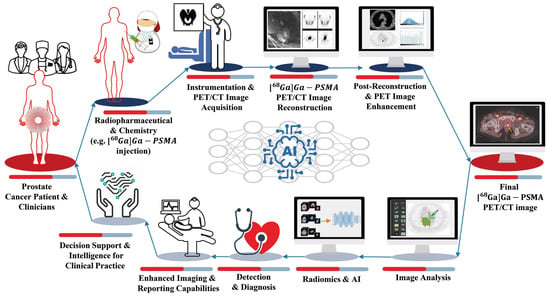
Radiotheranostics refers to the pairing of radioactive imaging biomarkers with radioactive therapeutic compounds that deliver ionizing radiation. Given the introduction of very promising radiopharmaceuticals, the radiotheranostics approach is creating a novel paradigm in personalized, targeted radionuclide therapies (TRTs), also known as radiopharmaceuticals (RPTs). Radiotherapeutic pairs targeting somatostatin receptors (SSTR) and prostate-specific membrane antigens (PSMA) are increasingly being used to diagnose and treat patients with metastatic neuroendocrine tumors (NETs) and prostate cancer. In parallel, radiomics and artificial intelligence (AI), as important areas in quantitative image analysis, are paving the way for significantly enhanced workflows in diagnostic and theranostic fields, from data and image processing to clinical decision support, improving patient selection, personalized treatment strategies, response prediction, and prognostication. Furthermore, AI has the potential for tremendous effectiveness in patient dosimetry which copes with complex and time-consuming tasks in the RPT workflow.
- radiotheranostics
- radiomics
- artificial intelligence
- SSTR
- PSMA
- workflow
1. Introduction
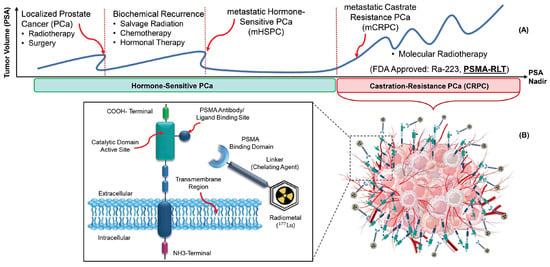
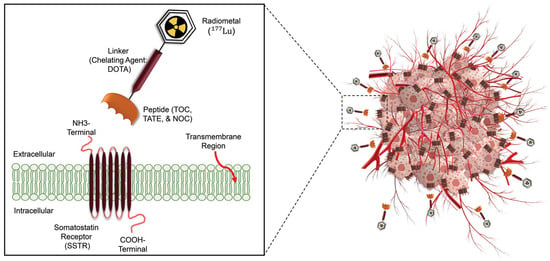
| Therapeutic Radioisotopes |
Diagnostic Radioisotopes-Pharmaceuticals | |
|---|---|---|
| SSTRs Target/NET | PSMA Target/mCRPC | |
| 177Lu | [68Ga]Ga-DOTA-TATE PET | [68Ga]Ga-PSMA-617 PET |
| [68Ga]Ga-DOTA-TOC PET | [68Ga]Ga-PSMA-I&T PET | |
| [68Ga]Ga-PSMA-11 PET | ||
| [64Cu]Cu-DOTA-TATE PET | [64Cu]Cu-PSMA-617 PET | |
| [64Cu]Cu-DOTA-TOC PET | ||
| No Clinical Match | [18F]PSMA-617 PET | |
| No Clinical Match | [44Sc]Sc-PSMA-617 PET | |
| 225Ac | [177Lu]Lu-DOTA-TATE SPECT | [177Lu]Lu-PSMA-617 SPECT |
| [177Lu]Lu-DOTA-TOC SPECT | ||
| 90Y | [177Lu]Lu-DOTA-TATE SPECT | [177Lu]Lu-PSMA-617 SPECT |
| [177Lu]Lu-DOTA-TOC SPECT | [177Lu]Lu-J591 SPECT | |
| [111In]In-DOTA-TATE SPECT | [111In]In-J591 SPECT | |
| [111In]In-DOTA-TOC SPECT | ||
2. Radiomics and AI Workflow
In precision medicine, radiomics is currently underway in research based on feature extraction from medical images. Radiomics paves the way to map multimodal imaging into quantitative information on a large scale [24]. Figure 3 depicts the steps required to build a predictive model from medical images [25,26][25][26]. The radiomics workflow begins with image acquisition and segmentation. After image post-processing, hundreds of radiomic features (RFs) are measured from segmented regions to provide raw data for developing the final model. The major categories of features are as follows: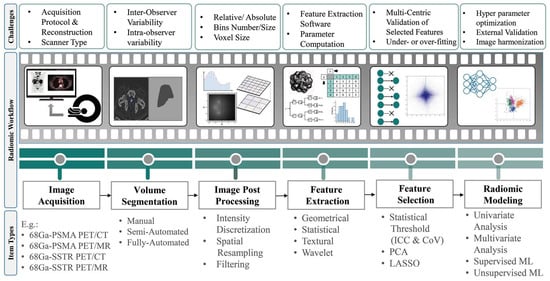
-
Geometric or shape features: based on the segmented regions.
