Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Camila Xu and Version 1 by Kubilay Kaptan.
Cement, being one of the most widely utilized materials for construction, plays a crucial role as the primary binder in concrete, leading to the formation of a durable, stone-like, hard material capable of withstanding various loads.
- construction and demolition waste
- carbon dioxide reduction
- cement production
1. Introduction
Cement, being one of the most widely utilized materials for construction, plays a crucial role as the primary binder in concrete, leading to the formation of a durable, stone-like, hard material capable of withstanding various loads [1,2,3,4][1][2][3][4].
The conventional kind of cement, known as ordinary Portland cement (OPC), primarily comprises over 90% Portland cement clinker. This particular type of cement is derived from readily accessible raw materials that are widely abundant and cost-effective, making it easily obtainable in nearly all regions [5].
This inexpensive mineral binder has rapid hardening properties in nearly all livable environments, enabling the creation of diverse structures [6]. Moreover, its user-friendly nature allows untrained individuals, including those lacking literacy skills, to utilize it effectively for self-construction purposes [5].
Cement constitutes approximately 10% of the total volume of concrete on a global scale, and approximately 50% of cement is allocated to produce concrete, while the remaining portion is designated for applications such as mortars, pastes, and pre-manufactured products [7].
1.1. Cement Production
1.1.1. Global Cement Production
The global output of cement has witnessed a significant rise over the years. Specifically, it has escalated from 0.94 billion tons in 1970 to 2.284 billion tons in 2005, further increasing to 4.05 billion tons in 2017 and reaching 4.1 billion tons in 2018 [8].
In the year 2017, the countries of China and India, which are recognized as the largest global manufacturers, collectively accounted for 64% of the global cement production. This equated to a total output of 2.61 million tons of cement out of the whole global production of 4.05 million tons [6]. In the year 2019, the primary producers of cement were China, India, the European Union, and the United States [9,10][9][10]. These four entities collectively accounted for 56.1%, 7.8%, 4.4%, and 2.2% of the total cement production, respectively [11].
Current cement consumption is about 4.2 billion tons per year [12], which is enough to produce almost 1.6 m3 of concrete per person. This amount, which is approximately half of the volume of food produced worldwide, is expected to reach approximately 6 billion tons by the end of 2050 [7,13][7][13].
1.1.2. Cement Production Stages
Cement is derived from a combination of limestone, clay, and sand, which serve as the primary sources of lime, silica, alumina, and iron [14]. Cement production by the dry manufacturing process consists of six stages [6,15,16,17,18][6][15][16][17][18].
During the initial phase, the raw materials necessary for the process are extracted through mining operations, such as limestone, clay, laterite, bauxite, iron ore, kaolinite, sandstone, and other similar inorganic materials. All of them, properly dosed, constitute the “Portland clinker crude” (PCC), the last six being in addition the fluxes or mineralizers of the first two (those with the highest dosage), to which, despite their very considerable lower dosage, they reduce their melting point so that they can chemically react more and better and thus form Portland clinker permanently. The second stage of the process entails the characterization of diverse raw materials and their proper dosage to create PCC. During the third stage, the PCC is introduced into a preheating chamber. During the fourth stage, the pre-heated decarbonized PCC is introduced into the rotary kiln to undergo the process of clinkerization at a temperature ≥1450 °C. During the fifth stage, the clinker that emerges from the kiln undergoes a quick cooling process facilitated using pressurized air. During the concluding phase, the cooled clinker is recovered from the cooling vessels and then transferred to the mills. The clinker is ground together with the optimum amount of setting regulator [19] (natural gypsum stone) into powder using a ball mill or roller mill, or a vertical mill, and the pulverized cement is transported to storage silos using a transportation system suitable for shipping (Figure 1). Nevertheless, if the composition of the ground material consists of a combination of natural and/or artificial pozzolans and/or GGBFS along with Portland clinker, the ideal quantity of setting regulator needs to be determined utilizing the R. Talero method [20].
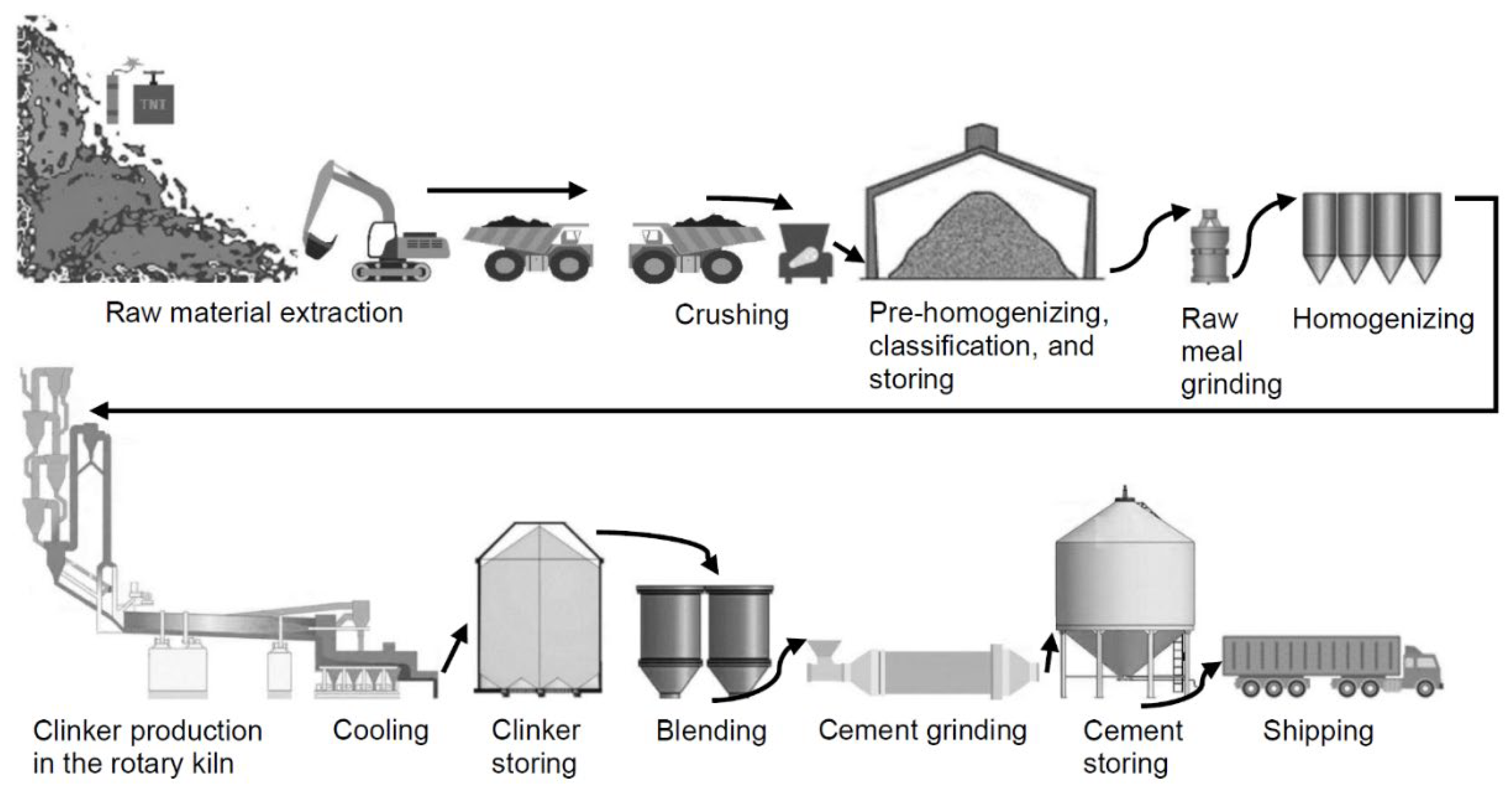
Figure 1.
Cement production.
The six phases can be condensed into three primary stages: raw meal preparation, clinker production, and finish grinding [21,22][21][22].
1.2. Environmental Impacts of Cement Production
In comparison to the year 1750, it has been observed that concentrations of CO2 in the Earth’s atmosphere have risen from 280 to 410 parts per million by volume (ppmV) [23,24,25,26,27,28,29][23][24][25][26][27][28][29]. This upward trajectory is projected to persist in the coming decades, potentially leading to a temperature rise of up to 5.8 °C during the present century [29,30][29][30].
Approximately 40% of worldwide CO2 emissions can be attributed to four key industries: power plants, iron and steel manufacturing, cement manufacturing, and chemicals and petrochemicals [29]. The cement sector is identified as the primary contributor of process emissions [16,31][16][31].
Based on the available worldwide CO2 emission data, cement plants made a substantial contribution of 2.9 billion tons of CO2 in the year 2021 [7]. This figure represents an almost fivefold increase when compared to the emission level of 0.57 billion tons recorded in 1990 [29,32][29][32].
CO2 Emissions from the Cement Industry
Cement production is a highly resource-intensive process that consumes significant amounts of energy and raw materials [16]. This process leads to the emission of CO2 through two primary pathways: direct emissions from the combustion of fossil fuels in the kiln and indirect emissions from the calcination process of the primary raw material, predominantly limestone [33]. Additionally, the consumption of electricity in cement production, particularly when generated from fossil fuel combustion, contributes to overall CO2 emissions [34].
The emission of CO2 during the manufacturing of one metric ton of Portland cement is predicted to range from 0.73 to 0.99 metric tons throughout various geographical regions [34]. It can be asserted that the manufacturing of one kilogram of Portland cement results in the emission of about one kilogram of CO2 into the atmosphere [35,36][35][36].
The global production of this product is responsible for approximately 5–9% of CO2 emissions [13,16,29,37,38,39,40,41][13][16][29][37][38][39][40][41]. Furthermore, it accounts for significant emissions of carbon monoxide (CO) and heavy metals [14]. In addition to CO2, CO, and heavy metals, the use of a substantial quantity of material has led to the excessive burden on deposits of these materials and the alteration of the environment. The production of Portland cement alone entails the consumption of approximately double the quantity of raw materials required to manufacture one metric ton of Portland cement [35].
As previously stated, the process of manufacturing Portland cement results in the emission of carbon dioxide through both direct and indirect means [39]. Indirect emissions are generated because of the calcination process, wherein limestone, the principal constituent of cement, undergoes heating [39,42,43][39][42][43]. The process of thermal decomposition causes the calcium carbonate present in limestone to undergo a chemical transformation, resulting in the formation of calcium oxide and the liberation of CO2 gas [39]. This procedure is responsible for approximately 50% of the total emissions generated during the manufacture of cement [32,41][32][41]. The production of cement involves subjecting limestone and other clay-like materials to high temperatures of approximately 1450 °C within a kiln [39,44][39][44]. Direct emissions arise because of the combustion of fossil fuels utilized for the purpose of heating the kiln, constituting approximately 40% of the total emissions associated with cement manufacturing [5,16,45][5][16][45]. The emissions associated with the quarrying of raw materials, their transportation, grinding processes [46], the electricity consumption for operating additional plant machinery, as well as the packaging and final delivery of cement, all contribute to the remaining 10% of the overall emissions [43,47][43][47].
Furthermore, a range of technological and managerial inefficiencies within the typical cement production process might result in additional CO2 emissions. Geographical location, technological factors, plant and manufacturing efficiency, the energy mix utilized for electricity generation, and the choice of kiln fuels all contribute to additional carbon dioxide CO2 emissions [29,38,39][29][38][39].
1.3. Construction and Demolition Waste
Construction waste results from building constructions and building renovations and consists of surplus material, unusable impaired or fractured material, cut-off pieces, processing waste, worn-out tools and accessories, dismantled shuttering, packaging, and waste produced by construction workers [48,49,50][48][49][50]. On the other hand, after the end of a structure’s life cycle, its demolition is crucial for the growth of cities where inadequate space is the major obstruction. CDW can also be generated in the aftermath of a natural disaster, which presents several significant challenges, such as transportation, storage in an appropriate location prior to processing, and disposal at landfill sites [51].
Overall, it can be stated that CDW is a type of solid waste generated on construction sites and during the entire or partial demolition of buildings and infrastructures [52,53,54,55,56,57,58][52][53][54][55][56][57][58].
1.3.1. CDW Composition and Generation
CDW consists primarily of inert and non-inert materials, such as gravel, concrete, sand, ceramic, tile, metal, plastic, glass, roofing materials, paper, cardboard, etc. The inert waste materials consist of soft and hard inert materials, whereas the noninert waste consists of residual waste and other materials such as metals, wood, plastic, and glass [53,59,60][53][59][60]. Inert fraction waste accounts for between 40 and 85 percent of total waste volume, excluding excavation soils [50,58,61][50][58][61].
It is estimated that the construction industry annually generates more than 3 billion tons of CDW worldwide [62,63,64][62][63][64]. This indicates that CDW accounts for approximately 36% of the world’s total waste production [65]. CDW in the United States rose from 50 million tons in 1980 to 600 million tons in 2018 [61]. More than 1.5 billion tons of CDW are produced annually in China [66[66][67],67], while in the European Union (EU), countries produce about 850 million tons/year, or 31% of the total waste generation in the EU [68].
1.3.2. Environmental Impacts of CDW
The generation of waste results in adverse externalities on the environment, even while a significant portion of CDW consists of inert materials that may not provide as significant a risk as hazardous waste [69,70][69][70]. The disposal of CDW in landfills causes landslides [71], depletes limited landfill resources, exacerbates energy consumption, amplifies greenhouse gas (GHG) emissions, poses public health concerns, and contaminates the environment [44,72,73,74,75][44][72][73][74][75].
In recent years, governmental bodies have enacted new regulations pertaining to the management of waste, encompassing responsibilities, disposal practices, and recycling efforts on a broader scale [76]. Consequently, the urban landscape is undergoing transformation through the establishment of recycling facilities, yet the current recovery rate for CDW remains very low [49,77][49][77]. The expansion of the worldwide population and the concurrent rise in sea levels have resulted in a reduction in the accessible land for dump sites, hence leading to an indirect escalation in the expenses associated with landfills [78].
2. Mitigation and Improvement Measures to Reduce CO2 in Portland Cement Production
Clinker is a transitional product in the production of cement, occurring before the mineral additions (MAs) to create the final cement product. As the temperature rises, the pre-calcined materials undergo physical and chemical transformations, causing them to liquefy and combine, resulting in the formation of lumps [39]. Thus, the manufacturing of cement emits greenhouse gases through both chemical and physical processes. The thermal decomposition of limestone releases CO2 by an endothermic chemical reaction, and the combustion of coal, fuel, or AF releases it as well (but exothermically), only that the transmission to the limestone of the heat generated at the same time, to decompose it and decarbonate it, is not carried out chemically but physically by the following ways: conduction, convection, and radiation. Although it is not possible to completely eliminate these emissions, the use of energy-saving technologies can help reduce physical emissions. Therefore, the cement industry had been actively engaged in the pursuit of techniques aimed at reducing CO2 emissions far in advance of the emergence of global warming as a prominent concern. To address this predicament, an increasing body of research has delved into the process of decarbonization within the cement sector, as outlined in Table 1.Table 1.
Relevant studies on CO
2
reduction methods for the cement industry.
| Reference | Region | Reviewed Methods |
|---|---|---|
| [81][79] | Global | Utilization of Afs/ARMs, supplementary cementitious materials (SCMs), and alternative low-carbon binders. |
| [5] | Global | Improving energy efficiency; use of Afs; clinker substitution by MAs/SCMs; utilization of carbon capture and storage (CCS), alternative clinkers, and alkali-activated materials; and improving the efficiency of cement use. |
| [82][80] | Global | Increased use of calcined clay and engineered filler with dispersants, introduction of new Portland clinker-based cement alternatives, use of alkali-activated materials, and improvement of the efficiency of cement use. |
| [16] | Global | Energy savings and the use of CCS and alternative materials (AFs, ARMs, and clinker substitute). |
| [14] | Global | Improving energy efficiency, material substitution, and the use of AFs and CCS. |
| [83][81] | Global | The use of CCS technologies, reduction of clinker/cement ratio, use of AFs, and pyro-processing improvements. |
| [84][82] | Global | Improving energy efficiency, changing fuel type, the use of CCS, substituting clinker, and improving cement use efficiency. |
| [22] | Global | Reduction of the clinker/cement ratio and the use of ARMs/AFs, energy efficiency improvements, the use of WHR and CCS, and the replacement of cement in concrete or mortar with alternative materials. |
| [85][83] | Global | Utilization of energy conservation approaches. |
| [86][84] | Global | Utilization of CCS, SCMs, and nanotechnology. |
| [87][85] | Global | Utilization of WHR, blended cements, efficiency improvements, and CCS. |
| [88][86] | Global | Utilization of CCS. |
| [10] | Global | Use of low-carbon cement technologies. |
| [89][87] | Asia | Improving energy efficiency, the use of AFs, reduction of the clinker-to-cement ratio, and utilization of emerging and innovative technologies (excess heat recovery, CCS, energy management systems, etc.) |
| [21] | China | The use of energy efficiency improvement technology, WHR, CCS, AFs, and clinker substitution. |
| [90][88] | China | The use of energy efficiency improvements, AFs, clinker substitution, and CCS. |
| [91][89] | China | Utilization of advanced efficiency technologies, ARMs, AFs, renewable electricity, CCS, and cement carbonation effects. |
| [92][90] | China | Use of energy efficiency, AFs, ARMs, and CCS. |
| [93][91] | Indonesia | The use of clinker substitutes, AFs, and WHR and upgrading kilns. |
| [94][92] | Indonesia | Improving energy efficiency and the use of clinker substitution, AFs, and CCS. |
| [95][93] | Japan | The use of energy and material efficiency strategies, AFs, reducing clinker-to-cement ratios, lowering transportation emissions, and decarbonizing electricity supply. |
| [96][94] | Japan | Reuse of building material waste. |
| [39] | Malaysia | The use of energy-efficient technologies, WHR, AFs (fuel switching/co-processing), alternative binders, and CCS. |
| [97][95] | Thailand | The use of WHR. |
| [38] | Hong Kong | The use of Afs and ARMs and the application of combined strategies. |
| [98][96] | USA | Improving energy efficiency. |
| [2] | Portugal | Use of alternative clinker technologies. |
| [99][97] | Poland | Improving energy efficiency and the use of waste as raw materials and MAs in cement production. |
| [100][98] | Italy and Germany | The use of AFs and ARMs. |
| [101][99] | Sweden | The use of CCS. |
2.1. Substitution of Alternative Fuels (AFs)
While AF substitution in the cement production process is not a novel concept [16[16][100],102], its prominence has grown considerably, and the utilization of AFs in cement manufacturing has received significant attention in recent years due to its efficacy in replacing the thermal energy derived from fossil fuels and mitigating pollutant emissions. The contemporary cement kiln exhibits a high degree of adaptability, enabling the cement industry to seamlessly transition between different fuel sources with moderate ease [5,16][5][16]. The cement rotary kiln possesses the capability to incinerate a diverse array of materials because of the extended durations spent at elevated temperatures, the inherent capacity of clinker to assimilate and confine impurities such as heavy metals within itself, and the alkaline conditions prevailing within the kiln [103][101]. The cement industry utilizes conventional fossil fuels, including coal, fuel oil, petroleum coke (petcoke), natural gas, and diesel, in its kilns and pre-heater systems to generate the elevated temperatures required for clinker production [46]. The aforementioned fuels account for over 94% of the thermal energy need in the worldwide cement industry [104][102]. The suitability of AFs is contingent upon various properties, including their physical state (solid, liquid, or gaseous), lower heating value, ash composition and content, toxicity (organic compounds, heavy metals), volatile content [105][103], humidity content, physical properties (scrap size, density, and homogeneity), content of circulating elements, grinding properties, storage/feeding capabilities, and calorific value [15,16,43,105,106,107][15][16][43][103][104][105]. The utilization of AFs offers several key benefits, namely enhanced energy recovery and the preservation of finite fossil fuel resources. These advantages result in the reduction of pollutant emissions, particularly CO2, and a projected decrease in the expenses associated with cement production [16,102,103,108,109][16][100][101][106][107]. Nevertheless, the adoption of AFs presents numerous problems as a result of the complexities associated with integrating supplementary fuel-saving methodologies. Furthermore, it is important to note that not all AFs guarantee a reduction in CO2 emissions due to their elevated carbon intensities [45,81,102,110,111][45][79][100][108][109]. AFs can be broadly categorized into three primary groups [112][110]. The first group comprises liquid AFs, encompassing materials such as waste oil, solvents, animal fat, and sewage sludge. The second group consists of solid AFs, which include waste tires (either chipped or whole), animal and bone meal, dried sewage sludge, scrap wood, and waste materials originating from various industries, such as the pulp, paper, cardboard, plastics, packaging, and textile industries. Lastly, the third group encompasses gas AFs, which encompass landfill gases, pyrolytic gases, and biogases. Typical AFs used by the cement industry include animal meat and bone meat [113[111][112][113][114][115][116][117][118][119],114,115,116,117,118,119,120,121], municipal solid waste [110,122[108][120][121][122][123][124][125][126],123,124,125,126,127,128], refuse derived fuel [129[127][128][129],130,131], waste tires [110,132[108][130][131][132],133,134], plastic waste [22[22][104][133],106,135], saw dust or wood [136[134][135],137], straw [138[136][137],139], agriculture and forest wastes [140[138][139][140],141,142], almond shells [143[141][142],144], olive residues [145][143], oil palm [146][144], food residue [147][145], rice husk ash [148][146], natural gas [149][147], biogas [150][148], sewage sludge [151[149][150][151],152,153], oil sludge [154][152], slaughterhouse residues [155][153], spent solvents [110][108], and solid recovered fuels [156,157][154][155]. It is projected that the global utilization of AFs will increase from 3% in 2006 to around 37% by 2050, resulting in a contribution of approximately 15% towards the intended overall reduction in CO2 emissions [5,34][5][34].2.2. Substitution of Alternative Raw Materials (ARMs)
The process of the decarbonation of commonly used raw materials, primarily limestone, results in the release of around 0.53 metric tons of CO2 for each metric ton of clinker produced [153][151]. Utilizing waste and by-products that include valuable minerals, such as calcium, silica, alumina, and iron, is a viable option to substitute for traditional raw materials, including clay, shale, and limestone [15,158][15][156]. The incorporation of alternative materials into the clinker recipe necessitates a prudent methodology, since any modification in the chemical composition of cement will have an impact on the ultimate quality of the product [22,159][22][157]. Various industrial by-products and waste-derived materials have been investigated as potential substitutes for limestone and clay in the production of cement. The objective is to minimize the utilization of natural resources, decrease CO2 emissions, and reduce heat consumption while ensuring that the manufacturing processes remain unaltered [159][157]. Some of the ARMs utilized in the raw meal for cement production are presented in Table 2.Table 2.
Typical ARMs utilized as a partial replacement in the raw meal.
2.2.2. Substitution of CDW as an ARM
The chemical and mineralogical properties of CDW are sufficient to qualify it as a viable substitute raw material in the limestone–clay mixture produced during the manufacturing process of Portland clinker. The composition of CDW typically includes calcium, silicon, aluminum, iron, and several trace elements, including magnesium, potassium, titanium, and sulfur. These minor elements have the potential to contribute to the development of the primary phases of Portland cement [163,169,180,290,291][161][167][178][288][289]. Furthermore, the substitution of CDW leads to a decrease in the generation of CO2. This waste serves as a source of CO2 that is separated from calcium oxide (CaO), thereby reducing the decarbonation of limestone that occurs during the flaring process in the manufacturing of clinker [163,180][161][178]. From the above-mentioned ARMs, CO2 emission related studies concerning CDW are listed in Table 3.Table 3.
CO
2
emissions for CDW substituted in the raw meal.
Table 4.
Minerals used as an addition or partial replacement for Portland cement to produce concrete.
