Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Tatsuo Yoshinobu and Version 2 by Rita Xu.
The artificial olfactory image was proposed by Lundström et al. in 1991 as a new strategy for an electronic nose system which generated a two-dimensional mapping to be interpreted as a fingerprint of the detected gas species.
- artificial olfactory image
- gas sensor
- electronic nose
- metal-oxide-semiconductor structure
1. Introduction
The concept of an artificial olfactory image was proposed by Lundström et al. in 1991 [1]. Following the discovery of the sensitivity of a Pd-gate metal–oxide–semiconductor (MOS) transistor to hydrogen in 1973, Prof. Ingemar Lundström paved the way for the realization and understanding of various gas sensors based on field-effect structures with catalytic metal gates and their applications to sensor arrays, electronic noses, and artificial olfactory images [2][3][2,3].
An artificial olfactory image is an image generated by a field-effect sensor device to be classified by pattern recognition to identify the composition of the gas sample under testing. From a technical point of view, it is realized by combining two technologies: a catalytic field-effect device, which generates a potential difference in response to the gas, and the scanned light pulse technique (SLPT) [4], which reads the two-dimensional distribution of the surface potential in the field-effect structure.
In some contexts, the artificial olfactory image is said to be the first example of a two-dimensional extension of the light-addressable potentiometric sensor (LAPS) proposed by Hafeman et al. in 1988 [5], a chemical sensor based on a mechanism of signal generation similar to that of the SLPT. It should be noted, however, that an artificial olfactory image does not carry information on the spatial distribution of the analyte. Unlike other imaging technologies based on a LAPS [6][7][8][9][6,7,8,9], in which the obtained image represents a map of the spatial distribution of the analyte, an artificial olfactory image is not a spatial map but a fingerprint of the gas. Despite this essential difference, the feasibility of image generation demonstrated in [1] inspired and promoted, directly and indirectly, the development of various chemical imaging technologies based on a semiconductor field-effect structure scanned by a light beam.
2. From SLPT to LAPS and Artificial Olfactory Images—How It Started
The SLPT is a method to visualize two-dimensional distributions of interface properties, such as the flat-band voltage and the interface state density in a metal–insulator–semiconductor (MIS) structure, as shown in Figure 1a [4]. A bias voltage Vbias is applied to the MIS structure so that the majority carriers in the semiconductor layer are driven away from the insulator–semiconductor interface to form a space charge layer (SCL). Under this situation, the semiconductor layer is illuminated with a chopped light beam with a photon energy larger than the bandgap of the semiconductor. When the light is switched on, the electrons and holes generated by the internal photoelectric effect in the semiconductor are separated by the electric field inside the SCL and a transient current flows in the external circuit until a steady state is reached. When the light is switched off, a transient current flows in the reverse direction. The amplitude of this alternating current Isig is recorded in the external circuit as a function of Vbias, and a current–voltage (Isig–Vbias) curve, as shown in Figure 1b, is obtained at each illuminated position. The amplitude of the current increases with the SCL’s thickness until strong inversion is reached. The in-plane distribution of the flat-band voltage in the MIS structure, for example, can be obtained by recording the horizontal shift in the Isig−Vbias curve at each position, or equivalently, by recording the change in Isig under a fixed value of Vbias chosen within the transition region of the curve.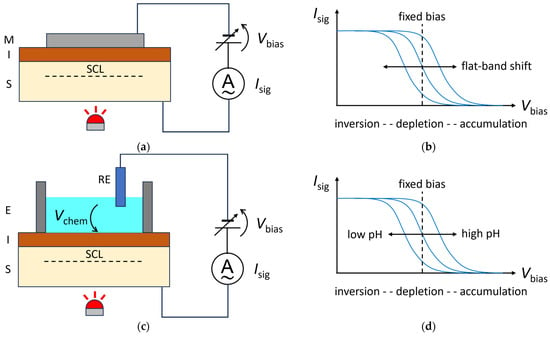
Figure 1. Comparison of SLPT (top) and LAPS (bottom) principles. (a) In SLPT, a chopped light beam illuminates an MIS structure under a bias voltage Vbias, and the amplitude of the photocurrent Isig is recorded. (b) The Isig−Vbias curve of SLPT shifts horizontally depending on the flat-band voltage of the MIS structure. Under a fixed value of Vbias, the current signal Isig varies depending on the local value of the flat-band voltage. (c) The sensing surface of a LAPS is the surface of the insulator, which is in contact with the solution under test, forming an electrolyte–insulator–semiconductor (EIS) structure. A potential difference Vchem is built up between the solution and the sensing surface, depending on the activity of the target analyte. The semiconductor layer is illuminated with a chopped light beam to generate the current signal Isig. (d) The Isig−Vbias curve of a LAPS shifts horizontally depending on the activity of the target analyte. Under a fixed value of Vbias, the current signal Isig varies depending on the local value of the activity of the target analyte.
