Biomarkers are vital in healthcare as they provide valuable insights into disease diagnosis, prognosis, treatment response, and personalized medicine. They serve as objective indicators, enabling early detection and intervention, leading to improved patient outcomes and reduced costs. Biomarkers also guide treatment decisions by predicting disease outcomes and facilitating individualized treatment plans. They play a role in monitoring disease progression, adjusting treatments, and detecting early signs of recurrence.
- biomarkers
- DNA
- RNA
- cancer
1. Introduction
2. Challenges Associated with Detecting Early-Stage Tumors
Successful cancer treatment depends on early detection [5][13]. Yet, physiological and mass transport barriers restrict the amount of biological indicators that can be released from early lesions [6][7][14,15]. Finding intrinsic biomarkers through blood and biofluid examination is the primary objective of ongoing research. To improve specificity, bioengineered sensors and synthetic markers are being developed. Imaging systems also aid in detecting and localizing tumors [8][9][10][16,17,18]. The typical spatial resolution of a positron emission tomography (PET) scanner is about 1 cm3, and hence, very small tumors (diameter < 5 mm) will be missed by the PET imagers. The typical blood draw is 5–10 mL, which is three orders of magnitude (1/1000th) smaller than the body’s total blood volume (~5 L). This means that the biomarkers shed by the tumor will be diluted > 1000 times when it is detected (Figure 1). Further, there are challenges in detecting genomic materials. For example, circulating tumor DNA (ctDNA) has a half-life of ~1.5 h. So, in a 24 h time period, it will undergo 16 half-lives. This means that by the time it is detected, only 0.0015% of the original materials will remain [9][10][11][12][17,18,19,20]. A potential ten-year window for early cancer detection is suggested by multicompartment models and studies on the genomic timeline. However, current screening techniques can find cancers that have been present for ten years or longer and are indolent. Cancers that spread quickly and aggressively and have poor clinical outcomes include triple-negative breast cancer and high-grade serous ovarian carcinoma. These problems are intended to be solved by activity-based or genetically encoded mechanisms for early detection in synthetic biomarker research.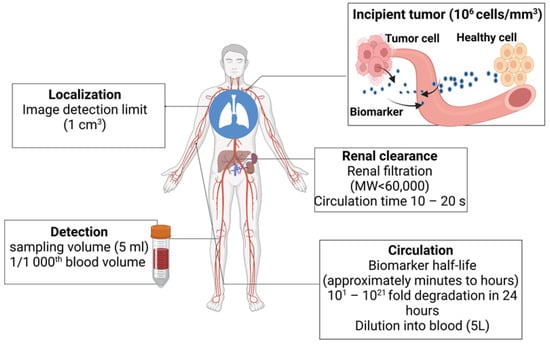
3. Biomarkers in Cancer Detection, Diagnosis, and Prognosis
3.1. Biofluid Biomarkers
3.2. Imaging Biomarkers
Tumor, node, metastasis (TNM) staging, objective response, and left ventricular ejection fraction are just a few of the imaging biomarkers (IBs) that are critical for clinical oncology [27][35]. Cancer research frequently uses imaging techniques like computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET), and ultrasonography. New IBs need to be validated and qualified in order to fill in the translational gaps [28][36]. A total of 14 important recommendations have been made by Cancer Research UK (CRUK) and the European Organization for Research and Treatment of Cancer (EORTC) to hasten the clinical translation of IBs [29][30][31][37,38,39]. These suggestions for achieving IB qualification emphasize parallel validation procedures, cost-effectiveness analysis, standardization, accreditation systems, precision evaluation, alternative validation frameworks, and multicenter studies [32][33][34][35][36][37][40,41,42,43,44,45]. IBs are derived from medical images. They offer non-invasive, cost-effective screening, tumor detection, patient progress, and therapy response monitoring tools [38][46]. Staging systems document the existence, dimensions, and quantity of abnormalities in tumor, lymph node, and additional metastatic locations to establish a structured categorical indicator of the patient’s disease severity. IBs have the ability to map tumor heterogeneity, monitor changes in tumors over time, and assess a person’s multiple lesions [39][40][41][47,48,49]. The evaluation of lesions at tumor, nodal, and metastatic sites using staging systems is crucial for the diagnosis and prognosis of cancer. The American Joint Committee on Cancer (AJCC) offers recommendations for precise and consistent reporting in radiology. TNM staging is frequently used and has prognostic value for a variety of cancer types. It is based on imaging modalities like CT, MRI, SPECT, and PET. TNM staging can occasionally be used to forecast treatment outcomes.3.3. Needle Biopsy