| MA | Mix Type | Optimum Substitution (wt.%) | References | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 165 | ,166,167,168,169][26][163][164][165][166][167], cement waste [170,171,172][168][169][170], recycled aggregates (RAs) [173,174][171][172], marble and brick waste [175][173], cement kiln dust [176,177][174][175], ceramic wastes [178,179,180][176][177][178], recycled mortar or paste [181][179], cellular concrete [182][180], asbestos cement tile waste [183][181], inorganic construction waste [184][182], dam fine sediments [185,186][183][184], and dredged sediments [187,188][185][186]. | |||||||||||
| Hydrated cement waste (HCW) | HCW is obtained as a by-product from the efficient separation of fine recycled concrete aggregates. CWp-A is prepared by replacing 30% weight of ordinary Portland powder by HCW. CWp-B is prepared with a higher amount of HCW, 55% in weight. | OPp | 76.00 | - | 24.00 | - | Significant reductions in CO | Manufacturing Industry |
Sewage sludge | |||
| Agricultural Industry | 2 | emissions connected with clinker/cement production are reported in both scenarios (low or high amounts of HCW). | ||||||||||
| [ | 189, | |||||||||||
| −5 | , 2.41 × 10 | −5 | , 2.3 × 10 | −5 | , 2.2 × 10−5, and 2.1 × 10−5 respectively. | . | 190,191 | CWp-A,192,193,194,195,196][187][188][189][190][191][192 | 53.00 | - | 17.00 | 30.00 |
| Sugarcane bagasse ash (SCBA) | ][193][194], blast furnace slag [197,198,199,200,201,202][195][196][197][198][199][200], lime sludge [13,203,204][13][201][202], steel slag [201,205,206][199] | Ordinary concrete | <25 | |||||||||
| [366][365] | ||||||||||||
| [428 | [203][204], stainless steel slag [207][205], basic oxygen furnace slag [208,209][206][207], calcium carbide slag [210][208], magnesium slag [211][209], water purification sludge [212][210], heavy metal-containing sludge [213][211], electric arc furnace slag [214][212], fly ash [197,198,200,215,216,195217,218,][196219,][198220,]221,222][[213][214][215][216][217][218][219][220], red mud [199,223,224,225][197][221][222][223], oil-based mud [226,[224227]][225], iron ore tailings [228,229][226][227], copper tailings [230][228], industrial hazardous waste [231][229], paper pulp waste [232,233][230][231], marine bio-refinery waste [234][232], glass waste [38,235][38][233], plastic waste [236][234], fiber-cement waste [237][235], black dross leached residue [238][236], and titanium dioxide waste [239][237]. | |||||||||||
| [ | 299 | CNSA | ,300,301,302,, | Ordinary concrete308][303,297][304,298305,306,][299][300][301][302][303][304]307[305][306] | 5, 10, 15, 20 | 10 | The embodied carbon of 5%, 10%, 15%, and 20% CSA is 4%, 7%, 11%, and 15% lower than that of the control mix. | Agricultural and Aquacultural Industries |
Wood ash [240,241][238][239], biomass ash [110][108], sugar filter mud [242,243][240][241], pulverized eggshell waste [244][242], bone ash [121][119], and pulverized oyster and scallop shell waste [245][243]. | |||
| Natural sources | Basalt rock [104,108,246][102][106][244], natural fluorapatite [247][245], meta-schist [248][246], Callovo-Oxfordian argillite [249][247], spent volcanic soil [18], calcined clay [250,251][248][249], and spent limestone sorbent [252][250]. | |||||||||||
| Other sources | Municipal solid waste [253,254,255][251][252][253], contaminated soil [256][254], and mining waste [257][255]. | |||||||||||
2.2.1. Consideration of CC as SCM: Replacement of Portland Cement by CC
The materials evaluated in Table 2 have the potential to partially substitute for Portland clinker by means of novel variations of already utilized SCMs. Among these materials, calcined clay (CC) deserves particular attention. By subjecting ordinary clay, which typically contains at least 40% kaolinite and is widely available in the earth’s crust, to moderate heat treatment (about 700 and 850 °C), it can be transformed into a pozzolanic material called CC [258,259][256][257]. CCs, especially when combined with limestone, are being recognized as a highly promising solution due to their excellent performance and the abundance of sufficient reserves of these materials [260][258]. Limestone calcined clay (LC2) and limestone calcined clay cement (LC3) systems exploit the synergistic effects of calcined clay and limestone, enabling a significant decrease of up to 50% in the utilization of clinker [261][259]. Nevertheless, the clays typically employed in LC3 systems consist of a minimum of 40% kaolinite [258,262][256][260]. Recently, there has been a significant increase in research [263,264,265,266,267,268,269,270,271][261][262][263][264][265][266][267][268][269] focused on the potential utilization of CC as an SCM in the manufacturing of cement, with a particular emphasis on advancing its economic viability [272][270]. Zhu et al. [273][271] conducted a study on the characteristics of LC2 blended cement and compared them with fly ash (FA) and granulated blast-furnace slag (GGBS). They reported that the normal consistency of LC2 blended cement was greatly raised and the substitution of LC2 at a rate of 60% resulted in an almost twofold increase in normal consistency. Dhandapani et al. [274][272] reported that concrete produced with LC3 had superior compressive strengths compared to concrete with equal combination proportions at all ages up to 1 year. The investigation carried out by Vaasudevaa et al. [275][273] involved the substitution of cement in concrete with a combination of LC2 at a proportion of 45%. They concluded that the compressive strength of steam-cured LC3 concrete after 1 day is comparable to that of OPC concrete, exhibiting a similar strength enhancement resulting from the steam curing conditions. The study carried out by Aramburo et al. [276][274] aimed to evaluate the mechanical properties and sulfate resistance of blended cements containing a significant amount of CC as pozzolanic material. The objective was to demonstrate that these cements can meet the requirements of CEM type IV/A-SR and IV/B-SR cements as defined by the EN 197-1:2011 standard. The results obtained validated the increase in sulfate resistance and the decrease in the mechanical strength of PC when it was replaced by CC (whose matrix clay was kaolin doped with ≈50% quartz) in quantities greater than 40%. They also stated that the blended cements with high percentages of CC replacement successfully met the specified requirements regarding compressive and flexural strengths without prejudice to its decrease observed with the increase in its replacement by PC. The reason for both opposing behaviors, sulfatic and mechanical strengths, was the same: the very high, early, and fast pozzolanic activity of its silica and reactive alumina contents especially (38.0% and 15.0%, respectively) [277[275][276][277],278,279], which excessively decreases the [Ca (OH)] in the liquid phase of its pastes. To verify this, the authors repeated the tests, replacing a small portion of the CC used with slaked lime powder (calcium hydroxide, Ca (OH)). Both behaviors contrasted again, but in the opposite direction; that is, the sulfate resistance decreased, and the mechanical strengths increased, as when the replacement by PC was ≤40%. This was similar to how it also increased its resistance to carbonation, which had also been significantly diminished and seriously compromised, with an increase in the replacement of CC by PC. The more impaired the material, the greater the 40% replacement was [280][278]. A study carried out by Yu et al. [281][279] investigated the practicality of creating a cost-effective and environmentally friendly cement by combining LC2 at a significant proportion of 50–80% relative to the weight of the cement. They reported that blended cements containing 50–60% LC2 exhibit satisfactory compressive strength, decreased hydration heat, reduced environmental effect, and lower material cost per unit strength but reduced workability in comparison to plain Portland cement. This contrasts quite a bit with the results of flexural and compressive strengths obtained by Arámburo et al. [276][274]. With regard to CO2 emissions, a review of the existing literature [82,260,267,281,282,283,284,285,286,287,288][80][258][265][279| ] | ||||||||||||||||
| [ | ||||||||||||||||
| 427] | Biomass fly ash (BFA) and coal fly ash (CFA) | Ordinary concrete | BFA/CFA: 10/10, 20/20, 30/30 | 30/30 | GWP impact values (kg CO2eq) are 7.84 × 102 | |||||||||||
| CWp-B | 25.00 | - | 20.00 | 55.00 | ||||||||||||
| for the control mix, 6.62 × 10 | 2 | for 10/10, 5.38 × 10 | 2 | for 20/20, and 4.15 × 10 | Eco-friendly concrete | |||||||||||
| 2 | for 30/30. | [422][<30 | 421] | |||||||||||||
| [429][ | [ | 309,310,311][307][308] | RHA[309] | Calcium aluminate cement concrete |
2.5, 5, 7.5, 10 | 5 | 428]5%RHA could reduce CO2 emissions by 18.75%. | SCBA and CSA | Ultra-high-strength concrete | SBA/CSA: 10/2, 20/2, 30/2, 10/4, 20/4, 30/4, 10/6, 20/6, 30/6, 10/8, 20/8, 30/8 | 20/4 | Considering the cost/MPa, the results show that the use of 20/4 had a higher lower cost per m3 in comparison with all concrete mixture. The reduction in concrete cost was 18.50% compared to the control mix. | [44] | Civil construction waste (CCW) | Reusable or recyclable aggregate waste materials, such as soil from earthworks, bricks, tiles, cladding plates, mortar, concrete, and curbs, are used for CCW. CCW0–10: concrete (1%), mortar (47%), rock (2%), ceramic (13%), and soil (37%); CCW10–20: concrete (41%), mortar (39%), rock (13%), and ceramic (7%); CCW20–40: concrete (57%), mortar (34%), rock (7%), and ceramic (2%). | C-REF |
| [ | 93.20 | 6.80 | - | 328.00 | 500.00 | - | - | |||||||||
| Self-compacting concrete | 15 | |||||||||||||||
| [307][305]430][429] | [ | 312][310] | SDA | Ordinary concrete | 5, 10, 15, 20 | <20 | Embodied carbon (kg CO | Corn cob ash (CCA) and glass powder (GP) as binary cementitious material (BCM)2 | Ordinary concrete | CCA/GP: 2.5/2.5, 5/5, 7.5/7.5, 10/10 | 5/5/kg) for SDA is 0.0014. The embodied carbon of concrete mixtures incorporating 20% SDA is approximately 20% lower than that of the concrete mixtures incorporating PC as the only binder. | Concrete mixtures incorporating 5%, 10%, 15%, and 20% BCM as partial replacement of Portland cement have 4.3%, 8.3%, 12.7%, and 16.8% lower embodied carbon control than the mixtures without BCM. Similarly, the incorporation of BCM into the mixtures led to a reduction of approximately 21% in the embodied energy of the concrete. | C-CCW-1 | 85.71 | - | |
| [307] | 14.29 | 326.00 | 488.00 | 0.60 | 2.40 | |||||||||||
| Ultra-high-strength concrete | [15–30 | 305][313,314,315][311][312][313] | SCBA | Portland fly ash cement concrete |
50, 60, 70 | 50 | ||||||||||
| [392][391] | Mixed cathode ray tubes (CRT) and mixed-container glass (MRF) | The CO | 2 | Ordinary concrete | MRF/CRT: 17/3 | 17/3-eq intensity values of control mix, BA50, BA60 and BA70 concretes were 9.65, 6.17, 6.73, and 7.67 kg CO | The GWP value is 1040 kg CO2-eq. for the control mix and 849 kg CO2-eq for 17/3. | C-CCW-4 | 89.53 | - | ||||||
| Rice husk ash (RHA) | Ordinary concrete | 10–25 | [80 | |||||||||||||
| [342] | , | 316 | ,317, | 10.47 | 318318.00 | 471.80 | [,3.00 | 5.60 | ||||||||
| 341] | 319 | ] | [314][315][316] | POFA and ESP[317 | Eco-friendly structural foamed concrete][318] | POFA/ESP: 20/5, 20/10, 20/15, 25/5, 25/10, 25/15 | 25/5 | CO2 emissions (kg CO2/m3) are 453.97 for control mix, 358.29 for 20/5, 339.61 for 20/10, 320.93 for 20/15, 339.04 for 25/5, 320.36 for 25/10, and 301.68 for 25/15. | C-CCW0–10 | 90.14 | - | 9.86 | 312.00 | 459.50 | ||
| Eco-friendly concrete | 4.90 | |||||||||||||||
| [431 | 5–15 | ][430][320,321, | 8.10 | |||||||||||||
| 322 | , | 323,324][319][320][321][322][323] | Cane bagasse ash (CBA) and waste glass (WG) | Green concrete | CBA/WG: 15/5, 10/10, 5/15 | 15/5 | Replacement of 20% of cement with CBA and WG showed reductions in CO2 emissions of about 20% compared to control mix. | C-CCW10–20 | 90.90 | - | 9.10 | 324.00 | ||||
| Self-compacting concrete | 5–15 | [325,326,327][324][325][326 | 488.00 | ] | 1.20 | 2.40 | ||||||||||
| C-CCW20–40 | 90.50 | - | 9.50 | 325.00 | 488.00 | |||||||||||
| Ultra-high-performance concrete | 20 | 0.90 | [328 | 2.40 | ||||||||||||
| , | 329 | [183][181] | Asbestos cement tile waste (ACW) | ACW in the form of aged tiles extracted from a roof. | CL-AC0 | 94.53 | 5.47 | - | 335.00 | 503.76 | - | - | ||||
| CL-AC24 | 72.05 | 3.94 | 24.01 | - | - | - | - | |||||||||
| CL-AC49 | 48.82 | 2.36 | 48.82 | 319.01 | 468.45 | 4.77 | 7.00 | |||||||||
| CL-AC74 | 24.82 | 0.72 | 74.46 | 303.06 | 434.84 | 9.53 | 13.68 | |||||||||
| CL-AC86 | 14.24 | - | 85.76 | - | - | - | - |
2.3. Replacement of MAs in Portland Cement
Due to the production of GHGs, a majority of concrete mixtures use SCMs either through the use of blended cements or by individually adding them to the mixer [217][215]. The incorporation of low-embodied carbon and low-energy elements in the substitution of Portland cement can significantly diminish the overall environmental consequences of binders and, as a result, of concrete [159,293][157][291]. These materials are commonly known as MAs or SCMs. When they are included into concrete and mixed with Portland cement, they create cementitious particles. However, on their own, they do not contain any cementitious compounds [217][215]. The selection of MAs for substituting Portland cement is contingent upon the geographical area and the specific solid waste or byproducts produced by industries or the presence of naturally occurring minerals in these regions [37]. The utilization of MAs as substitutes for Portland cement in concrete offers various sustainability benefits. MAs typically consist of industrial waste products, natural pozzolans, and activated minerals that possess either hydraulic or pozzolanic characteristics. When MAs are used alone or in contact with water, they generally do not exhibit substantial hydraulic reactions that contribute to the cementitious properties. Nevertheless, when exposed to alkaline aqueous conditions or in the presence of calcium hydroxide, fine particles undergo a chemical process known as the pozzolanic reaction. This reaction leads to the formation of hydration products that resemble those seen in Portland cement systems [200,294,295][198][292][293]. A wide variety of materials are available for use as MAs, including natural MAs (volcanic materials, including tuffs, ashes, pumicites, perlites, zeolites, etc.), calcined natural MAs (calcined kaolinite clay or metakaolin), LC3 materials (limestone calcined clay cement), by-product materials (agricultural wastes, CDW, ashes, glass, ferrous slags, non-ferrous slags, basic oxygen furnaces, and electric arc furnaces) [200,295,296,297,298][198][293][294][295]| 2 | ||||||||
| M-3/MPa, respectively. | ||||||||
| [ | ||||||||
| 317 | ||||||||
| ][316] | Ultra-high-performance concrete | |||||||
| [311][309] | SCBA and SF | Ecofriendly ternary concrete |
SCBA/SF: 10/10, 20/20, 30/30, 40/40, 50/50 | 30/30 and 20/20 | ||||
| ] | [ | 327][328] | ||||||
| Pervious concrete | 10–15 | [330][329] | ||||||
| Recycled aggregate concrete | 20 | [331,332][330][331] | ||||||
| Wood waste ash (WWA) | Ordinary concrete | 10 | [333,334,335,336,337][332][333][334][335][336] | |||||
| Self-compacting concrete | 10 | [338][337] | ||||||
| Palm oil fuel ash (POFA) | Ordinary concrete | 10–20 | [339,340,341][338][339][340] | |||||
| Eco-friendly structural foamed concrete | 25 | [342][341] | ||||||
| Lightweight concrete | ]10–15 | [343][342] | ||||||
| Nano silica (NS) | High-strength concrete | 1, 2, 3 | 2 | The climate change index for reference concrete is 534.26 kg CO | 2eq. The climate change index for HSC-NS1, HSC-NS2, and HSC-NS3 is 438.55, 426.70, and 415.56, respectively. | Sustainable lightweight foamed concrete | 20 | [344 |
| [348][347] | ] | [343 | POFA | ] | ||||
| Self-compacting concrete | 50, 60, 70 | 50–70 | The concrete specimens have up to 32–45% reduced carbon dioxide emissions. | Sustainable foamed concrete | ||||
| [ | 15 | 343][342][345][ | Lightweight concrete344 | 5, 10, 15, 20, 25 | 10–15 | Total CO2 emission values for mixes M0, M5, M10, M15, M20, and M25 were 0.477, 0.454, 0.430, 0.407, 0.384, and 0.361 CO2-e/m3, respectively. | ||
| [426][425] | Limestone | Ordinary concrete | 35–65 | <50 | The production of concretes made of limestone-rich cements exhibited roughly 25% less CO2 emissions. | |||
| [397][396] | Self-compacting concrete | 15, 25 | <25 | For control mix, CO2-eq is 5.69 × 102 kg/m3. For 15% and 25% replacement levels, CO2-eq is 4.87 × 102 and 25 4.34 × 102, respectively. |
Table 6.
CO
2
reduction through partial replacement of Portland cement with AM (ternary blended cements).
| Reference | MAs | Mix Type | Amounts of Substitution (wt.%) | Optimum Substitution (wt.%) | Results for CO2 Emissions | |||
|---|---|---|---|---|---|---|---|---|
| [427][426] | Brick dust (BD) and LP | Plain cement concrete | BD/LP: 15/5, 10/10, 7/13, 5/15 | 15/5 | Using PL and BD can save costs of cement in the range of 7–12.5%, which eventually reduces CO2 | |||
| The use of ETC concretes has a very significant sustainability impact by contributing to the reduction in CO | ||||||||
| 2 | ||||||||
| emissions caused by Portland cement. | ||||||||
| [ | ||||||||
| 432][431] | Limestone filler (LSF), calcined orange illitic clay (OIC), natural pozzolan (NP) and GGBS | Ordinary concrete | LSF/OIC: 20/7.5. LF/NP: 12.4/12.6. LF/GGBS: 6/22; 11/11 | 20% of LF | CO2 emissions (kg CO2/m3) for control mix is 399.8, 378.6 for 20/7.5, 380.6 for 12.4/12.6, 322.6 for 6/22, 341.7 for 11/11. | |||
| [397][396] | FA and LP | |||||||
| ] | ||||||||
| Self-consolidating concrete | FA/LP: 30/15, 40/15, 50/15, 60/15, 20/25, 30/25, 40/25, 50/25 | |||||||
| Self-compacting concrete | ||||||||
| <70 | ||||||||
| [ | 346 | ,347,348,349][345][346][347][348] | ||||||
| Self-consolidating high-strength concrete | <50 | [350,351][349][350] | ||||||
| <50% | CO | 2 | -eq (kg/m | 3 | ) for control mix is 5.69 × 102, 3.33 × 102 for 30/15, 2.82 × 102 for 40/15, 2.32 × 102 for 50/15, 1.83 × 102 for 60/15, 3.32 × 102 for 20/25, 2.81 × 102 for 30/25, 2.31 × 10 | Structural lightweight aggregate concrete | 37.5 | [352][351] |
| Recycled aggregate concrete | 20 | [331,332][330][331] | ||||||
| 2 | Palm oil clinker powder (POCP) | Environmentally friendly lightweight concrete | 15 | [353][352] | ||||
| Lightweight concrete | 15 | [354][353] | ||||||
| Recycled aggregate concrete | 15 | [331,332][330][331] | ||||||
| Eggshell powder (ESP) | Ordinary concrete | 10–15 | [355,[356,357,354 | |||||
| [ | ||||||||
| 333 | ||||||||
| ] | ||||||||
| [ | 332 | ] | ||||||
| Sawdust ash (SDA) | Ordinary concrete | 5–20 | [361,362][360][361] | |||||
| 20, 40, 60, 80 | Self-compacting concrete | 10 | [363,364][362][363] | |||||
| Coconut shell ash (CNSA) | Ordinary concrete | 10 | [365,366][364][365] | |||||
| 60 | Wheat straw ash (WSA) | Ordinary concrete | 5 | [317][316] | ||||
| Nano-POFA | Ordinary concrete | 10–20 | [367][366] | |||||
| Lightweight concrete | 15 | [354][353] | ||||||
| Nano-POCP | Semi-lightweight concrete | 10 | [368][367] | |||||
| The best environmental assessment results occur when the SCBA substitution rate is 80%. The global warming potential data decreased by 17.47%. | Nano-ESP | Ordinary concrete | 12.5 | [367][366] | ||||
| High-strength concrete | 5 | [369][368] | ||||||
| Aquacultural Industry | ||||||||
| Seashell powder (SSP) | Ordinary concrete | 5–15 | [370,371,372,373][369][370][371][372] | |||||
| High-strength concrete | 5 | [374][373] | ||||||
| Oyster shell powder (OSP) | ||||||||
| [382][381] | Ceramic | Ultra-high-performance concrete | 15, 25, 35, 45, 55 | Ordinary concrete | 5–15 | [375][374] | ||
| for 40/25, 1.82 × 10 | 2 | for 50/25. | ||||||
2.4. Substitution of CDW as a MA
Concrete, masonry, and brick wastes are prominent among the various waste fractions, exhibiting a significant proportion of approximately 80% in the overall global production of CDW [66,437,438,439][66][436][437][438]. Researchers have proposed the recycling of this prominent part to serve as a viable solution to address the sustainability issues encountered by the concrete industry [71,440,441,442,443,444,445,446][71][439][440][441][442][443][444][445]. The recycling process involves the conversion of CDW into a reduced-sized fraction through the utilization of mobile or fixed recycling plants [447][446]. The recycling process of CDW primarily results in the production of three distinct fractions [84,448,449,450][82][447][448][449]. One of these fractions includes a range of 25.00–5.00 mm, which is classified as recycled coarse aggregate (RCA). Another fraction falls within the range of 5.00–0.15 mm and is referred to as recycled fine aggregates (RFA). Lastly, there is a fraction that measures less than 0.15 mm, known as recycled powder (RP). It is important to highlight that in addition to the production of recycled coarse and fine aggregates, a significant quantity of fine recycled powder (RP), comprising approximately 15–35% of the total processed CDW mass, is generated [448,449][447][448]. This fine powder lacks a suitable destination and is typically disposed of in landfills [441,451][440][450]. The particulate matter emanating from cement mortar, concrete, or bricks typically has a fine texture. The observed range of diameters for the hybrid powder obtained from the crushing and sieving location of CDW was found to vary between 45 and 150 μm [441][440]. Although the application of RCA has gained increasing popularity in the past years, the possible use of RP as a partial replacement for Portland cement in concrete has received significant attention due to its tiny particle size and consequential reactivity [452][451]. Nevertheless, the efficacy of RPs is contingent upon their primary sources, which are impeded in their practical implementation due to their intricate components. When comparing RPs to Portland cement, it is observed that RPs exhibit a greater degree of irregularity and roughness in their shapes. Additionally, the little particles tend to cluster on the larger ones, resulting in a higher water consumption requirement to obtain a desired standard consistency [439,452][438][451]. The primary factor impeding the utilization of untreated RP derived from CDW in cementitious materials is its inherent low activity. The untreated powder is primarily comprised of inert hydrated materials, namely quartz or calcite [439,445][438][444]. Several modification approaches have been devised to enhance the characteristics of untreated RP, including mechanical activation [453[452][453],454], CO2 curing treatment [455[454][455][456][457],456,457,458], thermal treatment [445[444][458][459][460],459,460,461], tannic acid treatment [462][461], and chemical activators [463][462]. CDW-based material additions used as an addition or as a partial replacement of Portland cement to produce concrete are presented in Table 7.Table 7.
Substitution of CDW in Portland cement to produce concrete.
| Reference | CDW Type | Mix Type | Materials Used in the Mix | Treatment Method | Particle Size or Median Particle Size of CDW (d50) | Amount of Substitution (wt.%) |
|---|
Table 8.
Chemical properties of cementitious materials used (%).
| Reference | Cementitious Material Type | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | SO3 | Loss-on-Ignition (LOI) |
|---|---|---|---|---|---|---|---|---|---|---|
Table 9.
CO
2
reduction by the partial replacement of Portland cement with CDW.
| Reference | Label | Proportions | w/b a | SP b | 28 d Compressive Strength (MPa) | CO2 Emission (kg/m3 | Optimum Substitution (wt.%) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ) | Global Warming Potential (GWP) | ||||||||||||||||||
| [464][463] | Dehydrated cement paste (DCP) | Green ultra-high-performance concrete | Cement (PO 52.5), DCP, LP, SF, sand, superplasticizer (SP). | Heating | |||||||||||||||
| [464][463] | |||||||||||||||||||
| [ | OPC | <75 μm | 12.5, 25, 37.5, 50 | <25 | |||||||||||||||
| 464][ | 19.383 | 463 | 4.581 | 3.282 | ] | 63.074 | 2.786 | 0.175 | 1.027 | c | 3.498 | 1.540 | |||||||
| DCP0 | Control mix | 0.5 | 33.00 | 105.00 | 377.00 | 1.000 | [444][443] | RP | Ordinary concrete | Cement (OPC), RP, natural coarse aggregate (NCA), natural fine aggregate (NFA). | Repeated recycling | <150 μm | 10, 20, 30 | 10–20 | |||||
| DCP | 19.967 | 4.997 | 4.125 | 62.405 | 1.849 | 0.137 | 0.781 | 2.949 | 2.261 | [465][464] | Ground recycled concrete (GRC) | Structural concrete | Cement (OPC), GRC, mixed recycled CDW aggregate. | NA | Not stated | 10, 25 | 10 | ||
| [465][464] | OPC | 18.700 | 5.100 | 2.600 | 65.100 | 1.800 | 0.200 | 0.500 | 3.000 | 2.500 | [466][465] | Recycled brick powder (RBP) | |||||||
| GRC | 46.100 | Ultra-high-performance concrete | Cement (PII 52.5R), RBP, SF sand, SP. | NA | 3.800 | 1.500d50: 9.8 μm | 15, 30, 45 | 15 | |||||||||||
| 40.000 | 0.500 | 0.300 | 1.200 | 0.400 | 6.200 | [439][438] | RP | Ordinary concrete | Cement (OPC), RP, FA, sand, NCA, water reducing agent. | NA | |||||||||
| [467][466] | WBP | 36.510 | 23.440 | d50: 9.06 μm | 15, 30, 45 | 15–30 | |||||||||||||
| 15.140 | 4.530 | - | - | 1.510 | - | 4.520 | [467][466] | Waste brick powder (WBP) | Ordinary concrete | Cement (OPC-Grade C-53), WBP, natural aggregate (NA), sand. | NA | <75 μm | 5, 10 | ||||||
| [468][467] | 10 | ||||||||||||||||||
| OPC | 23.770 | 4.960 | 4.130 | 60.320 | 2.680 | 0.320 | 0.620 | 2.260 | 2.380 | [468][467] | Recycled concrete powder (RCP) | Green ultra-high-performance concrete | Cement (P.II 52.5R), RCP, SF, sand, SP. | NA | d50: 12.04 μm | 15, 30, 45 | 30 | ||
| RCP | 39.830 | 12.500 | 6.010 | 18.660 | 1.970 | 0.850 | 2.340 | 2.040 | 16.750 | [469][468] | RP | Green concrete | |||||||
| [469 | Cement (PO 42.5), RP (brick powder and concrete powder), NA, RA, river sand, SP. | ][468] | OPC | 19.240 | 4.080 | 3.250 | 62.470 | 4.190 | |||||||||||
| DCP50 | 12.5%DCP1 | 0.5 | 33.00 | 102.00 | 337.00 | 0.894 | |||||||||||||
| DCP100 | 25%DCP1 | 0.5 | 33.00 | 100.50 | 298.00 | NA | d50: 17.15 μm | 15, 30, 45 | 15 | ||||||||||
| - | - | 4.810 | - | [470][469] | Humid hardened concrete waste (HHCW) | Ordinary concrete | Cement (OPC- PI 52.5), HHCW, FA, GGBFS, machine-made sand, river sand, crushed stone, SP. | Multiple wet grinding | d50: 26.5 μm, 5.71 μm, and 2.52 μm | 5, 10, 15 | |||||||||
| 0.790 | 0.5 | 33.00 | 83.00 | 219.00 | 0.581 | ||||||||||||||
| [444][443] | Brick powder | 65.240 | 18.080 | 4.250 | |||||||||||||||
