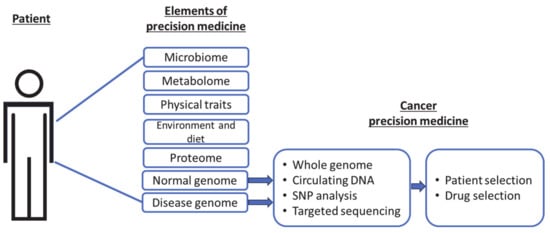
Precision cancer medicine primarily aims to identify individual patient genomic variations and exploit vulnerabilities in cancer cells to select suitable patients for specific drugs. These genomic features are commonly determined by gene sequencing prior to therapy, to identify individuals who would be most responsive. This precision approach in cancer therapeutics remains a powerful tool that benefits a smaller pool of patients, sparing others from unnecessary treatments. A limitation of this approach is that proteins, not genes, are the ultimate effectors of biological functions, and therefore the targets of therapeutics. An additional dimension in precision medicine that considers an individual’s cytokine response to cancer therapeutics is proposed. Cytokine responses to therapy are multifactorial and vary among individuals. Thus, precision is dictated by the nature and magnitude of cytokine responses in the tumor microenvironment exposed to therapy.
- precision medicine
- pharmacogenomics
- cytokines
- chemotherapy
- radiotherapy
1. Precision Cancer Medicine
2. Cytokine Pathways as Therapeutic Targets
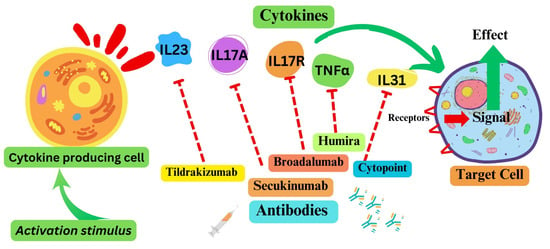
3. Cells of Cytokine Origin
4. Cytokine Responses in Solid-Tumors Therapy: An Emerging Theme in Cancer Precision Medicine
References
- Mathur, S.; Sutton, J. Personalized medicine could transform healthcare. Biomed. Rep. 2017, 7, 3–5.
- Sadée, W.; Dai, Z. Pharmacogenetics/genomics and personalized medicine. Hum. Mol. Genet. 2005, 14, R207–R214.
- Agúndez, J.A.G.; García-Martín, E. Editorial: Insights in Pharmacogenetics and Pharmacogenomics: 2021. Front. Pharmacol. 2022, 13, 907131.
- Wilke, R.A.; Reif, D.M.; Moore, J.H. Combinatorial Pharmacogenetics. Nat. Rev. Drug Discov. 2005, 4, 911–918.
- Shastry, B.S. Pharmacogenetics and the concept of individualized medicine. Pharmacogenomics J. 2005, 6, 16–21.
- Le Tourneau, C.; Delord, J.-P.; Gonçalves, A.; Gavoille, C.; Dubot, C.; Isambert, N.; Campone, M.; Trédan, O.; Massiani, M.-A.; Mauborgne, C.; et al. Molecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): A multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trial. Lancet Oncol. 2015, 16, 1324–1334.
- Warner, J. Giving Up on Precision Oncology? Not So Fast! Clin. Transl. Sci. 2017, 10, 128–129.
- Ghoreschi, K.; Balato, A.; Enerbäck, C.; Sabat, R. Therapeutics targeting the IL-23 and IL-17 pathway in psoriasis. Lancet 2021, 397, 754–766.
- Noack, M.; Miossec, P. Selected cytokine pathways in rheumatoid arthritis. Semin. Immunopathol. 2017, 39, 365–383.
- Agüero, R.; Woodbury, M.J.; Lee, K.; Johnsson, H.J.; Merola, J.F.; Armstrong, A.W. Interleukin (IL)-17 Versus IL-23 Inhibitors: Which Is Better to Treat Patients with Moderate-to-Severe Psoriasis and Mild Psoriatic Arthritis in Dermatology Clinics? J. Rheumatol. 2023, 50, 11–13.
