Compositional changes in soil microbes associated with decreases in abundance and species diversity were reported, especially in heavily contaminated areas of both Chernobyl and Fukushima, which may accompany explosions of radioresistant species. In Chernobyl, the population size of soil microbes remained low for at least 20 years after the accident, and the abundance of plant-associated microbes, which are related to the growth and defense systems of plants, possibly decreased. These reported changes in microbes likely affect soil conditions and alter plant physiology. These microbe-mediated effects may then indirectly affect insect herbivores through food-mass-mediated, pollen-mediated, and metabolite-mediated interactions. Metabolite-mediated interactions may be a major pathway for ecological impacts at low pollution levels and could explain the decreases in insect herbivores in Fukushima.
- radioactive pollution
- nuclear power plant accident
- Chernobyl
- Fukushima
- soil microbes
- plant-associated microbes
1. Introduction
2. Multiple Pathways for Biological Effects
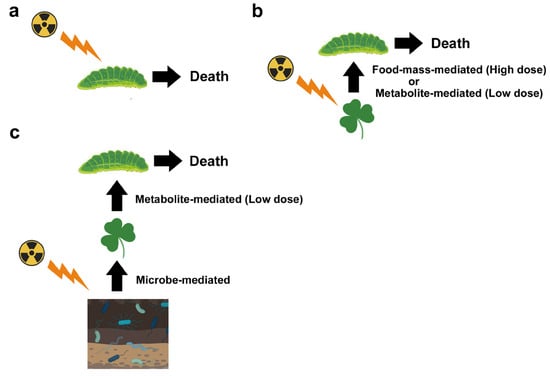
First, one should recognize that there are multiple pathways for biological effects of a nuclear accident. Here, one should be careful about the terminology. At the molecular level, “direct” damage occurs when ionizing radiation directly breaks the covalent bonds of biological molecules (especially those of DNA), and “indirect” damage occurs via reactive oxygen species (ROS) produced by water radiolysis [23][20]. At the individual (organismal) level, both direct and indirect molecular mechanisms for ionization are considered “direct” effects on a given organism. As a secondary effect of these “direct” effects, several “indirect” effects may occur. For example, the sole host plant of the pale grass blue butterfly may synthesize insect toxins in response to irradiation, and butterfly larvae die due to ingested toxins (Figure 1). Changes in metabolites in the host plant may be considered direct or primary effects, and if so, larval death is the secondary effect. However, the primary and secondary effects may not always be fixed. Plant metabolite changes may be caused by soil microbes and plant-associated microbes, and if so, these microbial changes are the primary effects; plant metabolite changes should be considered secondary effects, and larval death should be considered to occur through tertiary effects. In reality, soil microbes, plant-associated microbes, plants, and insects are exposed to radiation simultaneously. That is, these direct effects and indirect field effects work simultaneously in the field.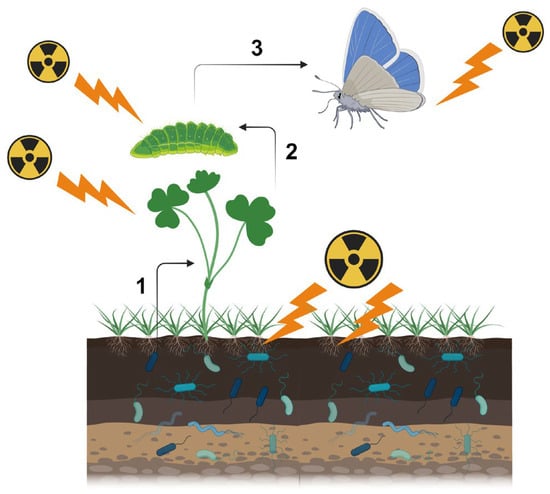
3. Soil Microbes and Soil Invertebrates
3.1. Chernobyl Studies
3.2. Fukushima Studies
The Fukushima nuclear accident occurred in March 2011, 25 years after the Chernobyl nuclear accident. Although advanced research methods and techniques have become more accessible, only a handful of studies on soil microbes have been documented in Fukushima. Ihara et al. (2021) [92][48] explored the soil bacterial community at the base of mugwort via high-throughput sequencing. They approached 1 km to the NPP at the closest location, where the 137Cs concentration in the soil sample was 563 kBq/kg (dry) in 2014. Notably, for comparison, soil samples were collected from four geographically remote sites with the same vegetation and land use. The authors demonstrated the following three points in terms of bacterial communities: at the most contaminated site, (i) the species diversity was lower, (ii) the composition was different, and (iii) the radioresistant bacterium Geodermatophilus bullaregiensis was more abundant. Similarly, Higo et al. (2019) [93][49] examined the community dynamics of the arbuscular mycorrhizal fungus colonizing the roots of napiergrass Pennisetum purpureum under different land uses (paddy field and grassland) before an accident within 30 km of the Fukushima NPP in 2013 and 2014. The deposition density of 137Cs was 3404 kBq/m2 in paddy fields and 3322 kBq/m2 in grasslands at the time of 2013. Illumina MiSeq sequencing data revealed that species diversity was lower in 2014 for both land-use types and that the species composition differed between sampling years and between land-use types. The most abundant family, Glomeraceae, may be tolerant of complex environments [94,95][50][51].3.3. Commonalities between Chernobyl and Fukushima
