Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Rosario Nicoletti and Version 2 by Wendy Huang.
The range of interactions between Cladosporium, a ubiquitous fungal genus, and insects, a class including about 60% of the animal species, is extremely diverse. Conventionally, Cladosporium species are not considered full-right representatives of the guild of entomopathogens, which is generally restricted to specialized fungi such as Beauveria, Metarhizium and Lecanicillium/Akanthomyces. However, like other fungi that are widely associated with crops such as Trichoderma and Talaromyces, the evidence is increasing that Cladosporium may also infect insects and cause epizootics in pest populations or promote plant defense reactions.
- interactions
- biocontrol
- Cladosporium
- insects
- fungi
- plant
1. Introduction
Fungi in the genus Cladosporium (Dothideomycetes, Cladosporiaceae) are ubiquitous and reported from any terrestrial and marine substrate, including all kinds of living organisms [1]. This is linked to their profuse sporulation, which allows the spread of conidia through atmospheric agents over long distances. Thus, the mere isolation of these fungi from plants and animals does not necessarily imply a symbiotic association. However, generality is not a rule, and in many cases, this pervasiveness subtends either occasional or more systematic biotic relationships that influence the fitness of the associated organisms in diverse ways [2][3][4][2,3,4].
Direct observations of the parasitic aptitude of insects are limited and essentially concern the case of C. cladosporioides on the sugarcane white wooly aphid (Ceratovacuna lanigera: Hemiptera, Aphididae); both light and electron microscopy at the host–parasite interface showed that nymphs and adults of the aphid were completely overgrown by the fungal mycelium, which penetrated and disrupted their powdery waxy coating [5].
However, circumstantial evidence of entomopathogenic aptitude in Cladosporium derives from several studies reporting on mortality induced by conidial suspensions administered at various concentrations and exposure times. In this regard, the available data concerning strains that proved to be effective against various targeted pests in experimental trials are summarized in Table 1.
Alternatively, the anti-insectan effect can be assessed through the addition of the fungi or their products to the laboratory diet. In this respect, when incorporated in the feed of larvae of the tobacco budworm (Chloridea virescens: Lepidoptera, Noctuidae), an isolate of C. cladosporioides was found to reduce larval and pupal weights by 56% and 7%, respectively; moreover, in preference tests, the caterpillars showed a marked tendency to avoid feed amended with the fungus [28][195]. Development of another noctuid moth, the tobacco cutworm (Spodoptera litura), was significantly prolonged when larvae were fed on a diet amended with ethyl acetate extract of C. uredinicola at concentrations of 1.25–2.00 μL g−1; moreover, at 2.00 μL g−1, a significantly higher number of adults emerged showing morphological deformities. At higher concentrations, significant reductions in adult emergence, longevity and reproductive potential were recorded. Finally, the toxicity of the ethyl acetate extract was further evidenced by a reduction in feed utilization by the larvae [29][196].
The ethyl acetate and methylene chloride extracts of a strain of C. cladosporioides were effective against nymphs and adults of the cotton aphid (Aphis gossypii: Hemiptera, Aphididae) [30][31][13,197]. Aphicidal effect was also displayed by formulations based on emulsions of culture filtrates of an endophytic strain of C. oxysporum endowed with proteolytic activity, which were more active than conidial suspensions against the black bean aphid (Aphis fabae: Hemiptera, Aphididae) [17][190]. In a subsequent experiment, formulations based on culture filtrates of this strain and two more endophytic isolates of C. echinulatum and Cladosporium sp. showed activity against the green peach aphid (Myzus persicae: Hemiptera, Aphididae), which increased at increasing concentrations. A significant reduction in the number of colonizing aphids and a relative increase in the number of winged adults were recorded. Moreover, the pretreatment of plants negatively influenced embryonic development, thus affecting fertility [32][198]. In the same study, consistent chitinolytic activity was determined in the culture filtrates of Cladosporium sp.; indeed, chitinases are considered a main factor in the bioactivity of fungal culture filtrates, as also documented for other strains of Cladosporium spp. [24][193], C. cladosporioides [12][33][27,48], C. tenuissimum and C. xanthocromaticum [12][48].
Even more, the anti-insectan effects of culture filtrates may depend on the presence of toxic compounds (Figure 1). Fungi in the genus Cladosporium are known as prolific producers of bioactive secondary metabolites [34][199], some of which have been detected as possible determinants of detrimental effects on insects. This is the case of bassianolide, a cycloligomer depsipeptide identified as a product of a strain related to the C. cladosporioides s.c. [35][200]. The alkaloid 3-(4β-hydroxy-6-pyranonyl)-5-isopropylpyrrolidin-2-one was identified in the ethyl acetate extracts of another strain of C. cladosporioides displaying aphicidal activity [30][13]. Another alkaloid, hydroxyquinoline, was identified as the potentially active product in the extracts of a strain of C. subuliforme [25][167]. The novel compound citreoviridin A was extracted from an isolate of C. herbarum from a marine sponge and found to inhibit the growth of larvae of the cotton leafworm (Spodoptera littoralis: Lepidoptera, Noctuidae) [36][201]. Chlorogenic acid, purified from an endophytic isolate of C. velox, displayed insecticidal activity by inducing significant mortality in the larvae of S. litura or adversely prolonging their developmental period. This phenolic compound, previously known to cause gut toxicity in lepidopterans [37][202], was characterized as an α-glucosidase inhibitor, performing a non-competitive type of inhibition in vitro; it also inhibited the activity of α-glycosidases in the gut of the larvae [38][39][203,204].
