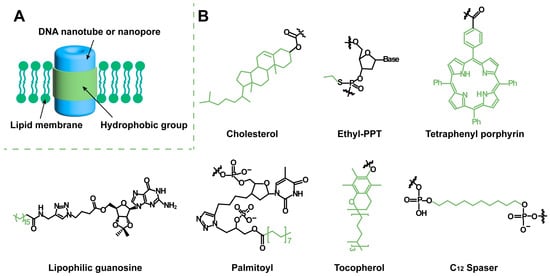
3.2. DNA Helix Bundle-Based Transmembrane Channels
Small-pore DNA-based artificial channels can be designed based on helix bundle (HB)-based nanotube assembly through the concatenation of multiple DNA strands
[16][24]. DNA helix bundles are prepared by cross-joining scaffolds and short strands in the middle or at the ends of the materials, and the helix bundles follow the structural layout of polygon arrays
[23][31]. The six DNA helix bundle (6-HB) nanotubes form approximately 2 nm-wide pores at the center, which are hydrophobically modified to maintain structural stability in the lipid bilayer and to support a constant transmembrane current. The reported conductivities of these 6-HB DNA transmembrane channels range from approximately 0.3 nS to 1.6 nS
[23][24][31,32]. Through molecular dynamics (MD) simulations, the following conclusions are found. (1) The chemical modification on the surface of the channel has an extremely large impact on the transport of water and ions across the membrane, and the type, number, and position of the hydrophobic group modification can directly affect the formation of transmembrane channels. (2) DNA nanochannels can be used to transfer charged solute pairs to antistatic gradients through electroosmosis. (3) The porous channel wall allows the transverse leakage of ions and water. (4) The central lumen of the DNA channel is cylindrical and filled with water and ions; the volumes of water at the opening regions of both ends fluctuate in time and exhibit mechanosensitive gating, creating a force sensor
[34][35][36][37][43,44,45,46].
3.3. DNA Tile-Based Transmembrane Channels
DNA tiles are short intersecting DNA strands that contribute to structural control. They have cohesive ends that can be programmed to self-assemble to form various DNA nanostructures by clinging together
[38][39][50,51]. DNA tiles have been widely used to prepare DNA nanotubes. The steps of their self-assembly mainly focus on the release of DNA input molecules to trigger the growth of nanostructures, which is a nonautonomous and irreversible reaction
[40][52]. With the maturation of construction technology, several methods have been designed to synthesize DNA tile nanotubes with adjustable, reversible, or controllable termination characteristics
[41][42][43][53,54,55]. By using DNA origami structures as seeds to construct channels, micron-long nanotubes can be obtained through the polymeric growth of DNA tiles. The hydrophobic unit on the seed can directly insert the nanotube into the membrane to form a transmembrane channel
[44][56]. An additional DNA origami channel cap can be used to terminate the aggregation of tiles. Conductivity measurements reveal that the conductance values of seeds and nanochannels are lower than their uncapped counterparts when the channel caps are attached. The results show that ions move from one end of the channel to the other and that there is partial leakage through the channel wall. However, the observation experiments of fluorescent dyes crossing lipid membranes confirm that molecular transport can occur through DNA nanochannels and that it is mainly end-to-end rather than across the channel wall
[45][57].
3.4. DNA Origami-Based Transmembrane Channels
DNA origami technology can be used for designing DNA-based channels; the shapes and sizes of the channels can be adjusted precisely, systematically, and abundantly
[11][19]. DNA origami, proposed by Rothemund in 2006, is a relatively new method for DNA assembly. Based on the principle of complementary base pairing, by utilizing the structural characteristics of DNA molecules, the long DNA strands folded in specific regions are fixed by short strands to construct the expected structure
[46][58]. Due to its simple experimental conditions and high assembly efficiency, DNA origami has become a popular technology for constructing artificial DNA nanochannels. Artificial DNA nanochannels have been comprehensively designed concerning their pore size, length, and morphology characteristics.