Imaging tests are essential in identifying and tracking cancer [42][52]. These examinations use various forms of energy, such as X-rays, sound waves, radioactive particles, or magnetic fields, to produce finely detailed images that reveal important details about the structure and location of the tumor [43][44][45][46][53,54,55,56]. It is crucial to remember that imaging tests do have their limitations. They cannot identify specific cancer cells, and their results are inconclusive. Imaging tests are typically validated by biopsy [47][57]. A cancer biopsy is a test for diagnosis employed to identify the kind and properties of the tumor cells and confirm or rule out the existence of cancer. Findings are crucial for making additional medical choices (grading of tumor; chemotherapy vs. radiation vs. immunotherapy) [48][49][50][51][58,59,60,61]. In accordance with the precise spot and accessibility of the suspicious region, biopsies can be carried out using a variety of approaches (Figure 24), including surgical biopsies, endoscopic biopsies, and needle biopsies [52][62]. Needle biopsy may employ a larger needle for collecting large tissue specimens or a fine needle aspiration for gathering a small sample from cells and fluid. A special needle with a suction mechanism is used in vacuum-assisted biopsy for acquiring tissue specimens. These methods provide versatility in gathering appropriate samples for analysis [53][54][63,64].
3.4. Tissue Imaging
4. Types of Cancer Biomarkers
4.1. Genetic Biomarkers
4.1.1. Mutations and Gene Alterations
4.1.2. Gene Expression Profiles
Gene-expression-profile-based cancer biomarkers involve analyzing the patterns of gene expression in cancer cells to provide insights into tumor behavior, prognosis, and treatment response. Here are some examples of gene-expression-profile-based cancer biomarkers: Oncotype DX in Breast Cancer: Oncotype DX is a genomic test that assesses the expression of a panel of about 16 genes involved in breast cancer. It provides a recurrence score (RS) that predicts the likelihood of disease recurrence and guides treatment decisions, particularly in early-stage hormone receptor-positive breast cancer. The genes in question are ERBB2 (also known as HER2), ESR1 (estrogen receptor 1), PGR (progesterone receptor), BIRC5 (survivin), SCUBE2 (signal peptide, CUB domain, EGF-like 2), STK15 (Aurora kinase A), BCL2 (B-cell lymphoma 2), MKI67 (Ki-67), GSTM1 (glutathione S-transferase mu 1), CD68 (cluster of differentiation 68), BAG1 (BCL2-associated athanogene 1), MMP11 (matrix metallopeptidase 11), CTSL2 (cathepsin L2), GRB7 (growth factor receptor-bound protein 7), GSTM1 (glutathione S-transferase mu 1), and CDKN1B (cyclin-dependent kinase inhibitor 1B) [61][73]. MammaPrint in Breast Cancer: MammaPrint is a gene-expression-based assay used to analyze the activity of a set of genes (~18 genes) in breast cancer. It provides a genomic risk score (RS) that helps determine the risk of distant metastasis and assists in treatment decision making, particularly in early-stage breast cancer. The list of genes includes AURKA (Aurora kinase A), BIRC5 (survivin), CCNB1 (cyclin B1), CDC2 (cell division cycle 2), CKS1B (CDC28 protein kinase regulatory subunit 1B), DLG7 (discs large homolog 7), ERBB2 (also known as HER2), ESR1 (estrogen receptor 1), FOXM1 (forkhead box M1), MMP11 (matrix metallopeptidase 11), MYBL2 (myb-related protein B), NDC80 (kinetochore protein NDC80 homolog), NEK2 (NIMA-related kinase 2), RACGAP1 (Rac GTPase-activating protein 1), RRM2 (ribonucleotide reductase M2 subunit), STK15 (Aurora kinase A), TYMS (thymidylate synthase), and UBE2C (ubiquitin-conjugating enzyme E2C) [62][74].4.1.3. DNA as a Cancer Biomarker