- Moyaert, H.; Van Brussel, L.; Borowski, S.; Escalada, M.; Mahabir, S.P.; Walters, R.R.; Stegemann, M.R. A blinded, randomized clinical trial evaluating the efficacy and safety of lokivetmab compared to ciclosporin in client-owned dogs with atopic dermatitis. Vet. Dermatol. 2017, 28, 593-e145.
- Maini, R.N.; Taylor, P.C.; Szechinski, J.; Pavelka, K.; Bröll, J.; Balint, G.; Emery, P.; Raemen, F.; Petersen, J.; Smolen, J.; et al. Double-blind randomized controlled clinical trial of the interleukin-6 receptor antagonist, tocilizumab, in European patients with rheumatoid arthritis who had an incomplete response to methotrexate. Arthritis Rheum. 2006, 54, 2817–2829.
- Cardona-Pascual, I.; Berlana, D.; Martinez-Valle, F.; Campany-Herrero, D.; Montoro-Ronsano, J.B. Effect of tocilizumab versus standard of care in adults hospitalized with moderate-severe COVID-19 pneumonia. Med. Clin. (Engl. Ed.) 2022, 158, 301–307.
- Antwi-Amoabeng, D.; Kanji, Z.; Ford, B.; Beutler, B.D.; Riddle, M.S.; Siddiqui, F. Clinical outcomes in COVID-19 patients treated with tocilizumab: An individual patient data systematic review. J. Med. Virol. 2020, 92, 2516–2522.
- WHO. A living WHO guideline on drugs to prevent COVID-19. Br. Med. J. 2021, 372, n526.
- West, W. Continuous infusion recombinant interleukin-2 (rIL-2) in adoptive cellular therapy of renal carcinoma and other malignancies. Cancer Treat. Rev. 1989, 16, 83–89.
- Berraondo, P.; Sanmamed, M.F.; Ochoa, M.C.; Etxeberria, I.; Aznar, M.A.; Pérez-Gracia, J.L.; Rodriguez-Ruiz, M.E.; Ponz-Sarvise, M.; Castañón, E.; Melero, I. Cytokines in clinical cancer immunotherapy. Br. J. Cancer 2019, 120, 6–15.
- Nagarsheth, N.; Wicha, M.S.; Zou, W. Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nat. Rev. Immunol. 2017, 17, 559–572.
- Sayana, S.; Khanlou, H. Maraviroc: A new CCR5 antagonist. Expert Rev. Anti-Infect. Ther. 2009, 7, 9–19.
- Ndegwa, S. Maraviroc (Celsentri) for multidrug-resistant human immunodeficiency virus (HIV)-1. Issues Emerg. Health Technol. 2007, 110, 1–8.
- Rallis, K.S.; Corrigan, A.E.; Dadah, H.; George, A.M.; Keshwara, S.M.; Sideris, M.; Szabados, B. Cytokine-based Cancer Immunotherapy: Challenges and Opportunities for IL-10. Anticancer. Res. 2021, 41, 3247–3252.
- Roufas, C.; Chasiotis, D.; Makris, A.; Efstathiades, C.; Dimopoulos, C.; Zaravinos, A. The Expression and Prognostic Impact of Immune Cytolytic Activity-Related Markers in Human Malignancies: A Comprehensive Meta-analysis. Front. Oncol. 2018, 8, 27.
- Liu, M.; Liu, L.; Song, Y.; Li, W.; Xu, L. Targeting macrophages: A novel treatment strategy in solid tumors. J. Transl. Med. 2022, 20, 586.
- Choudhry, H.; Helmi, N.; Abdulaal, W.H.; Zeyadi, M.; Zamzami, M.A.; Wu, W.; Mahmoud, M.M.; Warsi, M.K.; Rasool, M.; Jamal, M.S. Prospects of IL-2 in Cancer Immunotherapy. BioMed Res. Int. 2018, 2018, 9056173.
- Yui, M.A.; Sharp, L.L.; Havran, W.L.; Rothenberg, E.V. Preferential Activation of an IL-2 Regulatory Sequence Transgene in TCRγδ and NKT Cells: Subset-Specific Differences in IL-2 Regulation. J. Immunol. 2004, 172, 4691–4699.
