Microorganisms have a close relationship with humans, whether it is commensal, symbiotic, or pathogenic. It has been documented that microorganisms may influence the response to drug therapy. Pharmacomicrobiomics is an emerging field that focuses on the study of how variations in the microbiome affect the disposition, action, and toxicity of drugs. Two additional sciences have been added to complement pharmacomicrobiomics, namely toxicomicrobiomics, which explores how the microbiome influences drug metabolism and toxicity, and pharmacoecology, which refers to modifications in the microbiome as a result of drug administration. Additionally, the concept of "drug-infection interaction" is included to describe the influence of pathogenic microorganisms on drug response. This entry analyzes in detail each of these concepts.
- pharmacomicrobiomics
- toxicomicrobiomics
- pharmacoecology
- drug–infection interaction
- microbiome
1. Introduction
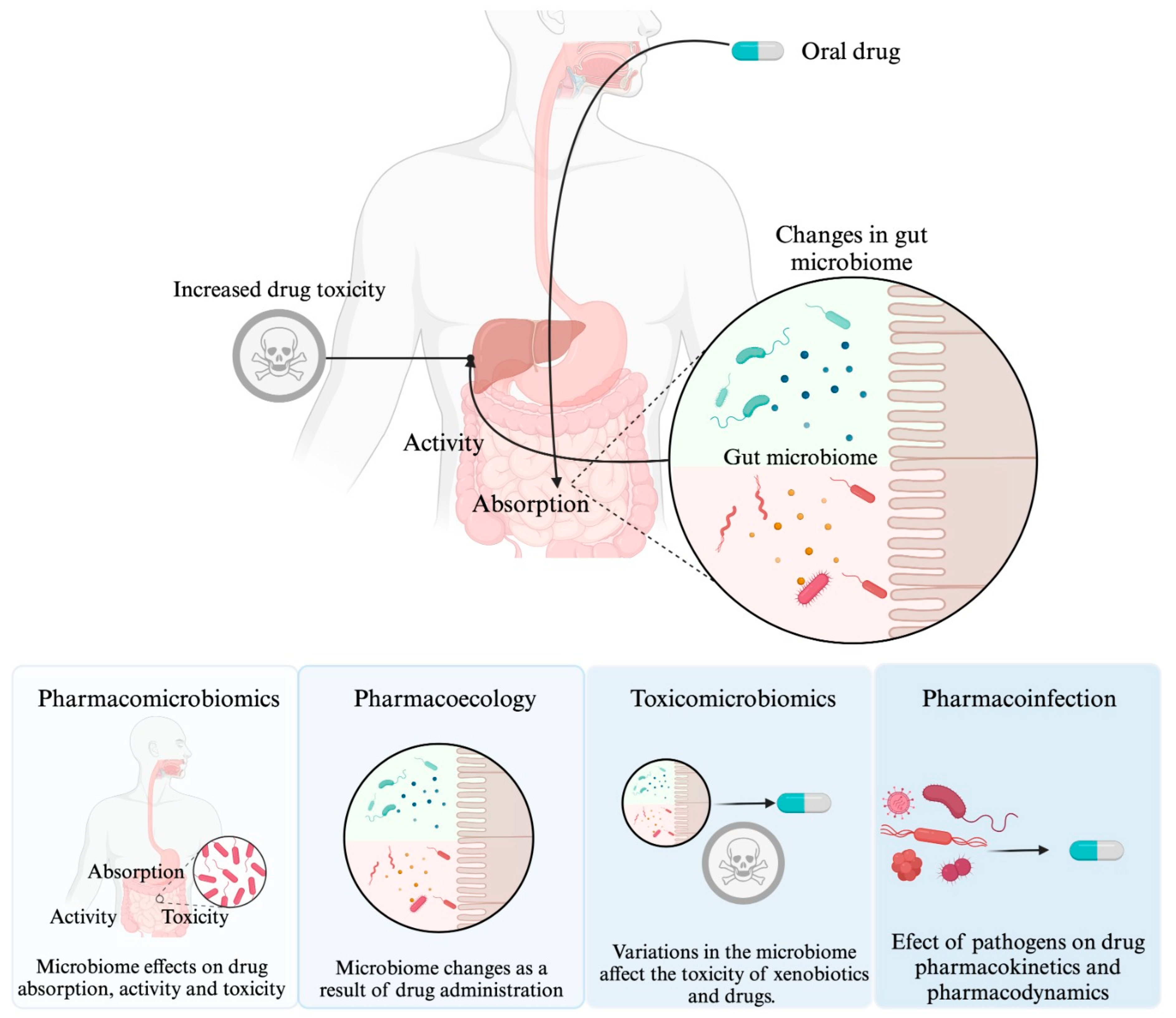
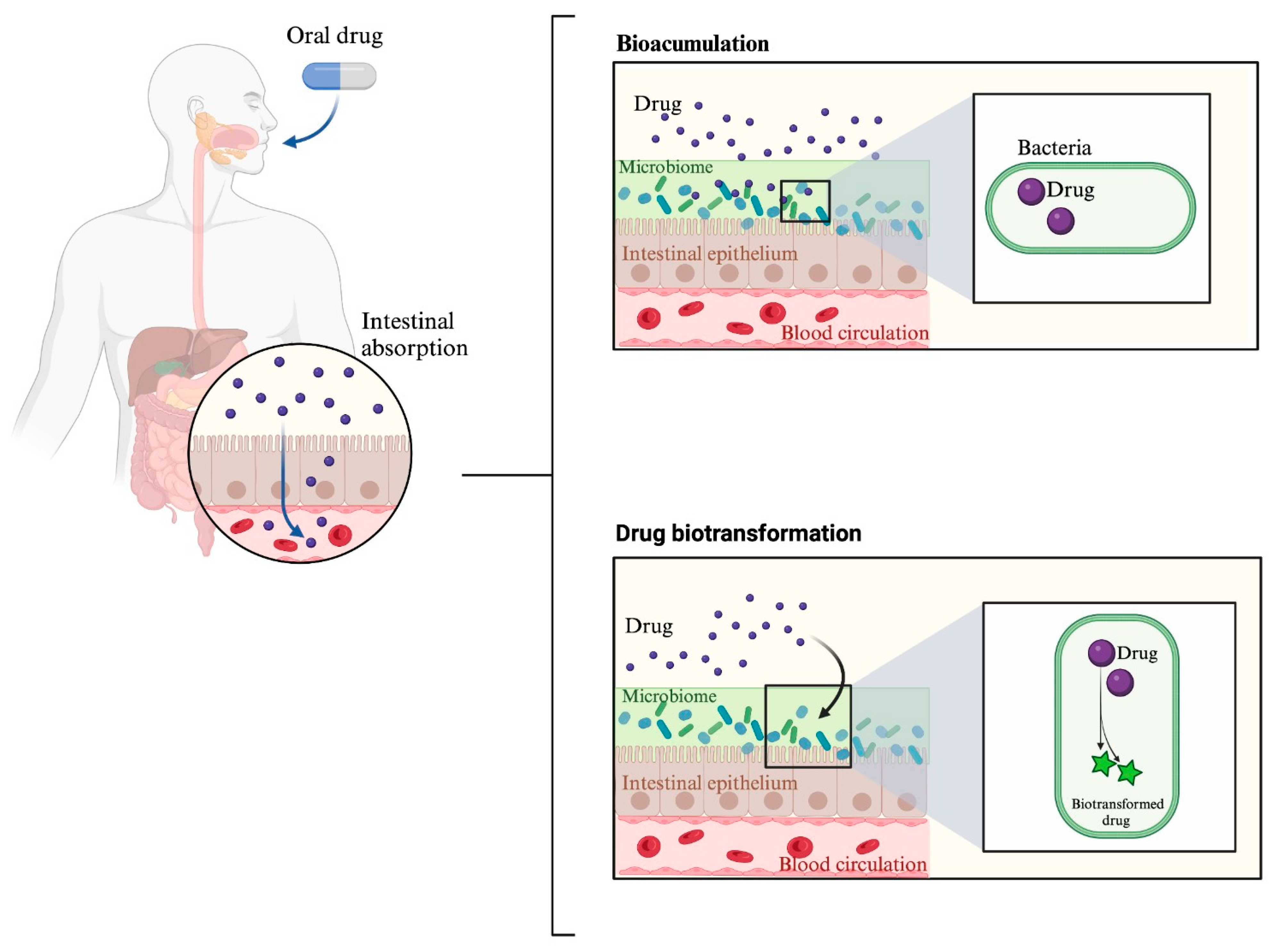
2. Effect of the Microbiome on Drug Response: “Pharmacomicrobiomics”
|
Drug Involved |
Affected Process |
Associated Microorganisms |
Clinical Effect |
Condition for Which Studied |
Organism in Which the Study Was Conducted |
|---|---|---|---|---|---|
|
Chemotherapy and/or immunotherapy |
Not described |
L. mucosae and L. salivarius |
Favorable response |
Metastatic/unresectable HER2-negative gastric/gastroesophageal junction adenocarcinoma |
|
|
FOLFOX regimen |
Drug metabolism |
Akkermansia muciniphila |
Better therapeutic effect |
Colon Cancer |
|
|
Hydrochlorothiazide |
Bioaccumulation |
Gram-negative enterobacteriaceae |
Impair glucose tolerance |
Metabolic control |
|
|
Mycophenolate mofetil |
Drug metabolism |
Bacteroides vulgatus, Bacteroides stercoris and Bacteroides thetaiotaomicron |
Graft-versus-host disease risk reduction |
Transplantation |
|
|
Simvastatin |
Drug metabolism and bioaccumulation |
Probiotic bacteria |
Alteration of simvastatin bioavailability and therapeutic effect |
Metabolic control |
|
|
Statins |
Drug metabolism |
Bacteroides |
Intense statin responses |
Metabolic control |
3. Effect of an Infection on Drug Response
3.1. Difference between Pharmacomicrobiomics and Drug–Infection Interaction
There are two fundamental differences between pharmacomicrobiomics and drug–infection interaction. The first one lies in the type of microorganism that influences the drug response. In pharmacomicrobiomics, the microbiome is the protagonist, whereas in drug–infection interaction, it involves a pathogenic microorganism [1013][2932][3033]. Pathogenic microorganisms are those capable of causing diseases, as they are transmissible and, in some cases, have