The initial markers tested for tumor staging were circulating DNA, as shown in Figure 35. Elevated concentrations of serum DNA have been linked to cancer (most particularly, metastatic cancer). Oncogene alterations, mismatch-repair gene mutations, and mutations in tumor suppressor genes can all be used as DNA biomarkers. In over 50% of sporadic malignancies, mutations in the p53 tumor suppressor gene are found, and mutations in the KRAS oncogene indicate metastatic spread [55][56][57][67,68,69]. A TP53 mutation passed down through the generations (Li–Fraumeni syndrome) raises the likelihood of acquiring several of the same malignancies. Several genes have single nucleotide polymorphisms, including RAD1, CYP1A1, and BRCA1/2 (breast cancer), PGS2 (lung cancer), and XRCC1, p53, and ATM (lung, head, and neck cancers). Diagnosis has been associated with mutations in DNA nucleotides in tumor promoters such as APC, RAS, and tumor suppressor genes. Tissue, sputum, serum, saliva, cerebrospinal fluid (CSF), bronchial tear, tumor cells circulating in the bone marrow, and blood are all potential sources of DNA [63][64][65][79,80,81]. Mutations in mitochondrial DNA have been postulated as diagnostics biomarkers for various malignancies [63][66][67][79,82,83]. Haplotype analysis was used to investigate the mitochondrial inheritance pattern in cancer patients. Researchers used polymerase chain reaction to look for critical polymorphic locations in the mitochondrial DNA in specimens from cancer patients and healthy subjects to see if there is a link connecting mitochondrial genotype and cancer. Nine mitochondrial genomic haplogroups have been described, namely, H, I, J, K, T, U, V, W, and X. U is linked with a high chance of developing renal and prostate cancer among these haplogroups [63][79].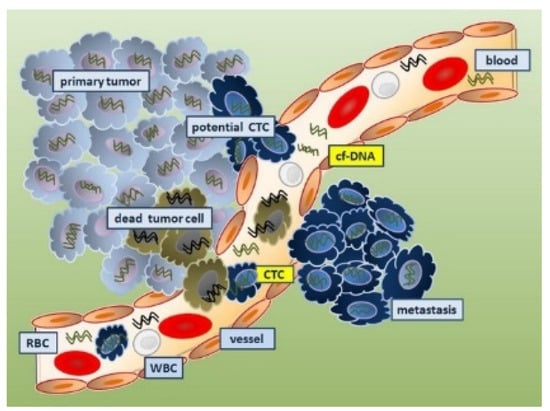
4.1.4. RNA as a Cancer Biomarker
4.1.5. Epigenetics as a Cancer Biomarker
Epigenetic alterations are potent biomarkers for cancer as they are frequent for specific genes, are stable, and can be detected in a minimally invasive mode. Numerous studies have discovered that DNA methyltransferases that insert methyl groups into cytosine groups of DNA are changed in cancer cells [81][112]. The hypermethylation of local CpG island promoter silences the tumor suppressor genes, stimulating their gene mutations. NKX2-6, SPAG6, PER1, and ITIH5 gene methylation was detected in breast cancer patients’ serum [82][113]. The hypermethylation of promoter p16 in serum DNA, for instance, is linked to recurring colorectal cancer. The methylation of the RASSF1A and p16Ink4 genes has been related to a 15-fold elevation in the comparative risk of lung cancer. The methylation status of multiple genes in clinical specimens might be a viable non-invasive technique for detecting smokers at risk of developing lung cancer [83][114].4.2. Protein Biomarkers
Proteins as Cancer Biomarkers
The proteome is a complex system made up of several proteins which interact with one another in dynamic intermolecular interactions and posttranslational alterations. Because they modulate molecular processes and pathways in normal and cancerous cells, proteomic markers are relevant to tumorigenesis and progression [84][85][131,132]. Proteins from pancreatic cancer can be found in a number of bodily fluids, including bile, pancreatic juice, urine, and fluid from pancreatic cysts, as shown in Figure 47. These proteins have a great deal of potential as useful biomarkers with a range of therapeutic applications, including early identification, illness staging, treatment prognosis, and in-flight patient monitoring. The majority of the FDA-approved cancer biomarkers in clinical usage are single proteins obtained from serum. HCG, AFP, and LDH are utilized to stage testicular cancer.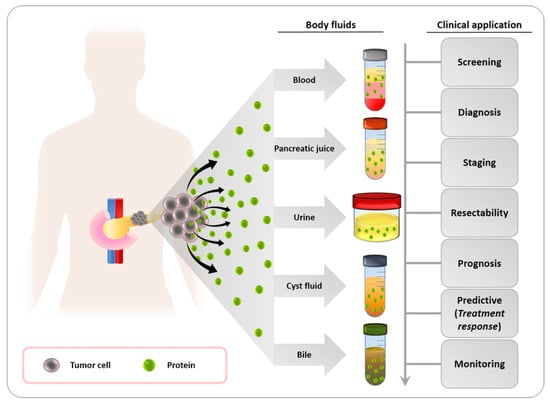
4.3. Metabolic Biomarkers
.3. Metabolic Biomarkers
4.3.1. Metabolites and Metabolic Pathways
4.3.2. Metabolic Imaging Techniques
