As an important barrier between the cytoplasm and the microenvironment of the cell, the cell membrane is essential for the maintenance of normal cellular physiological activities. An abnormal cell membrane is a crucial symbol of body dysfunction and the occurrence of variant diseases; therefore, the visualization and monitoring of biomolecules associated with cell membranes and disease markers are of utmost importance in revealing the biological functions of cell membranes. Due to their biocompatibility, programmability, and modifiability, DNA nanomaterials have become increasingly popular in cell fluorescence imaging in recent years. In addition, DNA nanomaterials can be combined with the cell membrane in a specific manner to enable the real-time imaging of signal molecules on the cell membrane, allowing for the real-time monitoring of disease occurrence and progression.
- DNA nanomaterials
- cell membrane
- fluorescence imaging
1. Introduction
2. Application of Cell Membrane Imaging
2.1. Monitoring Imaging Triggered by the Tumor Cell Microenvironment
The tumor microenvironment (TME) is a physical and biochemical system that plays a significant role in the occurrence, development, metastasis, and drug resistance of tumors [25][24]. In general, the tumor microenvironment’s physiological state is distinct from that of normal tissue. Tumor tissue can be distinguished from normal tissue according to a number of physiological characteristics, including the overexpression of ATP, an acidic pH, hypoxia, a high level of reactive oxygen species (ROS), and the overexpression of enzymes. Consequently, these characteristics have become increasingly desirable as diagnostic and therapeutic targets [26,27][25][26].2.1.1. ATP
Adenosine triphosphate (ATP) is a fundamental biomolecule involved in numerous biochemical synthesis and metabolic processes. For a deeper understanding of the related cellular processes, it is crucial to examine the distribution of, and changes in, ATP outside the cell [28][27]. Utilizing DNA tweezers and cleavage aptamers, Zhong et al. [30][28] proposed a ratiometric DNA nanoswitch (Figure 1A). The nanoswitch consists of three uniquely designed ssDNA chains that are attached to the cell membrane via cholesterol. First, the DNA tweezers are in the open state, causing the fluorescent groups to separate and produce a low fluorescence resonance energy transfer (FRET) ratio. FRET is a mechanism that describes the transfer of energy between a photosensitive chromophore donor and an acceptor, and it is widely used in biochemistry and other areas [31][29]. ATP imaging analysis alone is typically insufficient for sensitive analysis, so researchers frequently employ signal amplification for more sensitive ATP imaging. Catalytic hairpin assembly (CHA) and hybridization chain reaction (HCR) are two of the most widely used signal amplification strategies. The CHA is a reaction in which hairpin DNA can be activated by specific nucleic acid sequences and automatically forms a stable double-stranded body via thermodynamic entropy gain [32][30]. The HCR reaction requires the involvement of both hairpin DNAs, and in the presence of a target, one of the hairpin DNAs can be opened, which, in turn, triggers the opening of the other hairpin DNA to trigger the creation of the HCR, generating a long double-stranded tandem of DNA [33][31]. Wang et al. [34][32] proposed the signal amplification strategy of extracellular ATP-activated HCR amplification, which enables accurate and sensitive tumor cell detection. In complex biological matrices, this method has promising application potential. In addition, by altering the sequence of the corresponding aptamer, this method can be used to detect various types of tumor cells (Figure 1B).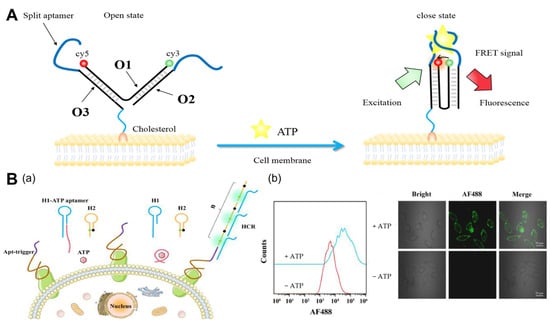
