dose oral prednisone (1 mg/kg/day) [50]. These results validate SC administration as a potential drug delivery route for small molecule suspensions.
These preclinical study results translated well into clinical trial outcomes. Specifically, in a phase 3 double-masked and randomized clinical trial for macula edema associated with noninfectious uveitis, 46.9% of the subjects treated with two SC injections of TA administered 12 weeks apart gained at least 15 Early Treatment Diabetic Retinopathy Study (ETDRS) letters from baseline in best corrected visual acuity (BCVA) at 24 weeks, compared to 15.6% in the sham control group [39] (
).
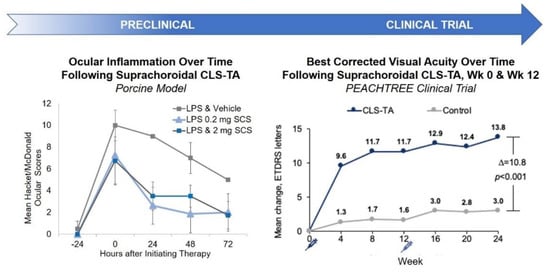
Preclinical and Clinical Results of SC Administration of Small Molecule Suspensions. CLS-TA: investigational formulation of triamcinolone acetonide developed by Clearside Biomedical (Alpharetta, GA, USA); LPS: lipopolysaccharides.
2.2. Tyrosine Kinase Inhibitor
Another small molecule suspension that has been evaluated for potential SC delivery is axitinib. Axitinib has several properties with attractive therapeutic potential. It is a highly potent tyrosine kinase inhibitor (TKI) with pan-VEGF inhibition and high binding affinities [51]. Pan-VEGF inhibition may have benefits over current specific VEGF-A inhibition for the treatment of nAMD, DR, and DME, as other VEGF ligands such as VEGF-C and VEGF-D, have been shown to be upregulated after anti-VEGF-A administration both locally, after the treatment of nAMD, and systemically [52][53]. This upregulation of other VEGF ligands could contribute to tachyphylaxis, a form of treatment resistance, and lead to refractory cases clinically. Axitinib has also been shown to more effectively inhibit angiogenic sprouts than anti-VEGF-A inhibition in an in vitro angiogenesis model [54]. One recent phase 2 clinical trial demonstrated that broad VEGF inhibition yielded a statistically significantly better visual outcome in nAMD than focused VEGF-A inhibition [55]. In addition, axitinib is a more highly potent TKI than others that have undergone assessment in ocular clinical trials [56] and has demonstrated more potent inhibition of murine corneal neovascularization, compared to other TKIs at the same dose [57]. Axitinib has been shown to not only inhibit angiogenesis, but also regress established neovascularization in a preclinical choroidal neovascularization model, which may be more relevant to potential clinical use [58]. Finally, axitinib has shown better biocompatibility with ocular cells, including retinal pigment epithelial cells, compared to other TKIs in an in vitro study, suggesting the potential for intrinsic safety benefits [59].
While axitinib, a poorly water-soluble small molecule (molecular weight: ~387 g/mol, solubility: 0.2 µg/mL in neutral pH aqueous media [60]), may not be well-suited for many forms of ocular delivery, it can be injected into the SCS as a stable suspension. In a rabbit PK and ocular tolerability study, a single bilateral SC injection of an axitinib suspension at 2 different doses (either 0.03 or 0.1 mg per eye) was administered to Dutch-Belted rabbits [61]. Axitinib was well-tolerated and optical coherence tomography (OCT) showed no evidence of choroidal/retinal degeneration through the duration of the study (10 weeks). More importantly, efficacious and sustained levels of axitinib, above the in vitro IC
for the VEGF 2 receptor, were observed in the posterior ocular tissues—specifically in the RCS and retina—throughout the duration of the study, while no axitinib was detected in the plasma or aqueous humor. Furthermore, when compared to an equivalent dose of IVT axitinib in this rabbit model, SC axitinib resulted in RCS levels that were 11 times greater, supporting the potential for favorable targeting of chorioretinal tissues, which are affected in common causes of vision loss, such as AMD and DME. In a separate rabbit PK and ocular tolerability study, a single SC axitinib suspension was administered at a dose of 4 mg/eye. Over the duration of this 91-day study, axitinib was quantifiable at all timepoints in the RCS, retina and the vitreous humor with the highest concentration detected in the RCS. The administered dose was generally well tolerated. Currently, a phase 1/2a clinical trial of suprachoroidally-administered axitinib, for the treatment of nAMD, is underway [62].