Metabolic imaging techniques are used to visualize and assess the metabolic activity of cancer cells. These techniques provide valuable information about tumor metabolism and can aid in cancer diagnosis, staging, treatment planning, and monitoring. Here are some commonly used metabolic imaging techniques in cancer: Positron Emission Tomography (PET): PET imaging utilizes radiolabeled tracers that are taken up by cells based on their metabolic activity. The most commonly used tracer in PET imaging is fluorodeoxyglucose (FDG), a glucose analog. FDG-PET measures glucose metabolism and is particularly useful in detecting and staging various cancers, including lung, colorectal, and breast cancers [97][163]. Magnetic Resonance Spectroscopy (MRS): MRS allows the non-invasive assessment of metabolite concentrations in tissues. It provides information on metabolites such as choline, creatine, and lactate, which are associated with cellular metabolism. MRS is used in brain tumor imaging to assess tumor grade, identify tumor margins, and monitor treatment response [98][164]. Magnetic Resonance Imaging with Hyperpolarized Substrates (HP-MRI): HP-MRI is an emerging technique that utilizes hyperpolarized substrates, such as pyruvate or fumarate, which are metabolized in real time to visualize metabolic pathways. This technique provides dynamic information on metabolic fluxes, such as glycolysis or TCA cycle activity, and holds promise for assessing tumor metabolism and treatment response [99][165]. Single-Photon Emission Computed Tomography (SPECT): SPECT imaging uses radiotracers that emit gamma rays to detect specific metabolic processes. SPECT can be used to assess various metabolic functions, such as blood flow, metabolism, and receptor binding. Examples include technetium-99m sestamibi for imaging myocardial perfusion and iodine-123 ioflupane for imaging dopamine transporter function in neuroendocrine tumors [100][166]. Dynamic Contrast-Enhanced Magnetic Resonance Imaging (DCE-MRI): DCE-MRI involves the administration of a contrast agent to evaluate the tumor’s vascularity and blood flow. By measuring the kinetics of contrast agent uptake and washout, DCE-MRI provides information on tumor perfusion, angiogenesis, and vascular permeability. It is used in various cancers, including breast, prostate, and brain tumors [101][167]. Optical Imaging: Optical imaging techniques, such as fluorescence imaging and bioluminescence imaging, can be used to assess metabolic processes at a cellular level. Fluorescent probes and reporter genes are utilized to visualize specific metabolic activities, such as pH, reactive oxygen species, or enzyme activity. Optical imaging is commonly employed in preclinical research and experimental studies. These metabolic imaging techniques offer complementary information about tumor metabolism and aid in understanding the biological characteristics of cancer cells.4.3.3. Molecular Probes and Contrast Agents
Molecular probes and contrast agents are invaluable tools in cancer research and clinical imaging. They are designed to specifically target and highlight certain molecular features or physiological processes associated with cancer. Here are some examples of molecular probes and contrast agents used in cancer: Fluorescent Probes: Fluorescent probes emit light at specific wavelengths when excited by the light of a different wavelength. They can be conjugated to antibodies or other targeting molecules to visualize specific cancer-related targets or processes. For example, fluorescently labeled antibodies can be used to target and detect specific proteins or receptors overexpressed in cancer cells [102][168]. Magnetic Resonance Imaging (MRI) Contrast Agents: MRI contrast agents enhance the contrast between normal and cancerous tissues in MRI scans. These agents often contain gadolinium, manganese, or iron oxide nanoparticles. They can help visualize tumor morphology, angiogenesis, and tissue perfusion. Examples include gadolinium-based contrast agents and superparamagnetic iron oxide nanoparticles (SPIONs) [103][169]. Positron Emission Tomography (PET) Tracers: PET tracers are radiolabeled molecules that are administered to patients and emit positrons, which can be detected by PET scanners. They are designed to target specific molecular pathways or processes associated with cancer. For example, fluorodeoxyglucose (FDG) is a radiolabeled glucose analog used to detect increased glucose metabolism in cancer cells, and 18F-fluorothymidine (FLT) is used to assess cell proliferation by targeting DNA synthesis [104][170]. Ultrasound Contrast Agents: Ultrasound contrast agents are microbubbles filled with gas that enhance the contrast during ultrasound imaging. These agents can help visualize blood flow, angiogenesis, and tumor vascularity. Microbubbles can be conjugated with targeting ligands to selectively bind to specific markers on cancer cells or blood vessels [105][171]. Near-Infrared (NIR) Imaging Probes: NIR imaging probes emit light in the near-infrared spectrum, which can penetrate deeper into tissues. They are used for the non-invasive imaging of tumors, lymph nodes, and other structures. NIR probes can target specific cancer markers or processes, allowing for real-time imaging during surgery or molecular imaging studies [106][172].4.4. Cells as Cancer Biomarkers
