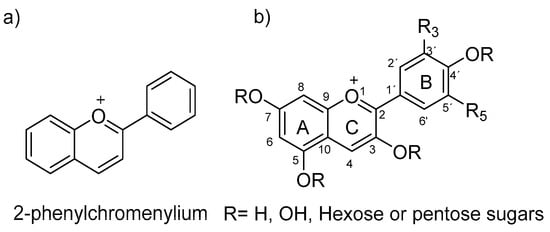
Within anthocyanins’ characteristics, they are known to be water- and alcohol-soluble organic compounds that have a three-ring heteroaromatic polyphenolic skeleton. Anthocyanins are glycosylated analogues, mainly at the C3 position, of anthocyanidins, both based on the 2-phenyl-benzopyrilium chromophore structure (flavylium ion in Figure 1), which shows an extended π conjugation, as well as the presence of a positive charge and several free -OH groups. These characteristics allow anthocyanins to absorb light in the visible region, which generates a great variety of dye colors, making them one of the most important natural pigments besides chlorophyll.
Generally, a molecule with more -OH groups generates a more intense blue color, while in those with -OR groups, the coloration turns red. As for the glycosyl substituents, their presence decreases the coplanarity of the B-ring, so they tend to be less stable, and the coloration is usually not as intense.
If, in addition to sugar, there is an acyl radical in the molecule, they are called acylated anthocyanins. An increase in glucosidic substitution and acylation with cinnamic acids allows them to be more stable and retain their characteristic color at alkaline pHs
[11].
Anthocyanins have received increasing attention from researchers in recent years mainly due to their importance in two main application sectors: the food industry and healthcare. Regarding food technology, the utility of anthocyanins extends beyond their role as a natural colorant to enhance the organoleptic properties of food. Their inherently reactive nature has led to their consideration as colorimetric indicators for assessing food quality in advanced smart food packaging technologies. Regarding healthcare and pharmaceutical applications, a large amount of highly interesting health-promoting effects of anthocyanins have been revealed in the last decade. Thus, the nutraceutical effect of the anthocyanin or anthocyanin-rich food intake has attracted research interest. However, their low stability and bioavailability hinder an efficient effect of anthocyanins in the human body. Consequently, research efforts have focused on the development of strategies to improve their stability, bioavailability, and color preservation. Among these strategies, encapsulation has become the most effective and explored one. Encapsulation is based on the development of a protective coverage of anthocyanins, which, in addition to improving their stability and bioavailability, could also provide advanced properties or performance (like selective or controlled release).
Accordingly, there exists a real need for immobilizing anthocyanins within appropriate substrates that fulfill the wide range of physical and chemical requirements specified for the mentioned applications. Among these substrate systems, polymers are versatile materials that show unique properties in a wide range of forms, like films, emulsions, hydrogels, mats, and nanoparticles, among others, that are of great interest in the food-packaging and healthcare sectors. In the light of this combination, the incorporation of anthocyanins within polymeric matrices has proliferated in recent years as a promising approach to utilize the synergistically positive properties of both substrates and active agents.
2. Anthocyanin-Loaded Polymers: Preparation and Characterization Methods
2.1. Films
Anthocyanin-loaded polymer films are obtained mainly by solution casting, a simple and cheap technique that uses aqueous solutions or organic solvents to dissolve anthocyanins as the active agent and polymers as the substrate for immobilization
[12][30]. In brief, polymer/s, plasticizer, and an anthocyanin extract are sequentially dissolved in solution at temperatures lower than 80 °C for a short period of time to minimize the degradation of the anthocyanins. Finally, the obtained solution is incubated onto flat Petri dishes at ~25–40 °C for 24–48 h. Other more sophisticated and expensive techniques have also been reported to fabricate anthocyanin films, such as electrospinning and layer-by-layer and 3D printing
[13][31]. Chitosan, cellulose, starch, zein, gelatin, pectin, agarose, xanthan gum, and combinations of these have been the most commonly employed polymer systems for the development of anthocyanin-loaded films. Glycerol and sorbitol are the most popular plasticizers used in anthocyanin-based films
[14][32].
Regarding films for monitoring food spoilage, it has been shown that combining polymers improves the physical properties of the material. For example, Nadi et al.
[15][33] showed that combining basil seed gum with chitosan and adding red cabbage extract as a colorimetric indicator improves properties such as solubility, water vapor permeability, and flexibility in the material.