developed the ability to evade cellular defenses. Only a small percentage of microbes are inherently pathogenic. Pathogenic microorganisms include some viruses, bacteria, prions, fungi, protozoa, and parasites [3033]. The second difference is that, unlike pharmacomicrobiomics, which modifies the drug response through bioaccumulation or metabolism, infections can alter the drug response mainly through inflammation and the regulation of CYP enzymes [1114][3134][3235].3.2. Inflammation as a Result of Infection Modifies Drug Response
Inflammation is a response to aggression, whether of endogenous or exogenous origin, and can manifest acutely or chronically. It plays a prominent role in numerous diseases, including infections [3336]. While inflammation is a complex and highly coordinated process involving multiple cell types and molecules operating in a cascading network, cytokines play a particularly relevant role in this process [3437]. For several years, it has been proven that inflammation has a significant impact on drug metabolism. This is partly because elevated levels of proinflammatory cytokines lead to a negative regulation of CYP enzymes, which play a fundamental role in drug metabolism [3134][3538][3639]. CYP enzymes are polymorphic proteins associated with a heme molecule and are capable of absorbing light at a wavelength of approximately 450 nm when exposed to carbon monoxide. These enzymes play an essential role in the biosynthesis of compounds such as steroids, prostacyclin, and thromboxane A2. While CYP enzymes are found in a wide variety of tissues, their expression is most prominent in the liver and small intestine. Regarding drug metabolism, a specific group of CYP enzymes, including CYP 1A2, 2B6, 2D6, 2C8, 2C9, 2C19, and 3A4, are responsible for metabolizing most drugs [3134][3740]. CYP enzymes during inflammation can be repressed by different mechanisms. These include the transcriptional downregulation of transcription factors, interference with nuclear transcription factor dimerization and translocation, alteration of C/EBP-enriched signaling in the liver, direct regulation by NF-κB, and various post-transcriptional mechanisms [3639]. It has recently been proposed that the reduction in CYP enzyme activity during inflammation, in the context of an infection, is due to a physiological response. This response involves a shift from a metabolic mode to a defensive mode, allowing the cell to concentrate its resources on fighting the infection [3841]. Regardless of the physiological cause, infections impact CYP enzyme activity due to the inflammatory process they trigger [3336][3942][4043]. A prominent example of this is the disease caused by SARS-CoV-2, known