- blue LED
- plant elongation
- mechanisms
- applications
- future directions
1. The Purity of Blue Light may Affect Plants’ Elongation Responses to This Light Wavelength
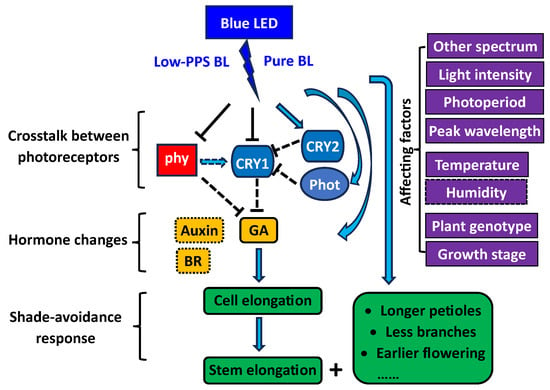
Blue LEDs normal emit more pure BL than the non-LED BL sources employed in prior studies, which can affect phytochrome activity and thus affecting plant elongation response. The non-LED BL sources emit impure BL due to the presence of low levels of other light wavelengths, such as a high ratio of R/FR light, that activate phytochromes, thereby making the BL exhibit more suppressive effect on elongation growth than R light [1]. For instance, the blue-colored fluorescent lamp, which was previously one of the commonly used BL sources, was reported to have a R/FR ratio of 1.87 [2]. The white fluorescent lamp filtered through blue acetates, another previously utilized BL source, did not contain >700 nm light due to the filters employed [3]. In contrast to BL from non-LED lighting, blue LED light exhibits a much lower phytochrome photostationary state (PPS, an indicator of phytochrome activity), estimated as 0.5, compared to 0.9 for red LED light, as per the method established by Sager et al. [4]. Although the threshold value of PPS required to elicit an active phytochrome response remains a matter of debate, it is generally agreed that a PPS < 0.6 may provoke an inactive response [5]. The lower phytochrome activity in plants could potentially account for the elongated plants observed under blue LED lighting. Thus, the researchers postulate that the effects of BL on plant elongation may, in some cases, be linked to phytochrome activity, which may differ under pure and impure BL sources.
To examine the hypothesis proposed above, the researchers created some impure BLs with different PPS by adding different ratios of R and FR to blue LEDs, in addition to the pure BL from blue LED. The results indicated that the pure BL promoted plant elongation compared to R light (Figure 1) [1]. However, the high-PPS impure BL had the opposite effect and inhibited elongation growth to a similar or greater extent than pure R light. The low-PPS impure BL restored the promotional effect observed with pure BL. The R/FR reversibility and the PPS changes suggest that the promotional effect observed with pure BL is linked to low phytochrome activity [1]. Under certain conditions, pure BL may need to co-act with R light to inhibit elongation growth by increasing phytochrome activity. Also, the promotional effect of impure BL increased gradually with the decreasing PPS values from 0.69 to 0.60. However, pure BL did not show a greater promotional effect on plant elongation than the lowest-PPS impure BL, despite a lower PPS value (0.49 vs. 0.60). It appears that the plant elongation promoted by BL gradually became saturated once the PPS values decreased below 0.60 [6][7][8]. It is possible that the deactivated phytochrome contributes to the maximum elongation promotion by BL [6].

Adding low-level R light to pure BL has a similar or greater inhibitory effect than that of R light on plant elongation, so it is also interesting to know how plant elongation responds to adding low-level other wavelengths, including ultraviolet-B (UVB), ultraviolet-A (UVA), green (G) or FR light, to pure BL, since BL from some non-LED lighting sources has also been found to contain small amounts of these light wavelengths. researchers'studies indicated that adding these low-level wavelengths to pure BL only slightly changed plant elongation compared with blue LED, but still promoted plant elongation compared with red LED[9][10]. The calculated PPS values of these impure BL were similarly low (<0.6) to those of B, but they were much less than those of high-PPS impure BL by adding red light to pure BL, indicating that phytochrome activity plays an important role in plant elongation mediated by BL [9][10]. It appears that among the wavelengths possibly contained in impure BL, low-level R light has the greatest contribution to the inhibitory effect of impure BL on plant elongation.