2.1.2. pH
In addition to the overexpression of ATP, extracellular pH (pHe) is a crucial microenvironmental factor in the development of tumors in the tumor microenvironment. The adjustment of pH value is essential for the maintenance of equilibrium in organisms. Several pathologies, such as ischemia, renal insufficiency, inflammation, and chronic lung disease, are typically associated with local pH fluctuations. Extracellular acidosis is becoming a universal indicator in the clinical diagnosis of tumors. To effectively distinguish normal cells from cancer cells, TME imaging must be sensitive. Nie et al. [38][33] designed a DNA tweezer composed of an i-motif to dynamically monitor pH changes in the cell microenvironment (Figure 2A). Double-stranded DNA can be hydrolyzed; it is not a suitable candidate for sensor functions in biological media. In contrast, framework nucleic acids have an excellent resistance to enzymatic hydrolysis and are widely employed in numerous sensors [41,42][34][35]. Yuan et al. [43][36] designed a programmable pH sensor that employs the tetrahedral DNA framework (TDF) structure as the skeleton and the DNA i-motif structure as the proton recognition probe (Figure 2B).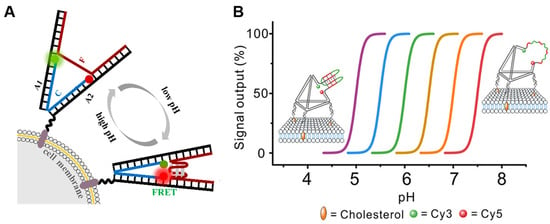
2.1.3. Metal Ions
2.2. Imaging of Cell Membrane Receptors
Receptors are a class of protein molecules that serve as subcellular cell surfaces or intracellular components. Receptors on the cell membrane play important roles in a variety of physiological and pathological processes and have become therapeutic targets for a large number of drugs [57,58][42][43]. G-protein-coupled receptors (GPCRs) and enzyme-linked receptors (especially RTKs) are the two largest known receptors among the thousands of known cell surface receptors in eukaryotes, and they also represent one of the hottest topics in the field of life science [59][44]. Recognizing extracellular stimuli and transducing cell signal pathways, cell surface receptors regulate fundamental cell behaviors, such as proliferation, migration, and differentiation [60][45]. Numerous membrane receptors, such as MUC1 [61][46], nucleolin [57][42], EGFR [62][47], epithelial cell adhesion molecules [63][48], etc., have been considered tumor markers and used for the specific imaging of cancer cells up to this point. The majority of these receptors are RTKs and participate in numerous intracellular signaling pathways associated with the growth and proliferation of cancer cells [64][49]. Ingeniously, Wang et al. [69][50] designed an i-motif with a hairpin as an acid response element and paired it with a tumor-targeting nucleic acid aptamer for the bispecific imaging and in situ drug release of tumor cells, dubbing it the “molecular doctor” (pH-Apt-MD) (Figure 4B). It uses the binding of, and structural changes in, nucleic acid aptamers to the i-motif, the dissociation of pH-Apt-MD, and the stimulation of FRET signaling between AF488 and Cy3 to achieve in situ drug release. The integration of tumor diagnosis and treatment is a major direction of development for cancer treatment at present. As a method of diagnosing tumors, the imaging of receptors on cell membranes has been widely reported, but there may be false positives. In contrast, multiple or distinct tumor markers in living cells are crucial for obtaining precise and multidimensional information regarding tumor cell types and tumor progression. Consequently, using receptors on the cell membrane as logical input signals can prevent false-positive results and enable the rapid and sensitive transmembrane sequential imaging of multilayer biomarkers that precisely distinguish tumor cell types.2.3. Imaging of Other Molecules