Declines in the abundance and species diversity and compositional differences in soil microbes have been reported both in Chernobyl and Fukushima, and some microbes with radioresistance or accumulation of 137Cs have been reported in both studies; however, in highly contaminated areas in Chernobyl, composition and species diversity may not follow these rules. Furthermore, the 137Cs concentration in leaf litter increased during the decomposition process in Chernobyl and Fukushima, and the movement of 137Cs in soil was potentially mediated by microbes to organisms in the soil and on the ground through trophic connections. As 137Cs is cycled and maintained in the environment over time, the ecological half-life becomes much longer than initially estimated. Species diversity is understood to decrease due to the simultaneous elimination of more radiosensitive species and also due to the increase in radioresistant species and immigrants [17][16]. In other words, radioresistance is a key trait for the abundance, species diversity, and composition of overall microbial communities in soil. Additionally, in general, radioresistant bacteria are resistant to ultraviolet rays [100,101][52][53] and dryness [102][54], suggesting that soil microbes adapt flexibly to various environmental stressors. Therefore, the abundance, species diversity, and composition of radioresistant microbes in the field seem to fluctuate on a large scale in response to stressor types.4. Plant-Associated Microbes
4.1. Chernobyl Studies
Studies on herbaceous plants were initiated as early as 1986 (see also Section 5). Geras’kin et al. (2002) [105][55] collected seeds of winter rye within 30 km of the Chernobyl NPP approximately 4 months after the accident, and after germination, the plants were subjected to cytogenetic tests. In a different study, germination of wild carrot seeds from maternal plants exposed to radiation in Chernobyl showed the lower gemination rate and other abnormal life-history traits [106][56]. However, plant-associated microbes have rarely been explored. Mousseau et al. (2014) [73][57] suggested that the reduced rate of litter mass loss and thicker forest floor (poor levels of decomposition in other words) in the 30 km zone of Chernobyl could have an effect on growth conditions for plants because free-living microbes strongly regulate plant productivity through mineralization during the decomposition process, which makes nutrients such as nitrogen and phosphorus available to plants [107][58]. Several papers have noted that the effect of radiation exposure on plants is a weakened defense system when radiation levels are relatively high. A decrease in the disease resistance of wheat, rye, and maize was observed within 10 km of the NPP, and in fact, brown rust and true mildew infection increased in winter wheat, corresponding to radioactive contamination [11][10]. Simultaneously, the emergence of a new causal agent of stem rust, Puccinia graminis, with a high frequency of more virulent clones was detected within 10 km [11,114][10][59]. Thus, the prevalence of plant diseases in Chernobyl could be caused by both reduced disease resistance and enhanced toxicity.4.2. Fukushima Studies
Within the context of even fewer studies on plant-associated microbes in Fukushima, Sakauchi et al. (2022) [125][60] subjected the field-picked creeping wood sorrel Oxalis corniculata to LC–MS analysis to quantify secondary metabolites. The radiation level ranged from nondetectable to 718 Bq/kg for the 137Cs radioactivity concentration in the leaves and from 0.04 μGy/h to 4.55 μGy/h for the ground dose rate at which the leaves grew. This study demonstrated that Oxalis leaves, which were field-picked in Fukushima and looked completely healthy to the naked eye, upregulated and downregulated secondary metabolites in response to low-dose radiation exposure [125][60]. In addition to these findings, Zhu et al. (2021) [108][61] studied contaminated soil samples containing three different 137Cs concentrations (low: 20–40 Bq/kg, medium: 40–60 Bq/kg, and high: >60 Bq/kg) from a historic nuclear test site in China and found that the richness of the endophytic bacteria in the roots of Kalidium schrenkianum was significantly greater only in low-radiation soil than in the control soil. Thus, endophytes could sensitively change their abundance in response to low-radiation exposure.4.3. Commonalities between Chernobyl and Fukushima
