Coatings to be applied on metallic heritage should satisfy complex requirements. This overview presents the main issues to be considered both from a Materials Science view and from a conservation-restoration one. The corrosion mechanism for outdoor bronze monuments is outlined with some of the research project which addressed the need for more affective treatments.
- Metallic Heritage
- Coating
- Atmospheric Corrosion
1. Introduction
Metal objects represents a very broad category in heritage conservation. It is something easy to realise just by having a look at the corrosion identification booklet published by Parks Canada [1] to “provide descriptions and helpful tips, accompanied by photographs, to anyone in charge of metal collections”. Selwyn [2] provided a deeper discussion about the known chemical and physical characteristics of “metals and alloys of interest for conservation professionals, along with the different form of corrosion problems indoors, outdoors, and in archaeological settings”. In her book, one can find an extensive discussion of metals and alloys of interest for cultural heritage (Table 1), main information on construction steps (Table 2)—which may influence the conservation—and on corrosion basic principles. Specific bibliographic references are provided there for each item.
Table 1.
Most used metals in heritage objects, with common minerals from which they are extracted, historical data and typical appearance (adapted from Tables 1.1, 1.2 and 1.5 in Ref. [2]).
| Metal | Mineral | Formula | Approximate Date of First Widespread Use | Typical Colours of Corrosion Products |
|---|
| aluminium | gibbsite | Al(OH)3 | 1800–1900 A.D. (Europe/USA) | colourless or white |
| copper | chalcocite | Cu2S | ~7000 B.C. (Near East) for native copper~ 5000 B.C. (Near East) for smelted copper |
Cu(I): red, black, colourless Cu(II): green, blue |
| gold | (native) | Au | 5000–4000 B.C. (Balkans) | – |
| iron | hematite | Fe2O3 | 1000–0 B.C. (Near East) | Fe(I,III): black Fe(III): red, yellow, orange |
| lead | galena | PbS | 6000–5000 B.C. (Near East/Balkans) | white, red yellow |
| nickel | pentlandite | (Ni,Fe)9S8 | 2000–1000 B.C. (Near East) for copper/nickel alloys | green |
| silver | argentite | Ag2S | 4000–3000 B.C. (Balkans/Near East) | black, white |
| tin | cassiterite | SnO2 | 4000–3000 B.C. (Near East) | black, white |
| zinc | smithsonite | ZnCO3 | 100–200 A.D. (Rome) for copper/zinc alloys 900–1000 A.D. (India) for zinc metal |
colourless or white |
Table 2.
Basic stages of metal objects construction techniques (adapted from Table 1.4 in Ref. [2]).
| Construction Step | Description |
|---|---|
| Forming and Shaping | production by pouring liquid metals into moulds (casting) and by mechanical deformation (forging, rolling; working such as milling, turning, spinning, grinding, stamping, cutting, drilling |
| Assembling | fitting components by welding, soldering, brazing, rivetting, bolting, crinping, gluing |
| Finishing | Completing surface appearance by plating, burnishing, polishing, etching, sand-blasting, painting, lacquering, engraving, chasing, embossing, enameling, patinating |
As a rule, the inherent instability of metallic heritage offers similar preservation challenges to those faced in civil engineering, automotive and construction industries [3]. The main source of this instability is the energy required to extract metals from their ores (smelting), which leaves metals in a high energy state with the tendency to return to the lower energy mineral state [2]. The actual behaviour of every metallic item is the result of complex interactions between the chemical–physical properties of the object and the particular environment around it [4]. These interactions may reach a static or dynamical equilibrium over time. It is thus essential to consider the specific environments all along the lifetime of a metallic heritage object in order to develop treatments and identify realistic conservation goals [4]. Through a deeper understanding of the complex chemical, thermodynamic and kinematic factors, several choices to mitigate the adverse effects of corrosion can be developed by modifying the environment or the surface finishing of the object, as addressed by Corrosion Science [5]. Corrosion is one of the major issues for metal heritage objects conservation, and methodologies and principles of corrosion science have been slowly entering conservation practice from the second half of the 20th century [3][6][7][8][9][10][3,6,7,8,9,10].
Although the basic laws underlying corrosion processes in the field of cultural heritage are the same as in industry, the criteria and priorities behind operational choices to mitigate the adverse effects of corrosion are deeply different [4][5][7][11][12][13][4,5,7,11,12,13]. The vast possibilities of interaction between distinct metals and types of environment give rise to unlimited combinations of corrosion forms, making it useless for conservation purposes to gather occurrences following traditional corrosion classifications. Since the exchange with its surroundings is so intensive, metallic heritage can be better understood and conserved when its context is considered. Accordingly, broad categories of heritage metal objects can be identified depending on the environment where they have been (or still are) [14]:
- Archaeological metals are characterised mainly by long burial in soil [15], waterlogged [12][15][16][12,15,16] or underwater [17]; they bear information on very long-term corrosion of metals [18][19][18,19]; the equilibrium state reached during burial may be broken when excavated, giving rise to new corrosion process if not properly treated [20].