2.3. Complement Inhibitor & Plasma Kallikrein Inhibitor
Additional small molecules have been investigated to target other signaling pathways, such as the complement system, a potential treatment target for non-neovascular age-related macular degeneration (dry AMD). Suprachoroidally administered A01017 (Achillion Pharamaceuticals, Blue Bell, PA, USA now AstraZeneca, Cambridge, UK), a potent small molecule complement factor D inhibitor [63], was well tolerated with sustained drug levels in posterior segment tissue of rabbits for up to 92 days. Similar to TA and axitinib, a high level of A01017 was observed in the RCS and retina throughout the entire duration of the study with an estimated half-life of approximately 66–76 days. Low or no quantifiable A01017 was detected in the vitreous humor, aqueous humor or plasma.
Yet another example of a small molecule suspension that has demonstrated preclinical ocular tolerability and sustained ocular drug levels is BCX4161 (BioCryst Pharmaceuticals, Durham, NC, USA), a potent and selective inhibitor of human plasma kallikrein, that is elevated in patients with diabetic macular edema [64]. BCX4161 was suprachoroidally administered to Dutch-Belted rabbits and was found to be well tolerated with sustained levels detected throughout the RCS and both central and peripheral retina over the 12-week study duration. Furthermore, the concentration of BCX4161 in the retina was 1 to 2 orders of magnitude higher than levels in the vitreous humor.
Collectively, these study results demonstrate that insoluble small molecule suspensions delivered via the SC route result in targeted high levels in chorioretinal tissues with potential efficacy benefits, compartmentalization away from unaffected tissues for potential safety benefits, and durability to potentially relieve treatment burden compared to current IVT therapies (
).
Summary of Preclinical and Clinical Investigations of Small Molecule Suspensions Administered into the Suprachoroidal Space.
| Therapeutic Agent | Therapeutic Class | Disease | Phase (Species) | Duration | Key Results | Ref |
|---|---|---|---|---|---|---|
| Triamcinolone Acetonide (TA) Suspensions | Corticosteroid | Macula Edema Associated with Uveitis | Phase 3 Clinical Study | 24 weeks | In a phase 3 clinical trial for macula edema associated with noninfectious uveitis, 46.9% of the subjects treated with 2 SC injections of TA gained at least 15 ETDRS letters from baseline at 24 weeks, compared to 15.6% in the sham group | [39] |
| Axitinib Suspensions | Tyrosine kinase inhibitor | nAMD | Preclinical (Rabbit) |
10 weeks | Efficacious and sustained level of axitinib, above IC50, was observed in the posterior ocular tissues throughout the duration of the study | [61] |
| A01017 | Complement inhibitor | non-neovascular AMD | Preclinical (Rabbit) |
92 days | High level of A01017 was observed in the RCS and retinal tissues throughout the entire duration of the study. | [63] |
| BCX4161 | Plasma kallikrein inhibitor | DME | Preclinical (Rabbit) |
12 weeks | BCX4161 was well tolerated and sustained levels were observed in the RCS, peripheral and central retinal tissues throughout the duration of the study | [64] |
References
- Patel, A. Ocular drug delivery systems: An overview. World J. Pharmacol. 2013, 2, 47.
- Edelhauser, H.F.; Rowe-Rendleman, C.L.; Robinson, M.R.; Dawson, D.G.; Chader, G.J.; Grossniklaus, H.E.; Rittenhouse, K.D.; Wilson, C.G.; Weber, D.A.; Kuppermann, B.D.; et al. Ophthalmic drug delivery systems for the treatment of retinal diseases: Basic research to clinical applications. Investig. Ophthalmol. Vis. Sci. 2010, 51, 5403–5420.