- Paliard, X.; Malefijt, R.d.W.; Yssel, H.; Blanchard, D.; Chrétien, I.; Abrams, J.; de Vries, J.; Spits, H. Simultaneous production of IL-2, IL-4, and IFN-gamma by activated human CD4+ and CD8+ T cell clones. J. Immunol. 1988, 141, 849–855.
- Hershko, A.Y.; Suzuki, R.; Charles, N.; Alvarez-Errico, D.; Sargent, J.L.; Laurence, A.; Rivera, J. Mast Cell Interleukin-2 Production Contributes to Suppression of Chronic Allergic Dermatitis. Immunity 2011, 35, 562–571.
- Granucci, F.; Vizzardelli, C.; Pavelka, N.; Feau, S.; Persico, M.; Virzi, E.; Rescigno, M.; Moro, G.; Ricciardi-Castagnoli, P. Inducible IL-2 production by dendritic cells revealed by global gene expression analysis. Nat. Immunol. 2001, 2, 882–888.
- Hinrichs, C.S.; Spolski, R.; Paulos, C.M.; Gattinoni, L.; Kerstann, K.W.; Palmer, D.C.; Klebanoff, C.A.; Rosenberg, S.A.; Leonard, W.J.; Restifo, N.P. IL-2 and IL-21 confer opposing differentiation programs to CD8+ T cells for adoptive immunotherapy. Blood 2008, 111, 5326–5333.
- Hromadnikova, I.; Li, S.; Kotlabova, K.; Dickinson, A.M. Influence of In Vitro IL-2 or IL-15 Alone or in Combination with Hsp 70 Derived 14-Mer Peptide (TKD) on the Expression of NK Cell Activatory and Inhibitory Receptors on Peripheral Blood T Cells, B Cells and NKT Cells. PLoS ONE 2016, 11, e0151535.
- Thommen, D.S.; Koelzer, V.H.; Herzig, P.; Roller, A.; Trefny, M.; Dimeloe, S.; Kiialainen, A.; Hanhart, J.; Schill, C.; Hess, C.; et al. A transcriptionally and functionally distinct PD-1+ CD8+ T cell pool with predictive potential in non-small-cell lung cancer treated with PD-1 blockade. Nat. Med. 2018, 24, 994–1004.
- Deng, H.; Liu, R.; Ellmeier, W.; Choe, S.; Unutmaz, D.; Burkhart, M.; Marzio, P.D.; Marmon, S.; Sutton, R.E.; Hill, C.M.; et al. Identification of a major co-receptor for primary isolates of HIV-1. Nature 1996, 381, 661–666.
- Simmons, G.; Wilkinson, D.; Reeves, J.D.; Dittmar, M.T.; Beddows, S.; Weber, J.; Carnegie, G.; Desselberger, U.; Gray, P.W.; Weiss, R.A.; et al. Primary, syncytium-inducing human immunodeficiency virus type 1 isolates are dual-tropic and most can use either Lestr or CCR5 as coreceptors for virus entry. J. Virol. 1996, 70, 8355–8360.
- Wu, L.; Gerard, N.P.; Wyatt, R.; Choe, H.; Parolin, C.; Ruffing, N.; Borsetti, A.; Cardoso, A.A.; Desjardin, E.; Newman, W.; et al. CD4-induced interaction of primary HIV-1 gp120 glycoproteins with the chemokine receptor CCR-5. Nature 1996, 384, 179–183.
- Tsimberidou, A.M.; Fountzilas, E.; Nikanjam, M.; Kurzrock, R. Review of precision cancer medicine: Evolution of the treatment paradigm. Cancer Treat. Rev. 2020, 86, 102019.
- Riedesser, J.E.; Ebert, M.P.; Betge, J. Precision medicine for metastatic colorectal cancer in clinical practice. Ther. Adv. Med. Oncol. 2022, 14, 17588359211072703.
- Ganesh, K. Optimizing immunotherapy for colorectal cancer. Nat. Rev. Gastroenterol. Hepatol. 2021, 19, 93–94.