| 21.700 | 5.100 | 3.400 | 65.000 | 1.400 | 0.300 | 0.550 | 1.500 | 1.050 | |||||||||||
| DCP150 | 37.5%DCP1 | 0.5 | 33.00 | 95.50 | 258.00 | 0.684 | |||||||||||||
| DCP200 | 50%DCP1 | c | NAC | Control mix | 0.45 | - | 36.80 | - | 1.000 | ||||||||||
| RP1 | 10%RP | 0.45 | - | 36.00 | - | 0.980 | |||||||||||||
| RP1 | 20%RP | 0.45 | - | 33.50 | - | 0.950 | HHCWS of 2.52 μm at the dosage of 10% | ||||||||||||
| 1.470 | 2.190 | ||||||||||||||||||
| RP1 | 30%RP | - | - | 0.340 | - | ||||||||||||||
| 0.45 | - | 27.00 | - | 0.930 | [471][470] | Ground recycled masonry aggregate (GR-RMA) | Ordinary concrete | Cement (CEM I 42.5 R OPC), GR-RMA, NA, MRA, natural sand, SP. | NA | Not stated | 25 | 25% GR and 25–50% MRA |
|||||||
| Concrete powder | 31.850 | 7.040 | 4.840 | 48.950 | 1.850 | - | - | 0.780 | - | ||||||||||
| RP2 | 10%RP | 0.45 | - | 35.50 | - | 0.950 | [472][471] | RCP | Ordinary concrete | Cement (CEM I 42.5), RCP, NA, SP. | NA | d50: 22μm | 10, 20, 30, 40, 50 | <10% | |||||
| [470][469] | OPC | 20.040 | 4.198 | 3.365 | 63.058 | 1.930 | 0.092 | 0.748 | 3.276 | 2.653 | [473][472] | RP | Sustainable concrete | ||||||
| RP2 | 20%RP | 0.45 | - | 32.00 | - | 0.900 | Cement (OPC), RP (RCP, RBP), NCA, NFA, FA, GGFBS, air entrainer admixture, water reducer admixture. | NA | d50: RCP: 11.8 μm, RBP: 13.4 μm | 20 | HHCW | 29.689 | 7.948 | 2.453 | 31.713 | 2.728 | 0.842RBP can provide equivalent |
1.078 | 0.685 |
| RP2 | strength and even better durability. | ||||||||||||||||||
| 21.986 | |||||||||||||||||||
| 30%RP | 0.45 | - | 27.50 | - | 0.850 | [474][473] | WP | WP concrete | Cement (OPC), WP (mixture of waste concrete and bricks), NA, sand. | [NA | 471d50: 12.54 μm | 15, 30, 45 | ][470] | GR-RMA | 60.00015 | ||||
| 19.000 | 6.000 | - | - | - | - | - | - | ||||||||||||
| RP3 | 10%RP | 0.45 | - | 34.00 | - | 0.930 | [475][474] | RP | Reactive powder concrete | Cement (PO 42.5), RP (abandoned clay bricks and cement solids), SF, SP. | NA | d50: 31.4 μm | 5, 10, 15, 25 | 10 | |||||
| [473][472] | OPC | 21.300 | 3.200 | 2.900 | 64.300 | 2.100 | 0.260 | 0.420 | 3.100 | 1.350 | [476][475] | Waste concrete powder (WCP) | Self-consolidating concrete | Cement (OPC), WCP, GGBFS, NCA, NFA, SP. | NA | d50: 90 μm | 15, 30, 45 | ||
| RCP | 51.000 | 10.130 | 5.360 | 26.310 | 1.380 | 1.230 | |||||||||||||
| RP3 | 20%RP | 0.45 | - | 31.50 | - | 0.850 | 15 | ||||||||||||
| 1.780 | 1.940 | 9.900 | |||||||||||||||||
| RP3 | 30%RP | 0.45 | - | 27.50 | - | 0.780 | [477][476] | RP | Ordinary concrete | RBPCement (OPC), RP (80% fired brick and 20% waste concrete), NA, sand, water reducer. | |||||||||
| [465][464 | NA | ] | <75 μm | 15, 30, 45 | 69.870 | 20.980 | <30% | ||||||||||||
| 3.610 | NAC | Control mix | 0.400 | 0.390 | 0.56 | 1.0–1.5% | 46.600.590 | 2.420 | 0.330 | 0.980 | [478][477] | Waste brick powder (WBP) | Ordinary concrete | ||||||
| 269.83 | 1.000 | Cement (type II OPC), WBP, NCA, sand. | NA | d50: 45 μm | 10, 15, 20, 25, 30, 40 | <20% | |||||||||||||
| [449][448] | OPC | 19.900 | 4.420 | 3.560 | 64.900 | 0.660 | 0.080 | 0.790 | 2.670 | - | |||||||||
| N10/0 | 10%GRC | 0.58 | 1.0–1.5% | 37.80 | 249.65 | 0.925 | [479][478] | Cement kiln dust (CKD) | Ordinary concrete | Cement (ASTM C 150 Type I and Type V), CKD, NCA, sand. | RP | 57.010NA | not stated | 10.930 | 3.450 | 21.3005, 10, 15 | 5 | ||
| 1.820 | 1.580 | 2.220 | 1.170 | - | |||||||||||||||
| N25/0 | 25%GRC | 0.60 | 1.0–1.5% | 27.70 | 218.43 | 0.810 | [480][479] | CKD | Self-consolidating Concrete | Cement (OPC Type I), CKD, NCA, sand, SP. | NA | not stated | [488] | 10, 20, 30, 40 | [487] | 20 | |||
| R0/50 | OPC | 0%GRC, 50% RA-CDW | 0.59 | 1.0–1.5% | 34.80 | 267.10 | 0.990 | [449][448] | CKDRP | Sustainable recycled concrete | Cement (PO42.5), RP, FA, NCA, NFA, river sand, SP. | NA | <45 μm | 15, 30 | 15 | ||||
| 11.690 | 3.250 | 2.400 | 44.900 | 0.800 | 0.290 | 0.500 | 0.000 | ||||||||||||
| R10/50 | 10%GRC, 50% RA-CDW | 0.61 | 36.000 | 1.0–1.5% | 32.80 | 246.94 | 0.915 | [481][480] | Ceramic (fired clay-based) fraction of CDW | Structural concrete | Cement (CEM I 42.5 R), ceramic (fired clay-based) fraction of CDW, NCA, RA, sand, SP. | ||||||||
| R25/50 | 25%GRC, 50% RA-CDW | 0.63 | NA | not stated | 1.0–1.5% | 23.30 | 216.7025, 50 | 25 | |||||||||||
| 0.803 | [482][481] | CKD | High performance self-compacting concrete | Cement (OPC), CKD, NCA, mineral sand, SP. | NA | <50 μm | 10, 20, 30 | <10% | |||||||||||
| [468][467] c | RCP0 | Control mix | 0.16 | 41.64 | 100.00 | 502.63 | 1.000 | [483][482] | Burnt clay and CKD | Blended concrete | Cement (OPC), burnt clay and CKD, NCA, NFA. | NA | <75 μm | 10, 20, 30, 40 | <20%CKD | ||||
| RCP15 | 15%RCP | 0.16 | 40.06 | 82.80 | 501.75 | 0.998 | [484][483] | CKD | Ordinary concrete | Cement (cement of Indian Standards (IS) mark 43 grade), CKD, NCA, NFA. | Bacterial treatment | not stated | 5, 10, 15 | 10% | |||||
| [ | |||||||||||||||||||
| RCP30 | 30%RCP | 0.16 | 39.08 | 96.10 | 500.86 | 0.996 | 485][484] | Clay brick powder (CBP) | Ordinary concrete | Cement (OPC), CBP (Recycled construction waste), natural sand. | NA | d50: 300 μm, 100 μm, 60 μm and 40 μm | 10, 20, 25, 30 | 10% | |||||
| [486 | |||||||||||||||||||
| RCP45 | 45%RCP | 0.16 | 37.13 | 88.30 | 499.27 | 0.993 | ][485] | Construction waste composite powder | Small-scale prefabricated concrete | Cement (42.5 OPC), CWBP (building demolition waste), NCA, sand. | NA | d50: 8–16 μm | |||||||
| [469 | 20, 30, 40 | ][468 | 30 | ||||||||||||||||
| ] | RAPC-0–0 | Control mix | 0.49 | 0.14 | 39.04 | - | [487][486] | GRC | Ordinary concrete | Cement (CEM I 42.5 R), GRC, NCA, MRA. | NA | <147 μm | 10, 25 | 25 | |||||
| 1.000 | |||||||||||||||||||
| RAPC-0–15 | 15%RP | 0.49 | 0.16 | 40.12 | - | 0.850 | [488][487] | CKD | Green concrete | Cement (OPC Type II), CKD, FA, river sand, NCA, SP. | NA | <45 μm | 10, 15, 20, 30, 40 | <20% | |||||
| RAPC-0–30 | 30%RP | 0.49 | 0.17 | 35.45 | - | 0.710 | [489][488] | CBP | Ordinary concrete | Cement (OPC), CBP (mainly, bricks and tiles), NA, recycled gravel. | NA | <63 μm | 25 | Green concrete | <20 | [ | |||
| RAPC-0–45 | 25 | 376][375] | |||||||||||||||||
| 25–35 | Compared to UHPC without CTWP, the energy intensity, and CO | 2 | emissions of UHPC with 55% CTWP were reduced by 41.0% and 33.1%, respectively.358 |
2.4.1. CO2 Reduction by the Partial Replacement of Portland Cement with CDW
Studies conducted regarding CO2| 45%RP | ||||||||
| 0.49 | ||||||||
| 0.16 | ||||||||
| 30.27 | ||||||||
| - | ||||||||
| 0.560 | ||||||||
| RAPC-30–0 | ||||||||
| 30%RA + 0%RP | ||||||||
| 0.49 | ||||||||
| 0.14 | ||||||||
| 41.17 | ||||||||
| - | ||||||||
| 1.000 | ||||||||
| RAPC-30–15 | ||||||||
| 30%RA + 15%RP | 0.49 | 0.16 | 43.29 | - | 0.850 | |||
| RAPC-30–30 | 30%RA + 30%RP | 0.49 | 0.17 | 37.45 | - | 0.700 | ||
| RAPC-30–45 | 30%RA + 30%RP | 0.49 | 0.16 | 31.32 | - | 0.560 | ||
| RAPC-50–0 | 50%RA + 0%RP | 0.49 | 0.14 | 36.44 | - | 0.990 | ||
| RAPC-50–15 | 50%RA + 15%RP | 0.49 | 0.16 | 37.28 | - | 0.850 | ||
| RAPC-50–30 | 50%RA + 30%RP | 0.49 | 0.17 | 33.56 | - | 0.700 | ||
| RAPC-50–45 | 50%RA + 45%RP | 0.49 | 0.16 | 29.56 | - | |||
| Periwinkle shell (PS) | ||||||||
| Ordinary concrete | ||||||||
| 5 | ||||||||
| [ | ||||||||
| 377 | ] | [ | 376] | |||||
| 0.550 | Scallop shell (SLS) | Ordinary concrete | <10 | [378][377] | ||||
| Manufacturing Industry | ||||||||
| Red ceramic waste (RCW) | Structural concrete | 20–40 | [379][378] | |||||
| RAPC–100–0 | 100%RA + 0%RP | Ceramic waste powder (CWP) | Ordinary concrete | 10–20 | [380][379] | |||
| Self-consolidating concrete | 15 | [381][380] | ||||||
| High-performance concrete | 25–35 | [382,383][381][382] | ||||||
| Recycled glass powder (RGP) | Ordinary concrete | 10–20 | [384,385,386,387,388,389,390,391][383][384][385][386][387][388][389][390] | |||||
| Environmentally friendly concrete | 25 | [392,393][391][392] | ||||||
| Self-compacting concrete | 24 | [394][393] | ||||||
| Fly ash (FA) | Ordinary concrete | 30 | [395,396][394][395] | |||||
| Self-compacting concrete | 10–55 | [325,397][324][396] | ||||||
| Pervious concrete | 10–15 | [330][329] | ||||||
| High-performance concrete | 30 | [398,399][397][398] | ||||||
| Granulated blast-furnace slag (GGBFS) | Ordinary concrete | <50 | [400][399] | |||||
| Recycled aggregate concrete | <20 | [401][400] | ||||||
| [423][422] | CLBA | Ordinary concrete | 10, 20, 30, 40 | <40 | CO2 released from limestone calcination is 0.37 kg for the control sample (CAC0), 0.33 kg for CAC10, 0.29 kg for CAC20, 0.26 kg for CAC30, and 0.22 kg for CAC40. | |||
| [396][395] | FA | Green structural concrete | 20, 40, 60, 80, 98 | <80 | Compared to commercial Grade 45 concrete, the proposed concrete shows a reduction in CO2 emission of around 70%. | |||
| [398][397] | High-strength concrete | 30, 40 | 30–40 | The replacement of FA0 with FA30 and FA40 could potentially reduce the carbon footprint by 22.1% and 21.9% per m3 of concrete, respectively. | ||||
| [424][423] | Ordinary concrete | 25 | 25 | Fly ash was found to be capable of reducing concrete CO2 emissions by 13% to 15% in typical concrete mixes. | ||||
| Sewage sludge ash (SSA) | ||||||||
| Ordinary concrete | ||||||||
| 10 | [ | 409 | ][408] | |||||
| Waste marble dust (WMD) | Ordinary concrete | <15 | ||||||
| [424][423] | GGBFS | Ordinary concrete | 40 | 40 | Replacing 40% of GGBS with Portland cement in 25 or 32 MPa concrete outputs results in a 22% reduction in CO2 emissions. | |||
| [404][403] | PPR | Ordinary concrete | 10, 20, 30, 40, 50 | 10–30 | For a compressive strength of 54 MPa at 91 days, the emission was reduced from 564 kg CO2-eq/m3 of concrete for the reference mixture to 473 kg CO2-eq/m3 of concrete (i.e., 16%) for 30% replacement and to 349 kg CO2-eq/m,359]][355][356][357][358] | |||
| Green concrete | 10–15 | [16,323][16][322] | ||||||
| Eco-friendly structural foamed concrete | 5 | [342][341] | ||||||
| Sustainable foamed concrete | 5 | [345][344] | ||||||
| Olive waste ash (OWA) | Ordinary concrete | 5 | [360][359] | |||||
| High-strength concrete | 5 | Steel slag (SS) | Ordinary concrete | 20 | [402][401] | |||
| High-early-strength concrete | 30 | [403][402] | ||||||
| 0.49 | 0.14 | 33.26 | - | 0.990 | ||||
| RAPC-100–15 | 100%RA + 15%RP | 0.49 | 0.16 | 35.18 | - | 0.840 | ||
| RAPC-100–30 | 100%RA + 30%RP | 0.49 | 0.17 | 28.36 | - | 0.690 | ||
| RAPC-100–45 | 100%RA + 30%RP | 0.49 | 0.16 | 22.79 | - | 0.550 | ||
| [471][470] | CC | Control mix | 0.45 | 6.20 | 51.2 | 407.00 | 1.000 | |
| C25 | 0%CDW + 25% MRA | 0.45 | 6.20 | 51.7 | 399.00 | 0.980 | ||
| C50 | 0%CDW + 50% MRA | 0.45 | 6.20 | 51.1 | 351.00 | 0.862 | ||
| R25/0 | 25%CDW | 0.45 | 6.20 | 46.1 | 335.00 | 0.823 | ||
| R25/25 | 25%CDW + 25% MRA | 0.45 | 6.20 | 45.7 | 327.00 | 0.803 | ||
| R25/R50 | 50%CDW + 50% MRA | 0.45 | 6.20 | 41.2 | 319.00 | 0.784 | ||
| [472][471] | RCP0 | Control mix | 0.55 | 3.00 | 51.60 | 333.00 | 1.000 | |
| RCP10 | 10%RCP | 0.55 | 3.00 | 41.30 | 304.00 | 0.913 | ||
| RCP20 | 20%RCP | 0.55 | 3.00 | 31.70 | 275.00 | 0.826 | ||
| RCP30 | 30%RCP | 0.55 | 3.00 | 22.80 | 246.00 | 0.739 | ||
| RCP40 | 40%RCP | 0.55 | 3.00 | 13.60 | 217.00 | 0.652 | ||
| RCP50 | 50%RCP | 0.55 | 3.00 | 10.00 | 188.00 | 0.565 | ||
| [449][448] d | Control | Control mix | 0.36 | 2.16% | 877.30 | 367.50 | 1.000 | |
| RP1 | 15%RP | 0.36 | 2.84% | 613.92 | 325.00 | 0.884 | ||
| RP2 | 30%RP | 0.36 | 3.52% | 786.23 | 278.00 | 0.756 | ||
| RP3 | 15%RP + 15%FA | 0.36 | 2.50% | 1298.73 | 275.60 | 0.750 | ||
| [488][487] | Ctrl-W37 | Control mix | 0.37 | 0.33 | 53.41 | 510.77 | 1.000 | |
| C5W37 | 5%CKD | 0.37 | 0.33 | 55.47 | 487.57 | 0.955 | ||
| C10W37 | 10%CKD | 0.37 | 0.33 | 52.13 | 464.36 | 0.909 | ||
| C15W37 | 15%CKD | 0.37 | 0.45 | 47.45 | 441.24 | 0.864 | ||
| C20W37 | 20%CKD | 0.37 | 0.54 | 41.42 | Silica fume (SF) | Ordinary concrete | 10 | [358][357] |
| Self-compacting concrete | 10 | [325][324] | ||||||
| Recycled aggregate concrete | 10 | [401][400] | ||||||
| Porcelain Tile Polishing Residue (PPR) | Ordinary concrete | 10–40 | [404][403] | |||||
| Self-compacting concrete | 25 | [405][404] | ||||||
| Electric Arc Furnace Dust (EAFD) | Ordinary concrete | 10 | [406][405] | |||||
| [ | 318 | , | 410,411][317][409][410] | |||||
| High-strength concrete | 15 | [412][411] | ||||||
| 3 | of concrete (i.e., 38%) for 50% addition. | |||||||
| [405][404] | Self-compacting concrete | 10, 20, 30 | <20 | For a compressive strength of 70 MPa, the incorporation of PPR would reduce the emission of CO2-eq/m3 of concrete by up to 17% when incorporating 127 kg of the residue per m3 of concrete. | Red mud (RM) | Ordinary concrete | 12 | [407][406] |
| Sustainable concrete | 10–15 | [408][407] | Titanium dioxide (TiO2) nanoparticles | Blended cement concrete | 3 | [413][412] | ||
| Coal bottom ash (CBA) | Sustainable concrete | 15 | [414][413] | |||||
| Copper Slag (CS) | Ordinary concrete | 10 | [396,415][395][414] | |||||
| Foundry sand waste (FSW) | Ordinary concrete | <30 | [416,417][415][416] | |||||
| Polyvinyl chloride (PVC) waste powder (WP) | Green concrete | 15–20 | [296][294] | |||||
| Others | ||||||||
| Limestone powder (LP) | Self-consolidating concrete | 55 | [397][396] | |||||
| Ultra-high-performance concrete | 54 | [418][417] | ||||||
| Metakaolin (MK) | High-performance concrete | 10 | [399][398] | |||||
| Volcanic ash (VA) | Ordinary concrete | 10–15 | [419][418] | |||||
| Crushed rock dust (CRD) | Ordinary concrete | 20 | [294][292] | |||||
| Municipal solid waste incineration ash (MSWI) | Ordinary concrete | <12 | [297,420][295][419] | |||||
CO2 Reduction through the Partial Replacement of Portland Cement with MA
CO2 reduction by the partial replacement of Portland cement with MAs is reviewed for two cases: first for binary blended cements in Table 5, and second for ternary blended cements in Table 6.Table 5.
CO
2
reduction through the partial replacement of Portland cement with AM (binary blended cements).
| Reference | MA | Mix Type | Amounts of Substitution (wt.%) | Optimum Substitution (wt.%) | Results for CO2 Emmisions | |
|---|---|---|---|---|---|---|
| [421][420] | Biochar rice husk (BRH) | Ordinary concrete | 5, 10, 15, 20 | Not stated | Global warming values (kg CO2eq) for BRH0%, BRH5%, BRH10%, BRH15% and BRH20% are 2.51 × 10 | |
| [ | ||||||
| 425 | ||||||
| ] | ||||||
| [ | ||||||
| 424 | ||||||
| ] | ||||||
| SF | ||||||
| High-strength concrete | ||||||
| 8, 10, 12 | ||||||
| 12 | The climate change index for reference concrete is 534.26 kg CO | 2 | eq. Values for HSC-SF8, HSC-SF10, and HSC-SF12 are 520.75, 495.11 and 453.15, respectively. | |||
| [425][424 | ||||||
| 418.10 | ||||||
| 0.819 | ||||||
| C30W37 | ||||||
| 30%CKD | ||||||
| 0.37 | ||||||
| 0.67 | ||||||
| 34.90 | ||||||
| 371.79 | ||||||
| 0.728 | ||||||
| C40W37 | ||||||
| 40%CKD | ||||||
| 0.37 | ||||||
| 1.63 | ||||||
| 28.09 | ||||||
| 326.07 | ||||||
| 0.638 | ||||||
| Ctrl-W40 | ||||||
| Control mix | ||||||
| 0.40 | ||||||
| 0.00 | ||||||
| 52.23 | ||||||
| 476.71 | ||||||
| 1.000 | ||||||
| C5W40 | 5%CKD | 0.40 | 0.00 | 49.52 | 457.94 | 0.961 |
| C10W40 | 10%CKD | 0.40 | 0.00 | 43.24 | 433.78 | 0.910 |
| C15W40 | 15%CKD | 0.40 | 0.00 | 37.97 | 412.32 | 0.865 |
| C20W40 | 20%CKD | 0.40 | 0.00 | 36.93 | 390.85 | 0.820 |
| C30W40 | 30%CKD | 0.40 | 0.33 | 34.94 | 348.16 | 0.730 |
| C40W40 | 40%CKD | 0.40 | 0.67 | 28.79 | 305.48 | 0.641 |
| Ctrl-W45 | Control mix | 0.45 | 0.00 | 50.14 | 430.36 | 1.000 |
| C5W45 | 5%CKD | 0.45 | 0.00 | 46.93 | 411.29 | 0.956 |
| C10W45 | 10%CKD | 0.45 | 0.00 | 44.76 | 392.20 | 0.911 |
| C15W45 | 15%CKD | 0.45 | 0.00 | 40.75 | 373.13 | 0.867 |
| C20W45 | 20%CKD | 0.45 | 0.00 | 37.53 | 354.05 | 0.823 |
| C30W45 | 30%CKD | 0.40 | 0.00 | 34.79 | 315.89 | 0.734 |
| C40W45 | 40%CKD | 0.40 | 0.33 | 28.59 | 277.96 | 0.646 |
| C5F15W37 | 5%CKD + 15%FA | 0.37 | 0.33 | 55.93 | 419.60 | 0.822 |
| C10F15W37 | 10%CKD + 15%FA | 0.37 | 0.33 | 48.64 | 396.52 | 0.776 |
| C5F15W40 | 5%CKD + 10%FA | 0.40 | 0.00 | 45.69 | 392.52 | 0.823 |
| C10F15W40 | 10%CKD + 15%FA | 0.40 | 0.00 | 46.44 | 371.28 | 0.779 |
| C5F15W45 | 5%CKD + 10%FA | 0.45 | 0.00 | 44.19 | 355.83 | 0.827 |
| C10F15W45 | 10%CKD + 15%FA | 0.45 | 0.00 | 40.03 | 336.44 | 0.782 |
a Water–binder ratio. b Superplasticizer; if % is not stated, the values are in kg/m3. c 28 day compressive stresses are approximately derived from the figure. d Static yield stresses (Pa) are given in the study.
In their study, Qian et al. [464][463] examined a viable approach to the production of environmentally friendly ultra-high-performance concrete (UHPC) through the integration of recycled concrete waste coarse aggregate material (RCWCM). By subjecting RCWCM to a heating treatment process, they produced DCP. Subsequently, DCP was employed in a progressive manner to substitute the Portland cement content, thereby being incorporated into the formulation of UHPC utilizing the modified Andreasen and Andersen particle packing model. The findings indicate that the substitution of up to 25% Portland cement with DCP does not significantly affect the compressive strength variation of UHPC. Moreover, the researchers utilized the EN ISO 14040 and EN ISO standards to evaluate the environmental impact of UHPC by employing the carbon footprint metric. To establish the sustainability and environmental cleanliness of the UHPC, this study undertook calculations to determine CO2 emissions per unit of green UHPC with varying DCP levels. Additionally, the ratio of CO2 emissions to compressive strength per unit of green UHPC was also evaluated. From the results, it can be noticed that the inclusion of DCP yields advantageous outcomes in enhancing the performance of UHPC from a sustainability perspective.
The objective of the study carried out by Kim and Jang [444][443] was to examine the feasibility of closed-loop recycling for construction waste. Specifically, the focus was on examining the impact of utilizing concrete powder, which is a byproduct of producing recycled aggregates, on the fresh and hardened mechanical properties of concrete. The authors assert that concretes produced using recycled materials such as RCA, RFA, and RP exhibit a lower cost compared to natural coarse aggregate (NAC). However, it is important to note that these recycled concretes also have reduced compressive strength. Additionally, it was asserted that the utilization of RP as a substitute for Portland cement yields environmental advantages, including reductions in CO2 emissions, the preservation of natural resources, and the mitigation of landfill usage.
Cantero et al. [465][464] examined the cumulative impact of using ground recycled concrete (GRC) as a Portland cement replacement along with the use of mixed recycled construction and demolition waste aggregate (RA-CDW) in the context of structural concrete. The mechanical performance of concrete mixes with GRC and recycled aggregate from CDW (RA-CDW) was shown to be inferior compared to mixes made solely with natural aggregate and cement. However, it is worth noting that the difference in performance was relatively smaller when considering the corresponding replacement ratios. The authors did not consider the emissions associated with manufacturing and transportation when assessing the environmental impact of the mixtures in terms of CO2 emissions from materials. These emissions were considered smaller than those created during material manufacturing. In accordance with the provided statistics, the implementation of GRC resulted in a reduction in CO2 emissions by 7.5% in N10/0, 18.7% in N25/0, 8.5% in R10/50, and 19.7% in R25/50. The utilization of GRC, in conjunction with RA-CDW, has been found to augment the environmental efficacy of concrete. When the replacement rate was set at 10%, the amount of CO2 released during the manufacturing process of concrete decreased by 8.5% compared to concrete produced with OPC and 100% natural aggregates (NA). Similarly, when the replacement rate was increased to 25%, the greenhouse gas emissions associated with GRC decreased by 19.7% compared to OPC-based concrete with 100% NA.
The study conducted by He et al. [468][467] aimed to evaluate the influence mechanism of RCP on the multi-scale properties of UHPC mixtures. The findings of the study revealed that the UHPC combination with 30% RCP exhibited a comparatively reduced strain in early-age autogenous shrinkage, along with the highest mechanical characteristics. The reference parameters used by the authors to assess UHPC’s positive environmental impact included the mixture’s total carbon emissions and non-renewable energy consumption (NREC). The study demonstrates that there is a decrease in the NREC per cubic meter of UHPC mixture when the RCP substitution ratio increases. In parallel, it can be observed that the augmentation in the substitution proportion of RCP leads to a corresponding reduction in the carbon emissions per unit volume of UHPC mixture.
The objective of the study of Wu et al. [469][468] was to examine the characteristics of pore structure, carbonation, and chloride ion permeability in recycled aggregate-powder concrete (RAPC). The findings of the study indicate that there is a positive correlation between the replacement rate of recycled aggregate (RA) and both the carbonation depth and chloride ion permeability of RAPC. The research indicates that the inclusion of 15% RP resulted in the enhanced performance of RAC. This addition has effectively addressed the issue of by-products generated during the manufacturing of RA, leading to cost reduction and a reduction in the adverse environmental effects associated with RAC production.
The durability of a concrete mixture containing ground recycled masonry aggregate (GR-RMA) as a partial replacement for cement and coarse mixed recycled aggregate (MRA), both obtained from CDW, was examined by Cantero et al. [471][470]. The investigation involved the indirect characterization of pore system permeability by utilizing important indicators of water transport. Based on the results obtained from the defined scenario, it was determined that the optimal combinations of mechanical efficiency and durability were observed in mixes with a 25% GR content as a replacement for Portland cement. Additionally, it was found that the mixes with the highest environmental benefits in terms of reducing CO2 emissions were those that included both 25% GR and 25% to 50% MRA.
In the study done by Pešta et al. [473][472], the researchers evaluated the environmental viewpoints pertaining to the utilization of RCP as a substitute for Portland cement. The findings from the assessment of mechanical properties indicate that RCP exhibits favorable characteristics as a substitute for Portland cement, particularly in scenarios with a low degree of replacement. Furthermore, the findings of the environmental assessment provide confirmation that the implementation of RCP resulted in a decrease in the adverse effects of climate change, as well as potential effects in other related domains.
Singh et al. [449][448] examined the practical application of recycled fines (RFs), namely RFA and RP, in the context of recycled concrete. The investigation focused on evaluating the fresh qualities (empirical and rheological) of the recycled concrete. The findings indicated that the decrease in slump was more pronounced in the series with RFA compared to RP. According to the authors, the inclusion of RF in concrete mixtures not only enhances material performance but also presents notable environmental advantages, specifically in mitigating carbon emissions linked to the production of concrete.
Bagheri et al. [488][487] utilized varying quantities of CKD, a waste material, and FA, a pozzolanic material, as replacements for Portland cement, both alone and in combination. The comparison between the Taguchi technique and experimental outcomes for the purpose of picking the most advantageous mixture designs revealed that the Taguchi approach demonstrated appropriate selections within the range of optimal experimental results taking into consideration the initial parameters. Furthermore, the values for the cost and CO2 emission factors of each plan were determined by considering the CO2 production cost associated with each material and the corresponding size of said material inside the relevant plan. The observed decrease in cost of 23% resulting from the substitution of Portland cement with cement additions, alongside the concurrent reduction in volume within the C40W45 mixture, was found to be statistically significant. Additionally, it is worth noting that the CO2 emission factor associated with the Ctrl-W37 value (510.8 kg/m3) exhibited a reduction of almost 50% when considering the C40F0W45 mixture (278 kg/m3).
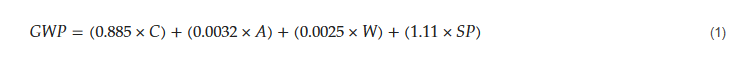 where C is the cement content of concrete (kg/m3), A is the aggregate content (kg/m3), W is the water content (kg/m3), and SP is the superplasticizer content (kg/m3).
Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9 and Figure 10 display the collected data on the GWP of the produced concrete, as well as the ratio of GWP to compressive strength per unit of the concrete, for various levels of CDW content. The second measure indicated above corresponds to the quantity of GWP per unit of strength. A higher GWP/compressive strength ratio in the produced concrete indicates a bigger quantity of carbon dioxide generated during the production of concrete, provided that the compressive strength remains constant. Based on the results depicted in the figures, it is evident that the inclusion of CDW has a positive impact on enhancing the performance of concrete from a sustainability perspective.
where C is the cement content of concrete (kg/m3), A is the aggregate content (kg/m3), W is the water content (kg/m3), and SP is the superplasticizer content (kg/m3).
Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8, Figure 9 and Figure 10 display the collected data on the GWP of the produced concrete, as well as the ratio of GWP to compressive strength per unit of the concrete, for various levels of CDW content. The second measure indicated above corresponds to the quantity of GWP per unit of strength. A higher GWP/compressive strength ratio in the produced concrete indicates a bigger quantity of carbon dioxide generated during the production of concrete, provided that the compressive strength remains constant. Based on the results depicted in the figures, it is evident that the inclusion of CDW has a positive impact on enhancing the performance of concrete from a sustainability perspective.