- Nayak, K.; Misra, M. A review on recent drug delivery systems for posterior segment of eye. Biomed. Pharmacother. 2018, 107, 1564–1582.
- Varela-Fernández, R.; Díaz-Tomé, V.; Luaces-Rodríguez, A.; Conde-Penedo, A.; García-Otero, X.; Luzardo-álvarez, A.; Fernández-Ferreiro, A.; Otero-Espinar, F.J. Drug delivery to the posterior segment of the eye: Biopharmaceutic and pharmacokinetic considerations. Pharmaceutics 2020, 12.
- Puliafito, C.A.; Wykoff, C.C. New Frontiers in Retina: Highlights of the 2020 angiogenesis, exudation and degeneration symposium. Int. J. Retin. Vitr. 2020, 6.
- Korobelnik, J.F.; Holz, F.G.; Roider, J.; Ogura, Y.; Simader, C.; Schmidt-Erfurth, U.; Lorenz, K.; Honda, M.; Vitti, R.; Berliner, A.J.; et al. Intravitreal aflibercept injection for macular edema resulting from central retinal vein occlusion: One-year results of the phase 3 GALILEO study. Ophthalmology 2014, 121, 202–208.
- Heier, J.S.; Clark, W.L.; Boyer, D.S.; Brown, D.M.; Vitti, R.; Berliner, A.J.; Kazmi, H.; Ma, Y.; Stemper, B.; Zeitz, O.; et al. Intravitreal aflibercept injection for macular edema due to central retinal vein occlusion: Two-year results from the COPERNICUS study. Ophthalmology 2014, 121, 1–2.
- Clark, W.L.; Boyer, D.S.; Heier, J.S.; Brown, D.M.; Haller, J.A.; Vitti, R.; Kazmi, H.; Berliner, A.J.; Erickson, K.; Chu, K.W.; et al. Intravitreal Aflibercept for Macular Edema Following Branch Retinal Vein Occlusion 52-Week Results of the VIBRANT Study. Ophthalmology 2016, 123, 330–336.
- Campochiaro, P.A.; Brown, D.M.; Pearson, A.; Chen, S.; Boyer, D.; Ruiz-Moreno, J.; Garretson, B.; Gupta, A.; Hariprasad, S.M.; Bailey, C.; et al. Sustained delivery fluocinolone acetonide vitreous inserts provide benefit for at least 3 years in patients with diabetic macular edema. Ophthalmology 2012, 119, 2125–2132.
- Heier, J.S.; Korobelnik, J.-F.; Brown, D.M.; Schmidt-Erfurth, U.; Do, D.V.; Midena, E.; Boyer, D.S.; Terasaki, H.; Kaiser, P.K.; Marcus, D.M.; et al. Intravitreal Aflibercept for Diabetic Macular Edema: 148-Week Results from the VISTA and VIVID Studies. Ophthalmology 2016, 123, 2376–2385.
- Brown, D.M.; Kaiser, P.K.; Michels, M.; Soubrane, G.; Heier, J.S.; Kim, R.Y.; Sy, J.P.; Schneider, S. Ranibizumab versus Verteporfin for Neovascular Age-Related Macular Degeneration. N. Engl. J. Med. 2006, 355, 1432–1444.
- Boyer, D.S.; Yoon, Y.H.; Belfort, R.; Bandello, F.; Maturi, R.K.; Augustin, A.J.; Li, X.Y.; Cui, H.; Hashad, Y.; Whitcup, S.M. Three-year, randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with diabetic macular edema. Ophthalmology 2014, 121, 1904–1914.
- Nguyen, Q.D.; Brown, D.M.; Marcus, D.M.; Boyer, D.S.; Patel, S.; Feiner, L.; Gibson, A.; Sy, J.; Rundle, A.C.; Hopkins, J.J.; et al. Ranibizumab for diabetic macular edema: Results from 2 phase iii randomized trials: RISE and RIDE. Ophthalmology 2012, 119, 789–801.
