Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Maria-Anna Gatou and Version 2 by Lindsay Dong.
Magnesium oxide (MgO) nanoparticles have excellent biocompatibility, stability, and diverse biomedical uses, such as antimicrobial, antioxidant, anticancer, and antidiabetic properties, as well as tissue engineering, bioimaging, and drug delivery applications. Magnesium oxide nanoparticles demonstrate substantial biocompatibility and display significant antibacterial, antifungal, anticancer, and antioxidant properties.
- magnesium oxide nanoparticles
- MgO
- Nanoparticles
1. Introduction
In recent times, nanotechnology has become a focal point in research owing to its diverse applications across scientific and technological domains. This field focuses on crafting nanoparticles (NPs) and harnessing their potential in areas like biomedicine, sensing, and catalysis [1]. The significance of NPs stems from their unique properties, such as small size, adaptable shapes, substantial surface area compared to volume, and outstanding magnetic, electronic, optical, and mechanical traits [2][3][4][5][2,3,4,5]. Yet, the distinctiveness of each nanoparticle primarily relies on the utilized synthesis method [6]. In general, nanoparticles have been synthesized using a variety physical, chemical, and green approaches [7][8][9][10][11][12][7,8,9,10,11,12].
Metal oxide nanoparticles represent a crucial category of nanomaterials extensively utilized today due to their distinct physical and chemical properties, finding applications across diverse fields like biosensing technology, tissue engineering, catalysis, food packaging, biomedicine, and environmental sciences [13]. The key members within the category of metal oxide nanoparticles include silicon dioxide (SiO2), ferric oxide (Fe2O3), copper oxide (CuO), zinc oxide (ZnO), titanium dioxide (TiO2), and magnesium oxide (MgO) [14]. Among these metal oxide nanoparticles, magnesium oxide (MgO) nanoparticles have gained considerable attention because of their exceptional biocompatibility, non-toxic nature, robust stability in abrupt conditions, and extensive applications, especially in biomedicine [15]. Furthermore, the United States Food and Drug Administration regards magnesium oxide as a safe material for human consumption [16]. MgO nanoparticles possess several advantageous physicochemical characteristics, such as enhanced ionic character, substantial specific surface area, distinctive crystal structures, as well as oxygen vacancies, enabling seamless interaction with various biological systems [17][18][17,18]. These nanoparticles have found widespread utility in diverse areas, including toxic waste remediation, paints, antiseptics, catalysis, superconductors, catalytic devices, semiconductors, additives in heavy fuel oils, refractory materials, adsorbents, reflective coatings, lithium-ion batteries, and more [19][20][21][19,20,21]. In the realm of biomedicine, magnesium oxide nanoparticles have been employed for stomach relief, heartburn alleviation, and bone regeneration [21][22][21,22], as well as for therapeutic applications, such as coated capsules, biological labeling, band-aids, blood collecting vessels, etc. [19]. Additionally, MgO nanoparticles have exhibited potential as antibacterial [21][22][23][21,22,23], fungicidal [24], anticancer [25][26][25,26], antioxidant [27], and antidiabetic [27] agents, as well as in applications such as tissue engineering [28][29][28,29], bioimaging [30], and drug delivery [31]. Hence, the pursuit of novel synthetic methods for producing magnesium oxide nanoparticles becomes imperative owing to their escalating usage in biomedicine.