4.4.1. Circulating Tumor Cells as Cancer Biomarkers
In the realm of cancer, circulating tumor cells (CTCs) are basic yet effective biomarkers. The existence of CTCs has been demonstrated to determine patient survival with invasive breast cancer at various periods during treatment [107][173]. Cancer treatment targets (CTTs) are better predictors of prognosis than traditional tumor markers (e.g., CA27-29). The prevalence of therapeutic targets on CTCs can also influence the choice of an effective treatment regime, and the impact of treatment can be assessed after the initial cycle of medication [108][174]. The prevalence of CTCs has been reported to predict patient survival with metastatic breast cancer at various periods throughout treatment [107][173]. For patients undergoing systemic therapy for metastatic breast cancer, CTC gives an early and accurate indication of the progression of the disease and survival. CTC counts have been confirmed to be a consistent indicator for prognosis and therapy response in patients with metastatic prostate cancer.4.4.2. Immune Cells as Cancer Biomarkers
The immune system can differentiate between self-antigen and foreign antigens, promoting the maintenance of immune tolerance and inducing defensive immunity towards foreign antigens. Across several tumor entities, such as colorectal cancer and liver metastases, immune cell count in scanned tissue has already been employed to identify reliable and clinically useful biomarkers [109][110][111][180,181,182]. Macrophages and T lymphocytes are the tumor site’s most prevalent immune cells linked to clinical effects [112][113][114][115][183,184,185,186]. The histopathological examination of tumor-infiltrating lymphoid cells has been confirmed to be a credible and prognostically useful biomarker [116][117][187,188]. T cells aid in thwarting immune pathologies by sustaining self-tolerance [118][119][144,145]. Studies reported that upregulated regulatory T-cells (T-regs) expression had been linked to poor immunological responses to tumor antigens in cancer patients, indicating that it may promote immune dysregulation and tumor progression [120][121][189,190]. T-regs have already been detected in large numbers in patients with lung, breast, pancreatic, skin, and liver cancers, either in the bloodstream or in the tumor [120][122][189,191]. The prevalence of T-regs, which impair tumor-specific T-cell immunity, was negatively related to survival in ovarian cancer patients [123][192]. T-regs are essential for the emergence of metastasis to lungs in breast cancer, according to Olkhanud et al. [124][193]. The infiltration of T-regs in primary tumor sites has also been correlated with the prevalence of circulating tumor cell cells in breast cancer patients, implying involvement in cancer cell dissemination [125][194].4.4.3. Cancer Stem Cells as Cancer Biomarkers
Within tumors, subpopulations of cancerous cells have long been identified that imitate the hierarchical developmental system of the healthy tissue from which cancer arises. The tumors are propelled and sustained by a small population of cells that can self-renew and produce the more differentiated cells that constitute the mass of the tumor [126][195]. Various researchers have termed the former subpopulation cancer stem cells (CSCs) to signify that exclusively these cells can produce new tumors when transplanted to animals with immune deficiency [127][196]. The cancer stem cell model has received a lot of attention recently. CSCs were first detected via research on acute myelogenous leukemia patients (AML). Numerous solid cancers, notably prostate cancer, glioblastoma, breast cancer, medulloblastoma, and melanoma, have been shown to contain CSCs [128][197]. Because CSC (cancer stem cell) destruction is expected