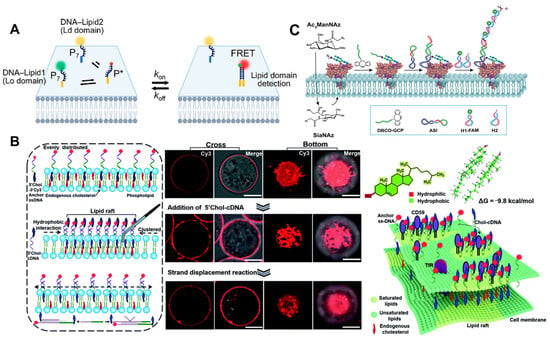
3. Conclusions
Although DNA nanomaterials are widely employed in cell surface fluorescence, the following obstacles must still be addressed. (1) Nowadays, most current imaging strategies involve imaging and analyzing only one or two targets, but using signal amplification techniques, such as HCR and CHA, to achieve the in situ accurate and highly sensitive imaging of multiple targets in the tumor microenvironment remains a challenge. (2) Aptamers must be designed specifically for each receptor. Currently, SELEX technology is used to screen all aptamers, and there are still receptors without aptamers. (3) The majority of DNA nanomaterials are attached to the cell membrane via covalent or noncovalent methods [44,85][57][58] but are susceptible to endocytosis. Even though some scientists have developed polymer molecular skeletons to increase the anchoring time of probes in cell membranes, long-term stability remains a problem. Monitoring intra- and extracellular signaling, cellular morphology, and structural changes requires prolonged in situ imaging, especially for slow-response events such as apoptosis. The creation of probes that can remain attached to the cell membrane for extended durations without being endocytosed remains an area of active research. (4) DNA nanomaterials are used not only for the fluorescence imaging of cell membranes but also for the functional regulation of cell membrane receptors. How to realize the integration of long-term imaging and the functional regulation of cell membranes should also be investigated. (5) FRET is dependent not only on the donor (D)–acceptor (A) separation distance and energetic resonance (i.e., D–A spectral overlap), but also on the orientation of the D emission and A absorption transition dipole moments [86,87][59][60].
References
- Yu, X.M.; Sha, L.J.; Liu, Q.; Zhao, Y.Y.; Fang, H.; Cao, Y.; Zhao, J. Recent Advances in Cell Membrane Camouflage-based Biosensing Application. Biosens. Bioelectron. 2021, 194, 113623.
- Feng, L.; Li, J.; Sun, J.; Wang, L.; Fan, C.; Shen, J. Recent Advances of DNA Nanostructure-Based Cell Membrane Engineering. Adv. Healthc. Mater. 2021, 10, 2001718.
- Qian, R.C.; Zhou, Z.R.; Guo, W.J.; Wu, Y.T.; Yang, Z.L.; Lu, Y. Cell Surface Engineering Using DNAzymes: Metal Ion Mediated Control of Cell-Cell Interactions. J. Am. Chem. Soc. 2021, 143, 5737–5744.
- Reading, E.; Hall, Z.; Martens, C.; Haghighi, T.; Findlay, H.; Ahdash, Z.; Politis, A.; Booth, P.J. Interrogating Membrane Protein Conformational Dynamics within Native Lipid Compositions. Angew. Chem. Int. Ed. 2017, 56, 15654–15657.
- Shi, P.; Wang, Y. Synthetic DNA for Cell-Surface Engineering. Angew. Chem. Int. Ed. 2021, 60, 11580–11591.
- Wang, H.; Wang, Y.S.; Wan, Y.Q.; Shang, J.H.; Wang, Q.; Jiang, Y.Q.; Liu, X.Q.; Wang, F. A Cellular Membrane-Confined Concatenate DNA Circuit for Non-Invasive Cell Modulation with High Accuracy and Efficiency. Adv. Funct. Mater. 2023, 33, 2302708.
- Zhang, S.Y.; Li, L.; Chen, J.Y.; Chen, Z.Q.; Zhang, W.L.; Lu, H.B. Quantitative Imaging of Gd Nanoparticles in Mice Using Benchtop Cone-Beam X-ray Fluorescence Computed Tomography System. Int. J. Mol. Sci. 2019, 20, 2315.