- Martin, D.F.; Maguire, M.G.; Ying, G.S.; Grunwald, J.E.; Fine, S.L.; Jaffe, G.J. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 2011, 364, 1897–1908.
- Varma, R.; Bressler, N.M.; Suñer, I.; Lee, P.; Dolan, C.M.; Ward, J.; Colman, S.; Rubio, R.G. Improved vision-related function after ranibizumab for macular edema after retinal vein occlusion: Results from the BRAVO and CRUISE trials. Ophthalmology 2012, 119, 2108–2118.
- Rosenfeld, P.J.; Brown, D.M.; Heier, J.S.; Boyer, D.S.; Kaiser, P.K.; Chung, C.Y.; Kim, R.Y. Ranibizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 2006, 355, 1419–1431.
- Diabetic Retinopathy Clinical Research Network; Wells, J.; Glassman, A.R.; Ayala, A.R.; Jampol, L.M.; Aiello, L.; Antoszyk, A.N.; Arnold-Bush, B.; Baker, C.; Bressler, N.M.; et al. Aflibercept, Bevacizumab, or Ranibizumab for Diabetic Macular Edema. N. Engl. J. Med. 2015, 372, 1193–1203.
- Ciulla, T.A.; Bracha, P.; Pollack, J.; Williams, D.F. Real-world Outcomes of Anti-Vascular Endothelial Growth Factor Therapy in Diabetic Macular Edema in the United States. Ophthalmol. Retin. 2018, 2, 1179–1187.
- Ciulla, T.A.; Hussain, R.M.; Pollack, J.S.; Williams, D.F. Visual Acuity Outcomes and Anti–Vascular Endothelial Growth Factor Therapy Intensity in Neovascular Age-Related Macular Degeneration Patients: A Real-World Analysis of 49 485 Eyes. Ophthalmol. Retin. 2020, 4, 19–30.
- Ciulla, T.A.; Huang, F.; Westby, K.; Williams, D.F.; Zaveri, S.; Patel, S.C. Real-world Outcomes of Anti-Vascular Endothelial Growth Factor Therapy in Neovascular Age-Related Macular Degeneration in the United States. Ophthalmol. Retin. 2018, 2, 645–653.
- Ciulla, T.; Pollack, J.S.; Williams, D.F. Visual acuity outcomes and anti-VEGF therapy intensity in macular oedema due to retinal vein occlusion: A real-world analysis of 15 613 patient eyes. Br. J. Ophthalmol. 2020.
- Ciulla, T.A.; Pollack, J.S.; Williams, D.F. Visual acuity outcomes and anti-VEGF therapy intensity in diabetic macular oedema: A real-world analysis of 28 658 patient eyes. Br. J. Ophthalmol. 2020.
- Amoaku, W.M.; Chakravarthy, U.; Gale, R.; Gavin, M.; Ghanchi, F.; Gibson, J.; Harding, S.; Johnston, R.L.; Kelly, S.; Lotery, A.; et al. Defining response to anti-VEGF therapies in neovascular AMD. Eye 2015, 29, 721–731.
- Kempen, J.H.; Altaweel, M.M.; Holbrook, J.T.; Jabs, D.A.; Louis, T.A.; Sugar, E.A.; Thorne, J.E. Randomized comparison of systemic anti-inflammatory therapy versus fluocinolone acetonide implant for intermediate, posterior, and panuveitis: The multicenter uveitis steroid treatment trial. Ophthalmology 2011, 118, 1916–1926.
- Yap, Y.C.; Papathomas, T.; Kamal, A. Results of intravitreal dexamethasone implant 0.7 mg (Ozurdex®) in non-infectious posterior uveitis. Int. J. Ophthalmol. 2015, 8, 835–838.