to be a crucial factor in achieving cure, their prevalence has enormous consequences on both cancer biology and treatment. Self-renewal, tumor-originating capacity, asymmetric cell division, and differentiation capacity are all features that identify potential CSCs [129][130][198,199]. CD24, CD133, CD166 (ALCAM), CD44, EpCAM, CD29, Lgr5, ALDH1B1, and ALDH1A1 are some of the cytoplasmic and surface markers which have been utilized to detect putative cancer CSCs. Metastatic colon malignancies from patient populations were associated with an elevated expression of ALDH1B1 (p = 0.001) compared with healthy colon tissue [131][200]. Other investigations have correlated the degree of CD24 expression in colorectal tumors to lymphovascular invasion and decreased survival rates [132][133][134][201,202,203]. The expression of CD44v9 is associated with initial stage lung adenocarcinoma and epidermal growth factor receptor mutations in lung malignancies [135][204]. CD44 variants are also found in gastric malignancies, where they stimulate tumor initiation [136][205]. Thus, a CSC biomarker has been suggested as a marker for diagnosis, interventional, and prognostic purposes.4.5. Lamins as Cancer Biomarkers
The nuclei of animal cells are identifiable by their well-defined chromatin compartmentalization and nuclear structure. In higher vertebrates, the intricate nuclear architecture has been associated with the surge in genomic intricacy and the demand for spatiotemporal control of gene expression. The nucleoplasm, nuclear pore complex, and lamina are the three main constituents of a standard multicellular nucleus. The lamina is a protein meshwork located on the inner nuclear membrane’s nucleoplasmic side. The main element of this lamina is a group of class V intermediate filaments proteins termed lamins which are abnormally expressed in tumors. Lamins control differentiation, apoptosis, gene expression, and DNA repair in a direct or indirect way. By analyzing abnormalities in the expression profile of lamins in different forms of malignancies, several researchers and cancer biologists were able to pinpoint the link between abnormal lamin expression and cancer subtype. The medication betulinic acid has anti-cancer properties in pancreatic cancer by limiting lamin B1 production, and it might be used as a biomarker for cancer. Scientists have looked into alterations in lamin patterns of expression in a variety of malignancy types in order to better understand the association between lamin transcription and cancer subgroups. Lamins, especially A-type lamins, communicate with transcription elements to control the growth and differentiation of cells [137][209]. In mature stem cells, the overexpression of the lamin A mutant inhibits the maturation and repair of tissue. The proliferation of cells is linked with decreased differentiation and zero or impaired gene expression for A-type lamins [138][139][210,211]. Lamins may function as indicators for cancer risk, forecasting the course and outcome of tumor growth. Nuclear lobulations and morphological alterations may result from lamin A depletion [140][212]. Colorectal malignancy, which has aberrant or misinterpreted lamin expression, is among the three most common malignancies worldwide. There is a strong correlation amongst lamin A/C expression, prognosis for patients, and the advancement of colorectal cancer, according to recent research. Death rates were almost twice as high in patients whose tumors tested positive for A-type lamin overexpression. Lamin A/C expression may serve as a risk signal for colorectal cancer-dependent mortality since it elevated T-plastin, reduced E-cadherin, and enhanced cell migration in colorectal cancer cells when GFP-lamin A was expressed ectopically.4.6. Galectins as Cancer Biomarkers