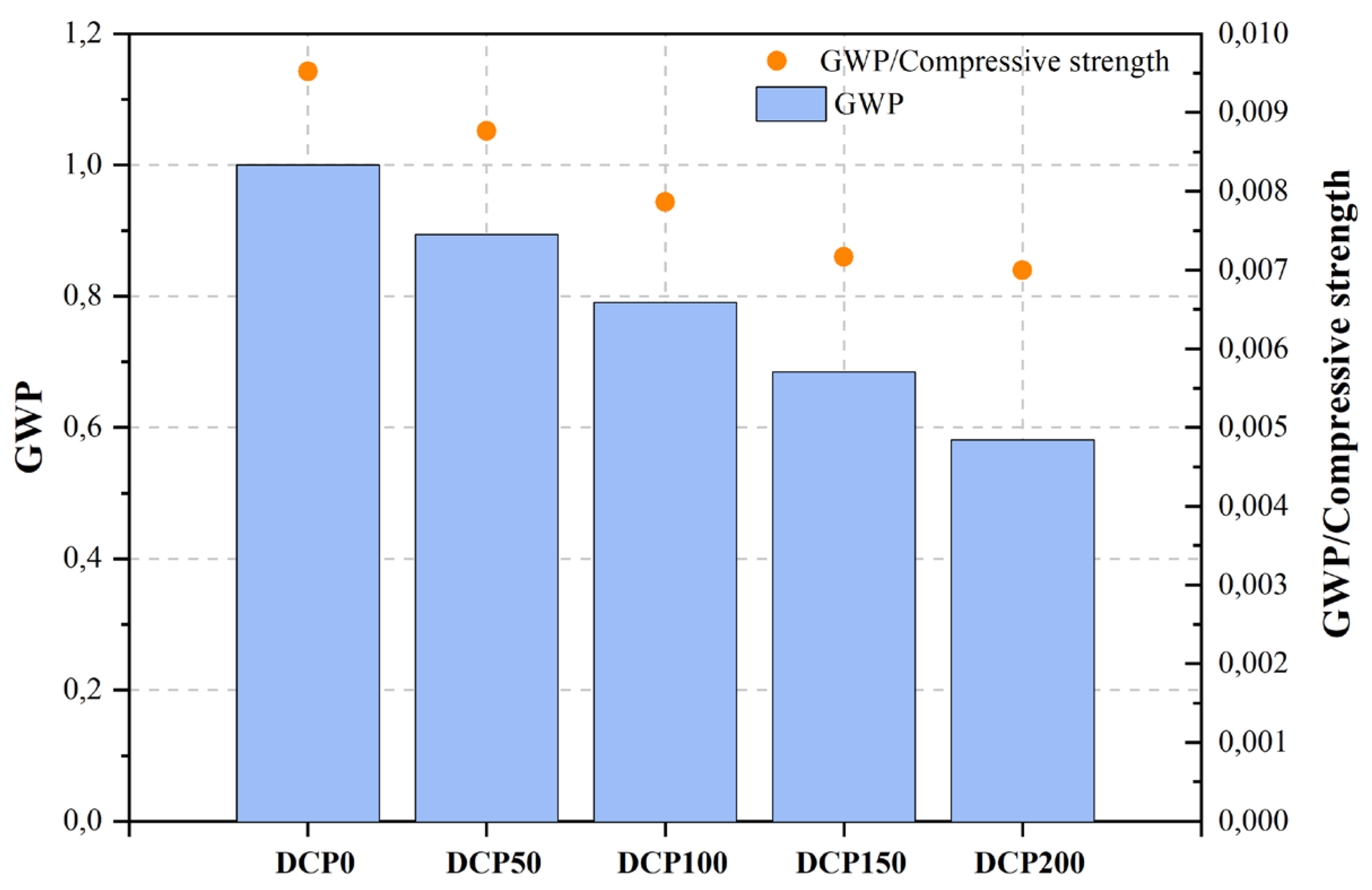
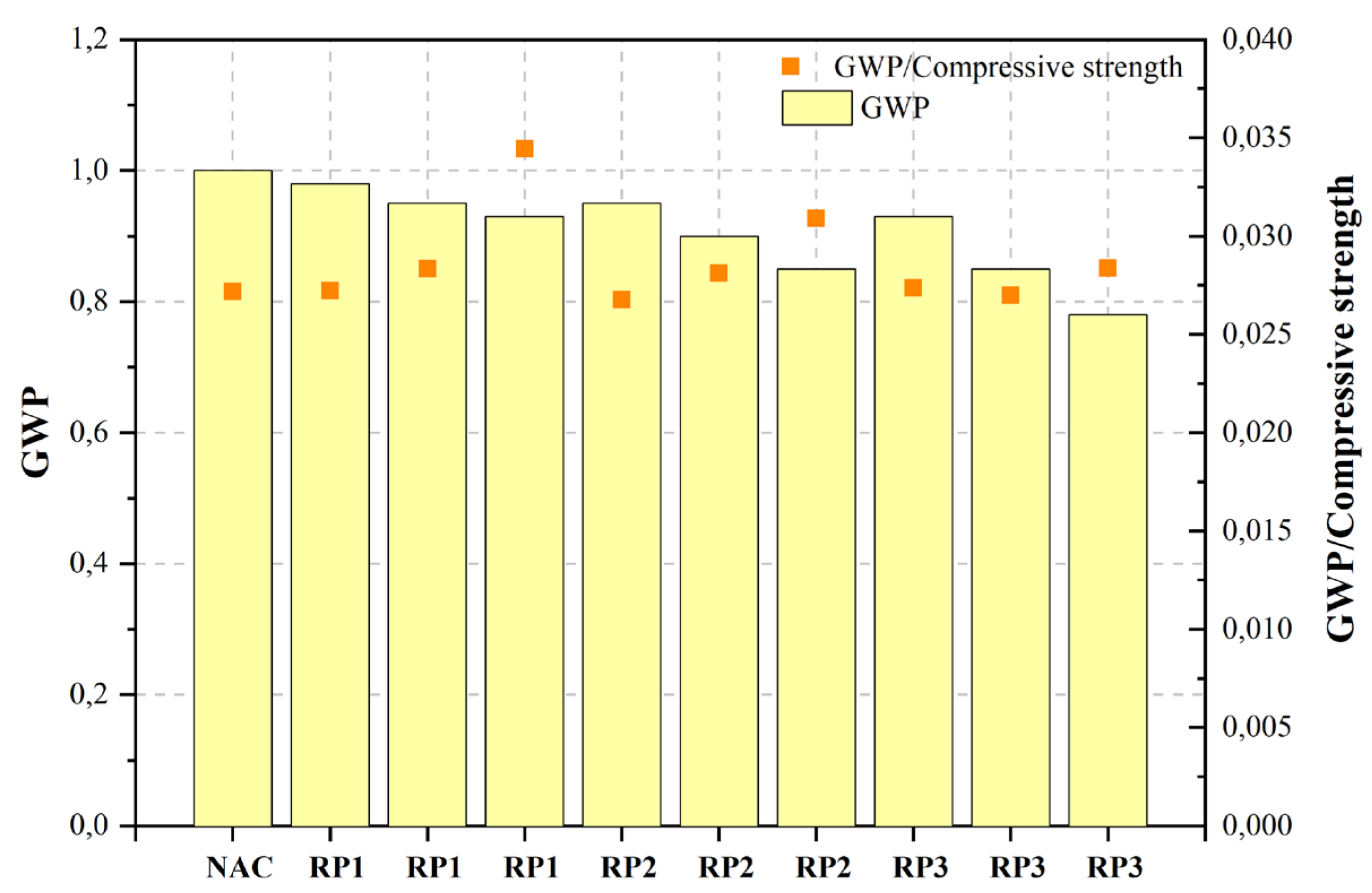
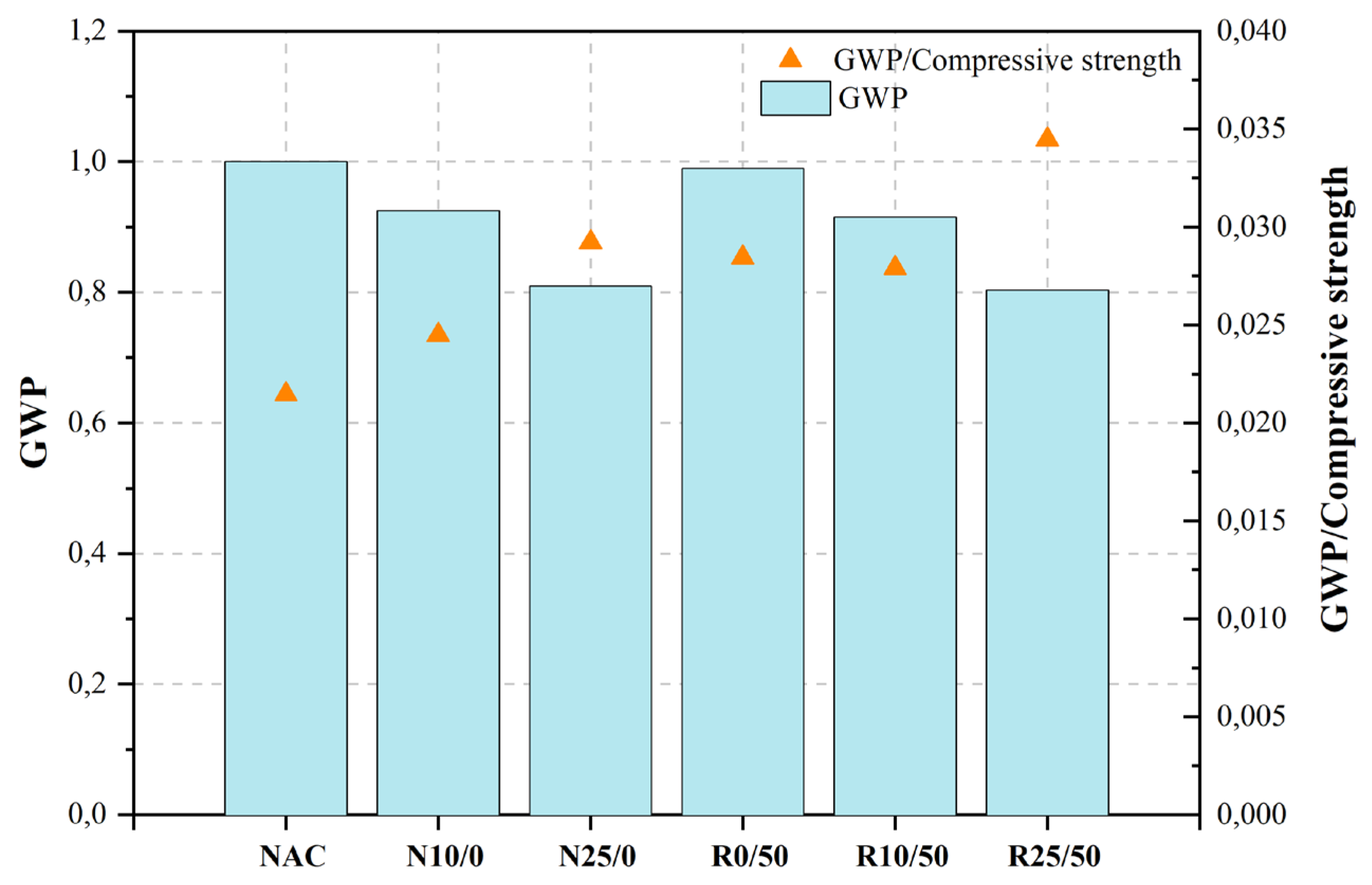
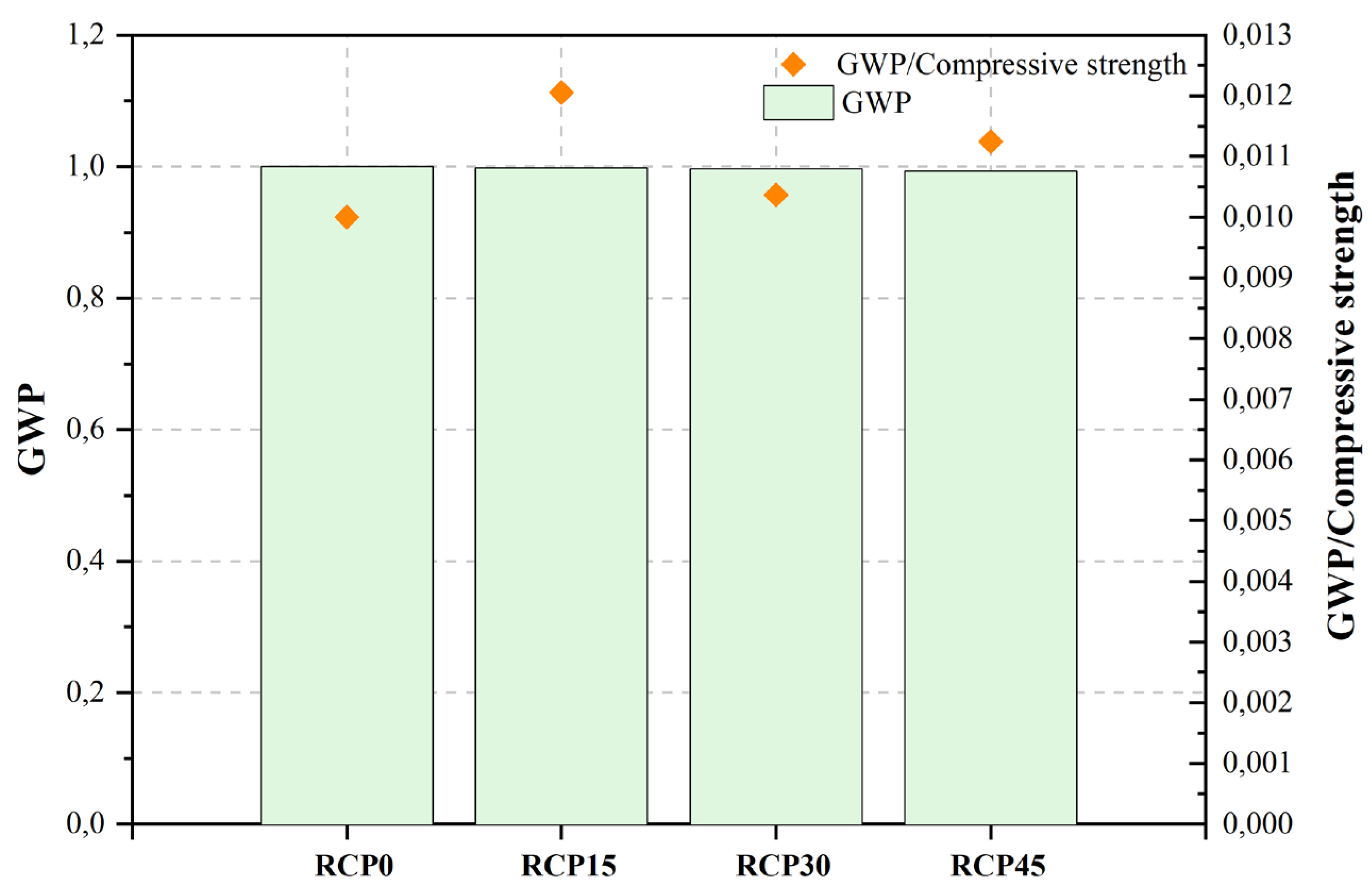
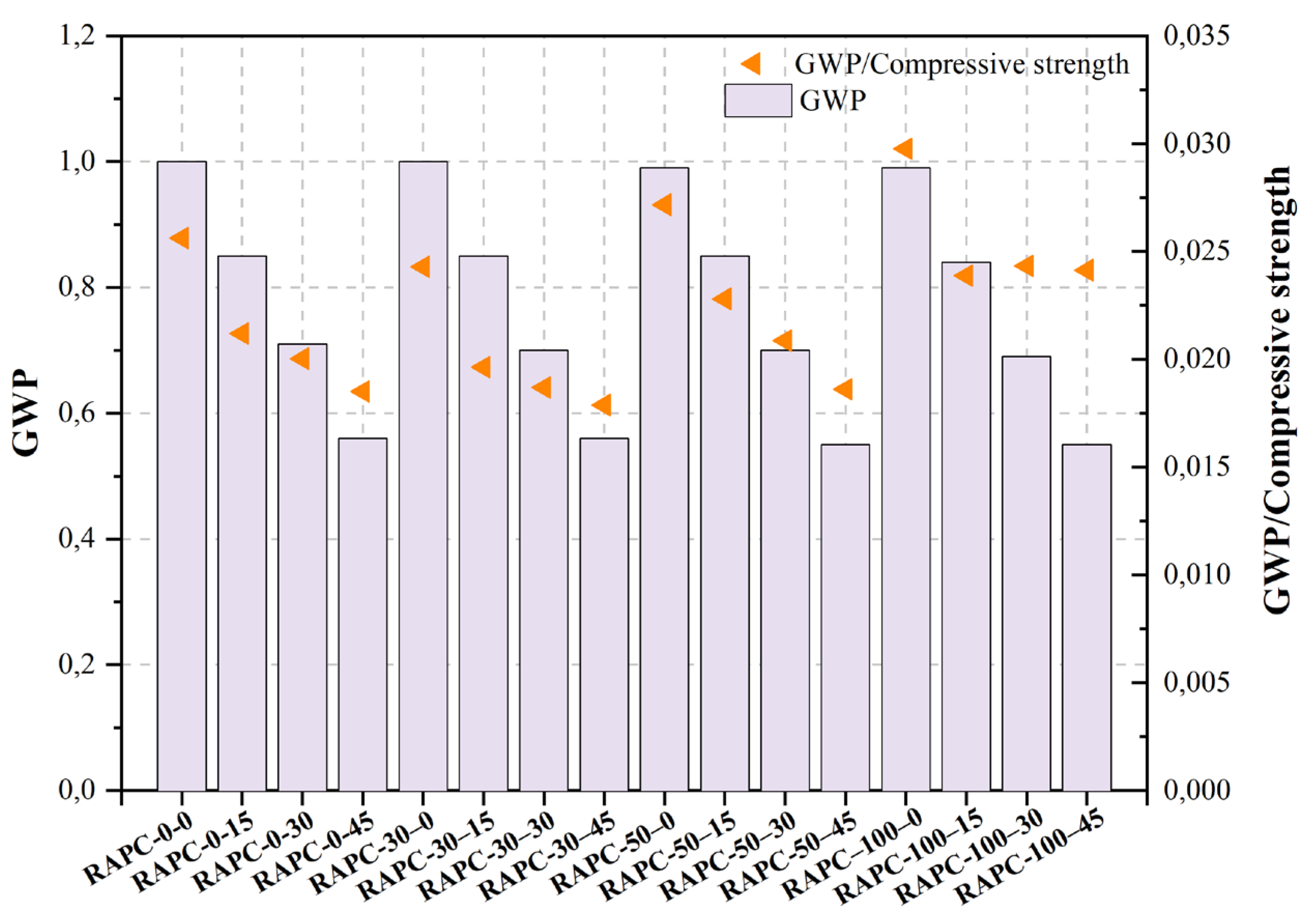
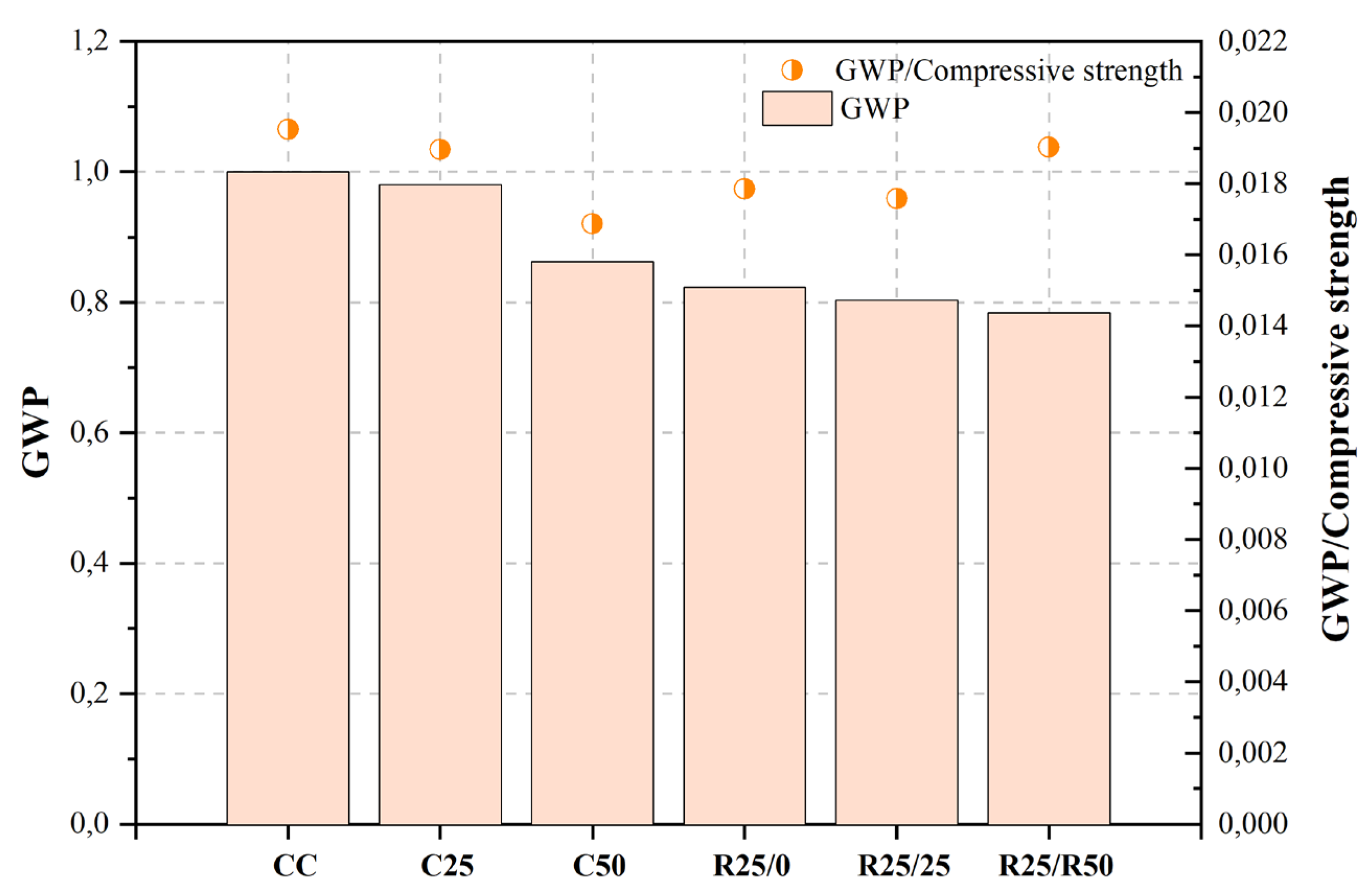
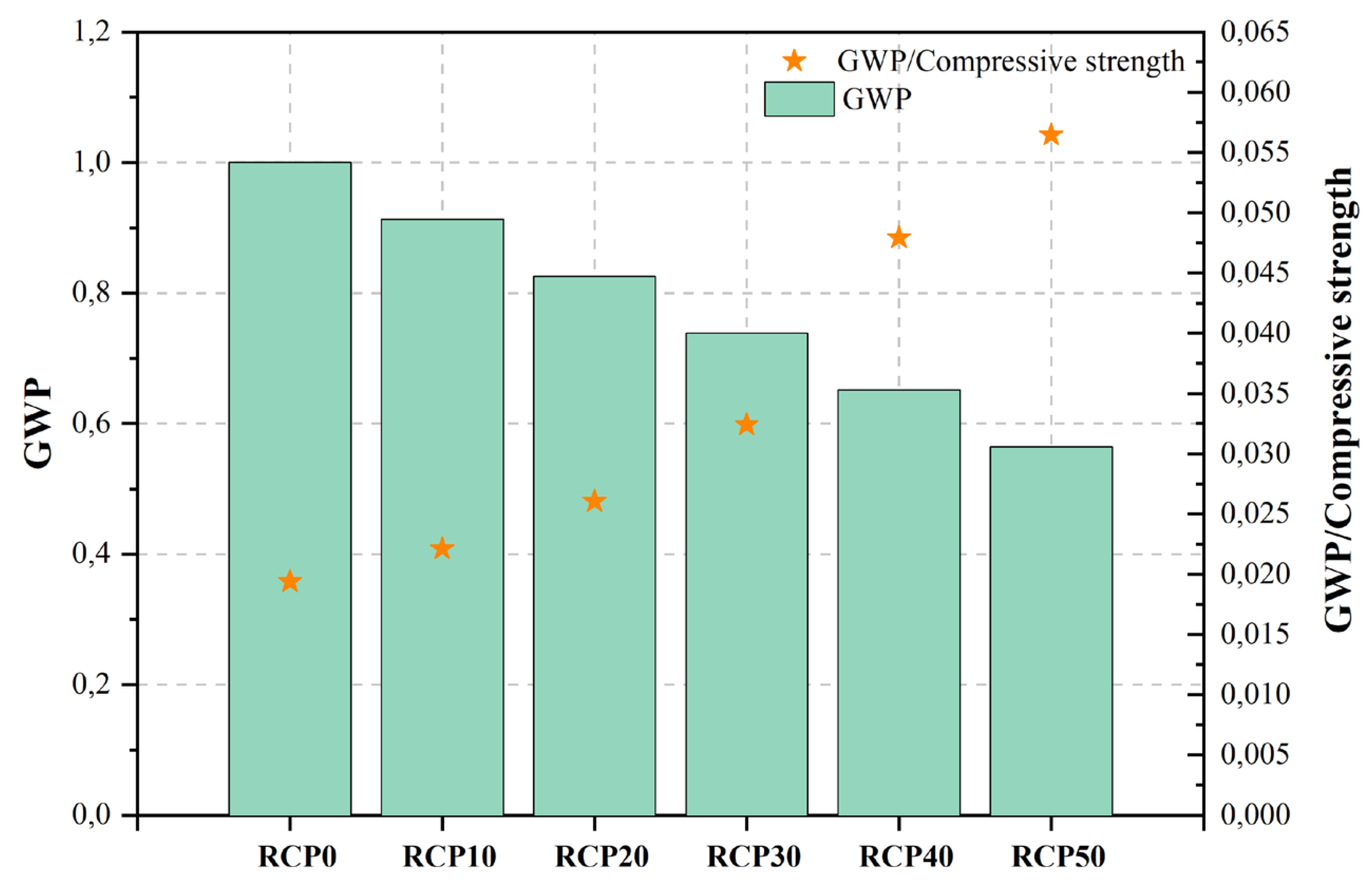
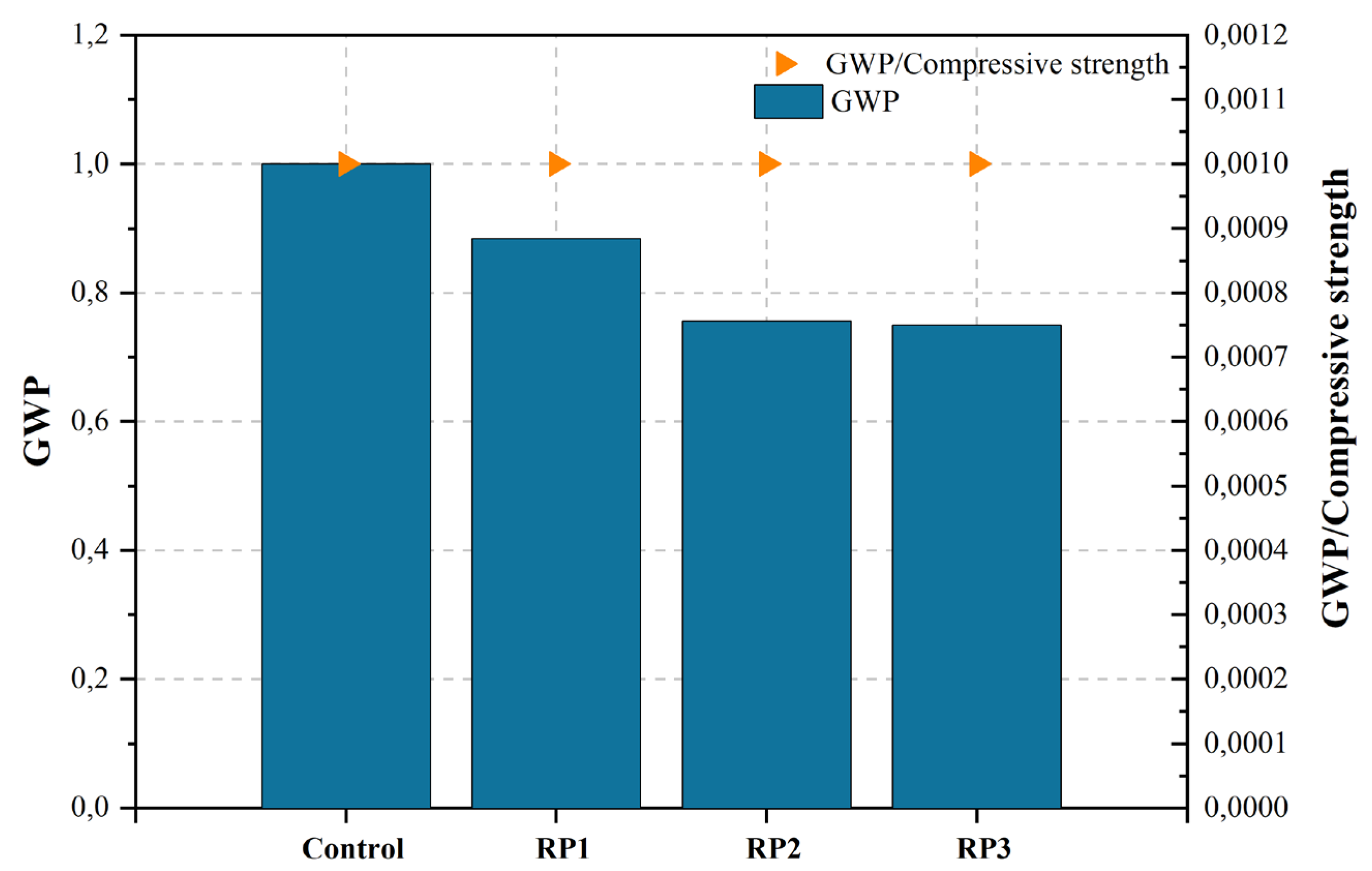
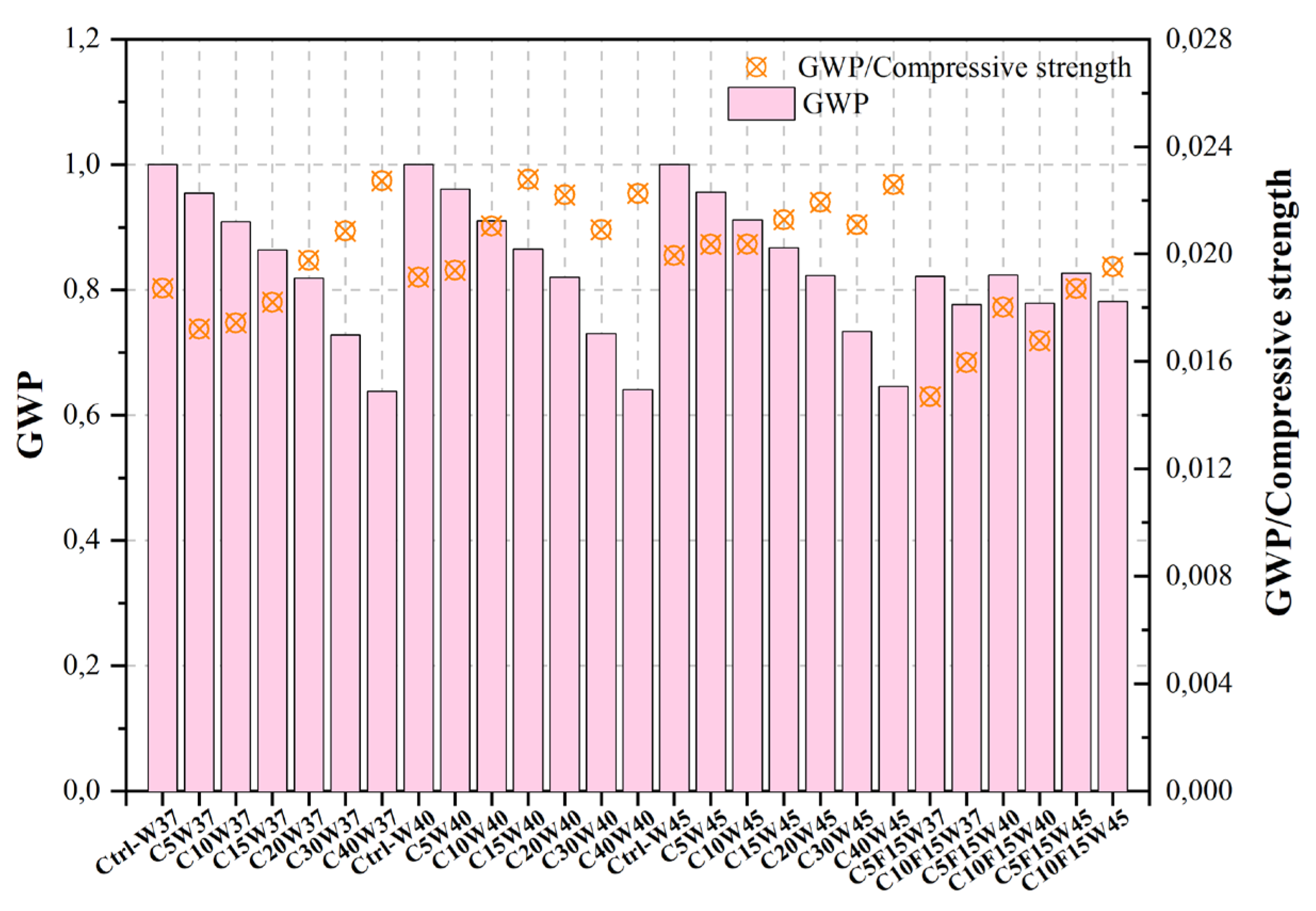 When comparing the identical strength conditions, the produced concrete including DCP [464][463], RP [463][462], GRC [465][464], RCP [468,472][467][471], RAPC [469][468], GR-RMA [471][470], RF [449][448], and CKD [488][487] exhibits a lower CO2 emission per unit volume compared to the reference sample. This indicates a higher efficiency in utilizing Portland cement in the produced concrete. In addition, the increase in all types of CDW contents leads to a significant reduction in carbon dioxide emissions during the production of the concrete.
