Branched short-chain fatty acids (BCFAs), the bacterial products of the catabolism of branched-chain amino acids, are proposed as markers for colonic protein fermentation. IBS is a gastrointestinal disorder characterized by low-grade inflammation and intestinal dysbiosis. The low-FODMAP diet (LFD) has increasingly been applied as first-line therapy for managing Irritable bowel syndrome (IBS)BS symptoms, although it decreases the production of short-chain fatty acids (SCFA), well known for their anti-inflammatory action. In parallel, high protein consumption increases BCFAs. Protein fermentation alters the colonic microbiome through nitrogenous metabolites production, known for their detrimental effects on the intestinal barrier promoting inflammation.
- branched-chain fatty acids
- short-chain fatty acids
- irritable bowel syndrome
- low-FODMAP diet
- gut microbiota
- inflammation
1. Introduction
The Role of BCFAs in Intestinal Inflammation
Intestinal inflammation disturbs normal growth in humans and animals and leads to bowel diseases [20][33]. Many commensal bacteria utilize BCFAs to survive in the varying milieus. Modulating membrane fluidity is essential for bacterial survival in a variety of environments, and many microorganisms use BCFAs in their membranes to modulate biophysical procedures [21][34]. BCFAs are taken up and incorporated into enterocyte membranes where they modulate the inflammatory response [22][23][24][35,36,37]. In detail, as shown in Figure 1, BCFAs affect the cell membrane’s fluidity: with high BCFAs concentrations, membrane falls into a disordered state, allowing better transport, membrane protein structure and functionality, and cellular signal transduction and trafficking.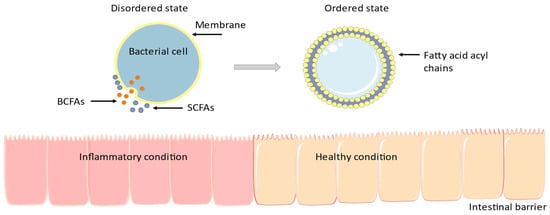
2. The Low-FODMAP Diet, IBS, and BCFAs
2.1. BCFAs & IBS
Human Studies
, Table 1
2.2. BCFAs, Inflammation, and Low-FODMAP Diet
2.2.1. Preclinical Trials
In 2019, Tuck et al. [32][51] performed experiments in mice with dextran sodium sulfate (DSS)-induced colitis during the inflammatory phase and after treatment. Animals were divided into three groups: two control treatments (“negative-control” and “positive-control”; with and without inflammation, respectively) and a “post-inflammatory” treatment group that mimicked quiescent IBD with IBS-like symptoms. After the recovery, mice were randomized to 2-week low-(0.51 g) or high-FODMAP (4.10 g) diets, respectively. In the positive-control and post-inflammatory treatment groups following the LFD, total levels of stool BCFAs were higher compared with those of the negative controls; statistical significance was reached for isobutyric and isovaleric acid. The results suggested that the higher proteolytic fermentation occurred in the LFD group. Considering the alterations of inflammatory markers, Myeloperoxidase (MPO) activity was lower in the negative-control group, in contrast to its higher levels in the positive-control group, regardless of the FODMAPs content. The research team created a histological score to evaluate the inflammatory status in the colon. The negative controls scored 0, but positive controls had higher scores only in the high-FODMAP diet group. Even if compared with the post-inflammatory group, positive control animals fed with a high-FODMAP diet had significantly higher histological scores. However, no significant differences were observed between low- and high-FODMAP diets. There were no significant changes regarding TNF-α, Granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-10, and IL-4. The same research team evaluated commercially available rodent diets across research institutions. Forty mice were randomized into four groups to assess the dietary impact on gut microbiota, SCFAs, and BCFAs profiles. Animals in Group A were euthanized at baseline (controls), mice in Group B received the breeding institution chow, mice in Group C received a low-gluten and LFD, and Group D consumed a high-gluten and FODMAP diet. In the LFD group, the BCFA stool levels were higher compared with those in the high-FODMAP group. More specifically, isovalerate and isobutyric acids levels were highest in the LFD group. The explanation is based on protein fermentation and reflects a trend for increased protein metabolism in low-carbohydrate diets.2.2.2. Clinical Trials
Halmos et al. [33][53] assessed the effects of an LFD versus a typical Australian diet on fecal biomarkers. This study included 33 participants (27 with IBS and 6 healthy controls). Volunteers followed two diets differing in FODMAP content (LFD contained 3.05 g, whereas the typical Australian diet contained 23.7 g of FODMAPs, respectively). At baseline, isovaleric and isobutyric levels were lower in IBS patients compared to controls, though no difference was noticed during the dietary intervention [33][53].
Recently, Nordin and her team investigated the effects of different dietary patterns on fecal microbiota, fecal fatty acids, and plasma metabolome in IBS symptoms. One hundred and three IBS patients were randomized into three groups in this double-blinded, placebo-controlled trial. Each group followed all the dietary plans (placebo, gluten, and FODMAP) but in a different sequence. Placebo consisted of 18 g of sucrose, gluten intervention contained 17.3 g of gluten, and the daily dose of FODMAP intake was 50 g. During the intervention, patients filled in questionnaires, and they underwent blood and fecal analyses and anthropometric measurements periodically. Results showed a reduction in plasma levels of isobutyrate in the FODMAP group compared to the placebo, while in feces, a decrease was observed in isovalerate after the gluten diet.