as COVID-19. This disease follows a progression divided into three stages: the viral invasion phase, the pulmonary immunoinflammatory phase, and the hyperinflammatory phase. Inflammation is its distinguishing feature, marked by increased NF-κB signaling, which in turn induces the production of proinflammatory cytokines such as IL-6, IL-2, TNF-α, and IFN-γ [4144]. In this context, it has been documented that the increased proinflammatory cytokines induced by COVID-19 impact drug metabolism as they interfere with the regulation of CYP enzymes and drug transporter expression [3942][4245][4346]. In humans, it has been observed that inflammation caused by COVID-19 reduces CYP3A activity, which, in turn, affects the metabolism of midazolam. Furthermore, two independent studies consistently reported abnormally elevated levels of lopinavir and ritonavir in COVID-19 patients, suggesting that this could be due to the negative regulation of CYP3A [4447][4548][4649]. Regarding COVID-19, the response to treatment is not solely related to the disease itself. In 2021 and 2022, two cases were reported, in which increased levels and adverse effects of clozapine were observed in patients who had been vaccinated against COVID-19. These specific vaccines were Moderna’s Spikevax and Pfizer-BioNTech’s vaccine. In both cases, the adverse reaction was associated with inflammation and CYP1A2 activity. It is important to note that this adverse reaction was short lived [4750][4851]. In the case of the human immunodeficiency virus (HIV), it has been described to affect CYP enzyme levels. People infected with HIV have shown a reduction in hepatic CYP3A4 and CYP2D6 enzyme activity compared to uninfected individuals [4952][5053]. However, the interpretation of these findings is not entirely conclusive, as other research has not found changes in drug metabolism in HIV patients, and, in other studies, an increase in CYP3A4 expression has even been observed in HIV patients receiving antiretroviral therapy [5154][5255]. For HIV patients, future research aimed at determining the impact of infection-generated cytokines on the pharmacokinetics of antiretroviral drugs should address various variables. This includes individual gene expression, the possible co-infection of HIV with hepatitis B, the presence of liver disease, the anti-inflammatory effects of drug therapy, and study design, among other factors. A consideration of these elements is crucial for obtaining clearer and more accurate results in this area of research [3134][5154]. As evidence continues to accumulate, it is likely that closer medical monitoring may be needed in the future for patients with an infection who are also being treated with drugs metabolized by CYP enzymes to prevent possible overdoses and toxicity.3.3. Other Mechanisms by Which Infections May Affect Drug Response