2. Biomedical Applications of MgO Nanoparticles
2.1. Antibacterial Activity
In recent times, the rise of bacterial resistance to commonly used antibiotics has significantly impacted the effective treatment of bacterial infections. The United Nations General Assembly has highlighted antibiotic resistance as a critical global peril that humanity confronts [32][147]. Consequently, exploring alternative strategies to combat bacterial growth has become imperative, with nanoparticles emerging as a promising solution, due to their strong antibacterial properties. Among these nanoparticles, MgO nanoparticles have garnered attention owing to their remarkable effectiveness in combating bacteria. Studies have indicated the potent MgO nanoparticles’ antibacterial effects against various strains, such as E. coli [33][134], S. aureus [34][148], P. aeruginosa, A. baumannii [35][149], and P. carotovorum [36][150]. Additionally, a proposed antibacterial mechanism of MgO nanoparticles is depicted in Figure 19. More specifically, magnesium oxide nanoparticles have the ability to stimulate reactive oxygen species (ROS) within bacteria. This process leads to oxidative stress, resulting in significant impairment to their membrane lipids, proteins, and nucleic acids [37][151].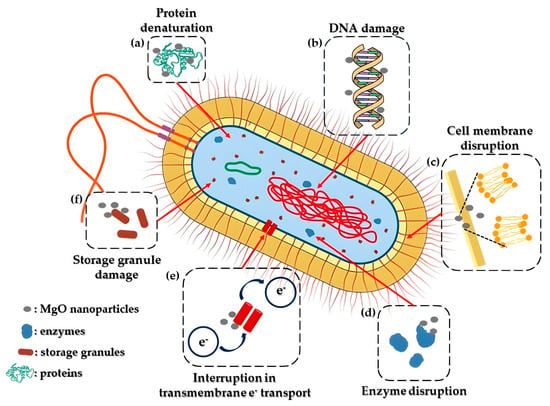
Figure 19. Representation of a feasible mechanism of MgO nanoparticles’ antibacterial activity. The entry of MgO nanoparticles into bacterial cells is facilitated by the disturbance of the bacterial cell membrane (c). Once inside the cytoplasm, these nanoparticles have the capacity to produce reactive oxygen species (ROS) or inflict direct harm on DNA and enzymes (b,d). This action results in protein denaturation and damage to the mitochondria (a,f). Additionally, they interfere with cellular memory and impede trans-tolerant electron transport (e). Consequently, the inflicted harm leads to the destruction of bacterial cells, prompting the release of their organelles and culminating in their eventual demise.
Makhluf and colleagues [38][152] demonstrated the antibacterial properties of powdered magnesium oxide prepared through a microwave-assisted synthetic approach, applying it to combat both Staphylococcus aureus and Escherichia coli cultures. The most substantial antibacterial effect was observed when subjecting both bacterial strains to 8 nm magnesium oxide nanoparticles. Following exposure to MgO for 60 min, less than one fifth of both cultures survived. Subsequently, after 4 h of treatment, the survival rates decreased significantly to less than 5% for Staphylococcus aureus and a mere 0.1% for Escherichia coli. In contrast, using 23 nm MgO resulted in a reduction in bacterial counts to approximately 40% for S. aureus and 35% for E. coli, comparatively less effective than the 8 nm particles.
Mechanism of MgO Nanoparticles’ Bactericidal Activity
The effectiveness of MgO nanoparticles in eradicating bacteria largely relies on reactive oxygen species (ROS) production. This reliance is specifically associated with factors such as the particle’s surface characteristics, polarity, crystal size, increased oxygen defects, morphology, the ability of molecules to chemically diffuse, as well as the release of Mg2+ ions. The bactericidal process involving magnesium oxide nanoparticles entails sterilization through particle release, a multifaceted mechanism, and absorption. The crystals’ size and their extensive surface area may contribute significantly to their potent antibacterial properties. Superoxide radicals, formed through reactions between H2O2 and ROS, inflict damage on cellular proteins and DNA, leading to cell death [39][160].
The mechanism underlying the antimicrobial efficacy of magnesium oxide nanostructures can be elucidated as follows. Primarily, a crucial antibacterial process involves a light-driven catalytic mechanism. Specifically, the creation of ROS on the nanoparticle surface, in the presence of light, initiates oxidative stress on microbial cells, ultimately leading to cellular demise. ROS comprises relatively low levels of toxic radicals like the superoxide anion radical (O2−), reactive hydroxyl radical (·OH), and a mild oxidizing agent, hydrogen peroxide (H2O2). A subsequent series of reactions also takes place, where the superoxide anion radical (•O2−) combines with hydrogen ions, forming the •HO2− radical, which then reacts with hydrogen ions to produce H2O2. This compound can interact with DNA, cellular proteins, and cell membranes, ultimately resulting in microbial death. The generation of a substantial amount of ROS hinges on the creation of smaller crystalline structures with increased specific surface areas and a corresponding rise in surficial defects.