2. Factors Affecting Plants’ Elongation Response to Pure Blue Light Relative to Red Light
2.1. Lighting Features
2.2. Plant Factors
2.3. Cultivation Conditions
3. Mechanisms Underlying Blue-LED-Promoted Plant Elongation
3.1. Shade-Avoidance Response
3.2. Hormone Changes
3.3. Involved Photoreceptors

4. Application of Blue LEDs in Mediating Plant Elongation for Controlled-Environment Production
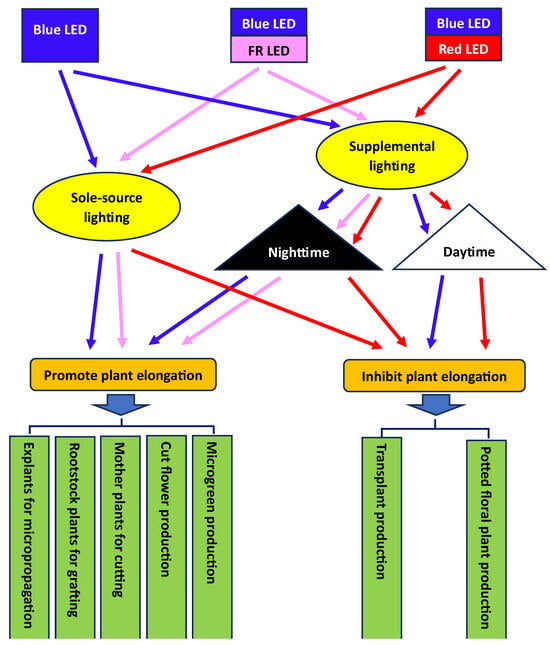
4.1. Plant Propagation
4.1.1. Promoting Explant Elongation for Micropropagation
4.1.2. Promoting Hypocotyl Elongation of Rootstock Plants for Grafting
4.1.3. Mediating Shoot Elongation of Mother Plants for Cuttings
4.2. Transplant Production
4.2.1. Sole-Source Lighting with a Combination of Blue and Red LEDs Can Produce Compact Transplants under Indoor Conditions
4.2.2. Supplemental Lighting with Blue LEDs Only or Their Combination with Red LEDs Can Produce Compact Transplants in Greenhouse Conditions
4.3. Floral Plant Production
4.3.1. Promoting Plant Compactness in Potted Floral Plant Production
4.3.2. Promoting Plant Elongation in Cut Flower Production
4.4. Microgreen Production
4.4.1. Application of Blue LEDs in Daytime Lighting to Promote Hypocotyl Elongation
RB-LEDs have been popularly used as daytime lighting sources for indoor-grown microgreens, and the BL proportion in RB-LEDs can be optimized to promote plant elongation while maintaining the yield and other quality traits. For daytime sole-source lighting at 300 µmol m−2 s−1 for 16 h d−1 during indoor microgreen production, the BL proportion in RB-LEDs (between 5–30%B) was optimized in terms of plant elongation, yield, and other appearance qualities for cabbage at 15%B, and at 5%B for kale, arugula, and mustard [90]. Although the BL proportion in RB-LEDs can be optimized, it can still cause shorter plants than blue LEDs. For example, under RB-LED lighting at 220 µmol m−2 s−1 for 18 h d−1, RB-LEDs with only 10%B reduced the hypocotyl length in Brassicaceae microgreens compared to blue LEDs only [91]. However, microgreens grown under either blue or red LEDs alone cannot meet the commercial requirements in terms of both plant height and appearance quality, so a potential approach using sequential lighting, with blue LEDs first to increase plant height and then red LEDs to improve leaf size and plant color (i.e., temporal combination of blue and red LEDs), has been suggested by the researchers' lab to address this problem [12]. Another approach developed by us to address the problem of short plants under RB-LED lighting is delaying the start of the lighting for several days—in other words, using early-stage dark treatment [92][93], since darkness, relative to RB-LED lighting, can also promote hypocotyl elongation during the early development stage of plants [94]. However, this approach is better for larger-seed species such as sunflowers, due to a potential yield loss in smaller-seed species such as arugula, despite the promotion of plant elongation in both species [92].4.4.2. Application of Blue LEDs in Nighttime Lighting to Promote Hypocotyl Elongation
For indoor-grown microgreens under electrical lighting, blue LEDs can be used for nighttime lighting to promote microgreen elongation without affecting yield or quality. For two microgreen species grown indoors under sole-source lighting with RB-LEDs (20%B) at a PPFD of 300 µmol m−2 s−1 during 16 h of daylight, nighttime lighting with blue LEDs alone at 20 µmol m−2 s−1 for 8 h or at 40 µmol m−2 s−1 for 4 h increased plant height by 34% and 18% for mustard and arugula, respectively, compared with no nighttime lighting [95]. Nighttime lighting with 20 µmol m−2 s−1 blue LEDs and 20 µmol m−2 s−1 FR LEDs together for 8 h further improved