- Kempen, J.H.; Jabs, D.A. Benefits of systemic anti-inflammatory therapy versus fluocinolone acetonide intraocular implant for intermediate uveitis, posterior uveitis, and panuveitis: Fifty-four-month results of the Multicenter Uveitis Steroid Treatment (MUST) Trial and Follow-up Study. Ophthalmology 2015, 122, 1967–1975.
- Cho, H.Y.; Kang, S.W.; Kim, Y.T.; Chung, S.E.; Lee, S.W. A three-year follow-up of intravitreal triamcinolone acetonide injection and macular laser photocoagulation for diffuse diabetic macular edema. Korean J. Ophthalmol. 2012, 26, 362–368.
- Yang, Y.; Bailey, C.; Loewenstein, A.; Massin, P. Intravitreal corticosteroids in diabetic macular edema: Pharmacokinetic considerations. Retina 2015, 35, 2440–2449.
- Sen, H.N.; Vitale, S.; Gangaputra, S.S.; Nussenblatt, R.B.; Liesegang, T.L.; Levy-Clarke, G.A.; Rosenbaum, J.T.; Suhler, E.B.; Thorne, J.E.; Foster, C.S.; et al. Periocular corticosteroid injections in uveitis: Effects and complications. Ophthalmology 2014, 121, 2275–2286.
- Kothari, S.; Foster, C.S.; Pistilli, M.; Liesegang, T.L.; Daniel, E.; Sen, H.N.; Suhler, E.B.; Thorne, J.E.; Jabs, D.A.; Levy-Clarke, G.A.; et al. The risk of intraocular pressure elevation in pediatric noninfectious uveitis. Ophthalmology 2015, 122, 1987–2001.
- James, E.R. The etiology of steroid cataract. J. Ocul. Pharmacol. Ther. 2007, 23, 403–420.
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The pathophysiology and treatment of glaucoma: A review. JAMA - J. Am. Med. Assoc. 2014, 311, 1901–1911.
- Burns, E.; Mulley, G.P. Practical problems with eye-drops among elderly ophthalmology outpatients. Age Ageing 1992, 21, 168–170.
- Gurwitz, J.H.; Glynn, R.J.; Monane, M.; Everitt, D.E.; Gilden, D.; Smith, N.; Avorn, J. Treatment for glaucoma: Adherence by the elderly. Am. J. Public Health 1993, 83, 711–716.
- Djebli, N.; Khier, S.; Griguer, F.; Coutant, A.L.; Tavernier, A.; Fabre, G.; Leriche, C.; Fabre, D. Ocular Drug Distribution After Topical Administration: Population Pharmacokinetic Model in Rabbits. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 59–68.
- Sakanaka, K.; Kawazu, K.; Tomonari, M.; Kitahara, T.; Nakashima, M.; Nishida, K.; Nakamura, J.; Sasaki, H.; Higuchi, S. Ocular pharmacokinetic/pharmacodynamic modeling for multiple anti-glaucoma drugs. Biol. Pharm. Bull. 2008, 31, 1590–1595.
- Goldstein, D.A.; Ciulla, T.A. Suprachoroidal Delivery of Suspensions of Tyrosine Kinase Inhibitor, Complement Inhibitor, and Corticosteroid: Preclinical and Clinical Correlates | IOVS | ARVO Journals. Invest. Ophthalmol. Vis. Sci. 2020, 61, 2898.
- Habot-Wilner, Z.; Noronha, G.; Wykoff, C.C. Suprachoroidally injected pharmacological agents for the treatment of chorio-retinal diseases: A targeted approach. Acta Ophthalmol. 2019, 97, 460–472.
- Yeh, S.; Khurana, R.N.; Shah, M.; Henry, C.R.; Wang, R.C.; Kissner, J.M.; Ciulla, T.A.; Noronha, G. Efficacy and Safety of Suprachoroidal CLS-TA for Macular Edema Secondary to Noninfectious Uveitis: Phase 3 Randomized Trial. Ophthalmology 2020, 127, 948–955.