- Wang, Z.L.; Xue, X.D.; He, Y.X.; Lu, Z.W.; Jia, B.; Wu, H.; Yuan, Y.; Huang, Y.; Wang, H.; Lu, H.W.; et al. Novel Redox-Responsive Polymeric Magnetosomes with Tunable Magnetic Resonance Property for In Vivo Drug Release Visualization and Dual-Modal Cancer Therapy. Adv. Funct. Mater. 2018, 28, 1802158.
- Miao, X.Y.; Mao, R.; You, Y.J.; Zhou, H.C.; Qiu, C.; Li, X.H.; Chen, Z.H.; Ren, J.; Chen, M.H.; Wang, P.; et al. Intracolic Ultrasound Molecular Imaging: A Novel Method for Assessing Colonic Tumor Necrosis Factor-α Expression in Inflammatory Bowel Disease. Mol. Med. 2021, 27, 119.
- Wozniak, M.; Ploska, A.; Siekierzycka, A.; Dobrucki, L.W.; Kalinowski, L.; Dobrucki, I.T. Molecular Imaging and Nanotechnology-Emerging Tools in Diagnostics and Therapy. Int. J. Mol. Sci. 2022, 23, 2658.
- Li, F.; Li, J.; Dong, B.; Wang, F.; Fan, C.; Zuo, X. DNA Nanotechnology-empowered Nanoscopic Imaging of Biomolecules. Chem. Soc. Rev. 2021, 50, 5650–5667.
- Xu, Y.W.; Lv, Z.; Yao, C.; Yang, D.Y. Construction of Rolling Circle Amplification-based DNA Nanostructures for Biomedical Applications. Biomater. Sci. 2022, 10, 3054–3061.
- Meng, H.M.; Liu, H.; Kuai, H.L.; Peng, R.Z.; Mo, L.T.; Zhang, X.B. Aptamer-integrated DNA Nanostructures for Biosensing, Bioimaging and Cancer Therapy. Chem. Soc. Rev. 2016, 45, 2583–2602.
- Wang, K.; Gao, H.Q.; Zhang, Y.W.; Yan, H.Y.; Si, J.H.; Mi, X.Y.; Xia, S.A.; Feng, X.Q.; Liu, D.B.; Kong, D.L.; et al. Highly Bright AIE Nanoparticles by Regulating the Substituent of Rhodanine for Precise Early Detection of Atherosclerosis and Drug Screening. Adv. Mater. 2022, 34, 2106994.
- Ma, W.; Zhan, Y.; Zhang, Y.; Mao, C.; Xie, X.; Lin, Y. The Biological Applications of DNA Nanomaterials: Current Challenges and Future Directions. Signal Transduct. Target. Ther. 2021, 6, 351.
- Nishikawa, M.; Tan, M.M.; Liao, W.Q.; Kusamori, K. Nanostructured DNA for the Delivery of Therapeutic Agents. Adv. Drug Deliv. Rev. 2019, 147, 29–36.
- Zhang, J.J.; Li, F.; Yang, D.Y. DNA: From Carrier of Genetic Information to Polymeric Materials. Trans. Tianjin Univ. 2019, 25, 301–311.
- Nicolson, F.; Ali, A.; Kircher, M.F.; Pal, S. DNA Nanostructures and DNA-Functionalized Nanoparticles for Cancer Theranostics. Adv. Sci. 2020, 7, 2001669.
- Wang, D.X.; Wang, J.; Wang, Y.X.; Du, Y.C.; Huang, Y.; Tang, A.N.; Cui, Y.X.; Kong, D.M. DNA Nanostructure-based Nucleic Acid Probes: Construction and Biological Applications. Chem. Sci. 2021, 12, 7602–7622.
- Song, L.; Zhuge, Y.; Zuo, X.L.; Li, M.; Wang, F. DNA Walkers for Biosensing Development. Adv. Sci. 2022, 9, 2200327.
- Wu, X.H.; Wu, T.T.; Liu, J.B.; Ding, B.Q. Gene Therapy Based on Nucleic Acid Nanostructure. Adv. Healthc. Mater. 2020, 9, 2001046.