- Zhao, C.; Wu, L.; Liang, D.; Chen, H.; Ji, S.; Zhang, G.; Yang, K.; Hu, Y.; Mao, B.; Liu, T.; et al. Identification of immune checkpoint and cytokine signatures associated with the response to immune checkpoint blockade in gastrointestinal cancers. Cancer Immunol. Immunother. 2021, 70, 2669–2679.
- Botticelli, A.; Pomati, G.; Cirillo, A.; Scagnoli, S.; Pisegna, S.; Chiavassa, A.; Rossi, E.; Schinzari, G.; Tortora, G.; Di Pietro, F.R.; et al. The role of immune profile in predicting outcomes in cancer patients treated with immunotherapy. Front. Immunol. 2022, 13, 974087.
- Lim, J.U.; Yoon, H.K. Potential predictive value of change in inflammatory cytokines levels subsequent to initiation of immune checkpoint inhibitor in patients with advanced non-small cell lung cancer. Cytokine 2020, 138, 155363.
- Achyut, B.R.; Yang, L. Transforming Growth Factor-β in the Gastrointestinal and Hepatic Tumor Microenvironment. Gastroenterology 2011, 141, 1167–1178.
- Desoteux, M.; Maillot, B.; Bévant, K.; Ferlier, T.; Leroux, R.; Angenard, G.; Louis, C.; Sulpice, L.; Boudjema, K.; Coulouarn, C. Transcriptomic evidence for tumor-specific beneficial or adverse effects of TGFβ pathway inhibition on the prognosis of patients with liver cancer. FEBS Open Bio 2023, 13, 1278–1290.
- Dranoff, G. Cytokines in cancer pathogenesis and cancer therapy. Nat. Rev. Cancer 2004, 4, 11–22.
- DeMaria, M.; O’Leary, M.N.; Chang, J.; Shao, L.; Liu, S.; Alimirah, F.; Koenig, K.; Le, C.; Mitin, N.; Deal, A.M.; et al. Cellular Senescence Promotes Adverse Effects of Chemotherapy and Cancer Relapse. Cancer Discov. 2017, 7, 165–176.
- Guillon, J.; Petit, C.; Toutain, B.; Guette, C.; Lelièvre, E.; Coqueret, O. Chemotherapy-induced senescence, an adaptive mechanism driving resistance and tumor heterogeneity. Cell Cycle 2019, 18, 2385–2397.
- Sriram, G.; Milling, L.E.; Chen, J.-K.; Kong, Y.W.; Joughin, B.A.; Abraham, W.; Swartwout, S.; Handly, E.D.; Irvine, D.J.; Yaffe, M.B. The injury response to DNA damage in live tumor cells promotes antitumor immunity. Sci. Signal. 2021, 14, eabc4764.
- Lotti, F.; Jarrar, A.M.; Pai, R.K.; Hitomi, M.; Lathia, J.; Mace, A.; Gantt, G.A., Jr.; Sukhdeo, K.; DeVecchio, J.; Vasanji, A.; et al. Chemotherapy activates cancer-associated fibroblasts to maintain colorectal cancer-initiating cells by IL-17A. J. Exp. Med. 2013, 210, 2851–2872.
- Reers, S.; Pfannerstill, A.C.; Rades, D.; Maushagen, R.; Andratschke, M.; Pries, R.; Wollenberg, B. Cytokine changes in response to radio-/chemotherapeutic treatment in head and neck cancer. Anticancer. Res. 2013, 33, 2481–2489.
- Edwardson, D.W.; Parissenti, A.M.; Kovala, A.T. Chemotherapy and Inflammatory Cytokine Signalling in Cancer Cells and the Tumour Microenvironment. Adv. Exp. Med. Biol. 2019, 1152, 173–215.
- Van der Sijde, F.; Dik, W.A.; Mustafa, D.A.M.; Vietsch, E.E.; Besselink, M.G.; Debets, R.; Koerkamp, B.G.; Haberkorn, B.C.M.; Homs, M.Y.V.; Janssen, Q.P.; et al. Serum cytokine levels are associated with tumor progression during FOLFIRINOX chemotherapy and overall survival in pancreatic cancer patients. Front. Immunol. 2022, 13, 898498.