- Yeh, S.; Kurup, S.K.; Wang, R.C.; Foster, C.S.; Noronha, G.; Nguyen, Q.D.; Do, D.V. Suprachoroidal Injection of Triamcinolone Acetonide, Cls-Ta, for Macular Edema due to Noninfectious Uveitis: A Randomized, Phase 2 Study (DOGWOOD). Retina 2019, 39, 1880–1888.
- Campochiaro, P.A.; Wykoff, C.C.; Brown, D.M.; Boyer, D.S.; Barakat, M.; Taraborelli, D.; Noronha, G. Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occlusion: Results of the Tanzanite Study. Ophthalmol. Retin. 2018, 2, 320–328.
- Barakat, M.R.; Wykoff, C.C.; Gonzalez, V.; Hu, A.; Marcus, D.; Zavaleta, E.; Ciulla, T.A. Suprachoroidal CLS-TA plus Intravitreal Aflibercept for Diabetic Macular Edema: A Randomized, Double-Masked, Parallel-Design, Controlled Study. Ophthalmol. Retin. 2021, 5, 60–70.
- Henry, C.R.; Shah, M.; Barakat, M.R.; Dayani, P.; Wang, R.C.; Khurana, R.N.; Rifkin, L.; Yeh, S.; Hall, C.; Ciulla, T. Suprachoroidal CLS-TA for non-infectious uveitis: An open-label, safety trial (AZALEA). Br. J. Ophthalmol. 2021.
- Block, L.H.; Patel, R.N. Solubility and Dissolution of Triamcinolone Acetonide. J. Pharm. Sci. 1973, 62, 617–621.
- Edelhauser, H.F.; Patel, S.R.; Meschter, C.; Dean, R.; Powell, K.; Verhoeven, R. Suprachoroidal Microinjection Delivers Triamcinolone Acetonide to Therapeutically-Relevant Posterior Ocular Structures and Limits Exposure in the Anterior Segment. Invest. Ophthalmol. Vis. Sci. 2013, 54, 5063.
- Edelhauser, H.F.; Verhoeven, R.S.; Burke, B.; Struble, C.B.; Patel, S.R. Intraocular Distribution and Targeting of Triamcinolone Acetonide Suspension Administered Into the Suprachoroidal Space. Invest. Ophthalmol. Vis. Sci. 2014, 55, 5259.
- Gilger, B.C.; Abarca, E.M.; Salmon, J.H.; Patel, S. Treatment of acute posterior uveitis in a porcine model by injection of triamcinolone acetonide into the suprachoroidal space using microneedles. Invest. Ophthalmol. Vis. Sci. 2013, 54, 2483–2492.
- Chen, M.; Li, X.; Liu, J.; Han, Y.; Cheng, L. Safety and pharmacodynamics of suprachoroidal injection of triamcinolone acetonide as a controlled ocular drug release model. J. Control. Release 2015, 203, 109–117.
- Patel, S.; Carvalho, R.; Mundwiler, K.; Meschter, C.; Verhoeven, R. Evaluation of Suprachoroidal Microinjection of Triamcinolone Acetonide in a Model of Panuveitis in Albino Rabbits. Invest. Ophthalmol. Vis. Sci. 2013, 54, 2927.
- Noronha, G.; Blackwell, K.; Gilger, B.C.; Kissner, J.; Patel, S.R.; Walsh, K.T. Evaluation of suprachoroidal CLS-TA and oral prednisone in a porcine model of uveitis. Invest. Ophthalmol. Vis. Sci. 2015, 56, 3110.
- Lledó Riquelme, M.; Campos-Mollo, E.; Fernández-Sánchez, L. Topical axitinib is a potent inhibitor of corneal neovascularization. Clin. Experiment. Ophthalmol. 2018, 46, 1063–1074.
- Lieu, C.H.; Tran, H.; Jiang, Z.-Q.; Mao, M.; Overman, M.J.; Lin, E.; Eng, C.; Morris, J.; Ellis, L.; Heymach, J.V.; et al. The Association of Alternate VEGF Ligands with Resistance to Anti-VEGF Therapy in Metastatic Colorectal Cancer. PLoS ONE 2013, 8, e77117.