When comparing the identical strength conditions, the produced concrete including DCP [464][463], RP [463][462], GRC [465][464], RCP [468,472][467][471], RAPC [469][468], GR-RMA [471][470], RF [449][448], and CKD [488][487] exhibits a lower CO2 emission per unit volume compared to the reference sample. This indicates a higher efficiency in utilizing Portland cement in the produced concrete. In addition, the increase in all types of CDW contents leads to a significant reduction in carbon dioxide emissions during the production of the concrete.
2.4.2. Evaluation of CO2 Emissions with Respect to Compressive Strength
For the studies presented in Table 9, the global warming potential (GWP) was calculated (Equation (2)) using the environmental parameter presented by Khodabakhshian et al. [490][489]. The GWP was formulated to quantify the alteration in the greenhouse effect resulting from human-caused emissions and absorptions.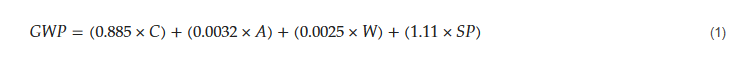









References
- Schneider, M.; Romer, M.; Tschudin, M.; Bolio, H. Sustainable cement production—Present and future. Cem. Concr. Res. 2011, 41, 642–650.
- Antunes, M.; Santos, R.; Pereira, J.; Rocha, P.; Horta, R.; Colaço, R. Alternative clinker technologies for reducing carbon emissions in cement industry: A critical review. Materials 2021, 15, 209.
- Mohamad, N.; Muthusamy, K.; Embong, R.; Kusbiantoro, A.; Hashim, M. Environmental impact of cement production and Solutions: A review. Mater. Today Proc. 2022, 48, 741–746.
- Khan, K.; Ahmad, W.; Amin, M.; Aslam, F.; Ahmad, A.; Al-Faiad, M. Comparison of Prediction Models Based on Machine Learning for the Compressive Strength Estimation of Recycled Aggregate Concrete. Materials 2022, 15, 3430.
- Environment, U.N.; Scrivener, K.; John, V.; Gartner, E. Eco-efficient cements: Potential economically viable solutions for a low-CO2 cement-based materials industry. Cem. Concr. Res. 2018, 114, 2–6.
- Abdul-Wahab, S.; Al-Dhamri, H.; Ram, G.; Chatterjee, V. An overview of alternative raw materials used in cement and clinker manufacturing. Int. J. Sustain. Eng. 2021, 14, 743–760.
- Belaïd, F. How does concrete and cement industry transformation contribute to mitigating climate change challenges? Resour. Conserv. Recycl. Adv. 2022, 15, 200084.
- Nwankwo, C.; Bamigboye, G.; Davies, I.; Michaels, T. High volume Portland cement replacement: A review. Constr. Build. Mater. 2020, 260, 120445.
- Uwasu, M.; Hara, K.; Yabar, H. World cement production and environmental implications. Environ. Dev. 2014, 10, 36–47.
- Guo, Y.; Luo, L.; Liu, T.; Hao, L.; Li, Y.; Liu, P.; Zhu, T. A review of low-carbon technologies and projects for the global cement industry. J. Environ. Sci. 2023, 136, 682–697.
- ECA (European Cement Association). Global Cement Production. Available online: https://cembureau.eu/ (accessed on 10 September 2023).
- Yang, K.; Jung, Y.; Cho, M.; Tae, S. Effect of supplementary cementitious materials on reduction of CO2 emissions from concrete. J. Clean. Prod. 2015, 103, 774–783.
- Singh, G.; Subramaniam, K. Production and characterization of low-energy Portland composite cement from post-industrial waste. J. Clean. Prod. 2019, 239, 118024.
- Salas, D.; Ramirez, A.; Rodríguez, C.; Petroche, D.; Boero, A.; Duque-Rivera, J. Environmental impacts, life cycle assessment and potential improvement measures for cement production: A literature review. J. Clean. Prod. 2016, 113, 114–122.
- World Business Council for Sustainable Development. Guidelines for Co-Processing Fuels and Raw Materials in Cement Manufacturing, Version 2.0; WBCSD: Geneva, Switzerland, 2014.
- Benhelal, E.; Zahedi, G.; Shamsaei, E.; Bahadori, A. Global strategies and potentials to curb CO2 emissions in cement industry. J. Clean. Prod. 2013, 51, 142–161.
- Baidya, R.; Ghosh, S.; Parlikar, U. Sustainability of cement kiln co-processing of wastes in India: A pilot study. Environ. Technol. 2017, 38, 1650–1659.
- Navia, R.; Rivela, B.; Lorber, K.; Méndez, R. Recycling contaminated soil as alternative raw material in cement facilities: Life cycle assessment. Resour. Conserv. Recycl. 2006, 48, 339–356.
- ASTM C 563-95; Standard Test Method for Optimum SO3 in Hydraulic Cement Using 24-h Compressive Strength. ASTM International: West Conshohocken, PA, USA, 2001.
- Talero, R. Contribution to the Analytical and Physicochemical Study of the System: Pozzolanic Cements-Gypsum-Water (at 20 ± 2 °C). Ph.D. Thesis, Ftad. de CC. Químicas, Universidad Complutense de Madrid, Madrid, Spain, 1986.
- Xu, J.; Yi, B.; Fan, Y. A bottom-up optimization model for long-term CO2 emissions reduction pathway in the cement industry: A case study of China. Int. J. Greenh. Gas Control. 2016, 44, 199–216.
- Mikulčić, H.; Klemeš, J.; Vujanović, M.; Urbaniec, K.; Duić, N. Reducing greenhouse gasses emissions by fostering the deployment of alternative raw materials and energy sources in the cleaner cement manufacturing process. J. Clean. Prod. 2016, 136, 119–132.
- Hussain, J.; Khan, A.; Zhou, K. The impact of natural resource depletion on energy use and CO2 emission in Belt & Road Initiative countries: A cross-country analysis. Energy 2020, 199, 117409.
- Blankendaal, T.; Schuur, P.; Voordijk, H. Reducing the environmental impact of concrete and asphalt: A scenario approach. J. Clean. Prod. 2014, 66, 27–36.
- Mishra, U.; Sarsaiya, S.; Gupta, A. A systematic review on the impact of cement industries on the natural environment. Environ. Sci. Pollut. Res. 2022, 29, 18440–18451.
- Kwon, E.; Ahn, J.; Cho, B.; Park, D. A study on development of recycled cement made from waste cementitious powder. Constr. Build. Mater. 2015, 83, 174–180.
- Saravanan, A.; Vo, D.; Jeevanantham, S.; Bhuvaneswari, V.; Narayanan, V.; Yaashikaa, P.; Swetha, S.; Reshma, B. A comprehensive review on different approaches for CO2 utilization and conversion pathways. Chem. Eng. Sci. 2021, 236, 116515.
- Olivier, J.; Schure, K.; Peters, J. Trends in Global CO2 and Total Greenhouse Gas Emissions 2017 Report; PBL Netherlands Environmental Assessment Agency: The Hague, The Netherlands, 2017.
- Benhelal, E.; Shamsaei, E.; Rashid, M. Challenges against CO2 abatement strategies in cement industry: A review. J. Environ. Sci. 2021, 104, 84–101.
- Usman, M.; Balsalobre-Lorente, D.; Jahanger, A.; Ahmad, P. Pollution concern during globalization mode in financially resource-rich countries: Do financial development, natural resources, and renewable energy consumption matter? Renew. Energy 2022, 183, 90–102.
- Afif, M.; Afif, A.; Apostoleris, H.; Gandhi, K.; Dadlani, A.; Ghaferi, A.; Torgersen, J.; Chiesa, M. Ultra-cheap renewable energy as an enabling technology for deep industrial decarbonization via capture and utilization of process CO2 emissions. Energies 2022, 15, 5181.
- Chaudhury, R.; Sharma, U.; Thapliyal, P.; Singh, L. Low-CO2 emission strategies to achieve net zero target in cement sector. J. Clean. Prod. 2023, 417, 137466.
- Song, D.; Yang, J.; Chen, B.; Hayat, T.; Alsaedi, A. Life-cycle environmental impact analysis of a typical cement production chain. Appl. Energy 2016, 164, 916–923.
- Zhu, Q. CO2 Abatement in the Cement Industry; IEA Clean Coal Centre: London, UK, 2011.
- Adesina, A. Recent advances in the concrete industry to reduce its carbon dioxide emissions. Environ. Chall. 2020, 1, 100004.
- Ghalandari, V.; Majd, M.; Golestanian, A. Energy audit for pyro-processing unit of a new generation cement plant and feasibility study for recovering waste heat: A case study. Energy 2019, 173, 833–843.
- Kermeli, K.; Edelenbosch, O.; Crijns-Graus, W.; Van Ruijven, B.; Mima, S.; Van Vuuren, D.; Worrell, E. The scope for better industry representation in long-term energy models: Modeling the cement industry. Appl. Energy 2019, 240, 964–985.
- Hossain, M.; Poon, C.; Lo, I.; Cheng, J. Comparative LCA on using waste materials in the cement industry: A Hong Kong case study. Resour. Conserv. Recycl. 2017, 120, 199–208.
- Ishak, S.; Hashim, H. Low carbon measures for cement plant–a review. J. Clean. Prod. 2015, 103, 260–274.
- Emissions, B.Z. Zero Carbon Industry Plan: Rethinking Cement; Beyond Zero Emissions: Melbourne, VIC, Australia, 2017.
- Rissman, J.; Bataille, C.; Masanet, E.; Aden, N.; Morrow III, W.; Zhou, N.; Elliott, N.; Dell, R.; Heeren, N.; Huckestein, B.; et al. Technologies and policies to decarbonize global industry: Review and assessment of mitigation drivers through 2070. Appl. Energy 2020, 266, 114848.
- Chen, K.; Wang, J.; Yu, B.; Wu, H.; Zhang, J. Critical evaluation of construction and demolition waste and associated environmental impacts: A scientometric analysis. J. Clean. Prod. 2021, 287, 125071.
- Baidya, R.; Ghosh, S. Low carbon cement manufacturing in India by co-processing of alternative fuel and raw materials. Energy Sources A Recovery Util. Environ. Eff. 2019, 41, 2561–2572.
- Costa, F.; Ribeiro, D. Reduction in CO2 emissions during production of cement, with partial replacement of traditional raw materials by civil construction waste (CCW). J. Clean. Prod. 2020, 276, 123302.
- Ren, M.; Ma, T.; Fang, C.; Liu, X.; Guo, C.; Zhang, S.; Zhou, Z.; Zhu, Y.; Dai, H.; Huang, C. Negative emission technology is key to decarbonizing China’s cement industry. Appl. Energy 2023, 329, 120254.
- Usón, A.; López-Sabirón, A.; Ferreira, G.; Sastresa, E. Uses of alternative fuels and raw materials in the cement industry as sustainable waste management options. Renew. Sustain. Energy Rev. 2013, 23, 242–260.
- Fairbairn, E.; Americano, B.; Cordeiro, G.; Paula, T.; Toledo Filho, R.; Silvoso, M. Cement replacement by sugar cane bagasse ash: CO2 emissions reduction and potential for carbon credits. J. Environ. Manag. 2010, 91, 1864–1871.
- Tam, V.; Soomro, M.; Evangelista, A. A review of recycled aggregate in concrete applications (2000–2017). Constr. Build. Mater. 2018, 172, 272–292.
- Wu, H.; Zuo, J.; Zillante, G.; Wang, J.; Yuan, H. Status quo and future directions of construction and demolition waste research: A critical review. J. Clean. Prod. 2019, 240, 118163.
- Menegaki, M.; Damigos, D. A review on current situation and challenges of construction and demolition waste management. Curr. Opin. Green Sustain. Chem. 2018, 13, 8–15.
- Medina, C.; Banfill, P.; De Rojas, M.; Frías, M. Rheological and calorimetric behaviour of cements blended with containing ceramic sanitary ware and construction/demolition waste. Constr. Build. Mater. 2013, 40, 822–831.
- Ginga, C.; Ongpeng, J.; Daly, M. Circular economy on construction and demolition waste: A literature review on material recovery and production. Materials 2020, 13, 2970.
- Islam, R.; Nazifa, T.; Yuniarto, A.; Uddin, A.; Salmiati, S.; Shahid, S. An empirical study of construction and demolition waste generation and implication of recycling. Waste Manag. 2019, 95, 10–21.
- Mália, M.; De Brito, J.; Pinheiro, M.; Bravo, M. Construction and demolition waste indicators. Waste Manag. Res. 2013, 31, 241–255.
- Yuan, H.; Shen, L. Trend of the research on construction and demolition waste management. Waste Manag. 2011, 31, 670–679.
- Merino, M.; Gracia, P.; Azevedo, I. Sustainable construction: Construction and demolition waste reconsidered. Waste Manag. Res. 2010, 28, 118–129.
- Kabirifar, K.; Mojtahedi, M.; Wang, C.; Tam, V. A conceptual foundation for effective construction and demolition waste management. Clean. Eng. Technol. 2020, 1, 100019.
- Reis, G.; Quattrone, M.; Ambrós, W.; Cazacliu, B.; Sampaio, C. Current applications of recycled aggregates from construction and demolition: A review. Materials 2021, 14, 1700.
- Zheng, L.; Wu, H.; Zhang, H.; Duan, H.; Wang, J.; Jiang, W.; Dong, B.; Liu, G.; Zuo, J.; Song, Q. Characterizing the generation and flows of construction and demolition waste in China. Constr. Build. Mater. 2017, 136, 405–413.
- Zhang, C.; Hu, M.; Maio, F.; Sprecher, B.; Yang, X.; Tukker, A. An overview of the waste hierarchy framework for analyzing the circularity in construction and demolition waste management in Europe. Sci. Total Environ. 2022, 803, 149892.
- Sáez, P.; Osmani, M. A diagnosis of construction and demolition waste generation and recovery practice in the European Union. J. Clean. Prod. 2019, 241, 118400.
- Oliveira, T.; Dezen, B.; Possan, E. Use of concrete fine fraction waste as a replacement of Portland cement. J. Clean. Prod. 2020, 273, 123126.
- Akhtar, A.; Sarmah, A. Construction and demolition waste generation and properties of recycled aggregate concrete: A global perspective. J. Clean. Prod. 2018, 186, 262–281.
- Villoria-Sáez, P.; Porras-Amores, C.; Merino, M. Estimation of construction and demolition waste. In Advances in Construction and Demolition Waste Recycling Management, Processing and Environmental Assessment, 1st ed.; Pacheco-Torgal, F., Ding, Y., Calengo, F., Tuladhar, R., Koutamanis, A., Eds.; Woodhead Publishing: Cambridge, UK, 2020; pp. 13–30.
- Oikonomou, N. Recycled concrete aggregates. Cem. Concr. Compos. 2005, 27, 315–318.
- Huang, B.; Wang, X.; Kua, H.; Geng, Y.; Bleischwitz, R.; Ren, J. Construction and demolition waste management in China through the 3R principle. Resour. Conserv. Recycl. 2018, 129, 36–44.
- Hoang, N.; Ishigaki, T.; Kubota, R.; Yamada, M.; Kawamoto, K. A review of construction and demolition waste management in Southeast Asia. J. Mater. Cycles Waste Manag. 2020, 22, 315–325.
- Fischer, C.; Werge, M.; Reichel, A. EU as a Recycling Society. In European Topic Centre on Resource Waste Management; Working Paper 2/2009; European Environment Agency: Copenhagen, Denmark, 2009.
- Mohammed, M.; ElKady, H.; Abdel-Gawwad, H. Utilization of construction and demolition waste and synthetic aggregates. J. Build. Eng. 2021, 43, 103207.
- Mah, C.; Fujiwara, T.; Ho, C. Environmental impacts of construction and demolition waste management alternatives. Chem. Eng. Trans. 2018, 63, 343–348.
- Alsheyab, M. Recycling of construction and demolition waste and its impact on climate change and sustainable development. Int. J. Environ. Sci. Technol. 2022, 19, 2129–2138.
- Ferronato, N.; Moresco, L.; Lizarazu, G.; Portillo, M.; Conti, F.; Torretta, V. Comparison of environmental impacts related to municipal solid waste and construction and demolition waste management and recycling in a Latin American developing city. Environ. Sci. Pollut. Res. 2021, 22, 1–5.
- Tafesse, S.; Girma, Y.; Dessalegn, E. Analysis of the socio-economic and environmental impacts of construction waste and management practices. Heliyon 2022, 8, 09169.
- Sun, X.; Xu, Y.; Liu, Y.; Nai, C.; Dong, L.; Liu, J.; Huang, Q. Evolution of geomembrane degradation and defects in a landfill: Impacts on long-term leachate leakage and groundwater quality. J. Clean. Prod. 2019, 224, 335–345.
- Nodehi, M.; Taghvaee, V. Applying circular economy to construction industry through use of waste materials: A review of supplementary cementitious materials, plastics, and ceramics. Circ. Econ. Sustain. 2022, 2, 987–1020.
- Contreras, M.; Teixeira, S.; Lucas, M.; Lima, L.; Cardoso, D.; Da Silva, G.; Gregório, G.; De Souza, A.; Dos Santos, A. Recycling of construction and demolition waste for producing new construction material (Brazil case-study). Constr. Build. Mater. 2016, 123, 594–600.
- Long, H.; Liao, Y.; Cui, C.; Liu, M.; Liu, Z.; Li, L.; Hu, W.; Yan, D. Assessment of popular techniques for co-processing municipal solid waste in Chinese cement kilns. Front. Environ. Sci. Eng. 2022, 16, 51.
- Ferronato, N.; Rada, E.; Portillo, M.; Cioca, L.; Ragazzi, M.; Torretta, V. Introduction of the circular economy within developing regions: A comparative analysis of advantages and opportunities for waste valorization. J. Environ. Manag. 2019, 230, 366–378.
- Gartner, E.; Hirao, H. A review of alternative approaches to the reduction of CO2 emissions associated with the manufacture of the binder phase in concrete. Cem. Concr. Res. 2015, 78, 126–142.
- Miller, S.; John, V.; Pacca, S.; Horvath, A. Carbon dioxide reduction potential in the global cement industry by 2050. Cem. Concr. Res. 2018, 114, 115–124.
- Ali, M.; Saidur, R.; Hossain, M. A review on emission analysis in cement industries. Renew. Sustain. Energy Rev. 2011, 15, 2252–2261.
- Rocha, J.; Toledo Filho, R.; Cayo-Chileno, N. Sustainable alternatives to CO2 reduction in the cement industry: A short review. Mater. Today Proc. 2022, 57, 436–439.
- Sahoo, N.; Kumar, A.; Samsher, S. Review on energy conservation and emission reduction approaches for cement industry. Environ. Dev. 2022, 44, 100767.
- Poudyal, L.; Adhikari, K. Environmental sustainability in cement industry: An integrated approach for green and economical cement production. Resour. Environ. Sustain. 2021, 4, 100024.
- Kunche, A.; Mielczarek, B. Application of system dynamic modelling for evaluation of carbon mitigation strategies in cement industries: A comparative overview of the current state of the art. Energies 2021, 14, 1464.
- Plaza, M.; Martínez, S.; Rubiera, F. CO2 capture, use, and storage in the cement industry: State of the art and expectations. Energies 2020, 13, 5692.
- Ahmed, M.; Bashar, I.; Alam, S.; Wasi, A.; Jerin, I.; Khatun, S.; Rahman, M. An overview of Asian cement industry: Environmental impacts, research methodologies and mitigation measures. Sustain. Prod. Consum. 2021, 28, 1018–1039.
- Wang, Y.; Höller, S.; Viebahn, P.; Hao, Z. Integrated assessment of CO2 reduction technologies in China’s cement industry. Int. J. Greenh. Gas Control 2014, 20, 27–36.
- Dinga, C.; Wen, Z. China’s green deal: Can China’s cement industry achieve carbon neutral emissions by 2060? Renew. Sustain. Energy Rev. 2022, 155, 111931.
- Zhang, C.; Yu, B.; Chen, J.; Wei, Y. Green transition pathways for cement industry in China. Resour. Conserv. Recycl. 2021, 166, 105355.
- Panjaitan, T.; Dargusch, P.; Wadley, D.; Aziz, A. Meeting international standards of cleaner production in developing countries: Challenges and financial realities facing the Indonesian cement industry. J. Clean. Prod. 2021, 318, 128604.
- Junianto, I.; Sunardi; Sumiarsa, D. The Possibility of Achieving Zero CO2 Emission in the Indonesian Cement Industry by 2050: A Stakeholder System Dynamic Perspective. Sustainability 2023, 15, 6085.
- Watari, T.; Cao, Z.; Hata, S.; Nansai, K. Efficient use of cement and concrete to reduce reliance on supply-side technologies for net-zero emissions. Nat. Commun. 2022, 13, 4158.
- Ghacham, A.; Pasquier, L.; Cecchi, E.; Blais, J.; Mercier, G. Valorization of waste concrete through CO2 mineral carbonation: Optimizing parameters and improving reactivity using concrete separation. J. Clean. Prod. 2017, 166, 869–878.
- Jaiboon, N.; Wongsapai, W.; Daroon, S.; Bunchuaidee, R.; Ritkrerkkrai, C.; Damrongsak, D. Greenhouse gas mitigation potential from waste heat recovery for power generation in cement industry: The case of Thailand. Energy Rep. 2021, 7, 638–643.
- Worrell, E.; Martin, N.; Price, L. Potentials for energy efficiency improvement in the US cement industry. Energy 2000, 25, 1189–1214.
- Deja, J.; Uliasz-Bochenczyk, A.; Mokrzycki, E. CO2 emissions from Polish cement industry. Int. J. Greenh. Gas Control 2010, 4, 583–588.
- Supino, S.; Malandrino, O.; Testa, M.; Sica, D. Sustainability in the EU cement industry: The Italian and German experiences. J. Clean. Prod. 2016, 112, 430–442.
- Garðarsdóttir, S.; Normann, F.; Skagestad, R.; Johnsson, F. Investment costs and CO2 reduction potential of carbon capture from industrial plants–A Swedish case study. Int. J. Greenh. Gas Control 2018, 76, 111–124.
- Beguedou, E.; Narra, S.; Afrakoma Armoo, E.; Agboka, K.; Damgou, M. Alternative Fuels Substitution in Cement Industries for Improved Energy Efficiency and Sustainability. Energies 2023, 16, 3533.
- Rahman, A.; Rasul, M.; Khan, M.; Sharma, S. Recent development on the uses of alternative fuels in cement manufacturing process. Fuel 2015, 145, 84–99.
- Hassaan, M. Basalt rock as an alternative raw material in Portland cement manufacture. Mater. Lett. 2001, 50, 172–178.
- Tsiliyannis, A. Alternative fuels in cement manufacturing: Modeling for process optimization under direct and compound operation. Fuel 2012, 99, 20–39.
- Bourtsalas, A.; Zhang, J.; Castaldi, M.; Themelis, N.; Karaiskakis, A. Use of non-recycled plastics and paper as alternative fuel in cement production. J. Clean. Prod. 2018, 181, 8–16.
- Chatterjee, A.; Sui, T. Alternative fuels–effects on clinker process and properties. Cem. Concr. Res. 2019, 123, 105777.
- Favier, A.; De Wolf, C.; Scrivener, K.; Habert, G. A Sustainable Future for the European Cement and Concrete Industry: Technology Assessment for Full Decarbonisation of the Industry by 2050; ETH Zurich: Zurich, Switzerland, 2018.
- Pardo, N.; Moya, J.; Mercier, A. Prospective on the energy efficiency and CO2 emissions in the EU cement industry. Energy 2011, 36, 3244–3254.
- Kusuma, R.; Hiremath, R.; Rajesh, P.; Kumar, B.; Renukappa, S. Sustainable transition towards biomass-based cement industry: A review. Renew. Sustain. Energy Rev. 2022, 163, 112503.
- Rahman, A.; Rasul, M.; Khan, M.; Sharma, S. Assessment of energy performance and emission control using alternative fuels in cement industry through a process model. Energies 2017, 10, 1996.
- Thomas, S. Use of alternative fuels in the cement industry. In Refra Technik Seminar; REFRATECHNIK: Duesseldorf, Germany, 2010; p. 47.
- Cascarosa, E.; Gasco, L.; García, G.; Gea, G.; Arauzo, J. Meat and bone meal and coal co-gasification: Environmental advantages. Resour. Conserv. Recycl. 2012, 59, 32–37.
- Gulyurtlu, I.; Boavida, D.; Abelha, P.; Lopes, M.; Cabrita, I. Co-combustion of coal and meat and bone meal. Fuel 2005, 84, 2137–2148.
- Fryda, L.; Panopoulos, K.; Vourliotis, P.; Kakaras, E.; Pavlidou, E. Meat and bone meal as secondary fuel in fluidized bed combustion. Proc. Combust. Inst. 2007, 31, 2829–2837.