The incorporation of anthocyanins into films can modify the technological and functional properties of the film. These changes depend on the physical or chemical interactions between the film-forming polymer and the anthocyanin extract, which can lead to structural modifications. The interaction between biopolymers and anthocyanin-rich extracts depends on the nature, chemical characteristics, and concentration of both the polymer and extract, as well as the structural properties of the active components. The addition of anthocyanin-rich extracts can influence the thickness, color, opacity, solubility, water vapor permeability, and mechanical properties of the films
[16][35]. Thickness is typically analyzed because, in most cases, it is modified and, since it hinders the penetration of water, it is a key parameter to take into consideration
[13][31].
2.2. Mats and Fibers
It is known that nanofibrous mats have a high surface-to-volume ratio, small pore size, and high porosity compared to polymer films, which makes electrospun nanofibrous mats very attractive materials for the manufacture of substrates for the immobilization of anthocyanins
[17][36].
The electrospinning process involves the application of an electric field to create fine polymer fibers from a solution containing anthocyanins. Anthocyanins can decrease the conductivity of the polymer solution, and consequently, as Jiang et al.
[18][29] observed by SEM microscopy, heterogeneous fibers are formed.
Anthocyanin nanofiber mats made by electrospinning are used as tools in the field of active/smart packaging where they have been studied to improve their stability, simplify the slow and controlled release of antimicrobial and antioxidant substances
[19][37], and as spoilage sensors
[20][38]. When it comes to healthcare applications, nanofibrous mats of anthocyanins have found their major application as smart wound dressings that can monitor the wound healing process
[21][39].
2.3. Hydrogels
Hydrogels have been widely used as a method of encapsulating water-soluble active ingredients. The thermo-degradation and stability against sudden pH changes of anthocyanins could be inhibited by encapsulating them within hydrogel systems. This has been demonstrated in the process of simulated digestion and may be very useful in the prevention of intestinal diseases
[22][23][40,41].
Recent research has demonstrated the versatility of these materials that allow the easy incorporation of multiple active agents. For instance, Lotfinia et al.
[24][42] combined alginate in the form of a hydrogel with honey and red cabbage extract, and could observe improved mechanical properties, the antibacterial activity attributed to the honey, as well as the antioxidant properties and good activity against pH changes from the red cabbage.
Polysaccharides have been traditionally employed in anthocyanin hydrogel preparations due to the simplicity of their corresponding gelation process. This is the case for alginate, which forms cold gels through Ca
2+-induced cross-linking
[25][43], or starch
[26][44], another widely used material for these gel-based encapsulation systems.
2.4. Polyelectrolyte Complexes
Polyelectrolytes are macromolecular materials that possess repeating units and dissociate into highly charged polymeric molecules in aqueous solution, forming either positively or negatively charged polymeric chains
[27][45]. There are numerous compounds that can serve as biopolyelectrolytes such as proteins, polysaccharides, and their derivatives. Biopolyelectrolyte complexes can be formed by the titration of a biopolyelectrolyte solution in another biopolyelectrolyte solution with the opposite charge under agitation. Among the advantages of this methodology are the simplicity, quickness, and the fact that it does not require high energy, chemical crosslinkers, specialized equipment, or organic solvents. An advantage of these materials is that polymeric chains can generate dense interconnecting networks, which is useful for inhibiting the penetration of polar reactive compounds and the attack on charged anthocyanins
[28][46].
2.5. Nanoparticles
As in the general case of polymeric nanoparticle preparations, the methods commonly used for the preparation of anthocyanin nanocarriers can be classified as the emulsification cross-linking method, ionic cross-linking method, covalent cross-linking method, and self-assembly methods.
The emulsification crosslinking method is a widely used method. A carrier particle is usually large (tens to hundreds of microns) with a highly homogeneous particle distribution. Anthocyanin nanoparticles are divided into ionically crosslinked and covalently crosslinked depending on the crosslinking method. Covalently cross-linked carriers are more stable. Studies have shown that the type of polymer employed has a relevant effect on the crosslinking efficiency.
Ionic nanoparticles could be easily formulated through complexation of two different charged biopolymers in diluted solutions. Biopolymers such as alginate, chitosan, whey, and soy protein are commonly extruded as a carrier solution through a needle or nozzle into a gelation solution containing the specified ions. This is a mild process that has shown a high encapsulation efficiency and effective protection of anthocyanins under gastric conditions
[29][50].
2.6. Emulsions
Emulsion-based systems are intended to overcome the low stability and bioavailability of anthocyanins. Among the most used systems are nanoemulsions and microemulsions that have been studied in vivo and in vitro as anthocyanin delivery systems
[30][31][32][33][51,52,53,54].
Nanoemulsions are colloidal dispersions formed by droplets of a liquid dispersed in another liquid that, despite being immiscible, can be stabilized with a surfactant layer. In nanoemulsions, particles showing sizes from 20 to 500 nm can avoid emulsification, flocculation, or precipitation during storage
[34][35][36][55,56,57]. Some of the benefits of nanoemulsions are the control of the release rate and the avoidance of decomposition or degradation of the encapsulated anthocyanins
[37][58].