- A large variety of historic objects (such as scientific instruments, fine arts, historic pieces, ethnographic specimens, etc.) is conserved indoors (museums, monumental buildings, collections); the main preservation strategy in this environment is preventive conservation [4][21][4,21], such as humidity control; critical parameter to consider are dangers from “off-gassing” materials used to build display cases and rooms, as well as air pollution introduced by visitors [21].
- Outdoors monumental and architectural items (sculptures, roofs and decorative objects, functional artifacts and industrial heritage) are mainly subject to weather conditions, pollution and climate change [4][6][14][22][23][24][25][26][27][4,6,14,22,23,24,25,26,27].
2. Coatings for Outdoor Bronze Monuments
In order to apply correctly the principles of Corrosion Science to the conservation of metal heritage objects, each specific metal or alloy, construction technique, and particular corrosion problem encountered in a museum, outdoor, or archaeological setting should be carefully considered [2][3][4][15][18][22][2,3,4,15,18,22]. The specific characteristics of heritage objects are the manifold result of a complex and often not completely known history. This can lead to inappropriate treatments when coatings checked for a specific context are adopted in another one without taking into account all the relevant features [28]. For this reason, the topic of protective treatments for metallic cultural heritage can be wide and complex and oversimplified research data may be useless for the conservation of metal heritage objects purposes. Accordingly, from now on the discussion will be restricted to outdoors artifacts. Within this category, the distinctive features relative to bronze sculptures will be considered in order to enlighten the specific characteristics required for conservation treatments [29].
Environmental changes produced by the industrial era raised problems in many fields. Increased pollution and acid rain effects on materials were widely recognised [30] and many research efforts addressed this topic, which is the field of Atmospheric Corrosion [31]. Outdoor copper and copper alloy statues started suffering from a sharp decrease in surface stability and localised corrosion endangering their artistic and aesthetic content [22]. This triggered a growing interaction between conservator-restorers and the scientific community [6][7][8][9][10][25][32][6,7,8,9,10,25,32]. Over the last decades, this allowed a number of improvements: A growing understanding of the electrochemical processes typical of outdoor bronze monuments [33][34][35][36][37][38][39][33,34,35,36,37,38,39]; the dissemination of the “Theory of Restoration” by Brandi as a tool to lead choices on new conservation practices [7][11][40][41][7,11,40,41]; more awareness about possible strategies to deal with the changing equilibria and available resources [3][42][3,42].
The present understanding of copper and copper alloys atmospheric corrosion ([31], chapters 8, 13, 14; appendix E, K), can be roughly described as the formation of a passivating cuprite layer in clean humid air which evolves into a more complex surface layer (patina) according to the main pollutants present in the surrounding atmosphere [3][25][34][3,25,34]. Green basic copper hydroxysulfates (mainly brochantite) form in SO2 rich atmosphere, while basic hydroxychlorides (atacamite, clinoatacamite) dominate marine environments [43]. Alloying elements play a key role in the corrosion mechanism, which differs from pure copper [34][39][44][45][34,39,44,45]. A decuprification process was identified [37][44][45][37,44,45], along with cyclic corrosion similar to the “bronze disease” traditionally associated with archaeological copper alloys [2][35][45][46][2,35,45,46]. The growth of this surface layer on outdoor bronze sculptures and architectonic elements exposed to different weather and pollution conditions is the result of a specific timeline. Several factors determine its local composition and texture, such as the solubility of corrosion products, sheltered and unsheltered exposure, the changing weather (relative humidity, temperature, light and Ultra Violet (UV) radiation, time of wetness), the composition and concentration of air pollutants (Figure 1).