In Chernobyl, various adverse effects have been observed on plants, including morphological changes, disturbances in growth, suppressed reproductive ability, death, disease, and pest infections [13][12]. A few of these events have also been observed in Fukushima [131,132,133,134][62][63][64][65]. It is highly likely that plant-associated microbes are involved in these observations, although no causal relationship has been demonstrated thus far. No reports of poor growth, disease, or pest infections were available in Fukushima, despite the large number of crop fields and fruit trees in the contaminated area. Partially considering Mousseau et al. (2014) [73][57] and Zhu et al. (2021) [108][61], plant-associated microbes may become less common in relatively severely contaminated areas in Chernobyl and Fukushima, possibly leading to growth failure and low immunity in plants. For example, arbuscular mycorrhizal fungi (AMF) that colonize plant roots and form symbiotic associations with 80% of terrestrial plant species are generally accepted to contribute to plant growth by facilitating the production of growth hormones and phosphorus uptake. The antibacterial endophytic fungus Streptomyces galbus improved resistance to Pestalotia disease, root rot, and anthracnose and was inoculated for practical use on flowering plants such as Rhododendron [135][66]. On the other hand, based on the LC–MS analysis of Oxalis leaves in Fukushima, the abundance of Streptomyces sp., which produces antibiotics, did not always decrease [125][60].5. Plants and Insect Herbivores
5.1. Food-Mass-Mediated Indirect Effects
Early studies, mostly conducted in 1986 at the time of the Chernobyl accident, reported reproductive degradation in various herbaceous plants [11][10]: a reduced number of seeds or a lower germination rate in winter wheat, cocksfoot Dactylis glomerata, and ribwort plantain Plantago lanceolata, and sterility in winter wheat, winter ryes, and wild vetch Vicia cracca. Taskaev et al. (1992) [146][67] observed no effect on the seeds of 15 species within 30 km of the Chernobyl NPP. Boratyński et al. (2016) [106][56] conducted a germination experiment using seeds of the wild carrot Daucus carota, collected from an abandoned field within 10 km from the Chernobyl NPP in 2012, and showed that the more radiation the maternal plants were exposed to, the longer the time that the seeds took to germinate and produce leaves and the lower the germination rate. Therefore, it is reasonable to speculate that the overall mass of phytocoenoses decreased around the Chernobyl NPP in heavily contaminated areas. The perturbation of phytocoenoses causes severe impacts on insect herbivores, which have no other option but to eat plants. Generalist herbivores may converge on surviving radioresistant plants. As a result, interspecies competition necessarily becomes more intense. In the case of specialist herbivores, survival will be difficult if their host plants are sensitive to radiation. This indirect effect through food loss, which many be called the food-mass-mediated effect, was mentioned in the early 1970s based on irradiation experiments [57][35]. The United Nations Scientific Committee on Effects of Atomic Radiation (UNSCEAR) 1996 report provided the example of a booklice, Psocoptera [17][16]. In this respect, the smaller population sizes of not only insect herbivores but also other various terrestrial organisms in Chernobyl could suggest insufficient amounts of food available for the following organisms, although direct effects on these organisms cannot be excluded: spiders [54[32][33],55], cicadas [54][32], dragonflies [54,55][32][33], butterflies [54[32][33],55], grasshoppers [54[32][33],55], bark beetles [17[16][33],55], bumblebees [54,55[32][33][34],56], booklice [17][16], springtails [17][16], soil invertebrates [57[35][36],58], reptiles [56][34], birds [54[32][37],59], and mammals [56][34].5.2. Pollen-Mediated Indirect Effects