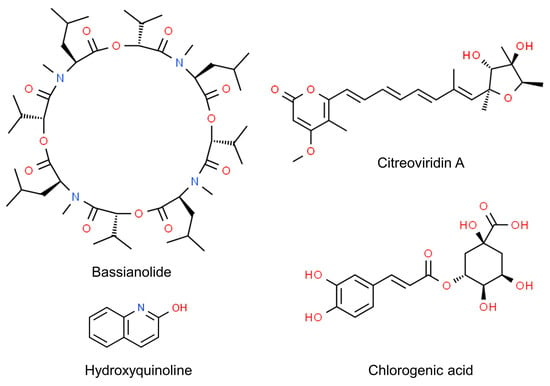 The importance of secondary metabolites for entomopathogenic aptitude in Cladosporium has been further affirmed after a study carried out on strains associated with the Chinese white wax scale (Ericerus pela: Hemiptera, Coccidae). This insect is known to be infected by Cladosporium spp. related to C. sphaerospermum and C. langeronii, which kill the scales after dramatically altering their microbiome [40][34]. However, the scales were later found to also harbor a non-infective Cladosporium. Genome sequencing showed that the non-infective strain is related to C. cladosporioides and has a larger genome size than a pathogenic one, which is more related to C. sphaerospermum. Particularly, the former has specific genes involved in nutrition pathways that are absent in the pathogen. Conversely, the latter possesses genes participating in the biosynthetic pathways of mycotoxins, such as asperfuranone, emericellamide and fumagillin. These genes were not found in the non-pathogenic strain, which, on the other hand, presented genes associated with reduced virulence [3].
The importance of secondary metabolites for entomopathogenic aptitude in Cladosporium has been further affirmed after a study carried out on strains associated with the Chinese white wax scale (Ericerus pela: Hemiptera, Coccidae). This insect is known to be infected by Cladosporium spp. related to C. sphaerospermum and C. langeronii, which kill the scales after dramatically altering their microbiome [40][34]. However, the scales were later found to also harbor a non-infective Cladosporium. Genome sequencing showed that the non-infective strain is related to C. cladosporioides and has a larger genome size than a pathogenic one, which is more related to C. sphaerospermum. Particularly, the former has specific genes involved in nutrition pathways that are absent in the pathogen. Conversely, the latter possesses genes participating in the biosynthetic pathways of mycotoxins, such as asperfuranone, emericellamide and fumagillin. These genes were not found in the non-pathogenic strain, which, on the other hand, presented genes associated with reduced virulence [3].
Table 1.
Reported effectiveness of conidial suspensions of
Cladosporium
in inducing mortality on insect pests.
| Cladosporium | Species | Source | Insect Targets | Country | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| C. cladosporioides | Bemisia | sp. | Bemisia | sp. | Egypt | [6] | [18] | |||
| Brevicoryne brassicae | B. brassicae | Egypt | [7] | [20] | ||||||
| Culex quinquefasciatus | C. quinquefasciatus | Iraq | [8] | [28] | ||||||
| endophytic | Duponchelia fovealis | Brazil | [9] | [188] | ||||||
| Kermes | sp. | Hemiberlesia pitysophila | China | [10] | [45] | |||||
| Lycorma delicatula | Tenebrio molitor | USA | [11] | [46] | ||||||
| Myzus persicae | M. persicae | Iraq | [12] | [48] | ||||||
| Nilaparvata lugens | Bemisia tabaci | Bangladesh | [13] | [49] | ||||||
| Pulvinaria aurantii | Aphis fabae | Iran | [14] | [157] | ||||||
| Sitophilus oryzae | Rhyzopertha dominica | Sitophilus zeamais | Trogoderma granarium | Pakistan | [15] | [56] | ||||
| soil | Metopolophium dirhodum | Egypt | [16] | [189] | ||||||
| C. oxysporum | endophytic | A. fabae | Algeria | [17] | [190] | |||||
| endophytic | Chilo partellus | India | [18] | [191] | ||||||
| Planococcus citri | Pseudococcus longispinus | Pulvinaria aethiopica | Toxoptera citricida | Trioza erytreae | South Africa | [19] | [75] | |||
| unknown | Aphis craccivora | India | [20] | [192] | ||||||
| C. sphaerospermum | endophytic | D. fovealis | Brazil | [9] | [188] | |||||
| Cladosporium | sp. | Helicoverpa armigera | Aphis gossypii | B. tabaci | H. armigera | Australia | [21] | [130] | ||
| Spodoptera frugiperda | S. frugiperda | China | [22] | [169] | ||||||
| Cladosporium | spp. | several species of sap-sucking Hemiptera | A. craccivora | A. gossypii | B. tabaci | Egypt | [23][24] | [98,193] | ||
| C. subuliforme | Diaphorina citri | D. citri | China | [25] | [167] | |||||
| C. tenuissimum | M. persicae | M. persicae | Iraq | [12] | [48] | |||||
| Trachymela sloanei | S. frugiperda | China | [26] | [194] | ||||||
| C. uredinicola | A. gossypii | B. tabaci | A. gossypii | B. tabaci | Egypt | [27] | [170] | |||
| Bemisia | sp. | Bemisia | sp. | Egypt | [6] | [18] | ||||
| C. xanthocromaticum | M. persicae | M. persicae | Iraq | [12] | [48] |

Figure 1.
Chemical structure of
Cladosporium
secondary metabolites displaying anti-insectan effects.