Galectins are a class of beta-galactoside-binding lectins widely found in all species. The genesis, progression, and pathological aggressiveness of tumors are linked to aberrant tumor-associated galectin expression. Rather than being a carcinoma diagnostic biomarker, galectin-3 is more of a malignancy function-related biomarker that can be applied in conjunction with certain other metabolic biomarkers. It is released into the tumor stroma and promotes tumor growth and angiogenesis [141][223]. Galectin-3 protein expression was much higher in breast tumor tissues relative to precancerous tissue, and triple-negative breast tumors have significantly higher levels of galectin-3 expression than other subtypes of breast cancer [142][143][224,225]. Evidence from a variety of cancer types suggests that the expression of galectin-1 is frequently higher in tumor tissues in contrast with healthy or benign tissues. Malignancies of the reproductive organs, gastrointestinal tract, lymphatic malignancies, myeloproliferative tumors, respiratory and urinary system, thyroid, and skin tumors all exhibit this pattern [144][145][146][147][148][149][150][151][152][153][154][155][229,230,231,232,233,234,235,236,237,238,239,240]. Although three studies found that galectin-1 expression was decreased in head and neck squamous cell carcinoma, cancers of the uterus, and prostate cancer, these results do not agree with those of the majority of studies, which may indicate that patient demographics, tumor subtypes, or methodologies may differ [156][157][158][241,242,243]. The expression of galectin-7 varies between cancer types; it is expressed less in malignancies of the skin, cervix, and stomach and more in cancers of the gastrointestinal tract, breast, thyroid, larynx, and indolent lymphoproliferative diseases. The expression of galectin-7 is also dependent on the subtype of cancer and the location of the disease inside the cell; it is absent in carcinomas of basal cells and present in squamous cell tumors, which are head and neck malignancies [159][160][161][162][163][244,245,246,247,248]. Malignant tissues release circulating galectins, which can be utilized as a biomarker for diagnosis. There have been reports of elevated amounts of galectin-1 and -3 in thyroid, pulmonary, skin, bladder, colon, and breast cancers. However, they are not very useful in diagnosing thyroid cancer. Glycoproteins that bind to lectin may potentially function as diagnostic markers [164][165][166][167][168][249,250,251,252,253].4.7. Carbohydrate Antigens as Cancer Biomarkers
Carbohydrate antigen (CA) biomarkers are cancer indicators that have been identified because of efforts to construct antibodies targeting extracts or cell lines derived from tumors. CA indicators are glycoproteins of high molecular weight. The most invariably utilized serum tumor biomarker for detecting malignancies of the digestive organs is CA19-9. The validated marker for detecting ovarian cancer recurrence and evaluating therapy response is CA-125 [169][304]. CA-125’s diagnosis sensitivity is limited, and it has been demonstrated that this glycoprotein is widely dispersed on the surface of cells in a variety of malignant or benign conditions other than ovarian cancer, leaving its efficacy in the diagnosis in jeopardy [170][115]. Carcinoembryonic antigen (CEA) is a glycoprotein found on the surface of cells that offers an important function in adhesion. CEA is produced by healthy mucosal cells, and its level in normal adults is as minimal as 2.5 ng/mL and as high as 5.0 ng/mL in people who smoke; but, in the existence of a tumor, it can reach 100 ng/mL.4.8. Viruses as Cancer Biomarkers
Hepatocellular carcinoma (HCC) is among the most widespread viral-induced tumors [171][309]. Over 80% of HCC cases are reported in underdeveloped nations. The risk factors are chronic hepatitis viral infections, caused primarily by the prevalent hepatitis B virus (HBV), and hepatitis C virus (HCV) infection in a small percentage of HCC patients (12–17%) [171][309]. HBV can induce tumorigenesis by genomic instability mediated by its frequent incorporation in host DNA [172][310]. Cervical is the second most prevalent cancer in women, accounting for most cancer-related fatalities worldwide, and chronic infection with particular strains of HPV is the most prevalent trigger for cervical cancer. HPV has been detected in a substantial amount in anal, oral, penile, esophageal, vulvar, and vaginal cancers, as well as a tiny portion in laryngeal, lung, and stomach cancers in some regions of the world [173][311]. Cervical carcinoma samples were utilized to diagnose, clone, and sequence papillomaviruses for the first time. Antibodies to HPV (E6 and E7) produced by participants act as biomarkers of an HPV-related carcinoma [174][312]. Due to the sheer rise in HPV-related disease, especially HPV16 infection, oropharyngeal squamous cell carcinoma (OPSCC) is presumed to be the third most prevalent malignancy in middle-aged, non-Hispanic, white men by 2045.4.9. Exosomes as a Cancer Biomarker