- Cascarosa, E.; Gasco, L.; Gea, G.; Sánchez, J.; Arauzo, J. Co-gasification of meat and bone meal with coal in a fluidised bed reactor. Fuel 2011, 90, 2798–2807.
- Ariyaratne, W.; Malagalage, A.; Melaaen, M.; Tokheim, L. CFD modelling of meat and bone meal combustion in a cement rotary kiln–Investigation of fuel particle size and fuel feeding position impacts. Chem. Eng. Sci. 2015, 123, 596–608.
- Kantorek, M.; Jesionek, K.; Polesek-Karczewska, S.; Ziółkowski, P.; Badur, J. Thermal utilization of meat and bone meals. Performance analysis in terms of drying process, pyrolysis and kinetics of volatiles combustion. Fuel 2019, 254, 115548.
- Kantorek, M.; Jesionek, K.; Polesek-Karczewska, S.; Ziółkowski, P.; Stajnke, M.; Badur, J. Thermal utilization of meat-and-bone meal using the rotary kiln pyrolyzer and the fluidized bed boiler–The performance of pilot-scale installation. Renew. Energy 2021, 164, 1447–1456.
- Cascarosa, E.; Gea, G.; Arauzo, J. Thermochemical processing of meat and bone meal: A review. Renew. Sustain. Energy Rev. 2012, 16, 942–957.
- Duvallet, T.; Jewell, R. Recycling of bone ash from animal wastes and by-products in the production of novel cements. J. Am. Ceram. Soc. 2023, 106, 3720–3735.
- Galvagno, S.; Casciaro, G.; Casu, S.; Martino, M.; Mingazzini, C.; Russo, A.; Portofino, S. Steam gasification of tyre waste, poplar, and refuse-derived fuel: A comparative analysis. Waste Manag. 2009, 29, 678–689.
- Rovira, J.; Mari, M.; Nadal, M.; Schuhmacher, M.; Domingo, J. Partial replacement of fossil fuel in a cement plant: Risk assessment for the population living in the neighborhood. Sci. Total Environ. 2010, 408, 5372–5380.
- Reza, B.; Soltani, A.; Ruparathna, R.; Sadiq, R.; Hewage, K. Environmental and economic aspects of production and utilization of RDF as alternative fuel in cement plants: A case study of Metro Vancouver Waste Management. Resour. Conserv. Recycl. 2013, 81, 105–114.
- Shumal, M.; Jahromi, A.; Ferdowsi, A.; Dehkordi, S.; Moloudian, A.; Dehnavi, A. Comprehensive analysis of municipal solid waste rejected fractions as a source of Refused Derived Fuel in developing countries (case study of Isfahan-Iran): Environmental Impact and sustainable development. Renew. Energy 2020, 146, 404–413.
- Kahawalage, A.; Melaeen, M.; Tokheim, L. Opportunities and challenges of using SRF as an alternative fuel in the cement industry. Clean. Waste Syst. 2023, 4, 100072.
- Sagala, G.; Kristanto, G.; Kusuma, M.; Rizki, S. Assessment of municipal solid waste as refuse derived fuel in the cement industry. Int. J. Adv. Sci. Eng. Inf. Technol. 2018, 8, 1062–1070.
- Świechowski, K.; Syguła, E.; Koziel, J.; Stępień, P.; Kugler, S.; Manczarski, P.; Białowiec, A. Low-temperature pyrolysis of municipal solid waste components and refuse-derived fuel—Process efficiency and fuel properties of carbonized solid fuel. Data 2020, 5, 48.
- Sharma, P.; Sheth, P.N.; Mohapatra, B. Recent Progress in Refuse Derived Fuel (RDF) Co-processing in Cement Production: Direct Firing in Kiln/Calciner vs Process Integration of RDF Gasification. Waste Biomass Valorization 2022, 13, 4347–4374.
- Pieper, C.; Liedmann, B.; Wirtz, S.; Scherer, V.; Bodendiek, N.; Schaefer, S. Interaction of the combustion of refuse derived fuel with the clinker bed in rotary cement kilns: A numerical study. Fuel 2020, 266, 117048.
- Stępień, P.; Pulka, J.; Serowik, M.; Białowiec, A. Thermogravimetric and calorimetric characteristics of alternative fuel in terms of its use in low-temperature pyrolysis. Waste Biomass Valorization 2019, 10, 1669–1677.
- Aranda, U.; Ferreira, G.; Bribián, Z.; Vásquez, Z. Study of the environmental performance of end-of-life tyre recycling through a simplified mathematical approach. Therm. Sci. 2012, 16, 889–899.
- Vasiliu, L.; Gencel, O.; Damian, I.; Harja, M. Capitalization of tires waste as derived fuel for sustainable cement production. Sustain. Energy Technol. Assess. 2023, 56, 103104.
- Kishan, G.; Sakthivel, M.; Vijayakumar, R.; Lingeshwaran, N. Life cycle assesment on tire derived fuel as alternative fuel in cement industry. Mater. Today Proc. 2021, 47, 5483–5488.
- Breyer, S.; Mekhitarian, L.; Rimez, B.; Haut, B. Production of an alternative fuel by the co-pyrolysis of landfill recovered plastic wastes and used lubrication oils. Waste Manag. 2017, 60, 363–374.
- Chakritthakul, S.; Kuprianov, V. Co-firing of eucalyptus bark and rubberwood sawdust in a swirling fluidized-bed combustor using an axial flow swirler. Bioresour. Technol. 2011, 102, 8268–8278.
- Steenari, B.; Lindqvist, O. Fly ash characteristics in co-combustion of wood with coal, oil or peat. Fuel 1999, 78, 479–488.
- Nielsen, C. Utilisation of straw and similar agricultural residues. Biomass Bioenergy 1995, 9, 315–323.
- Jensen, P.; Sander, B.; Dam-Johansen, K. Pretreatment of straw for power production by pyrolysis and char wash. Biomass Bioenergy 2001, 20, 431–446.
- Walker, N.; Bazilian, M.; Buckley, P. Possibilities of reducing CO2 emissions from energy-intensive industries by the increased use of forest-derived fuels in Ireland. Biomass Bioenergy 2009, 33, 1229–1238.
- Werther, J.; Saenger, M.; Hartge, E.; Ogada, T.; Siagi, Z. Combustion of agricultural residues. Prog. Energy Combust. Sci. 2000, 26, 1–27.
- Hossain, M.; Poon, C.; Wong, M.; Khine, A. Techno-environmental feasibility of wood waste derived fuel for cement production. J. Clean. Prod. 2019, 230, 663–671.
- Caballero, J.; Conesa, J.; Font, R.; Marcilla, A. Pyrolysis kinetics of almond shells and olive stones considering their organic fractions. J. Anal. Appl. Pyrolysis 1997, 42, 159–175.
- Rapagna, S.; Latif, A. Steam gasification of almond shells in a fluidised bed reactor: The influence of temperature and particle size on product yield and distribution. Biomass Bioenergy 1997, 12, 281–288.
- Rodríguez, G.; Lama, A.; Rodríguez, R.; Jiménez, A.; Guillén, R.; Fernández-Bolanos, J. Olive stone an attractive source of bioactive and valuable compounds. Bioresour. Technol. 2008, 99, 5261–5269.
- Shuit, S.; Tan, K.; Lee, K.; Kamaruddin, A. Oil palm biomass as a sustainable energy source: A Malaysian case study. Energy 2009, 34, 1225–1235.
- Papanikola, K.; Papadopoulou, K.; Tsiliyannis, C.; Fotinopoulou, I.; Katsiampoulas, A.; Chalarakis, E.; Georgiopoulou, M.; Rontogianni, V.; Michalopoulos, I.; Mathioudakis, D.; et al. Food residue biomass product as an alternative fuel for the cement industry. Environ. Sci. Pollut. Res. 2019, 26, 35555–35564.
- Henry, C.; Lynam, J. Embodied energy of rice husk ash for sustainable cement production. Case Stud. Chem. Environ. Eng. 2020, 2, 100004.
- Nhuchhen, D.; Sit, S.; Layzell, D. Alternative fuels co-fired with natural gas in the pre-calciner of a cement plant: Energy and material flows. Fuel 2021, 295, 120544.
- Yasmin, N.; Grundmann, P. Adoption and diffusion of renewable energy—The case of biogas as alternative fuel for cooking in Pakistan. Renew. Sustain. Energy Rev. 2019, 101, 255–264.
- Xie, L.; Tao, L.; Gao, J.; Fei, X.; Xia, W.; Jiang, Y. Effect of moisture content in sewage sludge on air gasification. J. Fuel Chem. Technol. 2010, 38, 615–620.
- Nipattummakul, N.; Ahmed, I.; Kerdsuwan, S.; Gupta, A. Hydrogen and syngas production from sewage sludge via steam gasification. Int. J. Hydrogen Energy 2010, 35, 11738–11745.
- Fang, P.; Tang, Z.; Huang, J.; Cen, C.; Tang, Z.; Chen, X. Using sewage sludge as a denitration agent and secondary fuel in a cement plant: A case study. Fuel Process. Technol. 2015, 137, 1–7.
- Huang, M.; Ying, X.; Shen, D.; Feng, H.; Li, N.; Zhou, Y.; Long, Y. Evaluation of oil sludge as an alternative fuel in the production of Portland cement clinker. Constr. Build. Mater. 2017, 152, 226–231.
- Kääntee, U.; Zevenhoven, R.; Backman, R.; Hupa, M. Cement manufacturing using alternative fuels and the advantages of process modelling. Fuel Process. Technol. 2004, 85, 293–301.
- Aldrian, A.; Sarc, R.; Pomberger, R.; Lorber, K.; Sipple, E. Solid recovered fuels in the cement industry–semi-automated sample preparation unit as a means for facilitated practical application. Waste Manag. Res. 2016, 34, 254–264.
- Thomanetz, E. Solid recovered fuels in the cement industry with special respect to hazardous waste. Waste Manag. Res. 2012, 30, 404–412.
- Wang, Y.; Yi, H.; Tang, X.; Wang, Y.; An, H.; Liu, J. Historical trend and decarbonization pathway of China’s cement industry: A literature review. Sci. Total Environ. 2023, 891, 164580.
- Coffetti, D.; Crotti, E.; Gazzaniga, G.; Carrara, M.; Pastore, T.; Coppola, L. Pathways towards sustainable concrete. Cem. Concr. Res. 2022, 154, 106718.
- Kara, M.; Kilic, Y.; Erenoglu, T. An experimental study on construction and demolition waste usage as secondary raw material for cement production. World J. Innov. Res. 2017, 2, 1–7.
- De Schepper, M.; Vernimmen, L.; De Belie, N.; De Buysser, K.; Van Driessche, I. The assessment of clinker and cement regenerated from completely recyclable concrete. In Proceedings of the 13th ICCC International Congress on the Chemistry of Cement, Madrid, Spain, 3–8 July 2011.
- Marroccoli, M.; Telesca, A.; Ibris, N.; Naik, T. Construction and demolition waste as raw materials for sustainable cements. In Proceedings of the Fourth International Conference on Sustainable Construction Materials and Technologies, Las Vegas, NV, USA, 7–11 August 2016.
- Galbenis, C.; Tsimas, S. Use of construction and demolition wastes as raw materials in cement clinker production. China Particuology 2006, 4, 83–85.
- Garzón, E.; Martínez-Martínez, S.; Pérez-Villarrejo, L.; Sánchez-Soto, P. Assessment of construction and demolition wastes (CDWs) as raw materials for the manufacture of low-strength concrete and bases and sub-bases of roads. Mater. Lett. 2022, 320, 132343.
- Liu, Y.; Yang, C.; Wang, F.; Hu, S.; Zhu, M.; Hu, C.; Lu, L.; Liu, Z. Evaluation on recycled clinker production and properties from regeneration of completely recycle concrete. Constr. Build. Mater. 2021, 301, 123882.
- De Schepper, M.; De Buysser, K.; Van Driessche, I.; De Belie, N. The regeneration of cement out of Completely Recyclable Concrete: Clinker production evaluation. Constr. Build. Mater. 2013, 38, 1001–1009.
- Diliberto, C.; Lecomte, A.; Mechling, J.; Izoret, L.; Smith, A. Valorisation of recycled concrete sands in cement raw meal for cement production. Mater. Struct. 2017, 50, 127.
- Haiyan, Y.; Zhixiao, R.; Cuina, Q.; Lintong, H. Experimental study on recycled cement prepared from waste concrete. Ferroelectrics 2021, 570, 218–227.
- Schoon, J.; De Buysser, K.; Van Driessche, I.; De Belie, N. Fines extracted from recycled concrete as alternative raw material for Portland cement clinker production. Cem. Concr. Compos. 2015, 58, 70–80.
- Gastaldi, D.; Canonico, F.; Capelli, L.; Buzzi, L.; Boccaleri, E.; Irico, S. An investigation on the recycling of hydrated cement from concrete demolition waste. Cem. Concr. Compos. 2015, 61, 29–35.
- Zhutovsky, S.; Shishkin, A. Recycling of hydrated Portland cement paste into new clinker. Constr. Build. Mater. 2021, 280, 122510.
- Miao, X.; Fang, Y.; Gong, Y.; Gu, Y.; Zhu, C. Recycling waste hardened mortar and paste from concrete to produce cement: State of the art. Mater. Res. Innov. 2015, 19 (Suppl. S6), S6-121–S6-124.
- Krour, H.; Trauchessec, R.; Lecomte, A.; Diliberto, C.; Barnes-Davin, L.; Bolze, B.; Delhay, A. Incorporation rate of recycled aggregates in cement raw meals. Constr. Build. Mater. 2020, 248, 118217.
- Kumar, G.; Deoliya, R. Recycled cement and recycled fine aggregates as alternative resources of raw materials for sustainable cellular light weight flowable material. Constr. Build. Mater. 2022, 326, 126878.
- Kirgiz, M. Use of ultrafine marble and brick particles as raw materials in cement manufacturing. Mater. Struct. 2015, 48, 2929–2941.
- Maslehuddin, M.; Al-Amoudi, O.; Shameem, M.; Rehman, M.; Ibrahim, M. Usage of cement kiln dust in cement products–research review and preliminary investigations. Constr. Build. Mater. 2008, 22, 2369–2375.
- Soares, E.; Castro-Gomes, J.; Sitarz, M.; Zdeb, T.; Hager, I.; Hassan, K.; Al-Kuwari, M. Feasibility for co-utilisation of Carbonated Reactive Magnesia Cement (CRMC) and industrial wastes in circular economy and CO2 mineralisation. Constr. Build. Mater. 2022, 323, 126488.
- Puertas, F.; García-Díaz, I.; Barba, A.; Gazulla, M.; Palacios, M.; Gómez, M.; Martínez-Ramírez, S. Ceramic wastes as alternative raw materials for Portland cement clinker production. Cem. Concr. Compos. 2008, 30, 798–805.
- Puertas, F.; García-Díaz, I.; Palacios, M.; Gazulla, M.; Gómez, M.; Orduña, M. Clinkers and cements obtained from raw mix containing ceramic waste as a raw material. Characterization, hydration and leaching studies. Cem. Concr. Compos. 2010, 32, 175–186.
- Puertas, F.; Barba, A.; Gazulla, M.; Gómez, M.; Palacios, M.; Martínez-Ramírez, S. Ceramic wastes as raw materials in Portland cement clinker fabrication: Characterization and alkaline activation. Mater. Constr. 2006, 56, 73–84.
- Liu, Y.; Yang, C.; Wang, F.; Hu, S.; Zhu, M.; Hu, C.; Lu, L. Performance evaluation of regenerated clinker from completely recyclable mortar. Constr. Build. Mater. 2021, 309, 125184.
- Schoon, J.; De Buysser, K.; Van Driessche, I.; De Belie, N. Feasibility study on the use of cellular concrete as alternative raw material for Portland clinker production. Constr. Build. Mater. 2013, 48, 725–733.
- Santos, T.; Cilla, M. Use of asbestos cement tile waste (ACW) as mineralizer in the production of Portland cement with low CO2 emission and lower energy consumption. J. Clean. Prod. 2022, 335, 130061.
- Kim, J.; Tae, S.; Kim, R. Theoretical study on the production of environment-friendly recycled cement using inorganic construction wastes as secondary materials in South Korea. Sustainability 2018, 10, 4449.
- Faure, A.; Smith, A.; Coudray, C.; Anger, B.; Colina, H.; Moulin, I.; Thery, F. Ability of two dam fine-grained sediments to be used in cement industry as raw material for clinker production and as pozzolanic additional constituent of portland-composite cement. Waste Biomass Valorization 2017, 8, 2141–2163.
- Faure, A.; Coudray, C.; Anger, B.; Moulin, I.; Colina, H.; Izoret, L.; Théry, F.; Smith, A. Beneficial reuse of dam fine sediments as clinker raw material. Constr. Build. Mater. 2019, 218, 365–384.
- Anger, B.; Moulin, I.; Commene, J.; Thery, F.; Levacher, D. Fine-grained reservoir sediments: An interesting alternative raw material for Portland cement clinker production. Eur. J. Environ. Civ. 2019, 23, 957–970.
- Aouad, G.; Laboudigue, A.; Gineys, N.; Abriak, N. Dredged sediments used as novel supply of raw material to produce Portland cement clinker. Cem. Concr. Compos. 2012, 34, 788–793.
- Rodríguez, N.; Granados, R.; Blanco-Varela, M.; Cortina, J.; Martínez-Ramírez, S.; Marsal, M.; Guillem, M.; Puig, J.; Fos, C.; Larrotcha, E.; et al. Evaluation of a lime-mediated sewage sludge stabilisation process. Product characterisation and technological validation for its use in the cement industry. Waste Manag. 2012, 32, 550–560.
- Valderrama, C.; Granados, R.; Cortina, J.; Gasol, C.; Guillem, M.; Josa, A. Comparative LCA of sewage sludge valorisation as both fuel and raw material substitute in clinker production. J. Clean. Prod. 2013, 51, 205–213.
- Lin, K.; Lo, K.; Hung, M.; Cheng, T.; Chang, Y. Recycling of spent catalyst and waste sludge from industry to substitute raw materials in the preparation of Portland cement clinker. Sustain. Environ. Res. 2017, 27, 251–257.
- Yen, C.L.; Tseng, D.H.; Lin, T.T. Characterization of eco-cement paste produced from waste sludges. Chemosphere 2011, 84, 220–226.
- Rezaee, F.; Danesh, S.; Tavakkolizadeh, M.; Mohammadi-Khatami, M. Investigating chemical, physical and mechanical properties of eco-cement produced using dry sewage sludge and traditional raw materials. J. Clean. Prod. 2019, 214, 749–757.
- Valderrama, C.; Granados, R.; Cortina, J. Stabilisation of dewatered domestic sewage sludge by lime addition as raw material for the cement industry: Understanding process and reactor performance. Chem. Eng. J. 2013, 232, 458–467.
- Lin, K.; Chiang, K.; Lin, C. Hydration characteristics of waste sludge ash that is reused in eco-cement clinkers. Cem. Concr. Res. 2005, 35, 1074–1081.
- Liu, G.; Yang, Z.; Chen, B.; Zhang, J.; Liu, X.; Zhang, Y.; Su, M.; Ulgiati, S. Scenarios for sewage sludge reduction and reuse in clinker production towards regional eco-industrial development: A comparative emergy-based assessment. J. Clean. Prod. 2015, 103, 371–383.
- Chen, I.; Juenger, M. Incorporation of waste materials into Portland cement clinker synthesized from natural raw materials. J. Mater. Sci. 2009, 44, 2617–2627.
- Baltakys, K.; Dambrauskas, T.; Rubinaite, D.; Siauciunas, R.; Grineviciene, A. Formation and hydration of eco-friendly cement using industrial wastes as raw materials. Sci. Rep. 2021, 11, 14742.
- Flegar, M.; Serdar, M.; Londono-Zuluaga, D.; Scrivener, K. Regional waste streams as potential raw materials for immediate implementation in cement production. Materials 2020, 13, 5456.
- Miller, S. Supplementary cementitious materials to mitigate greenhouse gas emissions from concrete: Can there be too much of a good thing? J. Clean. Prod. 2018, 178, 587–598.
- Zhang, T.; Yu, Q.; Wei, J.; Zhang, P. Efficient utilization of cementitious materials to produce sustainable blended cement. Cem. Concr. Compos. 2012, 34, 692–699.
- Jiang, W.; Li, X.; Lv, Y.; Jiang, D.; Liu, Z.; He, C. Mechanical and hydration properties of low clinker cement containing high volume superfine blast furnace slag and nano silica. Constr. Build. Mater. 2020, 238, 117683.
- Xu, W.; Xu, J.; Liu, J.; Li, H.; Cao, B.; Huang, X.; Li, G. The utilization of lime-dried sludge as resource for producing cement. J. Clean. Prod. 2014, 83, 286–293.
- Ping, L.; Zhao, G.; Lin, X.; Gu, Y.; Liu, W.; Cao, H.; Huang, J.; Xu, J. Feasibility and carbon footprint analysis of lime-dried sludge for cement production. Sustainability 2020, 12, 2500.
- Tsakiridis, P.; Papadimitriou, G.; Tsivilis, S.; Koroneos, C. Utilization of steel slag for Portland cement clinker production. J. Hazard. Mater. 2008, 152, 805–811.
- Gao, T.; Dai, T.; Shen, L.; Jiang, L. Benefits of using steel slag in cement clinker production for environmental conservation and economic revenue generation. J. Clean. Prod. 2021, 282, 124538.
- Iacobescu, R.; Angelopoulos, G.; Jones, P.; Blanpain, B.; Pontikes, Y. Ladle metallurgy stainless steel slag as a raw material in Ordinary Portland Cement production: A possibility for industrial symbiosis. J. Clean. Prod. 2016, 112, 872–881.
- Carvalho, S.; Vernilli, F.; Almeida, B.; Oliveira, M.; Silva, S. Reducing environmental impacts: The use of basic oxygen furnace slag in portland cement. J. Clean. Prod. 2018, 172, 385–390.
- Carvalho, S.; Vernilli, F.; Almeida, B.; Demarco, M.; Silva, S. The recycling effect of BOF slag in the Portland cement properties. Resour. Conserv. Recycl. 2017, 127, 216–220.
- Gong, X.; Zhang, T.; Zhang, J.; Wang, Z.; Liu, J.; Cao, J.; Wang, C. Recycling and utilization of calcium carbide slag-current status and new opportunities. Renew. Sustain. Energy Rev. 2022, 159, 112133.
- Li, H.; Huang, Y.; Yang, X.; Jiang, Z.; Yang, Z. Approach to the management of magnesium slag via the production of Portland cement clinker. J. Mater. Cycles Waste Manag. 2018, 20, 1701–1709.
- Chen, H.; Ma, X.; Dai, H. Reuse of water purification sludge as raw material in cement production. Cem. Concr. Compos. 2010, 32, 436–439.
- Shih, P.; Chang, J.; Lu, H.; Chiang, L. Reuse of heavy metal-containing sludges in cement production. Cem. Concr. Res. 2005, 35, 2110–2115.
- Bernardo, G.; Marroccoli, M.; Nobili, M.; Telesca, A.; Valenti, G. The use of oil well-derived drilling waste and electric arc furnace slag as alternative raw materials in clinker production. Resour. Conserv. Recycl. 2007, 52, 95–102.
- Liu, G.; Zhan, J.; Zheng, M.; Li, L.; Li, C.; Jiang, X.; Wang, M.; Zhao, Y.; Jin, R. Field pilot study on emissions, formations and distributions of PCDD/Fs from cement kiln co-processing fly ash from municipal solid waste incinerations. J. Hazard. Mater. 2015, 299, 471–478.
- Pan, J.; Huang, C.; Kuo, J.; Lin, S. Recycling MSWI bottom and fly ash as raw materials for Portland cement. Waste Manag. 2008, 28, 1113–1118.
- Vargas, J.; Halog, A. Effective carbon emission reductions from using upgraded fly ash in the cement industry. J. Clean. Prod. 2015, 103, 948–959.
- Krammart, P.; Tangtermsirikul, S. Properties of cement made by partially replacing cement raw materials with municipal solid waste ashes and calcium carbide waste. Constr. Build. Mater. 2004, 18, 579–583.
- Kleib, J.; Aouad, G.; Abriak, N.; Benzerzour, M. Production of Portland cement clinker from French municipal solid waste incineration bottom ash. Case Stud. Constr. Mater. 2021, 15, 00629.
- Lam, C.; Barford, J.; McKay, G. Utilization of municipal solid waste incineration ash in Portland cement clinker. Clean Technol. Environ. Policy. 2011, 13, 607–615.
- Lederer, J.; Trinkel, V.; Fellner, J. Wide-scale utilization of MSWI fly ashes in cement production and its impact on average heavy metal contents in cements: The case of Austria. Waste Manag. 2017, 60, 247–258.
- Wanga, L.; Jin, Y.; Nieb, Y.; Li, R. Recycling of municipal solid waste incineration fly ash for ordinary Portland cement production: A real-scale test resources. Resour. Conserv. Recycl. 2010, 54, 1428–1435.
- Liu, X.; Zhang, N. Utilization of red mud in cement production: A review. Waste Manag. Res. 2011, 29, 1053–1063.
- Ren, C.; Wang, W.; Yao, Y.; Wu, S.; Yao, X. Complementary use of industrial solid wastes to produce green materials and their role in CO2 reduction. J. Clean. Prod. 2020, 252, 119840.
- Ribeiro, D.; Labrincha, J.; Morelli, M. Potential use of natural red mud as pozzolan for Portland cement. Mater. Res. 2011, 14, 60–66.
- Abdul-Wahab, S.; Al-Dhamri, H.; Ram, G.; Black, L. The use of oil-based mud cuttings as an alternative raw material to produce high sulfate-resistant oil well cement. J. Clean. Prod. 2020, 269, 122207.
- Abdul-Wahab, S.; Al-Rawas, G.; Ali, S.; Al-Dhamri, H. Impact of the addition of oil-based mud on carbon dioxide emissions in a cement plant. J. Clean. Prod. 2016, 112, 4214–4225.
- Young, G.; Yang, M. Preparation and characterization of Portland cement clinker from iron ore tailings. Constr. Build. Mater. 2019, 197, 152–156.
- Luo, L.; Zhang, Y.; Bao, S.; Chen, T. Utilization of iron ore tailings as raw material for Portland cement clinker production. Adv. Mater. Sci. Eng. 2016, 2016, 1596047.
- Cheng, Y.; Qi, R.; Hou, J.; Huang, Q. Feasibility study on utilization of copper tailings as raw meal and addition for low carbon Portland cement production. Constr. Build. Mater. 2023, 382, 131275.
- Sadala, S.; Dutta, S.; Raghava, R.; Jyothsna, T.; Chakradhar, B.; Ghosh, S. Resource recovery as alternative fuel and raw material from hazardous waste. Waste Manag. Res. 2019, 37, 1063–1076.
- Buruberri, L.; Seabra, M.; Labrincha, J. Preparation of clinker from paper pulp industry wastes. J. Hazard. Mater. 2015, 286, 252–260.
- Simão, L.; Jiusti, J.; Lóh, N.; Hotza, D.; Raupp-Pereira, F.; Labrincha, J.; Montedo, O. Waste-containing clinkers: Valorization of alternative mineral sources from pulp and paper mills. Process Saf. Environ. Prot. 2017, 109, 106–116.
- Ravi, M.; Murugesan, B.; Jeyakumar, A.; Raparthi, K. A review on utilizing the marine biorefinery waste in construction raw materials to reduce land pollution and enhance green environment. Adv. Mater. Sci. Eng. 2021, 21, 43–62.
- Bădănoiu, A.; Moanță, A.; Dumitrescu, O.; Nicoară, A.; Trușcă, R. Waste Glass Valorization as Raw Material in the Production of Portland Clinker and Cement. Materials 2022, 15, 7403.
- Adela, Y.; Berhanu, M.; Gobena, B. Plastic wastes as a raw material in the concrete mix: An alternative approach to manage plastic wastes in developing countries. Int. J. Waste Resour. 2020, 10, 1–7.
- Schoon, J.; Van der Heyden, L.; Eloy, P.; Gaigneux, E.; De Buysser, K.; Van Driessche, I.; De Belie, N. Waste fibrecement: An interesting alternative raw material for a sustainable Portland clinker production. Constr. Build. Mater. 2012, 36, 391–403.
- Tsakiridis, P.; Oustadakis, P.; Agatzini-Leonardou, S. Black dross leached residue: An alternative raw material for Portland cement clinker. Waste Biomass Valorization 2014, 5, 973–983.
- Mariani, B.; Andrade, J.; Amorim, N.; Ribeiro, D. Effect of the incorporation of TiO2 waste (UOW) in the formation of the mineralogical phases of Portland clinker. Ambiente Construído 2019, 19, 57–71.
- Teker, E.; Andreas, L.; Cwirzen, A.; Habermehl-Cwirzen, K. Wood Ash as Sustainable Alternative Raw Material for the Production of Concrete—A Review. Materials 2023, 16, 2557.
