Vanillin, one of the most widely used and appreciated flavoring agents worldwide, is the main constituent of vanilla bean extract, obtained from the seed pods of various members belonging to the Orchidaceae family. Due to the great demand in the food confectionery industry, as well as in the perfume industry, medicine, and more, the majority of vanillin used today is produced synthetically, and only less than one percent of the world’s vanilla flavoring market comes directly from the traditional natural sources. The increasing global demand for vanillin requires alternative and overall sustainable new production methods, and the recovery from biobased polymers, like lignin, is an environmentally friendly alternative to chemical synthesis.
- flavor
- flavor authentication
- lignin depolymerization
- lignin valorization
1. Introduction

2. Types of Vanillin
Commercial vanillin can be categorized on the basis of its origin. A summary diagram of the main types of commercial vanillin including sources and market share is reported in Figure 2. The first question about vanillin production is whether it is a biobased product or not. In the first case, vanillin is defined as natural when the source is vanilla extract or vanilla pods or when it is a plant-based vanillin coming from the biotechnological treatment of compounds such as ferulic acid, eugenol, isoeugenol and guaiacol, obtained from natural fonts such as wood, rice, cloves or straw. On the other hand, if vanillin is not biobased, it is a synthetic vanillin obtained via chemical methods from products recovered from oil or from plant-based precursors. The market share for the different types of vanillin is reported in Figure 2. Synthetic vanillin accounts for 88% of the global market, whereas natural vanillin from pods accounts for only 1.5% and plant-based vanillin 11.5%.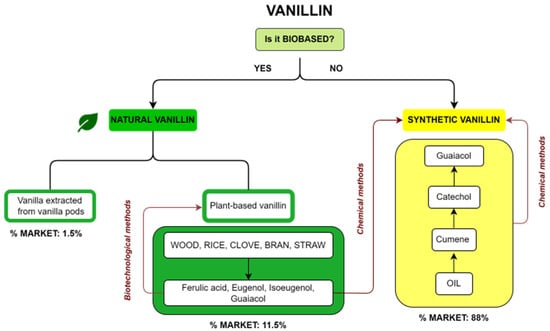
2.1. Natural Vanillin
2.1.1. Extraction from Vanilla Pods
Natural vanillin is recovered from the pod of a tropical orchid, especially the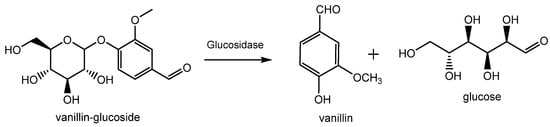
2.1.2. Plant-Based Vanillin
2.2. Synthetic Vanillin
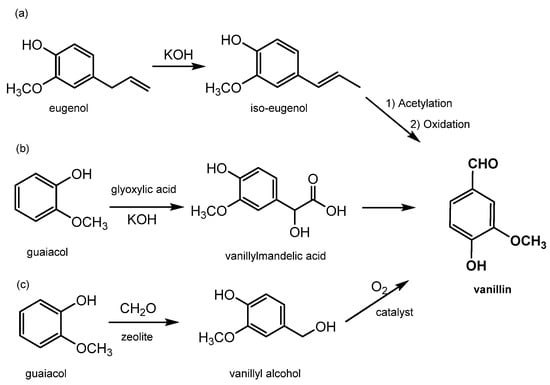
2.3. Vanillin Authentication
2.3.1. Chemical Fingerprint
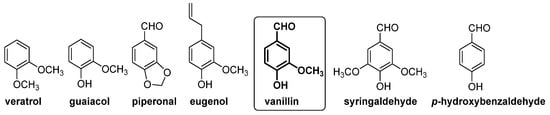
2.3.2. Isotopic Fingerprint
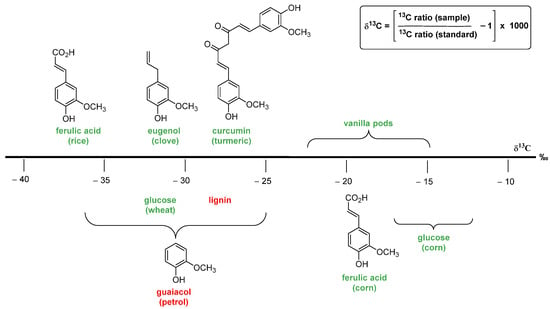
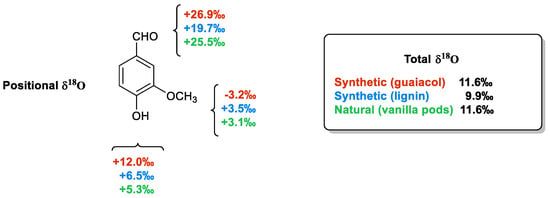
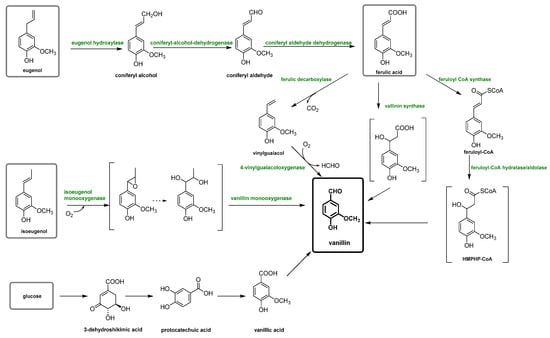
3. Vanillin Production from Lignin
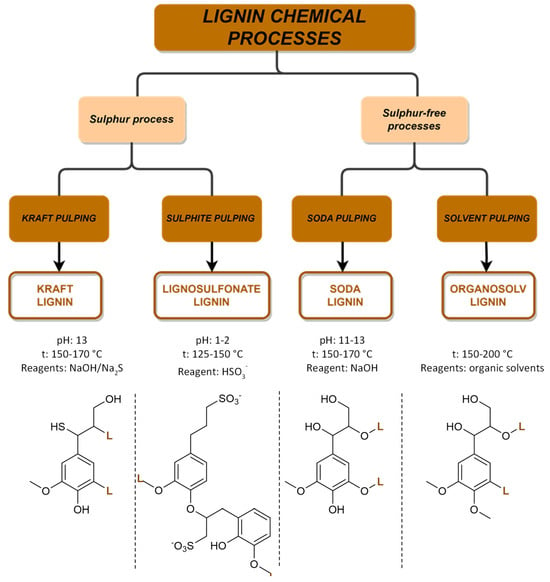
As the global economy continues to shift towards renewable feedstocks, there is a growing interest in developing new applications for lignin, which is driving commercial efforts. Moreover, from a circular economy perspective, the fractionation of lignocellulosic waste biomasses is also currently being highly investigated in order to recover and valorize the main components constituted by polysaccharides and lignin [59,60].