- Wang, H.M.; Luo, D.; Wang, H.; Wang, F.; Liu, X.Q. Construction of Smart Stimuli-Responsive DNA Nanostructures for Biomedical Applications. Chem. Eur. J. 2021, 27, 3929–3943.
- Li, C.; Tebo, A.G.; Gautier, A. Fluorogenic Labeling Strategies for Biological Imaging. Int. J. Mol. Sci. 2017, 18, 1473.
- Zhou, Z.X.; Lu, Z.R. Molecular Imaging of the Tumor Microenvironment. Adv. Drug Deliv. Rev. 2017, 113, 24–48.
- Zhang, Y.; Cheng, F.; Liang, R.; Shuai, X.J.; Nie, K.H.; Li, J.; Zeng, Q.L.; Huang, C.Z.; Li, C.M. In Situ Activation of the Receptor Aggregation for Cell Apoptosis by an AI-Au Intelligent Nanomachine via Tumor Extracellular Acidity. ACS Appl. Mater. Interfaces 2023, 15, 32262–32271.
- Park, H.; Saravanakumar, G.; Kim, J.; Lim, J.; Kim, W.J. Tumor Microenvironment Sensitive Nanocarriers for Bioimaging and Therapeutics. Adv. Healthc. Mater. 2021, 10, 2000834.
- Wang, Y.; Tang, L.H.; Li, Z.H.; Lin, Y.H.; Li, J.H. In Situ Simultaneous Monitoring of ATP and GTP using a Graphene Oxide Nanosheet-based Sensing Platform in Living Cells. Nat. Protoc. 2014, 9, 1944–1955.
- Yuan, J.; Deng, Z.W.; Liu, H.; Li, X.F.; Li, J.B.; He, Y.; Qing, Z.H.; Yang, Y.J.; Zhong, S.A. Cell-Surface-Anchored Ratiometric DNA Nanoswitch for Extracellular ATP Imaging. ACS Sens. 2019, 4, 1648–1653.
- Zong, H.; Wang, X.X.; Mu, X.J.; Wang, J.G.; Sun, M.T. Plasmon-Enhanced Fluorescence Resonance Energy Transfer. Chem. Rec. 2019, 19, 818–842.
- Yang, X.J.; Cui, M.R.; Li, X.L.; Chen, H.Y.; Xu, J.J. A Self-Powered 3D DNA Walker with Programmability and Signal-amplification for Illuminating MicroRNA in Living Cells. Chem. Commun. 2020, 56, 2135–2138.
- Bi, S.; Yue, S.Z.; Zhang, S.S. Hybridization Chain Reaction: A Versatile Molecular Tool for Biosensing, Bioimaging, and Biomedicine. Chem. Soc. Rev. 2017, 46, 4281–4298.
- Li, L.; Li, S.P.; Wang, J.; Wen, X.H.; Yang, M.; Chen, H.; Guo, Q.; Wang, K. Extracellular ATP-activated Hybridization Chain Reaction for Accurate and Sensitive Detection of Cancer Cells. Chin. Chem. Lett. 2023, 34, 108399.
- Zeng, S.; Liu, D.; Li, C.; Yu, F.; Fan, L.; Lei, C.; Huang, Y.; Nie, Z.; Yao, S. Cell-Surface-Anchored Ratiometric DNA Tweezer for Real-Time Monitoring of Extracellular and Apoplastic pH. Anal. Chem. 2018, 90, 13459–13466.
- Li, Z.Z.; Wang, J.B.; Zhou, Z.X.; O’Hagan, M.P.; Willner, I. Gated Transient Dissipative Dimerization of DNA Tetrahedra Nanostructures for Programmed DNAzymes Catalysis. ACS Nano 2022, 16, 3625–3636.
- Zhao, C.; Zhang, L.; Hu, Y.; Nie, C.; Chen, T.T.; Chu, X. Simultaneous Imaging and Visualizing the Association of Survivin mRNA and Telomerase in Living Cells by Using a Dual-Color Encoded DNA Nanomachine. Anal. Chem. 2023, 95, 1498–1504.