- Carević, I.; Serdar, M.; Štirmer, N.; Ukrainczyk, N. Preliminary screening of wood biomass ashes for partial resources replacements in cementitious materials. J. Clean. Prod. 2019, 229, 1045–1064.
- Li, H.; Xu, W.; Yang, X.; Wu, J. Preparation of Portland cement with sugar filter mud as lime-based raw material. J. Clean. Prod. 2014, 66, 107–112.
- Li, H.; Xu, J.; Wu, J.; Xu, W.; Xu, Y. Influence of sugar filter mud on formation of Portland cement clinker. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2013, 28, 746–750.
- Her, S.; Park, J.; Li, P.; Bae, S. Feasibility study on utilization of pulverized eggshell waste as an alternative to limestone in raw materials for Portland cement clinker production. Constr. Build. Mater. 2022, 324, 126589.
- Her, S.; Park, T.; Zalnezhad, E.; Bae, S. Synthesis and characterization of cement clinker using recycled pulverized oyster and scallop shell as limestone substitutes. J. Clean. Prod. 2021, 278, 123987.
- Abdel-Latief, M.; El-Sayed, M.; Shahien, M.; Zayed, A. A new insight upon the use of weathered basalt as alternative raw material in Portland clinker production. Ain Shams Eng. J. 2021, 12, 885–896.
- Boughanmi, S.; Labidi, I.; Megriche, A.; El Maaoui, M.; Nonat, A. Natural fluorapatite as a raw material for Portland clinker. Cem. Concr. Res. 2018, 105, 72–80.
- Korkmaz, A.; Hacıfazlıoğlu, H. An Alternative Raw Material for Portland Cement Clinker Preparation: Meta-Schist. Preprint. 2023. Available online: https://www.researchsquare.com/article/rs-2578354/v1 (accessed on 18 October 2023).
- Kleib, J.; Amar, M.; Aouad, G.; Bourbon, X.; Benzerzour, M.; Abriak, N. The Use of Callovo-Oxfordian Argillite as a Raw Material for Portland Cement Clinker Production. Buildings 2022, 12, 1421.
- Malacarne, C.; Longhi, M.; Silva, M.; Gonçalves, J.; Rodríguez, E.; Kirchheim, A. Influence of low-grade materials as clinker substitute on the rheological behavior, hydration and mechanical performance of ternary cements. Case Stud. Constr. Mater. 2021, 15, 00776.
- Onanga, G.; Manuku, E.; Khalifa, R.; Lofongo, D.; Preat, A.; Nkula, V.; Osomba, D. Production of an Eco-Cement by Clinker Substitution by the Mixture of Calcined Clay and Limestone, Songololo (DR Congo). J. Geosci. Environ. Prot. 2023, 11, 67–80.
- Telesca, A.; Calabrese, D.; Marroccoli, M.; Tomasulo, M.; Valenti, G.; Duelli, G.; Montagnaro, F. Spent limestone sorbent from calcium looping cycle as a raw material for the cement industry. Fuel 2014, 118, 202–205.
- Kikuchi, R. Recycling of municipal solid waste for cement production: Pilot-scale test for transforming incineration ash of solid waste into cement clinker. Resour. Conserv. Recycl. 2001, 31, 137–147.
- Liang, X.; Dang, W.; Yang, G.; Zhang, Y. Environmental feasibility evaluation of cement co-production using classified domestic waste as alternative raw material and fuel: A life cycle perspective. J. Environ. Manag. 2023, 326, 116726.
- Krammart, P.; Tangtermsirikul, S. A study on cement made by partially replacing cement raw materials with municipal solid waste ash and calcium carbide waste. Sci. Asia 2003, 29, 77–84.
- Li, Y.; Wang, H.; Zhang, J.; Wang, J.; Miao, W. The industrial practice of POPs contaminated soil as alternative raw materials in clinker production. Procedia Environ. Sci. 2012, 16, 641–645.
- Eugenio, T.; Narciso, C.; Fagundes, J.; Henriques, A.; Mendes, R. Study on the use of mining waste as raw material for extruded fiber cement production. J. Build. Eng. 2023, 63, 105547.
- Scrivener, K.; Martirena, F.; Bishnoi, S.; Maity, S. Calcined clay limestone cements (LC3). Cem. Concr. Res. 2018, 114, 49–56.
- Fernandez, R.; Martirena, F.; Scrivener, K. The origin of the pozzolanic activity of calcined clay minerals: A comparison between kaolinite, illite and montmorillonite. Cem. Concr. Res. 2011, 41, 113–122.
- Ferreiro, S.; Herfort, D.; Damtoft, J. Effect of raw clay type, fineness, water-to-cement ratio and fly ash addition on workability and strength performance of calcined clay–limestone Portland cements. Cem. Concr. Res. 2017, 101, 1–12.
- Krishnan, S.; Emmanuel, A.; Shah, V.; Parashar, A.; Mishra, G.; Maity, S.; Bishnoi, S. Industrial production of limestone calcined clay cement: Experience and insights. Green Mater. 2019, 7, 15–27.
- Scrivener, K.; Avet, F.; Maraghechi, H.; Zunino, F.; Ston, J.; Hanpongpun, W.; Favier, A. Impacting factors and properties of limestone calcined clay cements (LC3). Green Mater. 2019, 7, 3–14.
- Wild, S.; Khatib, J.; Jones, A. Relative strength, pozzolanic activity and cement hydration in superplasticised metakaolin concrete. Cem. Concr. Res. 1996, 26, 1537–1544.
- Malhotra, V. Mineral Admixtures, Concrete Construction Engineering Handbook; CRC Press: Boca Raton, FL, USA, 1997; Chapter 2.
- Sabir, B.; Wild, S.; Bai, J. Metakaolin and calcined clays as pozzolans for concrete: A review. Cem. Concr. Compos. 2001, 23, 441–454.
- Siddique, R.; Klaus, J. Influence of metakaolin on the properties of mortar and concrete: A review. Appl. Clay Sci. 2009, 43, 392–400.
- Aramburo, C.; Pedrajas, C.; Rahhal, V.; González, M.; Talero, R. Calcined clays for low carbon cement: Rheological behaviour in fresh Portland cement pastes. Mater. Lett. 2019, 239, 24–28.
- Pedrajas, C.; Aramburo, C.; Talero, R. Sulphate durability of low carbon cements with high contents of calcined clay. Reasons for the resistant phenomenon. Mater. Today Proc. 2023.
- Li, X.; Dengler, J.; Hesse, C. Reducing clinker factor in limestone calcined clay-slag cement using CSH seeding–A way towards sustainable binder. Cem. Concr. Res. 2023, 168, 107151.
- Rathnarajan, S.; Dhanya, B.; Pillai, R.; Gettu, R.; Santhanam, M. Carbonation model for concretes with fly ash, slag, and limestone calcined clay-using accelerated and five-year natural exposure data. Cem. Concr. Compos. 2022, 126, 104329.
- Bhattacherjee, S.; Jain, S.; Santhanam, M. A method to increase the workability retention of concrete with limestone calcined clay based cementitious system using a dispersing agent containing sodium hexametaphosphate. Cem. Concr. Compos. 2022, 132, 104624.
- Khan, M.; Nguyen, Q.; Castel, A. Performance of limestone calcined clay blended cement-based concrete against carbonation. Adv. Cement Res. 2019, 32, 481–491.
- Zhu, H.; Chen, W.; Cheng, S.; Yang, L.; Wang, S.; Xiong, J. Low carbon and high efficiency limestone-calcined clay as supplementary cementitious materials (SCMs): Multi-indicator comparison with conventional SCMs. Constr. Build. Mater. 2022, 341, 127748.
- Dhandapani, Y.; Sakthivel, T.; Santhanam, M.; Gettu, R.; Pillai, R. Mechanical properties and durability performance of concretes with Limestone Calcined Clay Cement (LC3). Cem. Concr. Res. 2018, 107, 136–151.
- Vaasudevaa, B.; Dhandapani, Y.; Santhanam, M. Performance evaluation of limestone-calcined clay (LC2) combination as a cement substitute in concrete systems subjected to short-term heat curing. Constr. Build. Mater. 2021, 302, 124121.
- Aramburo, C.; Pedrajas, C.; Talero, R. Portland cements with high content of calcined clay: Mechanical strength behaviour and sulfate durability. Materials 2020, 13, 4206.
- Trusilewicz, L.; Fernández-Martínez, F.; Rahhal, V.; Talero, R. TEM and SAED characterization of metakaolin. Pozzolanic activity. J. Am. Ceram. Soc. 2012, 95, 2989–2996.
- UNE 80225:2012 Standard; Methods of Testing Cement. Chemical Analysis. Determination of Reactive SiO Content in Cements, Pozzolans and Fly Ash. AENOR: Madrid, Spain, 2012.
- Talero, R. New Wet Chemical Analysis Method to Determine the Reactive Alumina Content of Natural and Artificial Pozzolans; Private Document; Rafael Talero: Madrid, Spain, 2017.
- Arámburo, C. Sulfatic and Mechanical-Strength Behavior of Portland Cements with High Pozzolan Contents (>40%): Its Chemical-Physical Basis and Justification of Other Possible Consequences. Ph.D. Thesis, UPM, Madrid, Spain, 2019.
- Yu, J.; Wu, H.; Mishra, D.; Li, G.; Leung, C. Compressive strength and environmental impact of sustainable blended cement with high-dosage Limestone and Calcined Clay (LC2). J. Clean. Prod. 2021, 278, 123616.
- Raut, S.; Olcun, S.; Butler, L. Evaluating the use of limestone calcined clay cement and recycled concrete aggregates for reducing the carbon footprint of concrete structures. Mater. Today Proc. 2023.
- Pillai, R.; Gettu, R.; Santhanam, M.; Rengaraju, S.; Dhandapani, Y.; Rathnarajan, S.; Basavaraj, A. Service life and life cycle assessment of reinforced concrete systems with limestone calcined clay cement (LC3). Cem. Concr. Res. 2019, 118, 111–119.
- Zhang, D.; Jaworska, B.; Zhu, H.; Dahlquist, K.; Li, V. Engineered Cementitious Composites (ECC) with limestone calcined clay cement (LC3). Cem. Concr. Compos. 2020, 114, 103766.
- Guo, M.; Gong, G.; Yue, Y.; Xing, F.; Zhou, Y.; Hu, B. Performance evaluation of recycled aggregate concrete incorporating limestone calcined clay cement (LC3). J. Clean. Prod. 2022, 366, 132820.
- Huang, W.; Kazemi-Kamyab, H.; Sun, W.; Scrivener, K. Effect of replacement of silica fume with calcined clay on the hydration and microstructural development of eco-UHPFRC. Mater. Des. 2017, 121, 36–46.
- Joseph, S.; Dhandapani, Y.; Geddes, D.; Zhao, Z.; Bishnoi, S.; Vieira, M.; Riding, K. Mechanical properties of concrete made with calcined clay: A review by RILEM TC-282 CCL. Mater. Struct. 2023, 56, 84.
- Barbhuiya, S.; Nepal, J.; Das, B. Properties, compatibility, environmental benefits and future directions of limestone calcined clay cement (LC3) concrete: A review. J. Build. Eng. 2023, 79, 107794.
- Gettu, R.; Patel, A.; Rathi, V.; Prakasan, S.; Basavaraj, A.; Palaniappan, S.; Maity, S. Influence of supplementary cementitious materials on the sustainability parameters of cements and concretes in the Indian context. Mater. Struct. 2019, 52, 10.
- Hoenig, V.; Schall, A.; Sultanov, N.; Papkalla, S.; Ruppert, J. Status and Prospects of Alternative Raw Materials in the European Cement Sector; The European Cement Research Academy (ECRA): Düsseldorf, Germany, 2022.
- He, Z.; Zhu, X.; Wang, J.; Mu, M.; Wang, Y. Comparison of CO2 emissions from OPC and recycled cement production. Constr. Build. Mater. 2019, 211, 965–973.
- Thapa, V.; Waldmann, D.; Simon, C. Gravel wash mud, a quarry waste material as supplementary cementitious material (SCM). Cem. Concr. Res. 2019, 124, 105833.
- Aprianti, E.; Shafigh, P.; Bahri, S.; Farahani, J. Supplementary cementitious materials origin from agricultural wastes–A review. Constr. Build. Mater. 2015, 74, 176–187.
- Kumar, N.; Ram, K. Experimental study on properties of concrete containing crushed rock dust as a partial replacement of cement. Mater. Today Proc. 2018, 5, 7240–7246.
- Juenger, M.; Siddique, R. Recent advances in understanding the role of supplementary cementitious materials in concrete. Cem. Concr. Res. 2015, 78, 71–80.
- Manjunatha, M.; Seth, D.; Balaji, K.; Bharath, A. Engineering properties and environmental impact assessment of green concrete prepared with PVC waste powder: A step towards sustainable approach. Case Stud. Constr. Mater. 2022, 17, 01404.
- Alderete, N.; Joseph, A.; Van den Heede, P.; Matthys, S.; De Belie, N. Effective and sustainable use of municipal solid waste incineration bottom ash in concrete regarding strength and durability. Resour. Conserv. Recycl. 2021, 167, 105356.
- Panesar, D.; Zhang, R. Performance comparison of cement replacing materials in concrete: Limestone fillers and supplementary cementing materials—A review. Construct. Build. Mater. 2020, 251, 118866.
- Cordeiro, G.; Toledo Filho, R.; Tavares, L.; Fairbairn, E. Ultrafine grinding of sugar cane bagasse ash for application as pozzolanic admixture in concrete. Cem. Concr. Res. 2009, 39, 110–115.
- Nuntachai, C.; Chai, J.; Kraiwood, K. Utilization of bagasse ash as a pozzolanic material in concrete. Constr. Build. Mater. 2009, 23, 3352–3358.
- Gar, P.; Suresh, N.; Bindiganavile, V. Sugar cane bagasse ash as a pozzolanic admixture in concrete for resistance to sustained elevated temperatures. Constr. Build. Mater. 2017, 153, 929–936.
- Jha, P.; Sachan, A.; Singh, R. Agro-waste sugarcane bagasse ash (ScBA) as partial replacement of binder material in concrete. Mater. Today Proc. 2021, 44, 419–427.
- Shafiq, N.; Hussein, A.; Nuruddin, M.; Al Mattarneh, H. Effects of sugarcane bagasse ash on the properties of concrete. In Proceedings of the Institution of Civil Engineers—Engineering Eustainability; Thomas Telford Ltd.: London, UK, 2016; Volume 171, pp. 123–132.
- Bahurudeen, A.; Kanraj, D.; Dev, V.; Santhanam, M. Performance evaluation of sugarcane bagasse ash blended cement in concrete. Cem. Concr. Compos. 2015, 59, 77–88.
- Zaheer, M.; Tabish, M. The Durability of Concrete Made Up of Sugar Cane Bagasse Ash (SCBA) as a Partial Replacement of Cement: A Review. Arab. J. Sci. Eng. 2023, 48, 4195–4225.
- Prabhath, N.; Kumara, B.; Vithanage, V.; Samarathunga, A.; Sewwandi, N.; Maduwantha, K.; Madusanka, M.; Koswattage, K. A review on the optimization of the mechanical properties of sugarcane-bagasse-ash-integrated concretes. J. Compos. Sci. 2022, 6, 283.
- Chindaprasirt, P.; Kroehong, W.; Damrongwiriyanupap, N.; Suriyo, W.; Jaturapitakkul, C. Mechanical properties, chloride resistance and microstructure of Portland fly ash cement concrete containing high volume bagasse ash. J. Build. Eng. 2020, 31, 101415.
- Neto, J.; de França, M.; de Amorim Junior, N.; Ribeiro, D. Effects of adding sugarcane bagasse ash on the properties and durability of concrete. Constr. Build. Mater. 2021, 266, 120959.
- Zareei, S.; Ameri, F.; Bahrami, N. Microstructure, strength, and durability of eco-friendly concretes containing sugarcane bagasse ash. Constr. Build. Mater. 2018, 184, 258–268.
- Yogitha, B.; Karthikeyan, M.; Reddy, M. Progress of sugarcane bagasse ash applications in production of Eco-Friendly concrete-Review. Mater. Today Proc. 2020, 33, 695–699.
- Landa-Ruiz, L.; Landa-Gomez, A.; Mendoza-Rangel, J.; Landa-Sanchez, A.; Ariza-Figueroa, H.; Méndez-Ramírez, C.; Santiago-Hurtado, G.; Moreno-Landeros, V.; Croche, R.; Baltazar-Zamora, M. Physical, mechanical and durability properties of ecofriendly ternary concrete made with sugar cane bagasse ash and silica fume. Crystals 2021, 11, 1012.
- Wagh, M.; Waghe, U. Development of self-compacting concrete blended with sugarcane bagasse ash. Mater. Today Proc. 2022, 60, 1787–1792.
- Rajasekar, A.; Arunachalam, K.; Kottaisamy, M.; Saraswathy, V. Durability characteristics of Ultra High Strength Concrete with treated sugarcane bagasse ash. Constr. Build. Mater. 2018, 171, 350–356.
- Huang, P.; Huang, B.; Li, J.; Wu, N.; Xu, Q. Application of sugar cane bagasse ash as filler in ultra-high performance concrete. J. Build. Eng. 2023, 71, 106447.
- Rukzon, S.; Chindaprasirt, P. Utilization of bagasse ash in high-strength concrete. Mater. Des. 2012, 34, 45–50.
- Alex, J.; Dhanalakshmi, J.; Ambedkar, B. Experimental investigation on rice husk ash as cement replacement on concrete production. Constr. Build. Mater. 2016, 127, 353–362.
- Ozturk, E.; Ince, C.; Derogar, S.; Ball, R. Factors affecting the CO2 emissions, cost efficiency and eco-strength efficiency of concrete containing rice husk ash: A database study. Constr. Build. Mater. 2022, 326, 126905.
- Bheel, N.; Awoyera, P.; Shar, I.; Sohu, S.; Abbasi, S.; Krishna Prakash, A. Mechanical properties of concrete incorporating rice husk ash and wheat straw ash as ternary cementitious material. Adv. Civ. Eng. 2021, 2021, 2977428.
- Varadharajan, S.; Jaiswal, A.; Verma, S. Assessment of mechanical properties and environmental benefits of using rice husk ash and marble dust in concrete. Structures 2020, 28, 389–406.
- Khan, R.; Jabbar, A.; Ahmad, I.; Khan, W.; Khan, A.; Mirza, J. Reduction in environmental problems using rice-husk ash in concrete. Constr. Build. Mater. 2012, 30, 360–365.
- Memon, M.; Jhatial, A.; Murtaza, A.; Raza, M.; Phulpoto, K. Production of eco-friendly concrete incorporating rice husk ash and polypropylene fibres. Environ. Sci. Pollut. Res. 2021, 28, 39168–39184.
- Thomas, B. Green concrete partially comprised of rice husk ash as a supplementary cementitious material–A comprehensive review. Renew. Sustain. Energy Rev. 2018, 82, 3913–3923.
- Gursel, A.; Maryman, H.; Ostertag, C. A life-cycle approach to environmental, mechanical, and durability properties of “green” concrete mixes with rice husk ash. J. Clean. Prod. 2016, 112, 823–836.
- Jhatial, A.; Goh, W.; Mo, K.; Sohu, S.; Bhatti, I. Green and sustainable concrete—The potential utilization of rice husk ash and egg shells. Civ. Eng. J. 2019, 5, 74–81.
- Lim, J.; Raman, S.; Lai, F.; Zain, M.; Hamid, R. Synthesis of nano cementitious additives from agricultural wastes for the production of sustainable concrete. J. Clean. Prod. 2018, 171, 1150–1160.
- Nuruddin, M.; Chang, K.; Azmee, N. Workability and compressive strength of ductile self compacting concrete (DSCC) with various cement replacement materials. Constr. Build. Mater. 2014, 55, 153–157.
- Sathurshan, M.; Yapa, I.; Thamboo, J.; Jeyakaran, T.; Navaratnam, S.; Siddique, R.; Zhang, J. Untreated rice husk ash incorporated high strength self-compacting concrete: Properties and environmental impact assessments. Environ. Chall. 2021, 2, 100015.
- Raisi, E.; Amiri, J.; Davoodi, M. Influence of rice husk ash on the fracture characteristics and brittleness of self-compacting concrete. Eng. Fract. Mech. 2018, 199, 595–608.
- Huang, H.; Gao, X.; Wang, H.; Ye, H. Influence of rice husk ash on strength and permeability of ultra-high performance concrete. Constr. Build. Mater. 2017, 149, 621–628.
- Hakeem, I.; Agwa, I.; Tayeh, B.; Abd-Elrahman, M. Effect of using a combination of rice husk and olive waste ashes on high-strength concrete properties. Case Stud. Constr. Mater. 2022, 17, 01486.
- Subramaniam, D.; Sathiparan, N. Comparative study of fly ash and rice husk ash as cement replacement in pervious concrete: Mechanical characteristics and sustainability analysis. Int. J. Pavement Eng. 2022, 16, 1–8.
- Alnahhal, M.; Alengaram, U.; Jumaat, M.; Abutaha, F.; Alqedra, M.; Nayaka, R. Assessment on engineering properties and CO2 emissions of recycled aggregate concrete incorporating waste products as supplements to Portland cement. J. Clean. Prod. 2018, 203, 822–835.
- Alabi, S.; Mahachi, J. Mechanical properties of sustainable concrete made with ceramic and sandcrete block wastes. Mater. Today Proc. 2022, 62, 44–48.
- Tamanna, K.; Raman, S.; Jamil, M.; Hamid, R. Utilization of wood waste ash in construction technology: A review. Constr. Build. Mater. 2020, 237, 117654.
- Siddique, R. Utilization of wood ash in concrete manufacturing. Resour. Conserv. Recycl. 2012, 67, 27–33.
- Hamid, Z.; Rafiq, S. A comparative study on strength of concrete using wood ash as partial replacement of cement. IOP Conf. Ser. Mater. Sci. Eng. 2020, 955, 012043.
- Al-Kharabsheh, B.; Arbili, M.; Majdi, A.; Ahmad, J.; Deifalla, A.; Hakamy, A. A review on strength and durability properties of wooden ash based concrete. Materials 2022, 15, 7282.
- Hamid, Z.; Rafiq, S. An experimental study on behavior of wood ash in concrete as partial replacement of cement. Mater. Today Proc. 2021, 46, 3426–3429.
- Bhat, J. Mechanical behaviour of self compacting concrete: Effect of wood ash and coal ash as partial cement replacement. Mater. Today Proc. 2021, 42, 1470–1476.
- Sooraj, V. Effect of palm oil fuel ash (POFA) on strength properties of concrete. Int. J. Sci. Res. 2013, 3, 1–7.
- Panchal, J.N.; Challagulla, S.P.; Kishore, I.S. Influence of palm oil fuel ash on strength properties of concrete. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1197, 012082.
- Hamada, H.; Jokhio, G.; Yahaya, F.; Humada, A.; Gul, Y. The present state of the use of palm oil fuel ash (POFA) in concrete. Constr. Build. Mater. 2018, 175, 26–40.
- Jhatial, A.; Goh, W.; Mastoi, A.; Rahman, A.; Kamaruddin, S. Thermo-mechanical properties and sustainability analysis of newly developed eco-friendly structural foamed concrete by reusing palm oil fuel ash and eggshell powder as supplementary cementitious materials. Environ. Sci. Pollut. Res. 2021, 28, 38947–38968.
- Islam, M.; Mo, K.; Alengaram, U.; Jumaat, M. Mechanical and fresh properties of sustainable oil palm shell lightweight concrete incorporating palm oil fuel ash. J. Clean. Prod. 2016, 115, 307–314.
- Alnahhal, A.; Alengaram, U.; Yusoff, S.; Singh, R.; Radwan, M.; Deboucha, W. Synthesis of sustainable lightweight foamed concrete using palm oil fuel ash as a cement replacement material. J. Build. Eng. 2021, 35, 102047.
- Jhatial, A. Thermomechanical evaluation of sustainable foamed concrete incorporating palm oil fuel ash and eggshell powder. J. Eng. Res. 2021, 9, 64–79.
- Al-Mughanam, T.; Aldhyani, T.; Alsubari, B.; Al-Yaari, M. Modeling of compressive strength of sustainable self-compacting concrete incorporating treated palm oil fuel ash using artificial neural network. Sustainability 2020, 12, 9322.
- Ranjbar, N.; Behnia, A.; Alsubari, B.; Birgani, P.; Jumaat, M. Durability and mechanical properties of self-compacting concrete incorporating palm oil fuel ash. J. Clean. Prod. 2016, 112, 723–730.
- Alsubari, B.; Shafigh, P.; Jumaat, M. Utilization of high-volume treated palm oil fuel ash to produce sustainable self-compacting concrete. J. Clean. Prod. 2016, 137, 982–996.
- Alsubari, B.; Shafigh, P.; Ibrahim, Z.; Alnahhal, M.; Jumaat, M. Properties of eco-friendly self-compacting concrete containing modified treated palm oil fuel ash. Constr. Build. Mater. 2018, 158, 742–754.
- Alsubari, B.; Shafigh, P.; Jumaat, M. Development of self-consolidating high strength concrete incorporating treated palm oil fuel ash. Materials 2015, 8, 2154–2173.
- Alsubari, B.; Shafigh, P.; Jumaat, M.; Alengaram, U. Palm oil fuel ash as a partial cement replacement for producing durable self-consolidating high-strength concrete. Arab. J. Sci. Eng. 2014, 39, 8507–8516.
- Shafigh, P.; Mahmud, H.; Jumaat, M.; Ahmmad, R.; Bahri, S. Structural lightweight aggregate concrete using two types of waste from the palm oil industry as aggregate. J. Clean. Prod. 2014, 80, 187–196.
- Ahmmad, R.; Alengaram, U.; Jumaat, M.; Sulong, N.; Yusuf, M.; Rehman, M. Feasibility study on the use of high-volume palm oil clinker waste in environmental friendly lightweight concrete. Constr. Build. Mater. 2017, 135, 94–103.
- Hamada, H.; Al-Attar, A.; Tayeh, B.; Yahaya, F. Optimizing the concrete strength of lightweight concrete containing nano palm oil fuel ash and palm oil clinker using response surface method. Case Stud. Constr. Mater. 2022, 16, 01061.
- Jhatial, A.; Sohu, S.; Memon, M.; Bhatti, N.; Memon, D. Eggshell powder as partial cement replacement and its effect on the workability and compressive strength of concrete. Int. J. Adv. Appl. Sci. 2019, 6, 71–75.
- Mahmood, L.; Rafiq, S.; Mohammed, A. A review study of eggshell powder as cement replacement in concrete. Sulaimania J. Eng. Sci. 2022, 9, 25–37.
- Hamada, H.; Tayeh, B.; Al-Attar, A.; Yahaya, F.; Muthusamy, K.; Humada, A. The present state of the use of eggshell powder in concrete: A review. J. Build. Eng. 2020, 32, 101583.
- Sohu, S.; Bheel, N.; Jhatial, A.; Ansari, A.; Shar, I. Sustainability and mechanical property assessment of concrete incorporating eggshell powder and silica fume as binary and ternary cementitious materials. Environ. Sci. Pollut. Res. 2022, 29, 58685–58697.
- Arif, S.; Rokiah, O.; Khairunisa, M.; Chong, B.; Chek, Y.; Youventharan, D.; Ramadhansyah, P.; Doh, S. Compressive Strength of Concrete containing Eggshell Powder as Partial Cement Replacement. IOP Conf. Ser. Earth Environ. Sci. 2021, 682, 012031.
- Tayeh, B.; Hadzima-Nyarko, M.; Zeyad, A.; Al-Harazin, S. Properties and durability of concrete with olive waste ash as a partial cement replacement. Adv. Concr. Constr. 2021, 11, 59–71.
- Ikponmwosa, E.; Falade, F.; Fashanu, T.; Ehikhuenmen, S.; Adesina, A. Experimental and numerical investigation of the effect of sawdust ash on the performance of concrete. J. Build. Pathol. Rehabil. 2020, 5, 15.
- Raheem, A.; Olasunkanmi, B.; Folorunso, C. Saw dust ash as partial replacement for cement in concrete. Organ. Technol. Manag. Constr. 2012, 4, 474–480.
- Elinwa, A.; Ejeh, S.; Mamuda, A. Assessing of the fresh concrete properties of self-compacting concrete containing sawdust ash. Constr. Build. Mater. 2008, 22, 1178–1182.
- Ayuba, S.; Uche, O.; Haruna, S.; Mohammed, A. Durability properties of cement–saw dust ash (SDA) blended self compacting concrete (SCC). Niger. J. Technol. 2022, 41, 212–221.