Artificial DNA nanochannels can be exploited for size-dependent, selective transmembrane transport. The pore diameter is critical for the selective delivery of substances of different sizes. Transmembrane channels with large pore sizes are required for the transport of macromolecular substances. To this end, long DNA strands that are arranged longitudinally are initially used as basic units to construct channels, and the inner diameters of these channels are <9 nm
[17][18][19][20][25,26,27,28]. A method of lateral assembly of DNA strands to form channels has been proposed; the same amount of DNA strands can be used to obtain artificial DNA nanochannels with a relatively large pore size (~35 nm)
[21][22][29,30].
The DNA nanochannels synthesized by Kjems et al.
[20][28] have flanks to adjust the cholesterol exposure on demand. The DNA channels are composed of a double-layer irregular hexagonal cylindrical DNA structure with 46 hydrophobic spots (17 on the walls and 29 on the flanks). Three programmable DNA lobes are on the three nonadjacent sides of the channel and connected approximately 12 nm from the bottom of the channel by a single-stranded DNA hinge. In the closed state, each flank is near the channel wall due to two stable strands that are complementary to the bottom single-strand portion of the channel. The flanking closure can protect the hydrophobic moiety from the aqueous environment and limit hydrophobicity-driven channel aggregation. When a key strand fully complementary to the single-stranded DNA is present around the channel, the flanks are opened and the cholesterol is exposed, thereby driving channel insertion into the membrane.
3.5. Other DNA-Based Transmembrane Channels
G-quadruplex, a DNA duplex with unique ion transport properties, is utilized to design transmembrane channels. Depending on the hydrogen bond between the nitrogen and oxygen atoms of guanosine and the π-π stacking between the bases, guanine (G)-rich DNA single strands can be deformed or aggregated to form a G-quadruplex structure with a central hole
[47][59]. Moreover, the stability of the G-quadruplex depends heavily on certain cations, such as K
+, Na
+, NH
4+, and Ca
2+ [48][60]. Within the central channel, each ion is completely dehydrated and interacts with the guanine carbonyl O
6 atom around the pore. This particular feature is reminiscent of the selectivity filters in K
+ ion channel proteins first noted by Feigon et al. Because of this structural similarity, the G-quadruplex is a candidate for the design of artificial ion channels for selective transmembrane transport of K
+ [49][50][51][61,62,63]. Dash et al.
[25][33] have used telomere DNA to form a G-quadruplex and additionally modified lipophilic guanosine to construct an artificial potassium ion transmembrane transport carrier.
4. Artificial Transmembrane Channels for Biosensing and Biomedical Applications
4.1. DNA-Based Transmembrane Channels for Biosensors
4.1.1. Single-Molecule Nanochannel Sensors
Transmembrane DNA channels have been proposed for use as single-molecule nanochannel sensors. In biomimetic sensing experiments, the translocation of analyte molecules leads to current changes in membrane pores and the duration and depth, which are related to the charges and sizes of the analytes. For example, DNA channels designed by Simmel et al.
[17][25] have been used for single-molecule sensing. A stable baseline current is detected on the lipid membrane containing the artificial DNA channels. The additions of the hairpin molecules at the beginning and ~30 min later show transient current blocking, in which the applied voltage can capture, decompress, and translocate the hairpin structure.
4.1.2. Ligand-Gated Artificial Transmembrane Channels
The DNA-based artificial transmembrane channels are mostly hollow tubes with openings. Voltage gating is observed in almost all DNA nanochannels, but channels with additional gates are highly flexible and controllable
[52][68]. Therefore, the design of artificial channels with ligand-gated opening or closing properties has become the focus of research. The reported artificial DNA nanochannels with gates can specifically recognize key strands. Howorka et al.
[53][66] built an artificial channel with a gate that can be opened with a key strand. The lock of the channel is tightly bound to the entrance by hybridizing with two docking sites to form a spiral bundle across the channel opening. The docking site is formed by the extension of two duplex support rods in opposite positions. The key can be hybridized with the lock strand to remove it, leaving the channel open. After being modified by cholesterol, such channels can be used to regulate the flow of small organic molecules (including many important drug compounds), which have broad application prospects in biomedicine.
To exploit the potential of DNA-based artificial channels as real-time smart sensing devices, Kjems et al.