- Liu, J.X.; Li, W.W.; Li, R.S.; Yin, X.Z.; He, S.L.; Hu, J.Q.; Ruan, S.C. Programmable DNA Framework Sensors for In Situ Cell-Surface pH Analysis. Anal. Chem. 2021, 93, 12170–12174.
- Aksentijevic, D.; Karlstaedt, A.; Basalay, M.V.; O’Brien, B.A.; Sanchez-Tatay, D.; Eminaga, S.; Thakker, A.; Tennant, D.A.; Fuller, W.; Eykyn, T.R.; et al. Intracellular Sodium Elevation Reprograms Cardiac Metabolism. Nat. Commun. 2020, 11, 4337.
- Deng, Z.; Gao, P.; Liu, H.; He, Y.; Zhong, S.; Yang, Y. Cell-Surface-Anchored DNA Sensors for Simultaneously Monitoring Extracellular Sodium and Potassium Levels. Anal. Chem. 2021, 93, 16432–16438.
- Anees, P.; Saminathan, A.; Rozmus, E.R.; Di, A.K.; Malik, A.B.; Delisle, B.P.; Krishnan, Y. Detecting Organelle-specific Activity of Potassium Channels with A DNA Nanodevice. Nat. Biotechnol. 2023, 1546–1696.
- Vodnala, S.K.; Eil, R.; Kishton, R.J.; Sukumar, M.; Yamamoto, T.N.; Ha, N.H.; Lee, P.H.; Shin, M.; Patel, S.J.; Yu, Z.Y.; et al. T Cell Stemness and Dysfunction in Tumors are Triggered by a Common Mechanism. Science 2019, 363, eaau0135.
- Eil, R.; Vodnala, S.K.; Clever, D.; Klebanoff, C.A.; Sukumar, M.; Pan, J.H.; Palmer, D.C.; Gros, A.; Yamamoto, T.N.; Patel, S.J.; et al. Ionic Immune Suppression within the Tumour Microenvironment Limits T Cell Effector Function. Nature 2016, 537, 539–543.
- Cheng, F.; Jiang, Y.J.; Kong, B.; Lin, H.R.; Shuai, X.J.; Hu, P.P.; Gao, P.F.; Zhan, L.; Huang, C.Z.; Li, C.M. Multi-Catcher Polymers Regulate the Nucleolin Cluster on the Cell Surface for Cancer Therapy. Adv. Healthc. Mater. 2023, 12, 2300102.
- Wang, L.P.; Liang, H.; Sun, J.; Liu, Y.C.; Li, J.Y.; Li, J.Y.; Li, J.; Yang, H.H. Bispecific Aptamer Induced Artificial Protein-Pairing: A Strategy for Selective Inhibition of Receptor Function. J. Am. Chem. Soc. 2019, 141, 12673–12681.
- Nishida, N.; Osawa, M.; Takeuchi, K.; Imai, S.; Stampoulis, P.; Kofuku, Y.; Ueda, T.; Shimada, I. Functional Dynamics of Cell Surface Membrane Proteins. J. Magn. Reson. 2014, 241, 86–96.
- He, F.; Wang, M.X.; Wang, J.Y.; Wang, H.H.; Nie, Z. An Extracellular miRNA-Responsive Artificial Receptor via Dynamic DNA Nano-assembly for Biomarker-Driven Therapy. Angew. Chem. Int. Ed. 2023, 135, e202305227.
- Li, L.L.; Lv, W.Y.; Xu, Y.T.; Li, Y.F.; Li, C.M.; Huang, C.Z. DNA Logic Nanodevices for the Sequential Imaging of Cancer Markers through Localized Catalytic Hairpin Assembly Reaction. Anal. Chem. 2022, 94, 4399–4406.
- Fang, Y.Y.; Li, Y.T.; Li, Y.Y.; He, R.; Zhang, Y.; Zhang, X.B.; Liu, Y.; Ju, H.X. In Situ Protease Secretion Visualization and Metastatic Lymph Nodes Imaging via a Cell Membrane-Anchored Upconversion Nanoprobe. Anal. Chem. 2021, 93, 7258–7265.