- Nagarajan, V.; Devi, S.; Manohari, S.; Santha, M. Experimental study on partial replacement of cement with coconut shell ash in concrete. Int. J. Sci. Res. 2014, 3, 651–661.
- Berenguer, R.; Capraro, A.; de Medeiros, M.; Carneiro, A.; De Oliveira, R. Sugar cane bagasse ash as a partial substitute of Portland cement: Effect on mechanical properties and emission of carbon dioxide. J. Environ. Chem. Eng. 2020, 8, 103655.
- Hamada, H.; Tayeh, B.; Yahaya, F.; Muthusamy, K.; Al-Attar, A. Effects of nano-palm oil fuel ash and nano-eggshell powder on concrete. Constr. Build. Mater. 2020, 261, 119790.
- Hamada, H.; Alattar, A.; Yahaya, F.; Muthusamy, K.; Tayeh, B. Mechanical properties of semi-lightweight concrete containing nano-palm oil clinker powder. Phys. Chem. Earth Parts A/B/C 2021, 121, 102977.
- Amin, M.; Attia, M.; Agwa, I.; Elsakhawy, Y.; Abu El-hassan, K.; Abdelsalam, B. Effects of sugarcane bagasse ash and nano eggshell powder on high-strength concrete properties. Case Stud. Constr. Mater. 2022, 17, 01528.
- Hamada, H.; Abed, F.; Tayeh, B.; Al Jawahery, M.; Majdi, A.; Yousif, S. Effect of recycled seashells on concrete properties: A comprehensive review of the recent studies. Constr. Build. Mater. 2023, 376, 131036.
- Bamigboye, G.; Nworgu, A.; Odetoyan, A.; Kareem, M.; Enabulele, D.; Bassey, D. Sustainable use of seashells as binder in concrete production: Prospect and challenges. J. Build. Eng. 2021, 34, 101864.
- Bamigboye, G.; Enabulele, D.; Odetoyan, A.; Kareem, M.; Nworgu, A.; Bassey, D. Mechanical and durability assessment of concrete containing seashells: A review. Cogent Eng. 2021, 8, 1883830.
- Tayeh, B.; Hasaniyah, M.; Zeyad, A.; Yusuf, M. Properties of concrete containing recycled seashells as cement partial replacement: A review. J. Clean. Prod. 2019, 237, 117723.
- Shetty, P.; Rao, A.; Pai, B.; Kamath, M. Performance of high-strength concrete with the effects of seashell powder as binder replacement and waste glass powder as fine aggregate. J. Compos. Sci. 2023, 7, 92.
- Ruslan, H.; Muthusamy, K.; Mohsin, S.; Jose, R.; Omar, R. Oyster shell waste as a concrete ingredient: A review. Mater. Today Proc. 2022, 48, 713–719.
- Liu, S.; Zhang, Y.; Liu, B.; Zou, Z.; Liu, Q.; Teng, Y.; Zhang, L. Sustainable Use of Waste Oyster Shell Powders in a Ternary Supplementary Cementitious Material System for Green Concrete. Materials 2022, 15, 4886.
- Ruslan, H.; Muthusamy, K.; Mohsin, S.; Kirgiz, M. Periwinkle Shell as Mixing Ingredient in Concrete: A Review. Construction 2021, 1, 21–30.
- El Mendili, Y.; Benzaama, M. Investigation of Mechanical and Thermal Performance of Concrete with Scallop Shells as Partial Cement Replacement: Alternative Binder and Life Cycle Assessment. CivilEng 2022, 3, 760–778.
- Pavesi, T.; Rohden, A.; Garcez, M. Supporting circular economy through the use of red ceramic waste as supplementary cementitious material in structural concrete. J. Mater. Cycles Waste Manag. 2021, 23, 2278–2296.
- El-Dieb, A.; Kanaan, D. Ceramic waste powder an alternative cement replacement–Characterization and evaluation. Sustain. Mater. Technol. 2018, 17, 00063.
- Subaşı, S.; Öztürk, H.; Emiroğlu, M. Utilizing of waste ceramic powders as filler material in self-consolidating concrete. Constr. Build. Mater. 2017, 149, 567–574.
- Xu, K.; Huang, W.; Zhang, L.; Fu, S.; Chen, M.; Ding, S.; Han, B. Mechanical properties of low-carbon ultrahigh-performance concrete with ceramic tile waste powder. Constr. Build. Mater. 2021, 287, 123036.
- Kannan, D.; Aboubakr, S.; El-Dieb, A.; Taha, M. High performance concrete incorporating ceramic waste powder as large partial replacement of Portland cement. Constr. Build. Mater. 2017, 144, 35–41.
- Du, H.; Tan, K. Properties of high volume glass powder concrete. Cem. Concr. Compos. 2017, 75, 22–29.
- Shi, C.; Zheng, K. A review on the use of waste glasses in the production of cement and concrete. Resour. Conserv. Recycl. 2007, 52, 234–247.
- Elaqra, H.; Abou Haloub, M.; Rustom, R. Effect of new mixing method of glass powder as cement replacement on mechanical behavior of concrete. Constr. Build. Mater. 2019, 203, 75–82.
- Rahman, S.; Uddin, M. Experimental investigation of concrete with glass powder as partial replacement of cement. Civ. Eng. Archit. 2018, 6, 149–154.
- Raydan, R.; Khatib, J.; Jahami, A.; El Hamoui, A.; Chamseddine, F. Prediction of the mechanical strength of concrete containing glass powder as partial cement replacement material. Innov. Infrastruct. Solut. 2022, 7, 311.
- Deschamps, J.; Simon, B.; Tagnit-Hamou, A.; Amor, B. Is open-loop recycling the lowest preference in a circular economy? Answering through LCA of glass powder in concrete. J. Clean. Prod. 2018, 185, 14–22.
- Kalakada, Z.; Doh, J.; Chowdhury, S. Glass powder as replacement of cement for concrete–an investigative study. Eur. J. Environ. Civ. 2022, 26, 1046–1063.
- Hilton, B.; Bawden, K.; Winnebeck, K.; Chandrasiri, C.; Ariyachandra, E.; Peethamparan, S. The functional and environmental performance of mixed cathode ray tubes and recycled glass as partial replacement for cement in concrete. Resour. Conserv. Recycl. 2019, 151, 104451.
- Khan, F.; Shahzada, K.; Ullah, Q.; Fahim, M.; Khan, S.; Badrashi, Y. Development of environment-friendly concrete through partial addition of waste glass powder (WGP) as cement replacement. Civ. Eng. J. 2020, 6, 2332–2343.
- Yu, J.; Lu, C.; Leung, C.; Li, G. Mechanical properties of green structural concrete with ultrahigh-volume fly ash. Constr. Build. Mater. 2017, 147, 510–518.
- younis Khudair, A.; Mohammed, M.K.; Hama, S.M. Optimization of glass powder content in self-compacting concrete as partial replacement of cement. IOP Conf. Ser. Mater. Sci. Eng. 2020, 928, 022140.
- da Silva, S.; de Oliveira Andrade, J. Investigation of mechanical properties and carbonation of concretes with construction and demolition waste and fly ash. Constr. Build. Mater. 2017, 153, 704–715.
- Dandautiya, R.; Singh, A. Utilization potential of fly ash and copper tailings in concrete as partial replacement of cement along with life cycle assessment. Waste Manag. 2019, 99, 90–101.
- Celik, K.; Meral, C.; Gursel, A.; Mehta, P.; Horvath, A.; Monteiro, P. Mechanical properties, durability, and life-cycle assessment of self-consolidating concrete mixtures made with blended Portland cements containing fly ash and limestone powder. Cem. Concr. Compos. 2015, 56, 59–72.
- Nath, P.; Sarker, P.; Biswas, W. Effect of fly ash on the service life, carbon footprint and embodied energy of high strength concrete in the marine environment. Energy Build 2018, 158, 1694–1702.
- Kumar, R.; Shafiq, N.; Kumar, A.; Jhatial, A. Investigating embodied carbon, mechanical properties, and durability of high-performance concrete using ternary and quaternary blends of metakaolin, nano-silica, and fly ash. Environ. Sci. Pollut. Res. 2021, 28, 49074–49088.
- Samad, S.; Shah, A.; Limbachiya, M. Strength development characteristics of concrete produced with blended cement using ground granulated blast furnace slag (GGBS) under various curing conditions. Sādhanā 2017, 42, 1203–1213.
- Habibi, A.; Ramezanianpour, A.; Mahdikhani, M. RSM-based optimized mix design of recycled aggregate concrete containing supplementary cementitious materials based on waste generation and global warming potential. Resour. Conserv. Recycl. 2021, 167, 105420.
- Jalil, A.; Khitab, A.; Ishtiaq, H.; Bukhari, S.; Arshad, M.; Anwar, W. Evaluation of steel industrial slag as partial replacement of cement in concrete. Civ. Eng. J. 2019, 5, 181–190.
- Kim, Y.; Hanif, A.; Usman, M.; Munir, M.; Kazmi, S.; Kim, S. Slag waste incorporation in high early strength concrete as cement replacement: Environmental impact and influence on hydration & durability attributes. J. Clean. Prod. 2018, 172, 3056–3065.
- Santos, H.; Jochem, L.; de Matos, P.; Casagrande, C.; Marinho, É.; Szeląg, M.; de Nóbrega, A. Porcelain Tile Polishing Residue in Concrete as an Additive or Replacement for Portland Cement. Appl. Sci. 2023, 13, 2824.
- de Matos, P.; Prudêncio, L., Jr.; de Oliveira, A.; Pelisser, F.; Gleize, P. Use of porcelain polishing residue as a supplementary cimentitious material in self-compacting concrete. Constr. Build. Mater. 2018, 193, 623–630.
- Alizadeh, M.; Momeni, M. The effect of the scrap/DRI ratio on the specification of the EAF dust and its influence on mechanical properties of the concrete treated by its dust. Constr. Build. Mater. 2016, 112, 1041–1045.
- Chavan, S.; Salokhe, S.; Nadagauda, P.; Patil, S.; Mane, K. An investigational study on properties of concrete produced with industrial waste red mud. Mater. Today Proc. 2021, 42, 733–738.
- Qureshi, H.; Ahmad, J.; Majdi, A.; Saleem, M.; Al Fuhaid, A.; Arifuzzaman, M. A Study on Sustainable Concrete with Partial Substitution of Cement with Red Mud: A Review. Materials 2022, 15, 7761.
- Nakic, D. Environmental evaluation of concrete with sewage sludge ash based on LCA. Sustain. Prod. Consum. 2018, 16, 193–201.
- Aliabdo, A.; Abd Elmoaty, M.; Auda, E. Re-use of waste marble dust in the production of cement and concrete. Constr. Build. Mater. 2014, 50, 28–41.
- Varadharajan, S. Determination of mechanical properties and environmental impact due to inclusion of flyash and marble waste powder in concrete. Structures 2020, 25, 613–630.
- Evram, A.; Akçaoğlu, T.; Ramyar, K.; Çubukçuoğlu, B. Effects of waste electronic plastic and marble dust on hardened properties of high strength concrete. Constr. Build. Mater. 2020, 263, 120928.
- Praveenkumar, T.; Vijayalakshmi, M.; Meddah, M. Strengths and durability performances of blended cement concrete with TiO2 nanoparticles and rice husk ash. Constr. Build. Mater. 2019, 217, 343–351.
- Bheel, N.; Khoso, S.; Baloch, M.; Benjeddou, O.; Alwetaishi, M. Use of waste recycling coal bottom ash and sugarcane bagasse ash as cement and sand replacement material to produce sustainable concrete. Environ. Sci. Pollut. Res. 2022, 29, 52399–52411.
- de Pedro, J.; Lagao, J.; Ongpeng, J. Life Cycle Assessment of Concrete Using Copper Slag as a Partial Cement Substitute in Reinforced Concrete Buildings. Buildings 2023, 13, 746.
- Makul, N.; Sua-Iam, G. Innovative utilization of foundry sand waste obtained from the manufacture of automobile engine parts as a cement replacement material in concrete production. J. Clean. Prod. 2018, 199, 305–320.
- Makul, N.; Sokrai, P. Influences of fine waste foundry sand from the automobile engine-part casting process and water-cementitious ratio on the properties of concrete: A new approach to use of a partial cement replacement material. J. Build. Eng. 2018, 20, 544–558.
- Huang, W.; Kazemi-Kamyab, H.; Sun, W.; Scrivener, K. Effect of cement substitution by limestone on the hydration and microstructural development of ultra-high performance concrete (UHPC). Cem. Concr. Compos. 2017, 77, 86–101.
- Meddah, M. Durability performance and engineering properties of shale and volcanic ashes concretes. Constr. Build. Mater. 2015, 79, 73–82.
- Vaičienė, M.; Simanavičius, E. The effect of municipal solid waste incineration ash on the properties and durability of cement concrete. Materials 2022, 15, 4486.
- Campos, J.; Fajilan, S.; Lualhati, J.; Mandap, N.; Clemente, S. Life cycle assessment of biochar as a partial replacement to Portland cement. IOP Conf. Ser. Earth Environ. Sci. 2020, 479, 012025.
- Abolhasani, A.; Samali, B.; Aslani, F. Rice husk ash incorporation in calcium aluminate cement concrete: Life cycle assessment, hydration and strength development. Sustainability 2022, 14, 1012.
- Ibeto, C.; Obiefuna, C.; Ugwu, K. Environmental effects of concretes produced from partial replacement of cement and sand with coal ash. Int. J. Environ. Sci. Technol. 2020, 17, 2967–2976.
- Flower, D.; Sanjayan, J. Green house gas emissions due to concrete manufacture. Int. J. Life Cycle Assess. 2007, 12, 282–288.
- Fallah-Valukolaee, S.; Mousavi, R.; Arjomandi, A.; Nematzadeh, M.; Kazemi, M. A comparative study of mechanical properties and life cycle assessment of high-strength concrete containing silica fume and nanosilica as a partial cement replacement. Structures 2022, 46, 838–851.
- Tayeh, B.; AlSaffar, D.; Askar, L.; Jubeh, A. Effect of incorporating pottery and bottom ash as partial replacement of cement. Karbala Int. J. Mod. Sci. 2019, 5, 9.
- Basit, A.; Khan, M.A.; Ahmed, I.; Khan, M.N.; Umar, M. Effect of brick dust and lime powder on the performance of plain cement concrete. IOP Conf. Ser. Mater. Sci. Eng. 2018, 414, 012008.
- Teixeira, E.; Mateus, R.; Camoes, A.; Bragança, L.; Branco, F. Comparative environmental life-cycle analysis of concretes using biomass and coal fly ashes as partial cement replacement material. J. Clean. Prod. 2016, 112, 2221–2230.
- Maglad, A.; Amin, M.; Zeyad, A.; Tayeh, B.; Agwa, I. Engineering properties of ultra-high strength concrete containing sugarcane bagasse and corn stalk ashes. J. Mater. Res. Technol. 2023, 23, 3196–3218.
- Bheel, N.; Adesina, A. Influence of binary blend of corn cob ash and glass powder as partial replacement of cement in concrete. Silicon 2021, 13, 1647–1654.
- Perez, O.; Florez, D.; Vergara, L.; Benavides, K. Innovative use of agro-waste cane bagasse ash and waste glass as cement replacement for green concrete. Cost analysis and carbon dioxide emissions. J. Clean. Prod. 2022, 379, 134822.
- Cordoba, G.; Barquero, M.; Bonavetti, V.; Irassar, E. Sustainability of concretes with binary and ternary blended cements considering performance parameters. Cement 2023, 13, 100077.
- Soliman, N.; Tagnit-Hamou, A. Development of ultra-high-performance concrete using glass powder–Towards ecofriendly concrete. Constr. Build. Mater. 2016, 125, 600–612.
- Rajendran, R.; Sathishkumar, A.; Perumal, K.; Pannirselvam, N.; Lingeshwaran, N.; Madavarapu, S. An experiment on concrete replacing binding material as waste glass powder. Mater. Today Proc. 2021, 47, 5447–5450.
- Soltanzadeh, F.; Emam-Jomeh, M.; Edalat-Behbahani, A.; Soltan-Zadeh, Z. Development and characterization of blended cements containing seashell powder. Constr. Build. Mater. 2018, 161, 292–304.
- Qin, Y.; Pang, X.; Tan, K.; Bao, T. Evaluation of pervious concrete performance with pulverized biochar as cement replacement. Cem. Concr. Compos. 2021, 119, 104022.
- Gálvez-Martos, J.; Styles, D.; Schoenberger, H.; Zeschmar-Lahl, B. Construction and demolition waste best management practice in Europe. Resour. Conserv. Recycl. 2018, 136, 166–178.
- Neto, R.; Gastineau, P.; Cazacliu, B.; Le Guen, L.; Paranhos, R.; Petter, C. An economic analysis of the processing technologies in CDW recycling platforms. Waste Manag. 2017, 60, 277–289.
- Xiao, J.; Ma, Z.; Sui, T.; Akbarnezhad, A.; Duan, Z. Mechanical properties of concrete mixed with recycled powder produced from construction and demolition waste. J. Clean. Prod. 2018, 188, 720–731.
- Yuan, F.; Shen, L.; Li, Q. Emergy analysis of the recycling options for construction and demolition waste. Waste Manag. 2011, 31, 2503–2511.
- Zhang, H.; Zhang, C.; He, B.; Yi, S.; Tang, L. Recycling fine powder collected from construction and demolition wastes as partial alternatives to cement: A comprehensive analysis on effects, mechanism, cost and CO2 emission. J. Build. Eng. 2023, 71, 106507.
- Zito, S.; Irassar, E.; Rahhal, V. Recycled Construction and Demolition Waste as Supplementary Cementing Materials in Eco-Friendly Concrete. Recycling 2023, 8, 54.
- Li, L.; Liu, Q.; Huang, T.; Peng, W. Mineralization and utilization of CO2 in construction and demolition wastes recycling for building materials: A systematic review of recycled concrete aggregate and recycled hardened cement powder. Sep. Purif. Technol. 2022, 298, 121512.
- Kim, J.; Jang, H. Closed-loop recycling of C&D waste: Mechanical properties of concrete with the repeatedly recycled C&D powder as partial cement replacement. J. Clean. Prod. 2022, 343, 130977.
- Wu, H.; Xu, J.; Yang, D.; Ma, Z. Utilizing thermal activation treatment to improve the properties of waste cementitious powder and its newmade cementitious materials. J. Clean. Prod. 2021, 322, 129074.
- Alghamdi, H. A review of cementitious alternatives within the development of environmental sustainability associated with cement replacement. Environ. Sci. Pollut. Res. 2022, 29, 42433–42451.
- Villanueva, A.; Delgado, L.; Luo, Z.; Eder, P.; Catarino, A.; Litten, D. Study on the Selection of Waste Streams for End-of-Waste Assessment; JRC Scientific and Technical Reports; European Commission: Seville, Spain, 2010; Volume 24362.
- Rocha, J.; Toledo Filho, R. The utilization of recycled concrete powder as supplementary cementitious material in cement-based materials: A systematic literature review. J. Build. Eng. 2023, 76, 107319.
- Singh, A.; Miao, X.; Zhou, X.; Deng, Q.; Li, J.; Zou, S.; Duan, Z. Use of recycled fine aggregates and recycled powders in sustainable recycled concrete. J. Build. Eng. 2023, 77, 107370.
- Gebremariam, A.; Vahidi, A.; Di Maio, F.; Moreno-Juez, J.; Vegas-Ramiro, I.; Łagosz, A.; Mróz, R.; Rem, P. Comprehensive study on the most sustainable concrete design made of recycled concrete, glass and mineral wool from C&D wastes. Constr. Build. Mater. 2021, 273, 121697.
- Sun, Z.; Liu, F.; Tong, T.; Qi, C.; Yu, Q. Hydration of concrete containing hybrid recycled demolition powders. J. Mater. Civ. Eng. 2017, 29, 04017037.
- Duan, Z.; Hou, S.; Xiao, J.; Li, B. Study on the essential properties of recycled powders from construction and demolition waste. J. Clean. Prod. 2020, 253, 119865.
- Mucsi, G.; Halyag Papné, N.; Ulsen, C.; Figueiredo, P.; Kristály, F. Mechanical activation of construction and demolition waste in order to improve its pozzolanic reactivity. ACS Sustain. Chem. Eng. 2021, 9, 3416–3427.
- Tang, Q.; Ma, Z.; Wu, H.; Wang, W. The utilization of eco-friendly recycled powder from concrete and brick waste in new concrete: A critical review. Cem. Concr. Compos. 2020, 114, 103807.
- Mehdizadeh, H.; Ling, T.; Cheng, X.; Pan, S.; Hung Mo, K. CO2 Treatment of Hydrated Cement Powder: Characterization and Application Consideration. J. Mater. Civ. Eng. 2021, 33, 04021041.
- Zajac, M.; Skocek, J.; Durdzinski, P.; Bullerjahn, F.; Skibsted, J.; Haha, M. Effect of carbonated cement paste on composite cement hydration and performance. Cem. Concr. Res. 2020, 134, 106090.
- Qin, L.; Gao, X. Recycling of waste autoclaved aerated concrete powder in Portland cement by accelerated carbonation. Waste Manag. 2019, 89, 254–264.
- Lu, B.; Shi, C.; Zhang, J.; Wang, J. Effects of carbonated hardened cement paste powder on hydration and microstructure of Portland cement. Constr. Build. Mater. 2018, 186, 699–708.
- Bogas, J.; Carriço, A.; Pereira, M. Mechanical characterization of thermal activated low-carbon recycled cement mortars. J. Clean. Prod. 2019, 218, 377–389.
- Ma, Z.; Shen, J.; Wu, H.; Zhang, P. Properties and activation modification of eco-friendly cementitious materials incorporating high-volume hydrated cement powder from construction waste. Constr. Build. Mater. 2022, 316, 125788.
- Ma, Z.; Yao, P.; Yang, D.; Shen, J. Effects of fire-damaged concrete waste on the properties of its preparing recycled aggregate, recycled powder and newmade concrete. J. Mater. Res. Technol. 2021, 15, 1030–1045.
- Wang, L.; Wang, J.; Wang, H.; Fang, Y.; Shen, W.; Chen, P.; Xu, Y. Eco-friendly treatment of recycled concrete fines as supplementary cementitious materials. Constr. Build. Mater. 2022, 322, 126491.
- Zhang, D.; Zhang, S.; Huang, B.; Yang, Q.; Li, J. Comparison of mechanical, chemical, and thermal activation methods on the utilisation of recycled concrete powder from construction and demolition waste. J. Build. Eng. 2022, 61, 105295.
- Qian, D.; Yu, R.; Shui, Z.; Sun, Y.; Jiang, C.; Zhou, F.; Ding, M.; Tong, X.; He, Y. A novel development of green ultra-high performance concrete (UHPC) based on appropriate application of recycled cementitious material. J. Clean. Prod. 2020, 261, 121231.
- Cantero, B.; Bravo, M.; De Brito, J.; del Bosque, I.; Medina, C. Mechanical behaviour of structural concrete with ground recycled concrete cement and mixed recycled aggregate. J. Clean. Prod. 2020, 275, 122913.
- He, Z.; Zhu, H.; Zhang, M.; Shi, J.; Du, S.; Liu, B. Autogenous shrinkage and nano-mechanical properties of UHPC containing waste brick powder derived from construction and demolition waste. Constr. Build. Mater. 2021, 306, 124869.
- Arif, R.; Khitab, A.; Kırgız, M.; Khan, R.; Tayyab, S.; Khan, R.; Anwar, W.; Arshad, M. Experimental analysis on partial replacement of cement with brick powder in concrete. Case Stud. Constr. Mater. 2021, 15, 00749.
- He, Z.; Han, X.; Zhang, M.; Yuan, Q.; Shi, J.; Zhan, P. A novel development of green UHPC containing waste concrete powder derived from construction and demolition waste. Powder Technol. 2022, 398, 117075.
- Wu, Y.; Liu, C.; Liu, H.; Hu, H.; He, C.; Song, L.; Huang, W. Pore structure and durability of green concrete containing recycled powder and recycled coarse aggregate. J. Build. Eng. 2022, 53, 104584.
- He, X.; Ma, Q.; Su, Y.; Zheng, Z.; Tan, H.; Peng, K.; Zhao, R. Humid hardened concrete waste treated by multiple wet-grinding and its reuse in concrete. Constr. Build. Mater. 2022, 350, 128485.
- Cantero, B.; del Bosque, I.; de Rojas, M.; Matías, A.; Medina, C. Durability of concretes bearing construction and demolition waste as cement and coarse aggregate substitutes. Cem. Concr. Compos. 2022, 134, 104722.
- Pešta, J.; Ženíšek, M.; Kočí, V.; Pavlů, T. Environmental perspectives of recycled concrete powder as cement replacement. AIP Conf. Proc. 2021, 2322, 020028.
- Likes, L.; Markandeya, A.; Haider, M.; Bollinger, D.; McCloy, J.; Nassiri, S. Recycled concrete and brick powders as supplements to Portland cement for more sustainable concrete. J. Clean. Prod. 2022, 364, 132651.
- Ma, Z.; Liu, M.; Duan, Z.; Liang, C.; Wu, H. Effects of active waste powder obtained from C&D waste on the micro properties and water permeability of concrete. J. Clean. Prod. 2020, 257, 120518.
- Zhu, P.; Mao, X.; Qu, W.; Li, Z.; Ma, Z. Investigation of using recycled powder from waste of clay bricks and cement solids in reactive powder concrete. Constr. Build. Mater. 2016, 113, 246–254.
- Kim, Y. Quality properties of self-consolidating concrete mixed with waste concrete powder. Constr. Build. Mater. 2017, 135, 177–185.
- Ma, Z.; Li, W.; Wu, H.; Cao, C. Chloride permeability of concrete mixed with activity recycled powder obtained from C&D waste. Constr. Build. Mater. 2019, 199, 652–663.
- Heidari, A.; Hasanpour, B. Effects of waste bricks powder of Gachsaran company as a pozzolanic material in concrete. Asian J. Civ. Eng. 2013, 14, 755–763.
- Maslehuddin, M.; Al-Amoudi, O.; Rahman, M.; Ali, M.; Barry, M. Properties of cement kiln dust concrete. Constr. Build. Mater. 2009, 23, 2357–2361.
- Abd El-Mohsen, M.; Anwar, A.; Adam, I. Mechanical properties of self-consolidating concrete incorporating cement kiln dust. HBRC J. 2015, 11, 1–6.
- Cantero, B.; Sáez del Bosque, I.; Matías, A.; Sánchez de Rojas, M.; Medina, C. Inclusion of construction and demolition waste as a coarse aggregate and a cement addition in structural concrete design. Arch. Civ. Mech. Eng. 2019, 19, 1338–1352.
- Najim, K.; Al-Jumaily, I.; Atea, A. Characterization of sustainable high performance/self-compacting concrete produced using CKD as a cement replacement material. Constr. Build. Mater. 2016, 103, 123–129.
- Al-Rezaiqi, J.; Alnuaimi, A.; Hago, A. Efficiency factors of burnt clay and cement kiln dust and their effects on properties of blended concrete. Appl. Clay Sci. 2018, 157, 51–64.
- Siddique, R.; Rajor, A. Influence of bacterial treated cement kiln dust on the properties of concrete. Constr. Build. Mater. 2014, 52, 42–51.
- Ge, Z.; Gao, Z.; Sun, R.; Zheng, L. Mix design of concrete with recycled clay-brick-powder using the orthogonal design method. Constr. Build. Mater. 2012, 31, 289–293.
- Xue, C.; Shen, A.; Guo, Y.; He, T. Utilization of construction waste composite powder materials as cementitious materials in small-scale prefabricated concrete. Adv. Mater. Sci. Eng. 2016, 2016, 8947935.
- Cantero, B.; Bravo, M.; de Brito, J.; Sáez del Bosque, I.; Medina, C. Thermal performance of concrete with recycled concrete powder as partial cement replacement and recycled CDW aggregate. Appl. Sci. 2020, 10, 4540.
- Bagheri, S.; Koushkbaghi, M.; Mohseni, E.; Koushkbaghi, S.; Tahmouresi, B. Evaluation of environment and economy viable recycling cement kiln dust for use in green concrete. J. Build. Eng. 2020, 32, 101809.
- Juan-Valdés, A.; Rodriguez-Robles, D.; Garcia-Gonzalez, J.; de Rojas Gómez, M.; Guerra-Romero, M.; De Belie, N.; Morán-del Pozo, J. Mechanical and microstructural properties of recycled concretes mixed with ceramic recycled cement and secondary recycled aggregates. A viable option for future concrete. Constr. Build. Mater. 2021, 270, 121455.
- Khodabakhshian, A.; de Brito, J.; Ghalehnovi, M.; Asadi Shamsabadi, E. Mechanical, environmental and economic performance of structural concrete containing silica fume and marble industry waste powder. Construct. Build. Mater. 2018, 169, 237–251.
More