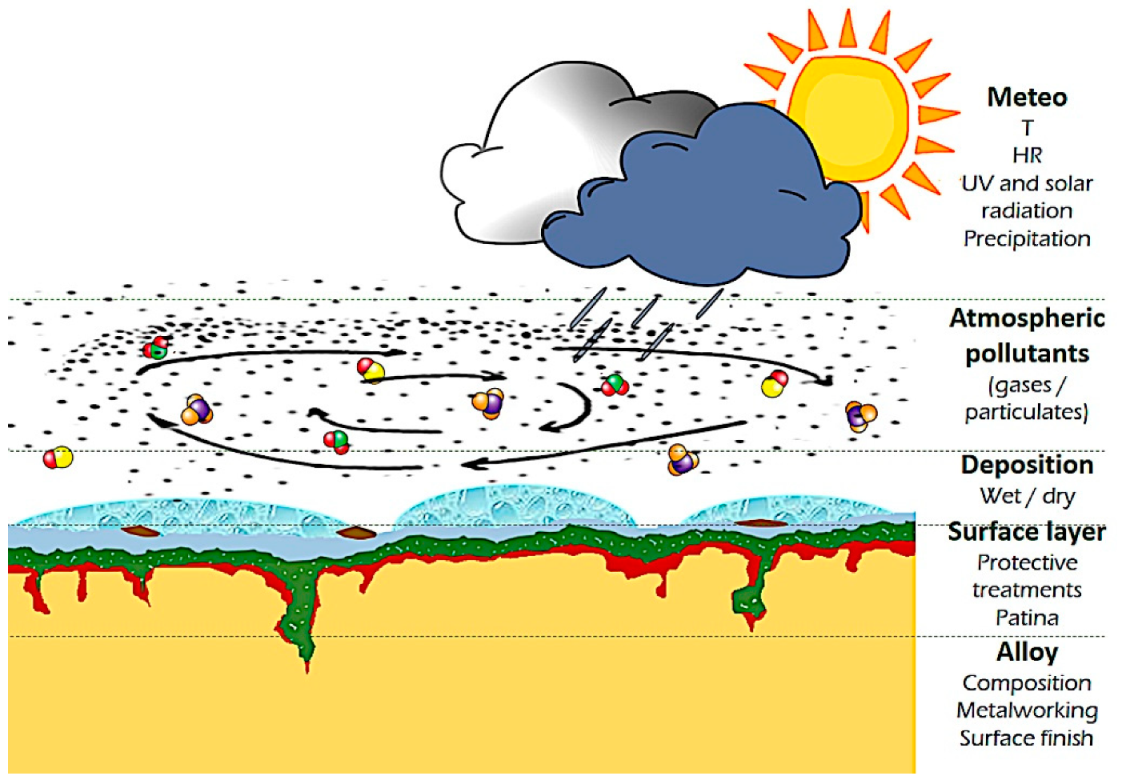
On a single monument, different conditions are often present, and the evolution of the overall result can be detrimental for aesthetic reasons if not for the material loss [42][45][42,45]. This leads to the necessity of restoration. The first step consists of cleaning [47][48][49][47,48,49]. The cleaning methodologies adopted should selectively remove only water-soluble compounds [47], atmospheric particulate deposits, hydrocarbon compounds coming from environmental pollution and other organic compounds from past treatments [50] or failed coatings [51]. At the same time, it should preserve the part of the patina valuable for aesthetic, historical and conservation reasons [48][52][48,52]. Afterward, the common practice consists in the use of treatments to prevent or reduce detrimental corrosion, which for outdoor bronze consist in the application of coatings that avoid the contact of the metal/patina layer with the actively corroding agents in the atmosphere (water, corroding ions) and/or other treatments (inhibitors) to reduce the electrochemical reaction rates [4][5][4,5]. Sooner or later, cleaning and application of a protective treatment should be repeated, according to environmental conditions, maintenance programs, etc. (Figure 2).