Microemulsions are mainly composed of water, oil, surfactants, and cosurfactants, and have better flowability, more uniform particle dispersion, and stronger stability than emulsions. Microemulsion particles have sizes between 10 and 100 nm, which can provide stable and uniformly dispersed systems
[38][59], which are useful properties for improving the absorption and bioavailability of anthocyanins and enable easy and multiple routes of administration
[39][40][60,61].
There are several studies that report the development of nanoemulsions and microemulsions with extracts from different sources of anthocyanins, such as mangosteen peel, Brazilian berry, purple sweet potato, cranberry, red cabbage, blueberry, and jaboticaba peel, among others, showing nanoparticles that are stable for long periods of time, ranging from 30 days to 3 months
[32][37][38][41][42][53,58,59,62,63].
2.7. Self-Assembled Liposomes, Proteins, Peptides, and Phospholipids
Today, liposomes, phospholipids, and proteins are used as anthocyanin nanocarriers and are formed by direct self-assembly. Proteins, as was commented above, have the drawback of being very sensitive to pH changes or temperature. Liposomes also have low stability and their phospholipids are prone to oxidation during long-term storage. Phospholipids have a lot of benefits including the ability to protect sensitive ingredients, increase the bioavailability of nutrients and the efficacy of food additives, and confine undesirable flavors
[43][65].
Amphiphilic peptides with a hydrophobic tail and a hydrophilic head are also used in molecular self-assembly. These amphiphilic peptides have good biocompatibility, biodegradable self-assembly, and chemical variability, which leads to a variety of nanostructures. Indeed, the addition of peptides to foods containing anthocyanins is recognized as the simplest way to improve the color stability of anthocyanins
[37][43][44][58,65,66].
2.8. Microencapsulates
Microencapsulation consists of the incorporation of the active agent within the encapsulants by noncovalent interactions formed during a mixing process that can include additional coating layers
[29][50]. The final drying step generally takes place by a freeze-drying or spray-drying methodology. Microencapsulation is an alternative to keep the properties of the anthocyanins intact and offer food processors a means of protecting sensitive food components
[45][46][47][48][49][50][72,73,74,75,76,77]. However, during the spray drying process, some heat-sensitive anthocyanins may lose their activity or degrade and, as a consequence of the rapid dehydration, they may change their crystal structure. Consequently, freeze-drying is often preferred to mitigate these potential issues and preserve the integrity of the anthocyanins
[51][49].
Microencapsulation allows the encapsulation of anthocyanins within a wide variety of encapsulating agents, such as polysaccharides, starches, inulin, maltodextrin or dextrose, corn syrups, arabic gum, mesquite gum, lipids, and proteins
[52][78].
2.9. Specific Characterization of Anthocyanin-Loaded Polymer
The characterization of anthocyanin-loaded polymers is crucial for understanding their structure and properties. Several techniques are used to obtain significant information on these materials.
UV-Vis spectroscopy is used to analyze the absorption spectra of these materials, providing information about their specific composition, color, stability, and optimum absorption range
[53][79].
FTIR-ATR is also a useful technique because it provides information regarding the interactions between the anthocyanin and the polymer matrix. The main changes are reported in two regions, between 1500 and 1600 cm
−1, corresponding to the C=C bonds, and around 3200 cm
−1, corresponding to the vibrations of the C-H bonds. In both cases, if the signals are shifted and/or widened, it confirms the immobilization of the anthocyanins in the polymer due the formation of strong interactions with the polymers
[12][54][55][30,80,81].
Using Thermo-Gravimetric Analysis (TGA), several studies have shown that the addition of anthocyanins to polymeric matrices or the encapsulation of anthocyanins in a polymer provides greater stability with respect to pure compounds
[56][57][58][82,83,84].
3. Anthocyanin-Based Polymers for Healthcare Applications
Owing to the vibrant and diverse color spectrum of anthocyanins, they have found multiple applications as natural dyes in various healthcare-related fields. These natural compounds present an intriguing potential as safe and biocompatible alternatives to synthetic dyes. Due to their ability to exhibit color shifts under different conditions, their potential use as biosensors has garnered attention in recent years. In addition, anthocyanins show excellent antioxidant and anti-inflammatory effects as well as potential in the prevention of cancer, neurodegenerative diseases, and diabetes, making them very interesting natural bioactive compounds. However, their limited bioavailability remains as one of the main disadvantages for this family of compounds. Therefore, the development of systems to increase the bioavailability and, consequently, the efficacy of anthocyanins is desirable.