Exosomes, which are the smallest (diameter of 30–150 nm) extracellular vesicles, are secreted by endothelial cells, erythrocytes, epithelial cells, dendritic cells, oligodendroglial cells, mesenchymal stem cells (MSCs), neural cells, and tumor cells [175][176][318,319]. Exosomes can serve as “cellular postmen” for carrying genomic material for inter- and intracellular communication because they are loaded with physiologically active components such as RNA, cytoplasmic proteins, cellular metabolites, and lipids [177][320]. Exosomes can be found in blood, breast milk, synovial fluid, amniotic fluid, urine, bronchoalveolar lavage fluid, pleural fluid, serum, and saliva [178][179][321,322]. Due to their widespread prevalence in physiological fluids and their resemblance to the contents of original cells, exosomes are potentially useful as circulating biomarkers for numerous kinds of cancers. By constructing or modulating the tumor microenvironment and encouraging angiogenesis and tumor invasion, tumor-derived exosomes (TEXs) serve a crucial role in tumorigenesis and progression [180][181][323,324]. TEXs contain a multitude of endogenous cargos which partly imitate the components and resemble the pathophysiological condition or signaling abnormalities of parent cells, rendering them potential biomarkers for early cancer detection. Exosomal proteins are emerging diagnosis and monitoring markers for cancers because there are plentiful cancer-related proteins in exosomes. In exosomes secreted from pancreatic cancer, overexpressed proteoglycan Glypican-1 (GPC-1) is found. Patients with pancreatic ductal adenocarcinoma (PDAC) were reported as having higher levels of exosomal protein 4 (CKAP 4) than healthy people. Exosomes containing CKAP 4 in the serum can be utilized as a potential biomarker for PDAC [182][325]. Trp5 (Transient Receptor Potential Channel 5) is overexpressed in exosomes from breast cancer, has a major function in drug resistance, and can be utilized to anticipate chemotherapy resistance in patients with breast cancer [183][326]. Exosomes have a double-layer lipid barrier that protects internal nucleic acids from being damaged. Consequently, exosomal nucleic acids can be potential indicators in cancer diagnostics. Hepatocellular carcinoma and other malignancies may benefit from exosomal miRNAs as potential serological markers [184][327]. Elevated exosomal miR-375 and miR-1290 levels in the plasma of prostate cancer (castration-resistant) patients were linked to a poor overall survival rate (OS) [185][328].4.10. Lipids as Cancer Biomarkers
Jiang and colleagues reported the lipid species that could be utilized as markers for early detection of breast cancer. In comparison to healthy controls, researchers noticed higher amounts of Phytosterol Diosgenin (DG), and Phosphatidylcholines (PC) in breast cancer samples. The level of Phosphatidylethanolamine (PE) was shown to be lower in breast cancer samples [27][35]. Prostate cancer patients have a 2.7-fold elevation in Lysophosphatidylcholine (LPC) relative to healthy subjects, according to Zhou and colleagues [28][36].
5. Emerging Technologies and Techniques
5.1. Liquid Biopsy
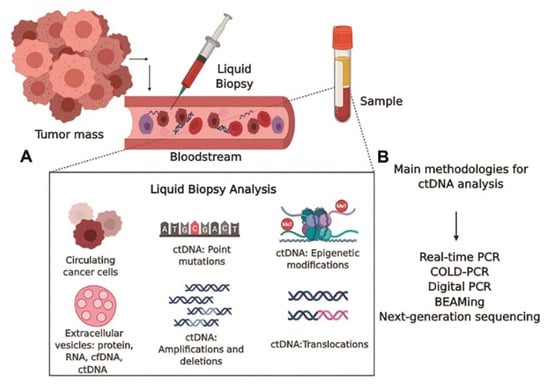
Liquid biopsy is a minimally invasive diagnostic procedure that involves the examination of numerous elements found in physiological fluids like blood or urine, including exosomes, circulating tumor cells, cell-free DNA (cfDNA), and proteins. With the help of this method, early cancer detection, treatment monitoring, and the discovery of potential therapeutic targets are made possible through insights into a patient’s molecular profile. Figure 512 provides an illustration of the methods used in liquid biopsy analysis. In this method, a single blood sample’s cfDNA/ctDNA profile is made up of both wild-type and genetically and epigenetically changed DNA fragments released by various tissues and organs through various pathways [186][413].