2.2. Antifungal Activity
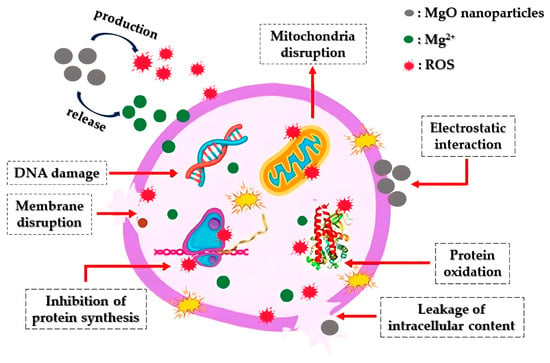
Apart from showcasing antibacterial effects, MgO nanoparticles have demonstrated significant antifungal capabilities against various pathogenic fungal strains (Figure 211). Fungi constitute natural pathogens as they present plenty of similarities with the host cell, inhibiting antifungal compounds’ growth [40][161]. Sierra-Fernandez and co-researchers [41][162] studied the antifungal activity of Zn-doped magnesium oxide nanoparticles synthesized through a facile sol-gel approach and compared it with that of pure ZnO and MgO nanoparticles. The as-mentioned nanoparticles presented enhanced antifungal efficiency compared to that of pure magnesium oxide or zinc oxide nanoparticles, restraining the growth of fungi Aspergillus niger, Penicillium oxalicum, Paraconiothyrium sp., and Pestalotiopsis maculans.
Figure 211. Diagrammatic representation of the way that MgO nanoparticles operate as an antifungal agent: Initially, the nanoparticles engage with fungal cell membranes through electrostatic interactions, leading to the disruption of both the membranes and the glucan matrix. Following this, they initiate the production of ROS and the release of Mg2+. Subsequently, they interfere with mitochondria by inducing DNA damage, subsequently impeding protein synthesis, disrupting proteins, leading to intracellular leakage, and ultimately resulting in the demise of fungal cells.
Moreover, De la Rosa-García and co-workers [42][163] evaluated the antifungal activity (towards strains of C. gloeosporioides) of pure ZnO and MgO, as well as ZnO/MgO and ZnO/Mg(OH)2 composites fabricated under different synthetic approaches (co-precipitation and hydrothermal). According to the acquired results, all tested nanoparticles at the tested concentrations significantly restrained conidia’s germination and led to the structural damage of the fungal cells, verifying that the as-mentioned nanoparticles could constitute promising fungicidal agents against C. gloeosporioides.
In addition, Castillo and his team [24] demonstrated through in vitro experiments that magnesium oxide nanoparticles, characterized by a diameter equal to 12 nm, presented fungistatic efficiency towards three filamentous fungal strains (T. reesei, A. niger, and C. cladosporioides), at concentrations ranging from 3 to 12 mg/mL.
2.3. Anticancer Activity
Cancer stands as one of the most lethal and intricate diseases known to date. The unregulated growth of cancerous cells detrimentally impacts neighboring healthy cells, leading to fatality [43][174]. Various treatments, including surgery, radiation therapy, and chemotherapy, have been proposed for combating cancer [44][175]. Nonetheless, the aforementioned approaches may harm normal cells, resulting in numerous side effects and potential disease recurrence [45][176]. Recently, there has been a rising focus on the development of nanoparticle-based nanomaterials, notably magnesium oxide nanoparticles, renowned for their potent anti-cancer properties. Figure 13 portrays a hypothetical anti-cancer mechanism associated with magnesium oxide nanoparticles. The advancement of synthesis methods and techniques has significantly propelled the application of magnesium oxide nanoparticles in anti-cancer therapy.
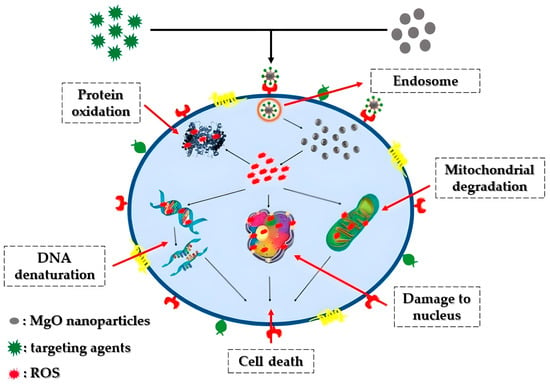

Figure 13. Representation of the potential anticancer mechanism involving MgO nanoparticles: Through electrostatic interactions with the cell surface, MgO nanoparticles gain entry into the cell via the intracellular pathway. Once inside, these nanoparticles prompt the formation of reactive oxygen species (ROS) within the cells, resulting in DNA damage, protein oxidation, and mitochondrial impairment, ultimately culminating in cell death.
