Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Valentina Biagioli and Version 2 by Lindsay Dong.
Nutrients play a crucial role in enriching and diversifying the microbiota, derived not only from solid food but also from maternal dietary patterns during gestation.
- microbiota
- human milk oligosaccharides
- complementary feeding
1. Introduction
From the first moments of life, the newborn’s body begins a mutualistic relationship with numerous microorganisms. This condition represents a true “superorganism” composed of bacteria, viruses, archaea, and unicellular eukaryotes. Bacteroidetes and Firmicutes are the major bacterial phyla, with subgroups such as Fusobacteria, Cyanobacteria, Proteobacteria, Verrucomicrobia, Actinobacteria, and a few others [1]. These microorganisms colonize our entire organism, including the integumentary system and mucosal cavities (e.g., pulmonary, oral, and vaginal); however, most are found in the gastrointestinal tract (GIT). Moreover, the colon contains more than 70% of the microorganisms colonizing the GIT [2]. The intestinal microbiota represents a “vital organ”, which, owning to the numerous connective pathways such as the neural, endocrine, immunological, and metabolic pathways, can communicate with areas that are anatomically distant from one another [3].
A balanced intestinal microbial ecosystem, called eubiosis, is effective in controlling a variety of infectious diseases. Therefore, balanced nutrition from qualitative and quantitative points of view and proper food processing to preserve their organoleptic and nutritional properties are fundamental for human health and for maintaining a healthy gut microbial composition [2]. On the contrary, the prolonged use of antibiotics causes an imbalance between the different strains that make up the microbiota, referred to as dysbiosis [4], a condition characterized by a shift in the composition of commensal bacterial strains toward a more pathogenic profile [2]. This change can lead to an increase in intestinal permeability, consequently triggering an inappropriate immune response [5]. This so-called “leaky gut” is often associated with conditions such as dysbiosis and gastrointestinal symptoms, which also appear to play a key role in the pathogenesis of autism spectrum disorder (ASD) [6][7][6,7]. Moreover, scientific evidence shows that microbiome dysbiosis correlates to the pathophysiology of allergies and autoimmune disorders [8]. Notably, the contribution of the human gut microbiota in early life is fundamental for the development and maturation of the infant mucosal and immune system (Figure 1).
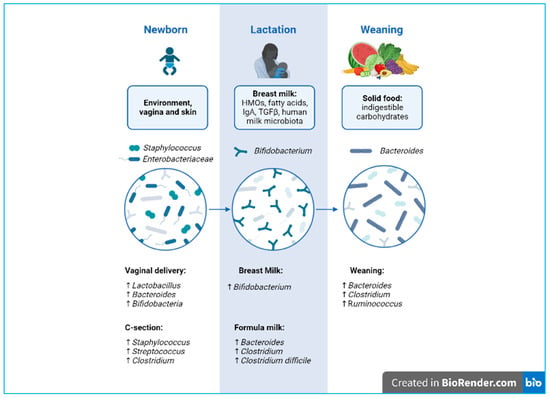
Figure 1.
Developmental phases of microbiota biodiversity from delivery to weaning. Created with
.
1.1. What Is the “Window of Opportunity”?
1.1.1. Maternal Nutrition and Infant Gut Microbiome: How Materno–fetal Exchange Influences the Development of Neonates
Pregnancy is a unique biological process that involves numerous physiological changes to support the health of both the mother and the fetus. Moreover, the placenta is a highly specialized organ, which divides the fetal and maternal environments. Historically, the placenta was considered a sterile organ, but, in recent years, new evidence has demonstrated the possible existence of a placental microbiome. In 2014, the first studies based on bacterial DNA demonstrated the existence of a placental microbiota even during full-term and normal pregnancies [9][24]. Metagenomics and metabolomics studies have shown that the maternal intestinal microbiota plays an important role in the transport of metabolites derived from commensal bacteria across the placental barrier and is therefore able to modulate fetal development [10][25]. Exogenous factors such as diet and drug intake, as well as endogenous factors such as maternal stress and alterations in the metabolic state, influence the maternal intestinal microbiota [11][26]. Even during a normal pregnancy, the maternal microbiota changes its composition. For example, Faecalibacterium is a producer of short-chain fatty acids (SCFAs), which tends to decrease in the last trimester of pregnancy [12][30]. SCFAs such as acetate, propionate, and butyrate are of fundamental importance as they provide 60–70% of the energy needs of