the promotional effect on elongation. The 8 h lighting with blue LEDs alone also increased the fresh weight of arugula by 12% compared to darkness. Additionally, nighttime treatments with blue LEDs, alone or in combination with FR LEDs, increased the chlorophyll content index, leafy index, or dry matter content, depending on the species [95]. During winter greenhouse microgreen production, overnight lighting with low-level blue LEDs alone can also promote plant elongation while improving the appearance quality and crop yield, without negatively affecting nutritional quality. For mustard and arugula microgreens, overnight lighting with 14 µmol m−2 s−1 from blue LEDs promoted stem elongation by 16% and 10%, respectively, and increased crop yield by 32% and 29%, respectively, compared to no overnight lighting [96]. Furthermore, blue LEDs increased the cotyledon area in mustard and the leaf mass unit area in arugula, and they enhanced the cotyledon color in both species, without affecting the total chlorophyll, carotenoid, and phenolic contents. However, overnight lighting with 14 µmol m−2 s−1 from FR LEDs did not have a positive effect on the above plant traits compared to blue LEDs [96].References
- Kong, Y.; Stasiak, M.; Dixon, M.A.; Zheng, Y. Blue light associated with low phytochrome activity can promote elongation growth as shade-avoidance response: A comparison with red light in four bedding plant species. Environ. Exp. Bot. 2018, 155, 345–359.
- Appelgren, M. Effects of light quality on stem elongation of Pelargonium in vitro. Sci. Hortic. 1991, 45, 345–351.
- Kigel, J.; Cosgrove, D.J. Photoinhibition of stem elongation by blue and red light. Plant Physiol. 1991, 95, 1049–1056.
- Sager, J.C.; Smith, W.O.; Edwards, J.L.; Cyr, K.L. Photosynthetic efficiency and phytochrome photoequilibria determination using spectral data. Trans. ASAE 1988, 31, 1882–1889.
- Stutte, G.W. Light-emitting diodes for manipulating the phytochrome apparatus. HortScience 2009, 44, 231–234.
- Kong, Y.; Schiestel, K.; Zheng, Y. Maximum elongation growth promoted as a shade-avoidance response by blue light is related to deactivated phytochrome: A comparison with red light in four microgreen species. Can. J. Plant Sci. 2019, 100, 314–326.
- Kong, Y.; Schiestel, K.; Zheng, Y. Does “blue” light invariably cause plant compactness? Not really: A comparison with red light in four bedding plant species during the transplant stage. Acta Hortic. 2020, 1296, 621–628.
- Kong, Y.; Schiestel, K.; Zheng, Y. Blue light associated with low phytochrome activity can promote flowering: A comparison with red light in four bedding plant species. Acta Hortic. 2020, 1296, 433–440.
- Kong, Y.; Schiestel, K.; Zheng, Y. Pure blue light effects on growth and morphology are slightly changed by adding low-level UVA or far-red light: A comparison with red light in four microgreen species. Environ. Exp. Bot. 2019, 157, 58–68.
- Kong, Y.; Zheng, Y. Growth and morphology responses to narrow-band blue light and its co-action with low-level UVB or green light: A comparison with red light in four microgreen species. Environ. Exp. Bot. 2020, 178, 104189.
- Johnson, R.E.; Kong, Y.; Zheng, Y. Elongation growth mediated by blue light varies with light intensities and plant species: A comparison with red light in arugula and mustard seedlings. Environ. Exp. Bot. 2020, 169, 103898.
- Kong, Y.; Kamath, D.; Zheng, Y. Blue versus red light can promote elongation growth independent of photoperiod: A study in four Brassica microgreens species. HortScience 2019, 54, 1955–1961.
- Kong, Y.; Masabni, J.; Niu, G. Effect of temperature variation and blue and red LEDs on the elongation of arugula and mustard microgreens. Horticulturae 2023, 9, 608.