3.1. Lignin Oxidation
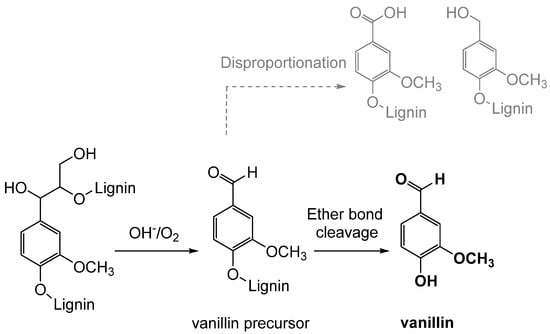
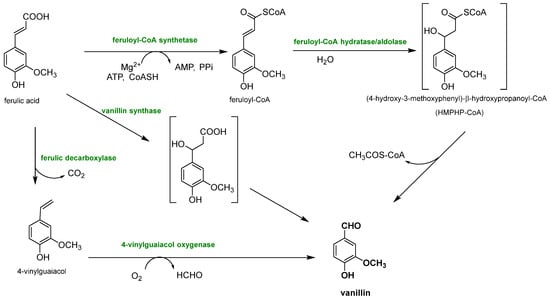
3.2. Biotechnological Lignin Transformations
3.3. Lignin Hydrothermal Liquefaction
3.4. Lignin Electrochemical Depolymerization
Electrochemical depolymerization is considered one of the most promising techniques for producing aromatic fine chemicals via a lignin cleavage. In fact, this process, which is an anodic degradation, is particularly environmentally friendly because it does not need intensive energy-consuming pre-treatments, such as high temperature and pressure, and can use renewable energy as a power source [53]. Moreover, this method overcomes the major issues associated with the production of bio-based vanillin linked to the use of toxic reagents and the presence of waste by-products that could contaminate the desired final vanillin. However, this technique is not widely exploited due to the low yield it produces which is conditioned by the nature of the electrode responsible for mass transport and flow distribution and by the electrochemically active area. Active nickel-based electrodes were tested in 2016 by Stiefel et al. on Kraft lignin [53]. They experimented with these electrodes at room temperature, ambient pressure and using a current of 8 A, obtaining a lignin degradation of 81% in 2 h, 87% in 3 h and 96% in 11 h [53]. The reduction of lignin molecular weight to 220 Da was achieved, but they recovered, using a membrane module containing polymeric tight ultrafiltration membranes in a constant-flow cross-flow mode, vanillin, acetovanillone and different carboxylic acids with an individual yield of these products lower than 0.5%. The authors reported that this method was still in development and the yields could be improved in order to use the technique on an industrial scale.3.4. Lignin Electrochemical Depolymerization
Electrochemical depolymerization is considered one of the most promising techniques for producing aromatic fine chemicals via a lignin cleavage. In fact, this process, which is an anodic degradation, is particularly environmentally friendly because it does not need intensive energy-consuming pre-treatments, such as high temperature and pressure, and can use renewable energy as a power source [98]. Moreover, this method overcomes the major issues associated with the production of bio-based vanillin linked to the use of toxic reagents and the presence of waste by-products that could contaminate the desired final vanillin. However, this technique is not widely exploited due to the low yield it produces which is conditioned by the nature of the electrode responsible for mass transport and flow distribution and by the electrochemically active area. Active nickel-based electrodes were tested in 2016 by Stiefel et al. on Kraft lignin [98]. They experimented with these electrodes at room temperature, ambient pressure and using a current of 8 A, obtaining a lignin degradation of 81% in 2 h, 87% in 3 h and 96% in 11 h [98]. The reduction of lignin molecular weight to 220 Da was achieved, but they recovered, using a membrane module containing polymeric tight ultrafiltration membranes in a constant-flow cross-flow mode, vanillin, acetovanillone and different carboxylic acids with an individual yield of these products lower than 0.5%. The authors reported that this method was still in development and the yields could be improved in order to use the technique on an industrial scale.3.5. Lignin Photocatalytic Depolymerization
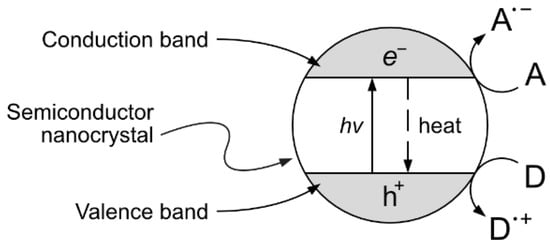
One interesting approach, although still at the experimental stage, is the production of vanillin via controlled oxidation of lignin using photocatalytic techniques. The implementation of this strategy is particularly appealing because of the potential sustainability of the resulting process. The selection of studies given in the following paragraphs can provide a good starting point for the development of further research. Heterogeneous photocatalysis is based on the photon-driven promotion of an electron from the valence band to the conduction band, resulting in a catalytic activated semiconductor crystal. The formed positive hole (h+) and free electron (e−) can then migrate from the bulk to the surface of the catalyst crystal, becoming available for redox reactions with suitable electron donors and acceptors (see Figure 123).