A version of the food mass-mediated effect is the pollen-mediated effect, in which the reproductive and pollination systems of plants are specifically affected via direct irradiation. Pollens are foods for some insects, but the relationships between plants and pollinating insects (i.e., bees, butterflies, and others) are more complex than the simple predator-prey relationship. A decrease in the plant population may occur slowly through low pollen viability, resulting in a decrease in pollinating and other related insects.5.3. Metabolite-Mediated Indirect Effect
In Fukushima, the pollution level was relatively low compared to that in Chernobyl. One of the main radionuclides detected when measured was 137Cs in both Chernobyl and Fukushima, and its released amount in Fukushima was estimated to be, at most, 40% of that in Chernobyl [154][68]. This is probably why plants in Fukushima seem to be healthy, at least to the naked eye; no deleterious effects on plants have been reported, although there are a few reports on morphological abnormalities [133,134,155][64][65][69]. In this sense, food mass-mediated indirect effects (Figure 3) may not occur in Fukushima between plants and insect herbivores. Pollination also does not seem to be affected much in Fukushima [156][70]. However, lower abundances of insects such as butterflies [54,157][32][71] and cicadas [54][32] have been reported along with an increasing radiation dose. They are insect herbivores.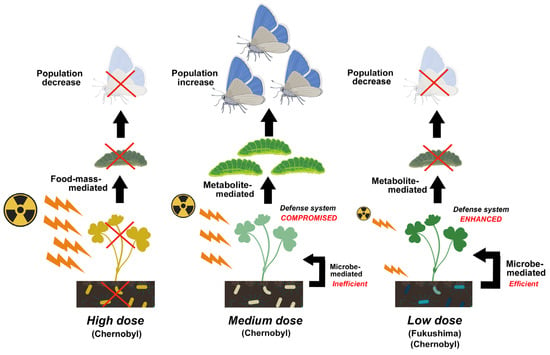
6. Conclusions
References
- Wagner, H.N., Jr. Atoms for peace (and health). J. Nucl. Med. 2004, 45, 24N.
- Babic, R.R.; Babic, G.S.; Babic, S.R.; Babic, N.R. 120 years since the discovery of x-rays. Med. Pregl. 2016, 69, 323–330.
- Nüsslin, F. Wilhelm Conrad Röntgen: The scientist and his discovery. Phys. Med. 2020, 79, 65–68.
- Brodsky, A.; Kathren, R.L.; Willis, C.A. History of the medical uses of radiation: Regulatory and voluntary standards of protection. Health Phys. 1995, 69, 783–823.
- Reed, A.B. History of radiation use in medicine. J. Vasc. Surg. 2011, 53 (Suppl. S1), 3S–5S.
- Scatliff, J.H.; Morris, P.J. From Röntgen to magnetic resonance imaging: The history of medical imaging. N. C. Med. J. 2014, 75, 111–113.
- Ma, L.; Kong, F.; Sun, K.; Wang, T.; Guo, T. From classical radiation to modern radiation: Past, present, and future of radiation mutation breeding. Front. Public Health 2021, 9, 768071.
- Riviello-Flores, M.d.l.L.; Cadena-Iñiguez, J.; Ruiz-Posadas, L.d.M.; Arévalo-Galarza, M.d.L.; Castillo-Juárez, I.; Hernández, M.S.; Castillo-Martínez, C.R. Use of gamma radiation for the genetic improvement of underutilized plant varieties. Plants 2022, 11, 1161.
- Whitmore, G.F. One hundred years of X rays in biological research. Radiat. Res. 1995, 144, 148–159.
- Geras’kin, S.A.; Fesenko, S.V.; Alexakhin, R.M. Effects of non-human species irradiation after the Chernobyl NPP accident. Environ. Int. 2008, 34, 880–897.
- Yoshida, N.; Kanda, J. Geochemistry. Tracking the Fukushima radionuclides. Science 2012, 336, 1115–1116.
- International Atomic Energy Agency (IAEA). Environmental Consequences of the Chernobyl Accident and their Remediation: Twenty Years of Experience; Radiological Assessment Reports Series No. 8; IAEA: Vienna, Austria, 2006.
- Aliyu, A.S.; Evangeliou, N.; Mousseau, T.A.; Wu, J.; Ramli, A.T. An overview of current knowledge concerning the health and environmental consequences of the Fukushima Daiichi Nuclear Power Plant (FDNPP) accident. Environ. Int. 2015, 85, 213–228.
- Ministry of the Environment (Government of Japan). Chapter 4. Concept of radiological protection. In BOOKLET to Provide Basic Information Regarding Health Effects of Radiation, 3rd ed.; Ministry of the Environment (Government of Japan): Tokyo, Japan, 2016; Available online: https://www.env.go.jp/en/chemi/rhm/basic-info/1st/04.html (accessed on 13 January 2024).