- Cabral, T.; Lima, L.H.; Luiz, L.G.; Polido, J.; Correa, É.P.; Oshima, A.; Duong, J.; Serracarbassa, P.; Regatieri, C.V.; Mahajan, V.B.; et al. Bevacizumab Injection in Patients with Neovascular Age-Related Macular Degeneration Increases Angiogenic Biomarkers. Ophthalmol. Retin. 2018, 2, 31–37.
- Giddabasappa, A.; Lalwani, K.; Norberg, R.; Gukasyan, H.J.; Paterson, D.; Schachar, R.A.; Rittenhouse, K.; Klamerus, K.; Mosyak, L.; Eswaraka, J. Axitinib inhibits retinal and choroidal neovascularization in in vitro and in vivo models. Exp. Eye Res. 2016, 145, 373–379.
- Jackson, T. OPT-302 Phase 2b in Wet AMD A Multicenter, Randomized, Double-Masked, Sham Controlled Study of Intravitreal OPT-302 in Combination with Ranibizumab, in Participants with Wet AMD. Available online: (accessed on 1 February 2021).
- Bhargava, P.; Robinson, M.O. Development of Second-Generation VEGFR Tyrosine Kinase Inhibitors: Current Status. Curr. Oncol. Rep. 2011, 13, 103–111.
- Yuan, X.; Marcano, D.C.; Shin, C.S.; Hua, X.; Isenhart, L.C.; Pflugfelder, S.C.; Acharya, G. Ocular Drug Delivery Nanowafer with Enhanced Therapeutic Efficacy. ACS Nano 2015, 9, 1749–1758.
- Kang, S.; Roh, C.R.; Cho, W.-K.; Park, K.C.; Yang, K.-J.; Choi, H.-S.; Kim, S.-H.; Roh, Y.-J. Antiangiogenic Effects of Axitinib, an Inhibitor of Vascular Endothelial Growth Factor Receptor Tyrosine Kinase, on Laser-Induced Choroidal Neovascularization in Mice. Curr. Eye Res. 2013, 38, 119–127.
- Thiele, S.; Liegl, R.G.; König, S.; Siedlecki, J.; Langer, J.; Eibl, K.; Haritoglou, C.; Kampik, A.; Kernt, M. Multikinase inhibitors as a new approach in neovascular age-related macular degeneration (AMD) treatment: In vitro safety evaluations of axitinib, pazopanib and sorafenib for intraocular use. Klin. Monbl. Augenheilkd. 2013, 230, 247–254.
- Research, C. for D.E. and Clinical Pharmacology and Biopharmaceutics Review(s) Application Number: 202324Orig1s000. Available online: (accessed on 15 February 2021).
- Muya, L.; Kansara, V.; Ciulla, T. Pharmacokinetics and Ocular Tolerability of Suprachoroidal CLS-AX (axitinib injectable suspension) in rabbits. Invest. Ophthalmol. Vis. Sci. 2020, 61, 4925.
- Biomedical, C. Clearside Biomedical Announces First Patients Enrolled in Phase 1/2a Clinical Trial of CLS-AX (axitinib injectable suspension) for the Treatment of Wet AMD. Available online: (accessed on 1 February 2021).
- Hancock, S.; Phadke, A.; Kansara, V.; Boyer, D.; Rivera, J.; Marlor, C.; Prodos, S.; Wiles, J.; McElheny, R.; Ciulla, T.A.; et al. Ocular Pharmacokinetics and Safety of Suprachoroidal A01017, Small Molecule Complement Inhibitor, Injectable Suspension in Rabbits. Invest. Ophthalmol. Vis. Sci. 2020, 61, 3694.
- Muya, L. Pharmacokinetics and Ocular Tolerability of suprachoroidal BCX4161 suspension, a selective plasma kallikrein inhibitor, in rabbits. Invest. Ophthalmol. Vis. Sci. 2021, 61, 4925.