[20][28] have designed a bolt on the inside of the channel. PEG is used as a plug that connects the toehold sequence with 8 nucleotides to partially block the gateway and allows small molecules (ATTO 655) to pass through. The unplugged strand is combined with the toehold-mediated strand to remove PEG so that the macromolecular material (dTMR-40k) can pass through the artificial DNA nanochannel. Another reversible gated protein transport membrane channel is constructed based on a horizontal routing DNA origami design strategy with a large pore size of 20.4 nm
[54][67]. The passageway opening is designed with a square cover, one side of which is attached to the cap by a flexure hinge. The other side of the cap carries two single strands that can be hybridized with the two single strands on the cap to form a complete double lock. Two key strands are added to the system to open the lock and lid. The lid is switched back to its closed state with a single-strand reverse key pair. This channel allows the precisely timed transport of folded proteins across the membrane.
4.1.3. Environmental Stimuli-Responsive Artificial Transmembrane Channels
Environmental stimuli-responsive artificial channels have been designed to be sensitive to temperature
[55][73], light
[56][57][58][74,75,76], and ions
[59][77]. The temperature-responsive DNA channel constructed by Howorka et al.
[55][73] has two main parts: a transmembrane barrel-shaped nanotube and a reversibly sealed lid at the top. Biphasic segments 1–4 are designed between the channel and the lid, with a designed melting temperature of approximately 40 °C for segments 2–4 and 62.8 °C for segment 1. The lid is hybridized to the two elongated rings of the channel at room temperature to block the mass influx. Temperatures higher than 40 °C selectively separate the lid from loop segments 2–4 to allow the cap to open. By adjusting the temperature, the lid of this channel can achieve reversible on/off functionality. Azobenzene is a reversible cis-trans photoisomerization switchable compound. The conversion of azobenzene from the trans isomer to the cis isomer can be triggered by light irradiation with a wavelength λ < 400 nm, and the reverse effect can be achieved by illumination at λ > 400 nm
[56][74]. The cis-trans isomerization of azobenzene can adjust the on/off state of the channel. Howorka et al. proposed a 6-HB-based DNA channel, and cis-azobenzene corresponded to the closed state
[57][75].
4.2. DNA-Based Transmembrane Channels for Biomedical Applications
4.2.1. Cell Mimics for Transmembrane Transport
Artificial DNA nanochannels can serve as synthetic cell membrane components to mimic transmembrane transport. Currently, the transport selectivity of DNA transmembrane channels is largely determined by their pore size. The molecules or ions smaller than their pore size are easily mass-transportable. Meanwhile, the negatively charged DNA ion channel has a poor transport capacity for negatively charged ions. Artificial DNA transmembrane channels with large pores have been proposed for the transmembrane transport of drugs, immune proteins, and so on. In 2016, Howorka et al.
[60][80] were inspired by organelles to create synthetic hybrid nanocontainers composed of polymersomes and DNA nanochannels. Nanocontainers exhibit size-dependent permeability. These containers enable the transport of the enzyme substrate across the membrane while retaining the relatively large enzyme inside the container. These nanodevices can be used to simulate the site where biocatalytic reactions occur. The 6-HB DNA nanochannels modified with three cholesterol molecules on this container mimic the protein channels in biofilms and enable specific substance transport.
4.2.2. Transmembrane Channels for Cell Death
Artificial transmembrane channels can selectively control ion transport across biological membranes, and artificial channels can disrupt cellular homeostasis of cell death. Howorka et al.
[61][81] designed a DNA channel with a highly hydrophobic 2-nm band composed of ethyl phosphorothioate (EP) at one end, which can penetrate the cell membrane and cause cell cytotoxicity. Tan et al.
[62][82] have found that phosphorothioate (PPT)-modified DNA nanochannels can be spontaneously inserted into the cell membrane, and they can transport ions and antitumor drugs to neurons and cancer cells, respectively. It has been proposed that their potency can be improved by specifically binding target cancer cells. Loading chemical toxins, such as doxorubicin, with DNA insertion enhances chemical toxicity.