Allergic diseases are one of the most common chronic conditions and their prevalence is on the rise. Environmental exposure, primarily prenatal and early life influences, affect the risk for the development and specific phenotypes of allergic diseases via epigenetic mechanisms. Exposure to pollutants, microorganisms and parasites, tobacco smoke and certain aspects of diet are known to drive epigenetic changes that are essential for immune regulation (e.g., the shift toward T helper 2-Th2 cell polarization and decrease in regulatory T-cell (Treg) differentiation).
- allergy
- epigenetic modifications
- environmental exposure
1. Introduction
2. Environmental Exposure and Epigenetic Regulation in Allergic Diseases
2.1. The Effect of Maternal and In Utero Exposure on Epigenetic Modifications
Maternal exposures, such as cigarette smoke, have already been shown to alter fetal lung development and immune function, contributing to an increased risk of respiratory disease [8][9][10][20,21,22]. Significant intrauterine exposures, including maternal diet and microbial exposure, are known to alter the risk of allergic disease in offspring. Studies show that prenatal exposures (diet, microbial infections, tobacco smoke and other pollutants) can epigenetically activate or silence immune-related genes, with significant effects on immune programming [11][12][13][14][23,24,25,26]. DNA methylation and histone modifications may control immune programming related to pro-allergic IL-4, IL-13 or counter-regulatory IFN-γ production [15][16][17][18][27,28,29,30]. Infections in the mother, the use of antibiotics, the consumption of raw or unprocessed cow’s milk and contact with molds and pets have also been associated with an increased risk of allergic respiratory diseases. Certain aspects of maternal diet, maternal stress and exposure to pollutants, improved hygiene, inadequate exposure to microorganisms and the modern Westernized diet can affect cellular homeostasis, and these factors can affect epigenetic regulation [19][20][35,36]. A schematic representation of the effects of environmental exposure on epigenetic changes and the pathophysiology of allergic diseases is shown in Figure 1.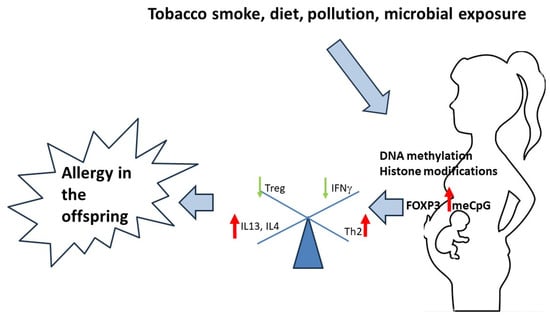
2.2. Exposure to Tobacco Smoke
2.3. Epigenetic Modifications and the Effects of Diet
One of the first epigenetic models in allergic diseases focused on the role of folic acid in the pathogenesis of these conditions, based on the ability of folic acid to epigenetically alter gene expression through its role as a methyl donor for DNA [24][13]. Supplementation of pregnant mice with a diet rich in folate resulted in altered gene methylation patterns and decreased transcriptional activity in the lung tissue of the offspring, with increased airway hyperresponsiveness, airway eosinophilia and production of chemokine and macrophage inflammatory proteins [25][41]. One of the genes involved was runt-related transcription factor 3, which has a protective role in the development of respiratory diseases via the induction of FOXP3+ Treg cells. Conversely, several studies reported that folic acid supplementation during pregnancy was associated with an increased risk of asthma in infants. However, the results of these studies should be considered in the context of other related nutrients, such as vitamins B2, B6, B12, methionine and choline [26][27][42,43]. The consumption of polyunsaturated fatty acids (PUFA) may influence epigenetic changes and contribute to protection against allergen sensitization or the development of allergic diseases [28][44]. Maternal vitamin D supplementation induces changes in DNA methylation in infants, specifically genes involved in collagen metabolism and apoptosis.2.4. Effects of Microbial Exposure to Epigenetic Regulation in Allergic Diseases
Recently, the interaction between the external and internal (for example, gut microbiome) environments and epigenetic regulation has become a special focus in studying the pathophysiologic mechanisms of allergic diseases. A number of studies demonstrated the relationship between changes in the biodiversity of gut, skin and airway microbiota with allergic diseases [29][30][31][46,47,48]. Moreover, the microbiome influences epigenetic regulation, cell differentiation and polarization, as well as homeostasis [32][49]. Short-chain fatty acids produced by commensal microorganisms may serve as signaling molecules in the processes of DNA methylation and histone modifications [33][50]. Although early postnatal exposure (mostly in the first six weeks of life) is considered the most significant source of direct microbial exposure to the developing infant, it is becoming increasingly clear that the epigenetic influences of bacteria are already substantial in utero. In humans, exposure to a higher degree of microbial load in rural environments has been proven to have a protective role against the development of allergic diseases [34][51].2.5. Exposure to Air Pollution and Epigenetic Effects in Allergic Diseases