- Lee, J.S.; Ae, C.; Lee, Y.; Kim, H.; Song, J.Y. Shorter wavelength blue light promotes growth of green perilla (Perilla frutescens). Int. J. Agric. Biol. 2014, 16, 1177–1182.
- Li, Y.; Zheng, Y.; Zheng, D.; Zhang, Y.; Song, S.; Su, W.; Liu, H. Effects of supplementary blue and UV-A LED lights on morphology and phytochemicals of Brassicaceae baby-leaves. Molecules 2020, 25, 5678.
- Li, Q.; Llewellyn, D.; Kong, Y.; Zheng, Y. Narrowband blue LEDs with different peak wavelengths similarly promote shade avoidance responses and have greater promotion effects than ultraviolet A and far red in two species of microgreens. Preprints 2023.
- Huche-Thelier, L.; Crespel, L.; Le Gourrierec, J.; Morel, P.; Sakr, S.; Leduc, N. Light signaling and plant responses to blue and UV radiations—Perspectives for applications in horticulture. Environ. Exp. Bot. 2016, 121, 22–38.
- Hirai, T.; Amaki, W.; Watanabe, H. Action of blue or red monochromatic light on stem internodal growth depends on plant species. Acta Hortic. 2006, 711, 345–350.
- Kong, Y.; Kamath, D.; Zheng, Y. Blue-light-promoted elongation and flowering are not artifacts from 24-h lighting: A comparison with red light in four bedding plant species. Acta Hortic. 2020, 1296, 659–666.
- Kong, Y.; Zheng, Y. Phototropin is partly involved in blue-light-mediated stem elongation, flower initiation, and leaf expansion: A comparison of phenotypic responses between wild Arabidopsis and its phototropin mutants. Environ. Exp. Bot. 2020, 171, 103967.
- Kong, Y.; Zheng, Y. Phytochrome contributes to blue-light-mediated stem elongation and flower initiation in mature Arabidopsis thaliana plants. Can. J. Plant Sci. 2021, 102, 449–458.
- Kong, Y.; Zheng, Y. Low-activity cryptochrome 1 plays a role in promoting stem elongation and flower initiation of mature Arabidopsis under blue light associated with low phytochrome activity. Can. J. Plant Sci. 2022, 102, 755–759.
- Innes, S.N.; Jakobsen, S.B.; Niday, A.; Ali, H.; Arve, L.E.; Torre, S. The aerial environment modulates plant responses to blue light. Acta Hortic. 2018, 1227, 525–532.
- Hahn, E.J.; Kozai, T.; Paek, K.Y. Blue and red light-emitting diodes with or without sucrose and ventilation affect in vitro Growth of Rehmannia glutinosa plantlets. J. Plant Biol. 2000, 43, 247–250.
- Izzo, L.G.; Mickens, M.A.; Aronne, G.; Gómez, C. Spectral effects of blue and red light on growth, anatomy, and physiology of lettuce. Physiol. Plant. 2021, 172, 2191–2202.
- Fukuda, N.; Fujita, M.; Ohta, Y.; Sase, S.; Nishimura, S.; Ezura, H. Directional blue light irradiation triggers epidermal cell elongation of abaxial side resulting in inhibition of leaf epinasty in geranium under red light condition. Sci. Hortic. 2008, 115, 176–182.
- Yanagi, T.; Okamoto, K.; Takita, S. Effects of blue, red, and blue/red lights of two different PPF levels on growth and morphogenesis of lettuce plants. Acta Hortic. 1996, 440, 117–122.
- Yavari, N.; Gazestani, V.H.; Wu, B.-S.; MacPherson, S.; Kushalappa, A.; Lefsrud, M.G. Comparative proteomics analysis of Arabidopsis thaliana response to light-emitting diode of narrow wavelength 450 nm, 595 nm, and 650 nm. J. Proteom. 2022, 265, 104635.
- Keuskamp, D.H.; Sasidharan, R.; Vos, I.; Peeters, A.J.; Voesenek, L.A.; Pierik, R. Blue-light-mediated shade avoidance requires combined auxin and brassinosteroid action in Arabidopsis seedlings. Plant J. 2011, 67, 208–217.