- International Atomic Energy Agency (IAEA). Effects of Ionizing Radiation on Plants and Animals at Levels Implied by Current Radiation Protection Standards; Technical Reports Series No. 332; IAEA: Vienna, Austria, 1992.
- United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR). Sources and Effects of Ionizing Radiation UNSCEAR 1996 Report to the General Assembly, with Scientific Annex; United Nations: New York, NY, USA, 1996.
- European Commission. Protection of the Environment; European Union: Luxembourg, 2014; Available online: https://energy.ec.europa.eu/system/files/2014-11/177_0.pdf (accessed on 13 January 2024).
- Hiyama, A.; Nohara, C.; Taira, W.; Kinjo, S.; Iwata, M.; Otaki, J.M. The Fukushima nuclear accident and the pale grass blue butterfly: Evaluating biological effects of long-term low-dose exposures. BMC Evol. Biol. 2013, 13, 168.
- Garnier-Laplace, J.; Geras’kin, S.; Della-Vedova, C.; Beaugelin-Seiller, K.; Hinton, T.G.; Real, A.; Oudalova, A. Are radiosensitivity data derived from natural field conditions consistent with data from controlled exposures? A case study of Chernobyl wildlife chronically exposed to low dose rates. J. Environ. Radioact. 2013, 121, 12–21.
- Otaki, J.M. Understanding low-dose exposure and field effects to resolve the field-laboratory paradox: Multifaceted biological effects from the Fukushima nuclear accident. In New Trends in Nuclear Science; Awwad, N.S., AlFaify, S.A., Eds.; InTechOpen: London, UK, 2018; pp. 49–71.
- Otaki, J.M.; Sakauchi, K.; Taira, W. The second decade of the blue butterfly in Fukushima: Untangling the ecological field effects after the Fukushima nuclear accident. Integr. Environ. Assess. Manag. 2022, 18, 1539–1550.
- Beaugelin-Seiller, K.; Della-Vedova, C.; Garnier-Laplace, J. Is non-human species radiosensitivity in the lab a good indicator of that in the field? Making the comparison more robust. J. Environ. Radioact. 2020, 211, 105870.
- Woodwell, G.M.; Sparrow, A.H. Predicted and observed effects of chronic gamma radiation on a near-climax forest ecosystem. Radiat. Botany 1963, 3, 231–237.
- Caffrey, E.; Leonard, M.; Napier, J.; Neville, D.; Higley, K. Radioecology: Why Bother? J. Environ. Prot. 2014, 5, 181–192.
- Bradshaw, C.; Kapustka, L.; Barnthouse, L.; Brown, J.; Ciffroy, P.; Forbes, V.; Geras’kin, S.; Kautsky, U.; Bréchignac., F. Using an Ecosystem Approach to complement protection schemes based on organism-level endpoints. J. Environ. Radioact. 2014, 136, 98–104.
- Brèchignac, F.; Doi, M. Challenging the current strategy of radiological protection of the environment: Arguments for an ecosystem approach. J. Environ. Radioact. 2009, 100, 1125–1134.
- Woodwell, G.M. Ecological Effects of Nuclear War; Brookhaven National Laboratory: Upton, NY, USA, 1963.
- Bréchignac, F.; Alexakhin, R.; Godoy, J.M.; Oughton, D.; Sheppard, S.; Strand, P. Integrating environment protection, a new challenge: Strategy of the International Union of Radioecology. Radioprotection 2008, 43, 339–356.
- Dalkvist, T.; Topping, C.J.; Forbes, V.E. Population-level impacts of pesticide-induced chronic effects on individuals depend more on ecology than toxicology. Ecotoxicol. Environ. Saf. 2009, 72, 1663–1672.
- Clements, W.H.; Rohr, J.R. Community responses to contaminants: Using basic ecological principles to predict ecotoxicological effects. Environ. Toxicol. Chem. 2009, 28, 1789–1800.
- Mothersill, C.; Seymour, C. Communication of ionising radiation signals—A tale of two fish. Int. J. Radiat. Biol. 2009, 85, 909–919.
- Møller, A.P.; Nishiumi, I.; Suzuki, H.; Ueda, K.; Mousseau, T.A. Differences in effects of radiation on abundance of animals in Fukushima and Chernobyl. Ecol. Indic. 2013, 24, 75–81.
- Møller, A.P.; Mousseau, T.A. Reduced abundance of insects and spiders linked to radiation at Chernobyl 20 years after the accident. Biol. Lett. 2009, 5, 356–359.