- Yang, L.L.; Meng, L.Y.; Song, J.Y.; Xiao, Y.; Wang, R.W.; Kang, H.Z.; Han, D. Dynamic Colloidal Nanoparticle Assembly Triggered by Aptamer-receptor Interactions on Live Cell Membranes. Chem. Sci. 2019, 10, 7466–7471.
- Jia, H.R.; Zhu, Y.X.; Duan, Q.Y.; Wu, F.G. Cell Surface-localized Imaging and Sensing. Chem. Soc. Rev. 2021, 50, 6240–6277.
- Ma, W.J.; Sun, H.H.; Chen, B.A.; Jia, R.C.; Huang, J.; Cheng, H.; He, X.X.; Huang, M.M.; Wang, K.M. Engineering a Facile Aptamer “Molecule-Doctor” with Hairpin- Contained I-Motif Enables Accurate Imaging and Killing of Cancer Cells. Anal. Chem. 2021, 93, 14552–14559.
- Sezgin, E.; Levental, I.; Mayor, S.; Eggeling, C. The Mystery of Membrane Organization: Composition, Regulation and Roles of Lipid Rafts. Nat. Rev. Mol. Cell Biol. 2017, 18, 361–374.
- Cheng, X.L.; Smith, J.C. Biological Membrane Organization and Cellular Signaling. Chem. Rev. 2019, 119, 5849–5880.
- Ali, A.A.; Bagheri, Y.; Tian, Q.; You, M.X. Advanced DNA Zipper Probes for Detecting Cell Membrane Lipid Domains. Nano Lett. 2022, 22, 7579–7587.
- Lingwood, D.; Simons, K. Lipid Rafts As a Membrane-Organizing Principle. Science 2010, 327, 46–50.
- Sun, L.L.; Su, Y.Y.; Wang, J.G.; Xia, F.; Xu, Y.; Li, D. DNA Nanotweezers for Stabilizing and Dynamically Lighting up a Lipid Raft on Living Cell Membranes and The Activation of T Cells. Chem. Sci. 2020, 11, 1581–1586.
- Li, J.Y.; Liu, S.Y.; Sun, L.Q.; Li, W.; Zhang, S.Y.; Yang, S.; Li, J.; Yang, H.H. Amplified Visualization of Protein-Specific Glycosylation in Zebrafish via Proximity-Induced Hybridization Chain Reaction. J. Am. Chem. Soc. 2018, 140, 16589–16595.
- Xun, K.Y.; Pei, K.; Liu, X.J.; Peng, X.Y.; Du, Y.L.; Qiu, L.P.; Tan, W.H. Cell-Membrane-Anchored DNA Nanoplatform for Programming Cellular Interactions. J. Am. Chem. Soc. 2019, 141, 18013–18020.
- Chandra, R.A.; Douglas, E.S.; Mathies, R.A.; Bertozzi, C.R.; Francis, M.B. Programmable Cell Adhesion Encoded by DNA Hybridization. Angew. Chem. Int. Ed. 2006, 45, 896–901.
- Mathur, D.; Díaz, S.A.; Hildebrandt, N.; Pensack, R.D.; Yurke, B.; Biaggne, A.; Li, L.; Melinger, J.S.; Ancona, M.G.; Knowlton, W.B.; et al. Pursuing Excitonic Energy Transfer with Programmable DNA-based Optical Breadboards. Chem. Soc. Rev. 2023, 52, 7848–7948.
- Cervantes-Salguero, K.; Biaggne, A.; Youngsman, J.M.; Ward, B.M.; Kim, Y.C.; Li, L.; Hall, J.A.; Knowlton, W.B.; Graugnard, E.; Kuang, W. Strategies for Controlling the Spatial Orientation of Single Molecules Tethered on DNA Origami Templates Physisorbed on Glass Substrates: Intercalation and Stretching. Int. J. Mol. Sci. 2022, 23, 7690.