Oxidative stress resulting from exposure to tobacco smoke and air pollution can have significant epigenetic effects by altering nuclear factor kB (NF-kB) activation or histone modification and chromatin remodeling of proinflammatory genes. As a potent source of oxidative stress, tobacco smoke (cigarette smoke) contributes to decreased histone deacetylase (HDAC) activity, leading to differential activation of NF-kB and expression of the proinflammatory cytokines IL-6 and IL-8 in peripheral lung tissue [35][12]. NF-kB is ubiquitously expressed in various cell types and can induce histone modifications that activate or silence inflammatory genes and other signal transduction pathways. During pregnancy, the induction of inflammatory genes can affect placental function and fetal programming. Oxidative stress resulting from chronic and high levels of exposure to traffic exhaust may also cause epigenetic changes during pregnancy.2.6. Exposure to Persistent Organic Pollutants and Epigenetic Effects
Organic products from industry and agriculture (including polychlorinated biphenyl compounds, organochlorine pesticides, dioxins and phthalates) contaminate modern homes, food, clothing and water sources and accumulate in human tissues with age. Although they have immunosuppressive effects in humans at high doses, low concentrations of these pollutants may more selectively inhibit Th1 immune responses and promote allergic Th2 immune responses through their “estrogenic” hormonal activity [36][60]. Some of these products have already been detected in breast milk, umbilical cord blood and placental tissue, indicating that they may affect early development. Studies have shown that persistent organic pollutants (especially organochlorine pesticides) are present in 94% of fat samples from mothers who have undergone caesarean section and in 62% of breast milk samples, which may be associated with immune reactions in both the mother and the newborn, especially the allergic inflammatory response [37][38][11,61]. Pesticides and other pollutants have also been linked to epigenetic effects, including differential methylation specific for each active ingredient (pesticide-specific methylation signals) and positive epigenetic age acceleration [39][40][41][62,63,64].2.7. Other Intrauterine Factors That Affect Epigenetic Modifications and Allergic Disease Risk
In the case of allergic diseases, the maternal phenotype is a known risk factor for diseases in the child. Maternal allergic status has a much stronger influence on allergic diseases and Th1 IFN-γ production in the newborn than paternal allergies [42][65]. Studies have also shown that maternal allergy status alters immune interactions between the mother and the fetus and reduces Th1 activation to the human leukocyte antigen mismatch of fetal alloantigens [43][66]. Many other changes in the intrauterine environment can directly affect gene expression in the placenta and potentially alter the offspring’s phenotype. Pre-eclampsia, the use of corticosteroids (and other medications, such as antibiotics) and stress have been associated with epigenetic changes affecting gene expression, placental immune function, growth retardation and congenital disabilities [44][67]. Maternal stress is critical for gene expression in the placenta, as adrenal glucocorticoid production modulates inflammatory gene expression via the hypothalamic–pituitary–adrenal axis, with recognized effects on glutathione metabolism and DNA methylation [45][68]. Inflammatory diseases during pregnancy can also alter the immune function of the placenta through the sex-specific production of cortisol [46][69]. The early induction of innate inflammatory genes in newborns (including IL-1b and TNFα) is closely linked to the later development of allergic diseases. These inflammatory mediators, important for immune programming, induce histone modifications and may be responsible for the alternative programming of neonatal immunity toward a pro-allergic response [47][70].2.8. Early Life Events and Epigenetic Changes Affecting Allergic Diseases
After birth, environmental influences also affect local events in the developing airways. Lower respiratory tract infections associated with wheezing in the first year of life are a crucial risk factor for asthma development at the age of six, both in non-atopic and atopic children [48][49][73,74]. This suggests that viral-induced inflammation of the lower airways early in the postnatal development can have profound long-term effects that are more pronounced than events later in life. Studies have shown that RSV infection in infancy leads to alterations in histone methylation, resulting in decreased proinflammatory cytokine production and an increase in Th2 cytokine production. Additionally, RSV infection alters DNA methylation of certain genes within the TGF-b superfamily, resulting in increased polarization of T cells toward a Th2 and Th17 phenotype.2.9. Effects of Exposure to Heavy Metals on Epigenetic Changes in Allergy
Heavy metals, such as mercury, cadmium, arsenic, chromium, nickel, copper and lead, are well-established environmental pollutants, due to their longevity in the ecosystem, toxicity and low doses required to exhibit a toxic effect and, most importantly, due to their bioaccumulation in organisms over time. Exposure to heavy metals, especially in utero and during childhood, is known to be harmful for human health, having both direct toxic and cumulative hazardous effects in the development of chronic conditions. Exposure to heavy metals from water, air or food has been linked to neurological and cardiovascular diseases, cancer, diabetes, hepatotoxicity, nephrotoxicity and immune disorders. It also causes substantial epigenetic changes, and these effects are persistent, even lifelong. Exposure to chromium, mercury, nickel, arsenic and lead is known to cause both gene-specific and global DNA methylation changes [50][51][52][81,82,83], and it affects all types of histone modifications (acetylation, phosphorylation, ubiquitination and methylation). Exposure to arsenic has been known to cause shifts in the immune responses toward a proinflammatory state [53][84]。2.10. The Changing Environment and Epigenetic Changes in Allergic Diseases