- Møller, A.P.; Mousseau, T.A. Efficiency of bio-indicators for low-level radiation under field conditions. Ecol. Indic. 2011, 11, 424–430.
- Turner, F.B. Effects of continuous irradiation on animal populations. Adv. Radiat. Biol. 1975, 5, 83–144.
- Lecomte-Pradines, C.; Bonzom, J.M.; Della-Vedova, C.; Beaugelin-Seiller, K.; Villenave, C.; Gaschak, S.; Coppin, F.; Dubourg, N.; Maksimenko, A.; Adam-Guillermin, C.; et al. Soil nematode assemblages as bioindicators of radiation impact in the Chernobyl Exclusion Zone. Sci. Total Environ. 2014, 490, 161–170.
- Møller, A.P.; Mousseau, T.A. Species richness and abundance of forest birds in relation to radiation at Chernobyl. Biol. Lett. 2007, 3, 483–486.
- Zhdanova, N.N.; Vasilevskaia, A.I.; Artyshkova, L.V.; Gavriliuk, V.I.; Lashko, T.N.; Sadovnikov, I.S. Kompleksy pochvennykh mikrokitsetov v zone vliianiia Chernobyl’skoĭ AES . Mikrobiol. Zh. 1991, 53, 3–9. (In Russian)
- Yablokov, A.V.; Nesterenko, V.B.; Nesterenko, A.V. Consequences of the Chernobyl catastrophe for public health and the environment 23 years later. Ann. N. Y. Acad. Sci. 2009, 1181, 318–326.
- Cordero, R.J.; Casadevall, A. Functions of fungal melanin beyond virulence. Fungal Biol. Rev. 2017, 31, 99–112.
- Dadachova, E.; Bryan, R.A.; Huang, X.; Moadel, T.; Schweitzer, A.D.; Aisen, P.; Nosanchuk, J.D.; Casadevall, A. Ionizing radiation changes the electronic properties of melanin and enhances the growth of melanized fungi. PLoS ONE 2007, 2, e457.
- Dadachova, E.; Casadevall, A. Ionizing radiation: How fungi cope, adapt, and exploit with the help of melanin. Curr. Opin. Microbiol. 2008, 11, 525–531.
- Romanovskaia, V.A.; Stoliar, S.M.; Malashenko, I.R.; Shatokhina, E.S. Vliianie radiatsii dlitel’nogo deĭstviia na raznoobrazie geterotrofnykh bakteriĭ v pochvakh 10-kilometrovoĭ zony Chernobyl’skoĭ atomnoĭ élektrostantsii . Mikrobiol. Z. 1996, 58, 3–12. (In Russian)
- Romanovskaia, V.A.; Sokolov, I.G.; Rokitko, P.V.; Chernaia, N.A. Ekologicheskie posledstviia radioaktivnogo zagriazneniia dlia pochvennykh bakteriĭ v 10-km zone ChAES . Mikrobiologiia 1998, 67, 274–280. (In Russian)
- Krivolutzkii, D.A.; Pokarzhevskii, A.D. Effects of radioactive fallout on soil animal populations in the 30 km zone of the Chernobyl atomic power station. Sci. Total Environ. 1992, 112, 69–77.
- Suzuki, J.; Egami, N. Mortality of the earthworms, Eisenia foetida, after γ-irradiation at different stages of their life history. J. Radiat. Res. 1983, 24, 209–220.
- Hertel-Aas, T.; Oughton, D.H.; Jaworska, A.; Bjerke, H.; Salbu, B.; Brunborg, G. Effects of chronic gamma irradiation on reproduction in the earthworm Eisenia fetida (Oligochaeta). Radiat. Res. 2007, 168, 515–526.
- Ihara, H.; Kumagai, A.; Hori, T.; Nanba, K.; Aoyagi, T.; Takasaki, M.; Katayama, Y. Direct comparison of bacterial communities in soils contaminated with different levels of radioactive cesium from the first Fukushima nuclear power plant accident. Sci. Total Environ. 2021, 756, 143844.
- Higo, M.; Kang, D.J.; Isobe, K. First report of community dynamics of arbuscular mycorrhizal fungi in radiocesium degradation lands after the Fukushima-Daiichi Nuclear disaster in Japan. Sci. Rep. 2019, 9, 8240.