colonocytes [13][31]; in particular, butyrate promotes cellular repair and regeneration processes, maintaining epithelial integrity [14][15][32,33]. The fetal microbiota composition is therefore strongly influenced by prenatal factors and strictly dependent on maternal lifestyle and habits. For example, smoking and a Western diet style rich in sugars, processed foods, and saturated fats negatively influence the fetal microbiota [16][34]. The intrauterine life and the first years of life represent a critical time to shape the newborn’s microbiota. In humans, the composition of the intestinal microbiota appears to stabilize around 2.5–4 years of life [17][35]. Before this stabilization occurs, the intestinal microbiota of the newborn appears to be much more susceptible to stimuli that modulate it [18][19][36,37].1.1.2. Vaginal Delivery and Vertical Transmission of Microbiota
During natural birth, the fetus passes through the maternal vaginal canal, coming into contact with the microbial ecosystem present (vaginal and fecal). This has a long-term influence on the composition and development of the newborn’s microbiota [20][38].1.1.3. Caesarean Section
During a cesarean (C-) section, the baby has increased contact with the mother’s integumentary system and with the microbes present in the hospital environment [21][39]. Contrary to vaginal births, C-section-delivered newborns show reduced microbiota diversity and richness, with significantly lower levels of bacterial genera such as Bifidobacterium, Lactobacillus, and Bacteroides, as well as higher levels of Staphylococcus. Interestingly, clinical studies highlight a relationship between C-section and immune disorders, such as allergies and asthma [22][40]. However, further studies are needed to clarify the long-term role of the altered microbiota.2. Human Milk and Neonatal Brain Development
2.1. Macro- and Micronutrient Composition
Breast milk is made up of macro- and micronutrients specialized in supporting the physiological and metabolic functions, neural development, and the intestinal microbiota of the newborn [23][49]. Two weeks after giving birth, human milk is considered fully mature. Mature milk contains 6.9–7.2% carbohydrates, 3–5% fats, 0.8–0.9% proteins, and 0.2% mineral components [24][44] (Figure 23).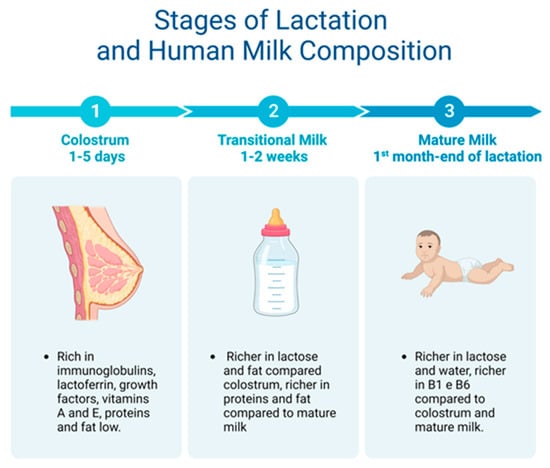
Figure 23. From colostrum to mature milk and their different properties.
2.2. Human Milk Oligosaccharides
Human milk oligosaccharides (HMOs) are complex, indigestible glycans present in high quantities in breast milk [25]. HMOs act as real prebiotics as they cross the intestinal lumen and are fermented by the intestinal microbiota [26]. After lactose and lipids, HMOs are the third most abundant components of human milk, with high quantities (20–25 ng/L) in HC, gradually decreasing to 5–15 ng/L in mature milk.
HMOs prevent infection with pathogenic bacteria such as
From colostrum to mature milk and their different properties.
2.2. Human Milk Oligosaccharides
Human milk oligosaccharides (HMOs) are complex, indigestible glycans present in high quantities in breast milk [50]. HMOs act as real prebiotics as they cross the intestinal lumen and are fermented by the intestinal microbiota [51]. After lactose and lipids, HMOs are the third most abundant components of human milk, with high quantities (20–25 ng/L) in HC, gradually decreasing to 5–15 ng/L in mature milk.
HMOs prevent infection with pathogenic bacteria such as
Salmonella
,
Campylobacter
, and
Listeria
and promote
Bifidobacterium [27], thus limiting the growth of bacteria potentially harmful to the newborn [28].
2.3. Other Bioactive Compounds
Some bioactive components of breast milk such as insulin-like growth factors (IGFs) 1 and 2 act as energy substrates for newborns, promoting the growth and development of various tissues. Among the bioactive components, there is lactoferrin, a glycoprotein that binds iron and has antimicrobial activity. Furthermore, secretory (S)IgA and SIgG are the most abundant immunoglobulins in milk, providing support to the newborn’s immune functions [29] by preventing the adhesion of pathogens to the surface of epithelial cells. SIgA is present at concentrations up to 12 mg/L in HC. [52], thus limiting the growth of bacteria potentially harmful to the newborn [48].