-
Statistical or intensity features: computed using intensity values in each image region.
-
Textural features (TFs): quantification of image intensity and regularity via mathematical functions.
-
Wavelet or high-order features: the image transformation process is essential to obtain these features.
References
- Li, T.; Ao, E.C.; Lambert, B.; Brans, B.; Vandenberghe, S.; Mok, G.S. Quantitative imaging for targeted radionuclide therapy dosimetry-technical review. Theranostics 2017, 7, 4551.
- Teker, F.; Elboga, U. Is SUVmax a useful marker for progression-free survival 177 in patients with metastatic GEP-NET receiving Lu-DOTATATE therapy? Hell. J. Nucl. Med. 2021, 24, 122–131.
- Huizing, D.; Aalbersberg, E.A.; Versleijen, M.W.; Tesselaar, M.E.; Walraven, I.; Lahaye, M.J.; de Wit van der Veen, B.J.; Stokkel, M.P. Early response assessment and prediction of overall survival after peptide receptor radionuclide therapy. Cancer Imaging 2020, 20, 57.
- Jeelani, S.; Reddy, R.J.; Maheswaran, T.; Asokan, G.; Dany, A.; Anand, B. Theranostics: A treasured tailor for tomorrow. J. Pharm. Bioallied Sci. 2014, 6 (Suppl. S1), S6.
- Sjögreen Gleisner, K.; Chouin, N.; Gabina, P.M.; Cicone, F.; Gnesin, S.; Stokke, C.; Konijnenberg, M.; Cremonesi, M.; Verburg, F.A.; Bernhardt, P. EANM dosimetry committee recommendations for dosimetry of 177Lu-labelled somatostatin-receptor-and PSMA-targeting ligands. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 1778–1809.
- Khazaei, Z.; Jarrahi, A.M.; Momenabadi, V.; Ghorat, F.; Adineh, H.; Sohrabivafa, M.; Goodarzi, E. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide stomach cancers and their relationship with the human development index (HDI). World Cancer Res. J. 2019, 6, e1257.
- Dy, G.W.; Gore, J.L.; Forouzanfar, M.H.; Naghavi, M.; Fitzmaurice, C. Global burden of urologic cancers, 1990–2013. Eur. Urol. 2017, 71, 437–446.
- Steele, C.B.; Li, J.; Huang, B.; Weir, H.K. Prostate cancer survival in the United States by race and stage (2001–2009): Findings from the CONCORD-2 study. Cancer 2017, 123, 5160–5177.
- Cooperberg, M.R.; Moul, J.W.; Carroll, P.R. The changing face of prostate cancer. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2005, 23, 8146–8151.
- Jones, W.; Griffiths, K.; Barata, P.C.; Paller, C.J. PSMA theranostics: Review of the current status of PSMA-targeted imaging and radioligand therapy. Cancers 2020, 12, 1367.
- Rahbar, K.; Afshar-Oromieh, A.; Jadvar, H.; Ahmadzadehfar, H. PSMA theranostics: Current status and future directions. Mol. Imaging 2018, 17, 1536012118776068.
- Ahmadzadehfar, H.; Aryana, K.; Pirayesh, E.; Farzanehfar, S.; Assadi, M.; Fallahi, B.; Shafiei, B.; Ayati, N.; Amoui, M. The Iranian Society of Nuclear Medicine practical guideline on radioligand therapy in metastatic castration-resistant prostate cancer using 177Lu-PSMA. Iran. J. Nucl. Med. 2018, 26, 2.
- Shakeri, S.; Askari, E.; Zarehparvar, S.; Farahmandfar, F.; Norouzbeigi, N.; Salek, R.; Aryana, K. Ga-PSMA-11 PET/CT for staging and patient management of high-risk prostate cancer: A single-center experience from Iran. Iran. J. Nucl. Med. 2022, 30, 33–39.
- Liberini, V.; Laudicella, R.; Balma, M.; Nicolotti, D.G.; Buschiazzo, A.; Grimaldi, S.; Lorenzon, L.; Bianchi, A.; Peano, S.; Bartolotta, T.V. Radiomics and artificial intelligence in prostate cancer: New tools for molecular hybrid imaging and theragnostics. Eur. Radiol. Exp. 2022, 6, 27.
- FDA. FDA Approves Pluvicto for Metastatic Castration-Resistant Prostate Cancer. 2022. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-pluvicto-metastatic-castration-resistant-prostate-cancer (accessed on 23 March 2022).
- Fallahi, B.; Khademi, N.; Karamzade-Ziarati, N.; Fard-Esfahani, A.; Emami-Ardekani, A.; Farzanefar, S.; Eftekhari, M.; Beiki, D. 99mTc-PSMA SPECT/CT versus 68Ga-PSMA PET/CT in the evaluation of metastatic prostate cancer. Clin. Nucl. Med. 2021, 46, e68–e74.