- Bever, J.D.; Richardson, S.C.; Lawrence, B.M.; Holmes, J.; Watson, M. Preferential allocation to beneficial symbiont with spatial structure maintains mycorrhizal mutualism. Ecol. Lett. 2009, 12, 13–21.
- Barto, E.K.; Hilker, M.; Müller, F.; Mohney, B.K.; Weidenhamer, J.D.; Rillig, M.C. The fungal fast lane: Common mycorrhizal networks extend bioactive zones of allelochemicals in soils. PLoS ONE 2011, 6, e27195.
- Arrange, A.A.; Phelps, T.J.; Benoit, R.E.; Palumbo, A.V.; White, D.C. Bacterial sensitivity to UV light as a model for ionizing radiation resistance. J. Microbiol. Methods 1993, 18, 127–136.
- Kawaguchi, Y.; Shibuya, M.; Kinoshita, I.; Yatabe, J.; Narumi, I.; Shibata, H.; Hayashi, R.; Fujiwara, D.; Murano, Y.; Hashimoto, H.; et al. DNA damage and survival time course of deinococcal cell pellets during 3 years of exposure to outer space. Front. Microbiol. 2020, 11, 2050.
- Musilova, M.; Wright, G.; Ward, J.M.; Dartnell, L.R. Isolation of radiation-resistant bacteria from Mars analog Antarctic Dry Valleys by preselection, and the correlation between radiation and desiccation resistance. Astrobiology 2015, 15, 1076–1090.
- Geras’kin, S.A.; Dikarev, V.G.; Zyablitskaya, Y.Y.; Oudalova, A.A.; Spirin, Y.V. Cytogenetic effects of radiation on agricultural plants observed in the Chernobyl region during the first years after the accident. KURRI-KR 2002, 79, 287–296.
- Boratyński, Z.; Arias, J.; Garcia, C.; Mappes, T.; Mousseau, T.A.; Møller, A.P.; Pajares, A.J.M.; Piwczyński, M.; Tukalenko, E. Ionizing radiation from Chernobyl affects development of wild carrot plants. Sci. Rep. 2016, 6, 39282.
- Mousseau, T.A.; Milinevsky, G.; Kenney-Hunt, J.; Møller, A.P. Highly reduced mass loss rates and increased litter layer in radioactively contaminated areas. Oecologia 2014, 175, 429–437.
- van der Heijden, M.G.; Bardgett, R.D.; van Straalen, N.M. The unseen majority: Soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310.
- Dmitriev, A.; Guscha, N.; Dyachenko, A. Effects of low dose radiation on plant-pathogen interactions in Chernobyl Zone. Radioprotection 2008, 43, 148.
- Sakauchi, K.; Taira, W.; Otaki, J.M. Metabolomic profiles of the creeping wood sorrel Oxalis corniculata in radioactively contaminated fields in Fukushima: Dose-dependent changes in key metabolites. Life 2022, 12, 115.
- Zhu, J.; Sun, X.; Zhang, Z.D.; Tang, Q.Y.; Gu, M.Y.; Zhang, L.J.; Hou, M.; Sharon, A.; Yuan, H.L. Effect of ionizing radiation on the bacterial and fungal endophytes of the halophytic plant Kalidium schrenkianum. Microorganisms 2021, 9, 1050.
- Hayashi, G.; Shibato, J.; Imanaka, T.; Cho, K.; Kubo, A.; Kikuchi, S.; Satoh, K.; Kimura, S.; Ozawa, S.; Fukutani, S.; et al. Unraveling low-level gamma radiation-responsive changes in expression of early and late genes in leaves of rice seedlings at Iitate Village, Fukushima. J. Hered. 2014, 105, 723–738.
- Rakwal, R.; Hayashi, G.; Shibato, J.; Deepak, S.A.; Gundimeda, S.; Simha, U.; Padmanaban, A.; Gupta, R.; Han, S.I.; Kim, S.T.; et al. Progress toward rice seed OMICS in low-level gamma radiation environment in Iitate Village, Fukushima. J. Hered. 2018, 109, 206–211.
- Watanabe, Y.; Ichikawa, S.; Kubota, M.; Hoshino, J.; Kubota, Y.; Maruyama, K.; Fuma, S.; Kawaguchi, I.; Yoschenko, V.I.; Yoshida, S. Morphological defects in native Japanese fir trees around the Fukushima Daiichi Nuclear Power Plant. Sci. Rep. 2015, 5, 13232.