- Keuskamp, D.H.; Keller, M.M.; Ballaré, C.L.; Pierik, R. Blue light regulated shade avoidance. Plant Signal. Behav. 2012, 7, 514–517.
- Smith, H.; Whitelam, G.C. The shade avoidance syndrome: Multiple responses mediated by multiple phytochromes. Plant Cell Environ. 1997, 20, 840–844.
- Fukuda, N.; Yoshida, T.; Olsen, J.; Senaha, C.; Jikumaru, Y.; Kamiya, Y. Short main shoot length and inhibition of floral bud development under red light can be recovered by application of gibberellin and cytokinin. Acta Hortic. 2012, 956, 215–222.
- Fukuda, N.; Ajima, C.; Yukawa, T.; Olsen, J.E. Antagonistic action of blue and red light on shoot elongation in petunia depends on gibberellin, but the effects on flowering are not generally linked to gibberellin. Environ. Exp. Bot. 2016, 121, 102–111.
- Fukuda, N. Advanced light control technologies in protected horticulture: A review of morphological and physiological responses in plants to light quality and its application. J. Dev. Sustain. Agric. 2013, 8, 32–40.
- Fukuda, N.; Ishii, Y.; Ezura, H.; Olsen, J.E. Effects of light quality under red and blue light emitting diodes on growth and expression of FBP28 in Petunia. Acta Hortic. 2011, 907, 361–366.
- Matsuo, S.; Nanya, K.; Imanishi, S.; Honda, I.; Goto, E. Effects of blue and red lights on gibberellin metabolism in tomato seedlings. Hortic. J. 2019, 88, 76–82.
- Keuskamp, D.H.; Pollmann, S.; Voesenek, L.A.; Peeters, A.J.; Pierik, R. Auxin transport through PIN-FORMED 3 (PIN3) controls shade avoidance and fitness during competition. Proc. Natl. Acad. Sci. USA 2010, 107, 22740–22744.
- Keller, M.M.; Jaillais, Y.; Pedmale, U.V.; Moreno, J.E.; Chory, J.; Ballaré, C.L. Cryptochrome 1 and phytochrome B control shade-avoidance responses in Arabidopsis via partially independent hormonal cascades. Plant J. 2011, 67, 195–207.
- Demotes-Mainard, S.; Peron, T.; Corot, A.; Bertheloot, J.; Le Gourrierec, J.; Pelleschi-Travier, S.; Crespel, L.; Morel, P.; Huche-Thelier, L.; Boumaza, R. Plant responses to red and far-red lights, applications in horticulture. Environ. Exp. Bot. 2016, 121, 4–21.
- Li, J.; Li, G.; Wang, H.; Deng, X.W. Phytochrome signaling mechanisms. Arab. Book/Am. Soc. Plant Biol. 2011, 9, e0148.
- Liu, B.; Yang, Z.; Gomez, A.; Liu, B.; Lin, C.; Oka, Y. Signaling mechanisms of plant cryptochromes in Arabidopsis thaliana. J. Plant Res. 2016, 129, 137–148.
- Ahmad, M.; Cashmore, A.R. The blue-light receptor cryptochrome 1 shows functional dependence on phytochrome A or phytochrome B in Arabidopsis thaliana. Plant J. 1997, 11, 421–427.
- Ahmad, M.; Jarillo, J.A.; Smirnova, O.; Cashmore, A.R. The CRY1 blue light photoreceptor of Arabidopsis interacts with phytochrome A in vitro. Mol. Cell 1998, 1, 939–948.
- Más, P.; Devlin, P.F.; Panda, S.; Kay, S.A. Functional interaction of phytochrome B and cryptochrome 2. Nature 2000, 408, 207–211.
- Hughes, R.M.; Vrana, J.D.; Song, J.; Tucker, C.L. Light-dependent, dark-promoted interaction between Arabidopsis cryptochrome 1 and phytochrome B proteins. J. Biol. Chem. 2012, 287, 22165–22172.
- Su, J.; Liu, B.; Liao, J.; Yang, Z.; Lin, C.; Oka, Y. Coordination of cryptochrome and phytochrome signals in the regulation of plant light responses. Agronomy 2017, 7, 25.
- Wang, Q.; Liu, Q.; Wang, X.; Zuo, Z.; Oka, Y.; Lin, C. New insights into the mechanisms of phytochrome–cryptochrome coaction. New Phytol. 2018, 217, 547–551.