- Harsini, S.; Fallahi, B.; Ziarati, N.K.; Razi, A.; Amini, E.; Emami-Ardekani, A.; Fard-Esfahani, A.; Parizi, M.K.; Farzanehfar, S.; Beiki, D. A Prospective Study on -PSMA PET/CT Imaging in Newly Diagnosed Intermediate-and High-Risk Prostate Cancer. Asia Ocean. J. Nucl. Med. Biol. 2021, 9, 101.
- Das, S.; Dasari, A. Epidemiology, incidence, and prevalence of neuroendocrine neoplasms: Are there global differences? Curr. Oncol. Rep. 2021, 23, 43.
- Taal, B.; Visser, O. Epidemiology of neuroendocrine tumours. Neuroendocrinology 2004, 80 (Suppl. S1), 3–7.
- Virgolini, I.; Ambrosini, V.; Bomanji, J.B.; Baum, R.P.; Fanti, S.; Gabriel, M.; Papathanasiou, N.D.; Pepe, G.; Oyen, W.; De Cristoforo, C. Procedure guidelines for pet/ct tumour imaging with 68Ga-dota-conjugated peptides: 68Ga-dota-toc, 68Ga-dota-noc, 68Ga-dota-tate. Eur. J. Nucl. Med. Mol. Imaging 2010, 37, 2004–2010.
- FDA. FDA Approves Lutetium Lu, F. 177 Dotatate for Treatment of GEP-NETS. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-lutetium-lu-177-dotatate-treatment-gep-nets (accessed on 26 January 2018).
- Miller, C.; Rousseau, J.; Ramogida, C.F.; Celler, A.; Rahmim, A.; Uribe, C.F. Implications of physics, chemistry and biology for dosimetry calculations using theranostic pairs. Theranostics 2022, 12, 232.
- Yousefirizi, F.; Decasez, P.; Amyar, A.; Ruan, S.; Saboury, B.; Rahmim, A. Artificial Intelligence-Based Detection, Classification and Prediction/Prognosis in PET Imaging: Towards Radiophenomics. arXiv 2021, arXiv:2110.10332.
- Gillies, R.J.; Kinahan, P.E.; Hricak, H. Radiomics: Images are more than pictures, they are data. Radiology 2016, 278, 563.
- Mayerhoefer, M.E.; Materka, A.; Langs, G.; Häggström, I.; Szczypiński, P.; Gibbs, P.; Cook, G. Introduction to radiomics. J. Nucl. Med. 2020, 61, 488–495.
- Lambin, P.; Leijenaar, R.T.; Deist, T.M.; Peerlings, J.; De Jong, E.E.; Van Timmeren, J.; Sanduleanu, S.; Larue, R.T.; Even, A.J.; Jochems, A. Radiomics: The bridge between medical imaging and personalized medicine. Nat. Rev. Clin. Oncol. 2017, 14, 749–762.
- Ray, S. A quick review of machine learning algorithms. In Proceedings of the 2019 International Conference on Machine Learning, Big Data, Cloud and Parallel Computing (COMITCon), Faridabad, India, 14–16 February 2019; IEEE: New York, NY, USA, 2019; pp. 35–39.
- Alzubaidi, L.; Zhang, J.; Humaidi, A.J.; Al-Dujaili, A.; Duan, Y.; Al-Shamma, O.; Santamaría, J.; Fadhel, M.A.; Al-Amidie, M.; Farhan, L. Review of deep learning: Concepts, CNN architectures, challenges, applications, future directions. J. Big Data 2021, 8, 1–74.
- Kumar, V.; Gu, Y.; Basu, S.; Berglund, A.; Eschrich, S.A.; Schabath, M.B.; Forster, K.; Aerts, H.J.; Dekker, A.; Fenstermacher, D. Radiomics: The process and the challenges. Magn. Reson. Imaging 2012, 30, 1234–1248.
- Reuzé, S.; Schernberg, A.; Orlhac, F.; Sun, R.; Chargari, C.; Dercle, L.; Deutsch, E.; Buvat, I.; Robert, C. Radiomics in nuclear medicine applied to radiation therapy: Methods, pitfalls, and challenges. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 1117–1142.
- Orlhac, F.; Nioche, C.; Klyuzhin, I.; Rahmim, A.; Buvat, I. Radiomics in PET imaging: A practical guide for newcomers. PET Clin. 2021, 16, 597–612.
- Bradshaw, T.J.; Boellaard, R.; Dutta, J.; Jha, A.K.; Jacobs, P.; Li, Q.; Liu, C.; Sitek, A.; Saboury, B.; Scott, P.J. Nuclear medicine and artificial intelligence: Best practices for algorithm development. J. Nucl. Med. 2022, 63, 500–510.