Behzadi and his team [46][177] reported that the tested magnesium oxide nanoparticles presented selective cytotoxicity against the K562 cell line, thus rendering them as a novel anticancer agent. Their study’s data revealed that MgO nanoparticle-mediated apoptosis was initiated through reactive oxygen species generation within the cancer cells.
Additionally, magnesium oxide nanoparticles synthesized through a simple sol-gel method and further modified with polyethylene glycol were successfully fabricated by Alfaro and his team [47][178] to be utilized as carrier for the anticancer drug 2-Methoxyestradiol for advancing its clinical utilization. According to their research’s results, the as-developed nanoparticles significantly reduced the viability of a prostate cancer cell line (LNCap), rendering the as-mentioned nanocomposite appropriate as a drug delivery system towards anticancer prostate therapy.
2.4. Antioxidant Activity
For a considerable duration, researchers have established that free radicals negatively impact human health, potentially leading to various illnesses including heart disease, arteriosclerosis, tumors, diabetes, and aging [48][187]. Consequently, the focus on understanding antioxidants has expanded to counteract the free radicals’ detrimental effects. However, despite their effectiveness, potent antioxidant agents also bring along several adverse effects. For instance, Edaravone, acknowledged as a free radical scavenger beneficial in preventing lipid oxidation and treating ischemic stroke, mitigates nerve cell damage. Yet, when utilized clinically, Edaravone induces numerous side effects, including liver and kidney toxicity, which can impact human health [49][188]. Hence, exploring antioxidant capabilities via enhanced magnesium oxide nanoparticles presents a possible solution to mitigate several associated drawbacks. Podder and colleagues [50][189] explored the antioxidant activity of three nano-MgO structures (i.e., nanoparticles, nanoplates, and nanorods). They reported the effective production of superoxide anions (•O2−) and hydroxyl radicals (•OH) at increased concentrations (>500 μg/mL) and the scavenging of •O2− at lower concentrations (40 μg/mL) for all examined nanostructures. More specifically, it was observed that magnesium oxide nanorods produce the most increased levels of superoxide anions, while magnesium oxide nanoparticles possessed the most enhanced ability (60%) to scavenge superoxide anions. Lastly, the researchers also reported a 100% scavenging ability of the nitrogen-centered free radical (DPPH) by magnesium oxide nanoplates, given their significantly enhanced specific surface area (342.2 m2/g). Magnesium oxide nanoparticles (42 nm) were successfully developed utilizing geranium leaf extract by Mylarappa et al. [51][191]. The antioxidant characteristics of the synthesized nanoparticles were evaluated using the DPPH method. Based on the obtained results, MgO nanoparticles displayed significant efficacy in scavenging free radicals, as demonstrated by their DPPH scavenging activities.2.5. MgO-Based Biosensors towards Diabetes Detection and Treatment
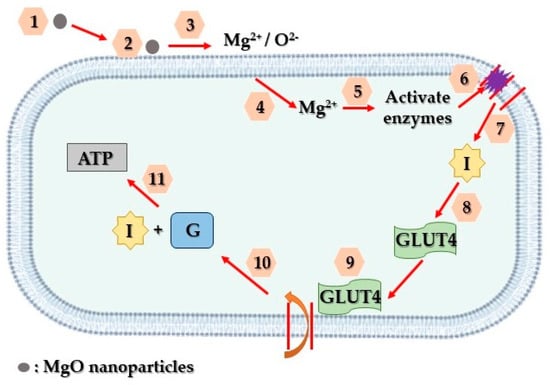
Diabetes comprises a collection of severe and enduring metabolic disorders associated with elevated blood glucose levels, contributing to increased rates of premature morbidity. In spite of advancements in life’s quality, there hasn’t been a decline in diabetes prevalence; instead, it continues to strain global healthcare systems [52][195]. Often diagnosed after irreversible organ damage due to prolonged hyperglycemia, diabetes stands among the most pressing global health challenges [52][195], alongside cancer, chronic respiratory issues, and cardiovascular diseases, causing approximately five million deaths annually in developed nations. Notably, cardiovascular disease (50%) and kidney failure (10–20%) account for the majority of these fatalities. Diabetes also leads to complications like blindness, lower limb amputations, and severe outcomes in viral infections, such as COVID-19 [53][196]. The primary diabetes types include insulin-dependent or juvenile diabetes (T1DM-Type 1 Diabetes Mellitus), non-insulin-dependent (T2DM-Type 2 Diabetes Mellitus), and gestational diabetes [54][197]. The initial and crucial