- Tung, H.T.; Luan, V.Q.; Van Anh, L.T.; Nhut, D.T. Stem elongation for plant micropropagation. In Plant Tissue Culture: New Techniques and Application in Horticultural Species of Tropical Region; Springer: Singapore, 2022; pp. 105–128.
- Luan, V.Q.; Huy, N.P.; Nam, N.B.; Huong, T.T.; Hien, V.T.; Hien, N.T.T.; Hai, N.T.; Thinh, D.K.; Nhut, D.T. Ex vitro and in vitro Paphiopedilum delenatii Guillaumin stem elongation under light-emitting diodes and shoot regeneration via stem node culture. Acta Physiol. Plant. 2015, 37, 136.
- Chia, P.; Kubota, C. End-of-day far-red light quality and dose requirements for tomato rootstock hypocotyl elongation. HortScience 2010, 45, 1501–1506.
- Vu, N.T.; Kim, Y.S.; Kang, H.M.; Kim, I.S. Influence of short-term irradiation during pre-and post-grafting period on the graft-take ratio and quality of tomato seedlings. Hortic. Environ. Biotechnol. 2014, 55, 27–35.
- Kamath, D.; Kong, Y.; Dayboll, C.; Zheng, Y. Dynamic versus concurrent lighting with red and blue light-emitting diodes as the sole light source can potentially improve campanula stock plant morphology for cutting production. HortScience 2021, 56, 1439–1445.
- Randall, W.C.; Lopez, R.G. Comparison of supplemental lighting from high-pressure sodium lamps and light-emitting diodes during bedding plant seedling production. HortScience 2014, 49, 589–595.
- Trivellini, A.; Toscano, S.; Romano, D.; Ferrante, A. LED lighting to produce high-quality ornamental plants. Plants 2023, 12, 1667.
- Izzo, L.G.; Mele, B.H.; Vitale, L.; Vitale, E.; Arena, C. The role of monochromatic red and blue light in tomato early photomorphogenesis and photosynthetic traits. Environ. Exp. Bot. 2020, 179, 104195.
- Li, Y.; Liu, Z.; Shi, Q.; Yang, F.; Wei, M. Mixed red and blue light promotes tomato seedlings growth by influencing leaf anatomy, photosynthesis, CO2 assimilation and endogenous hormones. Sci. Hortic. 2021, 290, 110500.
- Miao, Y.; Chen, Q.; Qu, M.; Gao, L.; Hou, L. Blue light alleviates ‘red light syndrome’ by regulating chloroplast ultrastructure, photosynthetic traits and nutrient accumulation in cucumber plants. Sci. Hortic. 2019, 257, 108680.
- Kamath, D.; Kong, Y.; Dayboll, C.; Blom, T.; Zheng, Y. Growth and morphological responses of gerbera seedlings to narrow-band lights with different light spectral combinations as sole-source lighting in a controlled environment. Can. J. Plant Sci. 2021, 101, 943–953.
- Wollaeger, H.M.; Runkle, E.S. Growth of impatiens, petunia, salvia, and tomato seedlings under blue, green, and red light-emitting diodes. HortScience 2014, 49, 734–740.
- Johkan, M.; Shoji, K.; Goto, F.; Hashida, S.; Yoshihara, T. Blue light-emitting diode light irradiation of seedlings improves seedling quality and growth after transplanting in red leaf lettuce. HortScience 2010, 45, 1809–1814.
- Hernandez, R.; Eguchi, T.; Kubota, C. Growth and morphology of vegetable seedlings under different blue and red photon flux ratios using light-emitting diodes as sole-source lighting. Acta Hortic. 2016, 1134, 195–200.
- Di, Q.; Li, J.; Du, Y.; Wei, M.; Shi, Q.; Li, Y.; Yang, F. Combination of red and blue lights improved the growth and development of eggplant (Solanum melongena L.) seedlings by regulating photosynthesis. J. Plant Growth Regul. 2021, 40, 1477–1492.
- Li, Y.; Xin, G.; Shi, Q.; Yang, F.; Wei, M. Response of photomorphogenesis and photosynthetic properties of sweet pepper seedlings exposed to mixed red and blue light. Front. Plant Sci. 2023, 13, 984051.