In the past decades, the rapid and growing climate changes have paralleled the several-fold increase in the prevalence of allergic diseases. Climate changes include global warming due to greenhouse gases coming from various human activities (such as industry, traffic and deforestation), pollution (air, water and soil), and increases in the frequency of extreme weather events (floods, storms, wild fires, heat waves, droughts, snowstorms and blizzards, etc.), which lead to reduced biodiversity and health hazards, affecting both the prevalence of infectious diseases due to their effects on the food and water supply and the prevalence of non-communicable diseases, with allergies being one of the most prominent.2.11. Expression of Small Non-Coding RNAs and Allergic Diseases
In patients with allergic diseases, miRNAs play a crucial role in the development, differentiation, maturation and activation of immune cells, and remodeling and deregulation of the airways [54][93]. There is evidence that miRNA expression differs in tissues, cells and biological fluids of allergic and healthy patients [55][56][57][58][94,95,96,97]. Since it is unclear whether the expression of non-coding RNA (ncRNA) molecules (miRNA, siRNA and long non-coding RNA) is transmissible across generations, it is more likely they serve as a more direct regulatory mechanism for gene expression patterns. For example, studies have indicated that let-7 miRNAs (a highly conserved group of at least nine miRNAs known to be involved in Toll-like receptor 4 (TLR4) signaling) may play a proinflammatory role, as targeted inhibition of let-7 miRNA suppressed Th2 cytokine production, lung eosinophil recruitment and airway hyperreactivity in in vivo models [59][98].3. Epigenetic Changes and Treatment Outcomes in Allergic Diseases
Different allergic diseases have different clinical manifestations within the same epithelial barrier organ (phenotypes), driven by distinct pathophysiological and molecular endotypes. Phenotyping of inflammatory profiles aims at enabling personalized treatment of allergic diseases. Identifying epigenetic changes associated with these phenotypes and endotypes can contribute to better management and possible prevention of allergic diseases, to the assessment of tolerance after immunotherapy and to the prediction of treatment success in early phases [1][60][61][62][1,105,106,107]. It is also evident that both stable and transient epigenetic modifications (DNA methylation, histone modifications and ncRNA expression) mediate environmental exposure in the development of asthma and the clinical features of the disease [37][11]. Therefore, epigenetic regulation has emerged as a potential mechanism for the effect of asthma-related pharmacological therapies. For example, inhaled corticosteroids (ICS), which have been used for decades to treat inflammation in acute and chronic asthma and chronic obstructive pulmonary disease (COPD), are thought to act, at least in part, via certain epigenetic modifications, such as histone acetylation and, according to recent studies, miRNA expression [63][64][108,109]. Corticosteroids bind intracellularly to glucocorticoid receptors, which are activated and bind to glucocorticoid response elements in the promoter regions of glucocorticoid-responsive genes. Corticosteroids exert their anti-inflammatory effects in part by inducing histone acetylation of anti-inflammatory genes (e.g., mitogen-activated protein kinase phosphatase-1 (MKP-1)) and by recruiting histone deacetylases (HDAC2) and causing deacetylation of proinflammatory genes (e.g., IL-8, NF-κB and activator protein-1 (AP-1)).4. Environmental Exposure, Epigenetic Changes and Protection from Allergy
Microbial exposure, both early in the postnatal development or in utero, seems to greatly affect the offspring’s susceptibility to allergic diseases. Other than microbial exposure, infections with other protozoa and parasites, such as helminths, seem to have an important role in immune modulation and development. This is reinforced by the fact that a great part of the immune responses involved in the defense from parasitic infections and allergic diseases overlap. Certain pattern recognition pattern members participate in the response to parasitic (e.g., helminth) infection and are responsible for driving T-cell polarization. Adult helminths secrete certain antigens that are known to activate a number of immune effector cells, including basophils, mast cells, eosinophils, Th2 cells and innate lymphocyte T cells 2 (ILC2), as well as induce the production of innate and adaptive cytokines. Additionally, lipids derived from helminths can stimulate the production of a number of inflammatory cytokines, such as TNFα, IL-6, IL-8, IL-10 and IL-12.
Other than differential DNA methylation, other epigenetic modifications have been associated with protection against the development of allergic diseases or the development of immune tolerance, such as decreased histone acetylation of Th2 genes in a murine food allergy study assessing acquired immune tolerance associated with raw milk consumption.