aspect of managing diabetes involves diagnosis. Presently, traditional methods such as assessing fasting plasma glucose (FPG) levels, conducting oral glucose tolerance tests (OGTT), and measuring hemoglobin A1c (HbA1c) levels [55][56][200,201] are employed for diabetes diagnosis. However, the aforementioned methods are often uncomfortable and painful for patients, due to blood withdrawal, leading to potential neglect of therapy. Additionally, periodic measurements might not capture significant fluctuations in glucose levels between testing intervals. Moreover, variations in measured values can occur due to factors like timing of testing, age, and an individual’s physiological state. These approaches are also unsuitable for continuous monitoring due to their laborious nature, prolonged diagnosis duration, increased blood withdrawal, and complex blood processing [57][202]. Notably, clinical signs of detrimental diabetes symptoms, like hyperglycemia, are usually observed only after the disease has progressed, hindering early intervention. To mitigate these complications, it is crucial to develop diagnostic tools that are more affordable, rapid, and widely accessible [58][199]. Addressing these challenges, various nanotechnologies focusing on diverse biomarkers have emerged, aiming to enable early and non-invasive diabetes detection. Analyzing specific biomarkers serves as an indicator for multiple diseases [59][203]. Lately, there has been a notable emphasis on advancing research related to diabetes treatment through the utilization of nanoparticles. Magnesium oxide nanoparticles, in particular, have gained substantial attention among various nanoparticles and are extensively employed in biomedical studies, particularly in diabetes treatment, through a suggested mechanism depicted in Figure 415.
Figure 415. Schematic representation of the way that MgO nanoparticles contribute to diabetes treatment. Firstly, MgO nanoparticles adhere to the surface of affected cells (1, 2, 3). Subsequently, they discharge Mg2+ and O2− ions, initiating the activation of internal enzymes (4, 5). These enzymes, in turn, facilitate the reversal of insulin resistance and facilitate the entry of glucose transporter 4 into the cell’s plasma membrane (6, 7, 8). Ultimately, this glucose transporter enables the absorption of glucose into the cells, where insulin functions to decrease glucose levels and generate ATP (9, 10, 11).
2.6. Tissue Engineering Applications
2.6.1. Bone Tissue Engineering
Enhanced biomaterials aimed at restoring bone integrity are necessary to address the escalating number of individuals grappling with deteriorating or damaged bones [60][61][62][63][209,210,211,212]. Presently, existing biomaterial solutions for this purpose involve invasive procedures that introduce permanent materials, potentially leading to prolonged issues within the body. Although progress has been made in bone defect regeneration using injectable cements and various scaffold materials, significant enhancements are still needed [64][65][66][213,214,215]. The ideal biomaterials for bone tissue regeneration should harmonize mechanically with surrounding tissue, lessening stress and strain discrepancies, while also possessing suitable chemical compositions and surface features that foster bone cell adhesion, growth, movement, and the production of proteins forming the extracellular matrix.
Furthermore, Suryavanshi and co-researchers [67][218] evaluated the suitability of electrospun polycaprolactone polymer composites loaded with magnesium oxide nanoparticles as scaffolds for bone-soft tissue engineering. Magnesium oxide nanoparticles were synthesized using a hydroxide precipitation sol-gel process. The nanocomposites exhibited significantly improved mechanical properties compared to the pure polymer samples, due to the even dispersion of MgO nanoparticles throughout the polymer fibers. In immersion tests, the nanocomposite scaffolds displayed notable bioactivity by developing a surface hydroxyapatite layer by the third day of incubation. The electrospun polymer mats loaded with magnesium oxide nanoparticles demonstrated enhanced in vitro biological performance with osteoblast-like MG-63 cells, showing increased adhesion, proliferation, and enhanced differentiation marker activity.