- Ren, M.; Liu, S.; Tang, C.; Mao, G.; Gai, P.; Guo, X.; Zheng, H.; Tang, Q. Photomorphogenesis and photosynthetic traits changes in rice seedlings responding to red and blue light. Int. J. Mol. Sci. 2023, 24, 11333.
- Wollaeger, H.M.; Runkle, E.S. Growth and acclimation of impatiens, salvia, petunia, and tomato seedlings to blue and red light. HortScience 2015, 50, 522–529.
- Mitchell, C.A.; Dzakovich, M.P.; Gomez, C.; Lopez, R.; Burr, J.F.; Hernández, R.; Kubota, C.; Currey, C.J.; Meng, Q.; Runkle, E.S. Light-Emitting Diodes in Horticulture. In Horticultural Reviews; Wiley-Blackwell: Hoboken, NJ, USA, 2015; Volume 43, pp. 1–88.
- Nanya, K.; Ishigami, Y.; Hikosaka, S.; Goto, E. Effects of blue and red light on stem elongation and flowering of tomato seedlings. Acta Hortic. 2012, 956, 261–266.
- Novičkovas, A.; Brazaitytė, A.; Duchovskis, P.; Jankauskienė, J.; Samuolienė, G.; Virsilė, A.; Sirtautas, R.; Bliznikas, Z.; Zukauskas, A. Solid-state lamps (LEDs) for the short-wavelength supplementary lighting in greenhouses: Experimental results with cucumber. Acta Hortic. 2010, 927, 723–730.
- Kang, C.; Zhang, Y.; Cheng, R.; Kaiser, E.; Yang, Q.; Li, T. Acclimating cucumber plants to blue supplemental light promotes growth in full sunlight. Front. Plant Sci. 2021, 12, 782465.
- Hernández, R.; Kubota, C. Tomato seedling growth and morphological responses to supplemental LED lighting red: Blue ratios under varied daily solar light integrals. Acta Hortic. 2012, 956, 187–194.
- Hernández, R.; Kubota, C. Growth and morphological response of cucumber seedlings to supplemental red and blue photon flux ratios under varied solar daily light integrals. Sci. Hortic. 2014, 173, 92–99.
- Gómez, C.; Mitchell, C.A. Growth responses of tomato seedlings to different spectra of supplemental lighting. HortScience 2015, 50, 112–118.
- Terfa, M.T.; Solhaug, K.A.; Gislerød, H.R.; Olsen, J.E.; Torre, S. A high proportion of blue light increases the photosynthesis capacity and leaf formation rate of Rosa× hybrida but does not affect time to flower opening. Physiol. Plant. 2013, 148, 146–159.
- Sabzalian, M.R.; Heydarizadeh, P.; Zahedi, M.; Boroomand, A.; Agharokh, M.; Sahba, M.R.; Schoefs, B. High performance of vegetables, flowers, and medicinal plants in a red-blue LED incubator for indoor plant production. Agron. Sustain. Dev. 2014, 34, 879–886.
- Gautam, P.; Terfa, M.T.; Olsen, J.E.; Torre, S. Red and blue light effects on morphology and flowering of Petunia× hybrida. Sci. Hortic. 2015, 184, 171–178.
- Islam, M.A.; Kuwar, G.; Clarke, J.L.; Blystad, D.-R.; Gislerød, H.R.; Olsen, J.E.; Torre, S. Artificial light from light emitting diodes (LEDs) with a high portion of blue light results in shorter poinsettias compared to high pressure sodium (HPS) lamps. Sci. Hortic. 2012, 147, 136–143.
- Poel, B.R.; Runkle, E.S. Spectral effects of supplemental greenhouse radiation on growth and flowering of annual bedding plants and vegetable transplants. HortScience 2017, 52, 1221–1228.
- Ouzounis, T.; Fretté, X.; Rosenqvist, E.; Ottosen, C.-O. Spectral effects of supplementary lighting on the secondary metabolites in roses, chrysanthemums, and campanulas. J. Plant Physiol. 2014, 171, 1491–1499.
- Park, Y.G.; Jeong, B.R. How supplementary or night-interrupting low-intensity blue light affects the flower induction in chrysanthemum, a qualitative short-day plant. Plants 2020, 9, 1694.