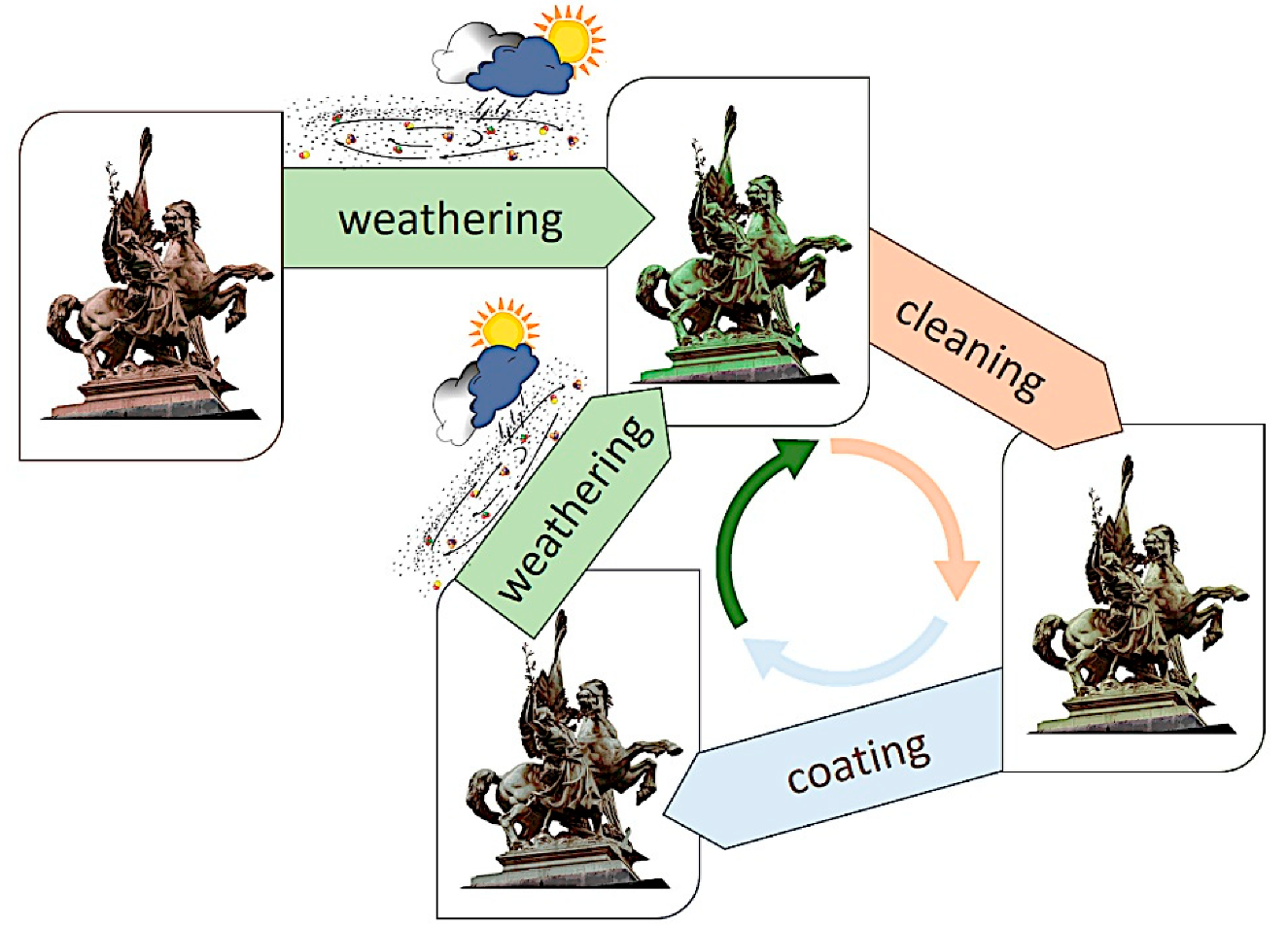
Since the end of the 20th Century, several projects have addressed the need for new/more effective protective treatments to be applied on metallic heritage objects.
The EU-FP3-funded project “New Conservation Methods for Outdoor Bronze Sculptures” [53] considered a new class of sol–gel derived coatings-organic inorganic copolymers, called ormocers (ORganically MOdified CERamics); addressed requirements were the use in outdoor bronze conservation with good protection against corrosion, and at the same time the right compromise between stability and reversibility of the cured coating [54].
After the public engagement following the refurbishment of the Statue of Liberty [55], some sponsors supported the research “Coating strategies for the protection of outdoor bronze art and ornamentation” [56], where the performance of 29 coatings on different copper alloy substrates was addressed and the importance of considering a coating system as a whole, and not only by its part, was pointed out.
In the mainframe of EU-FP6-funded project “EU-ARTECH”, traditional treatments performances (Incralac, waxes, Benzotriazole) were compared to innovative treatments ones on coupons with natural and artificial patina [56]; different commercial organo-silanes were tested, the use of limewater was considered to adjust pH toward alkalinity to inhibit bronze disease, and the possible use of fungi to transform unstable corrosion patinas to insoluble copper oxalate was investigated [40]; on copper lamina with natural green patina (mainly brochantite) the Dynasylan F8263 and SIVOClear showed a protective behaviour comparable to Incralac but without perceivable chromatic alterations [57][58][57,58]. Biopatina was further investigated in other projects [40][59][40,59].
Other EU-funded projects addressed the development of new and more effective protective treatments for outdoor bronze artworks [60][61][60,61]. Several efforts were performed to deepen the understanding of the forms and properties of outdoor bronze surface layers [36][39][44][48][62][36,39,44,48,62]. Increasing attention was paid to the characterisation and comparison of surface layers and treatments on artworks [63][64][65][66][67][63,64,65,66,67].
Until now, the transfer of innovative treatments in the practice of restoration of outdoor bronzes was quite poor, despite the considerable amount of laboratory studies in this field since the 1990s.