- Yoschenko, V.; Nanba, K.; Yoshida, S.; Watanabe, Y.; Takase, T.; Sato, N.; Keitoku, K. Morphological abnormalities in Japanese red pine (Pinus densiflora) at the territories contaminated as a result of the accident at Fukushima Dai-Ichi Nuclear Power Plant. J. Environ. Radioact. 2016, 165, 60–67.
- Shimizu, M. Endophytic actinomycetes: Biocontrol agents and growth promoters. In Bacteria in Agrobiology: Pant Growth Responses; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 201–220.
- Taskaev, A.I.; Frolova, N.P.; Popova, O.N.; Shevchenko, V.A. The monitoring of herbaceous seeds in the 30-km zone of the Chernobyl nuclear accident. Sci. Total Environ. 1992, 112, 57–67.
- Imanaka, T. Chernobyl and Fukushima: Comparison of accident process and radioactive contamination. Kagaku 2016, 86, 0252–0257. (In Japanese)
- Ludovici, G.M.; Chierici, A.; de Souza, S.O.; d’Errico, F.; Iannotti, A.; Malizia, A. Effects of ionizing radiation on flora ten years after the Fukushima Dai-ichi disaster. Plants 2022, 11, 222.
- Yoshioka, A.; Mishima, Y.; Fukasawa, K. Pollinators and other flying insects inside and outside the Fukushima evacuation zone. PLoS ONE 2015, 10, e0140957.
- Hiyama, A.; Taira, W.; Nohara, C.; Iwasaki, M.; Kinjo, S.; Iwata, M.; Otaki, J.M. Spatiotemporal abnormality dynamics of the pale grass blue butterfly: Three years of monitoring (2011–2013) after the Fukushima nuclear accident. BMC Evol. Biol. 2015, 15, 15.
- Cannon, G.; Kiang, J.G. A review of the impact on the ecosystem after ionizing irradiation: Wildlife population. Int. J. Radiat. Biol. 2022, 98, 1054–1062.
- Nohara, C.; Hiyama, A.; Taira, W.; Otaki, J.M. Robustness and radiation resistance of the pale grass blue butterfly from radioactively contaminated areas: A possible case of adaptive evolution. J. Hered. 2018, 109, 188–198.
- Hancock, S.; Vo, N.T.K.; Omar-Nazir, L.; Batlle, J.V.I.; Otaki, J.M.; Hiyama, A.; Byun, S.H.; Seymour, C.B.; Mothersill, C. Transgenerational effects of historic radiation dose in pale grass blue butterflies around Fukushima following the Fukushima Dai-ichi Nuclear Power Plant meltdown accident. Environ. Res. 2019, 168, 230–240.
- Hinton, T.G.; Alexakhin, R.; Balonov, M.; Gentner, N.; Hendry, J.; Prister, B.; Strand, P.; Woodhead, D. Radiation-induced effects on plants and animals: Findings of the United Nations Chernobyl Forum. Health Phys. 2007, 93, 427–440.
- Adam-Guillermin, C.; Hertal-Aas, T.; Oughton, D.; Blanchard, L.; Alonzo, F.; Armant, O.; Horemans, N. Radiosensitivity and transgenerational effects in non-human species. Ann. ICRP 2018, 47, 327–341.
- Horemans, N.; Spurgeon, D.J.; Lecomte-Pradines, C.; Saenen, E.; Bradshaw, C.; Oughton, D.; Rasnaca, I.; Kamstra, J.H.; Adam-Guillermin, C. Current evidence for a role of epigenetic mechanisms in response to ionizing radiation in an ecotoxicological context. Environ. Pollut. 2019, 251, 469–483.
- Gombeau, K.; Bonzom, J.M.; Cavalié, I.; Camilleri, V.; Orjollet, D.; Dubourg, N.; Beaugelin-Seiller, K.; Bourdineaud, J.P.; Lengagne, T.; Armant, O.; et al. Dose-dependent genomic DNA hypermethylation and mitochondrial DNA damage in Japanese tree frogs sampled in the Fukushima Daiichi area. J. Environ. Radioact. 2020, 225, 106429.
- Hancock, S.; Vo, N.T.K.; Goncharova, R.I.; Seymour, C.B.; Byun, S.H.; Mothersill, C.E. One-Decade-Spanning transgenerational effects of historic radiation dose in wild populations of bank voles exposed to radioactive contamination following the chernobyl nuclear disaster. Environ. Res. 2020, 80, 108816.