- Jeong, S.W.; Park, S.; Jin, J.S.; Seo, O.N.; Kim, G.S.; Kim, Y.H.; Bae, H.; Lee, G.; Kim, S.T.; Lee, W.S. Influences of four different light-emitting diode lights on flowering and polyphenol variations in the leaves of chrysanthemum (Chrysanthemum morifolium). J. Agric. Food Chem. 2012, 60, 9793–9800.
- Jeong, S.W.; Hogewoning, S.W.; van Ieperen, W. Responses of supplemental blue light on flowering and stem extension growth of cut chrysanthemum. Sci. Hortic. 2014, 165, 69–74.
- Singh, M.C.; van Ieperen, W.; Heuvelink, E.P. Effect of LEDs on flower bud induction in Chrysanthemum morifolium cv. Zembla. HortFlora Res. Spectr. 2013, 2, 185–188.
- SharathKumar, M.; Heuvelink, E.; Marcelis, L.F.; Van Ieperen, W. Floral induction in the short-day plant chrysanthemum under blue and red extended long-days. Front. Plant Sci. 2021, 11, 610041.
- Shimizu, H.; Ma, Z.; Tazawa, S.; Douzono, M.; Runkle, E.; Heins, R. Blue light inhibits stem elongation of chrysanthemum. Acta Hortic. 2006, 711, 363–368.
- Ganesh, S.; Jawaharlal, M.; Rajamani, K.; Thamaraiselvi, S. Responses of mixed light-emitting diode ratios on vegetative, flower regulation, and stalk elongation of cut chrysanthemum (Dendranthema grandiflora Tzvelev). J. Appl. Nat. Sci. 2021, 13, 496–503.
- Park, Y.G.; Muneer, S.; Jeong, B.R. Morphogenesis, flowering, and gene expression of Dendranthema grandiflorum in response to shift in light quality of night interruption. Int. J. Mol. Sci. 2015, 16, 16497–16513.
- Roh, Y.S.; Yoo, Y.K. Light quality of light emitting diodes affects growth, chlorophyll fluorescence and phytohormones of Tulip ‘Lasergame’. Hortic. Environ. Biotechnol. 2023, 64, 245–255.
- Amiri, A.; Kafi, M.; Kalate-Jari, S.; Matinizadeh, M.; Karaj, I. Tulip response to different light sources. J. Anim. Plant Sci. 2018, 28, 539–545.
- Kyriacou, M.C.; Rouphael, Y.; Di Gioia, F.; Kyratzis, A.; Serio, F.; Renna, M.; De Pascale, S.; Santamaria, P. Micro-scale vegetable production and the rise of microgreens. Trends Food Sci. Technol. 2016, 57, 103–115.
- Ying, Q.; Kong, Y.; Jones-Baumgardt, C.; Zheng, Y. Responses of yield and appearance quality of four Brassicaceae microgreens to varied blue light proportion in red and blue light-emitting diodes lighting. Sci. Hortic. 2020, 259, 108857.
- Brazaitytė, A.; Miliauskienė, J.; Vaštakaitė-Kairienė, V.; Sutulienė, R.; Laužikė, K.; Duchovskis, P.; Małek, S. Effect of different ratios of blue and red led light on brassicaceae microgreens under a controlled environment. Plants 2021, 10, 801.
- Kong, Y.; Zheng, Y. Early-stage dark treatment promotes hypocotyl elongation associated with varying effects on yield and quality in sunflower and arugula microgreens. Can. J. Plant Sci. 2021, 101, 954–961.
- Carvalho, S.D.; Folta, K.M. Sequential light programs shape kale (Brassica napus) sprout appearance and alter metabolic and nutrient content. Hortic. Res. 2014, 1, 8.
- Kong, Y.; Zheng, Y. Variation of phenotypic responses to lighting using a combination of red and blue light-emitting diodes versus darkness in seedlings of 18 vegetable genotypes. Can. J. Plant Sci. 2018, 99, 159–172.
- Ying, Q.; Kong, Y.; Zheng, Y. Applying blue light alone, or in combination with far-red light, during nighttime increases elongation without compromising yield and quality of indoor-grown microgreens. HortScience 2020, 55, 876–881.
- Ying, Q.; Kong, Y.; Zheng, Y. Overnight supplemental blue, rather than far-red, light improves microgreen yield and appearance quality without compromising nutritional quality during winter greenhouse production. HortScience 2020, 1, 1468–1474.