3.3.1. Alterations in Gastrointestinal Motility and Drug Absorption
It has been proposed that gastrointestinal infections may affect the availability of certain drugs due to various factors, such as changes in intestinal transit speed or the pH of gastrointestinal fluids; however, information is limited [5356][5457]. In the context of alterations in intestinal transit caused by an infection, it is important to consider infectious diarrhea. While this condition can influence the absorption of a medication, its specific impact can vary considerably, depending on several factors, including the severity of the diarrhea, its duration, the overall health of the individual, and the underlying infectious agent, as infectious diarrhea can be caused by viral, bacterial, or parasitic infections [5558]. Overall, the evidence supporting the impact of an infection that causes diarrhea on drug absorption is limited. As a result of this research, very few studies directly addressed the impact of infectious diarrhea on drug absorption. The lack of research directly addressing the mechanisms by which infectious diarrhea affects drug absorption is largely due to the fact that most patients with acute diarrhea typically present mild and transient symptoms. Additionally, in severe cases of diarrhea, the priority is to immediately address the clinical condition rather than evaluating whether diarrhea might modify the absorption of a prescribed medication [5659]. Further research is needed to fully understand the impact of infectious diarrhea on drug absorption and its implications in clinical practice.3.3.2. Pharmacological Effect Mimicry
A poorly described mechanism by which an infection might modify the response to a drug is through mimicry of the drug effect. An example of this is human adenovirus 36 (HAdV-36), which has been associated with obesity and changes in glucose and lipid metabolism, with long-term effects, such as the irreversible expansion of adipose tissue, even after the resolution of the acute phase of infection [5760]. HAdV-36 increases peroxisome proliferator-activated receptor-γ (PPAR-γ) expression in the same way as thiazolidinediones, which are used to increase insulin sensitization in patients with type 2 diabetes mellitus [5861][5962][6063]. This virus could potentially influence the response to drugs used in lipid or glucose control, although there is no evidence so far that HAdV-36 modifies the response to drugs such as metformin [6164]. To the researchers' knowledge, there is no other virus that can mimic the effect of a drug.3.3.3. Unknown Mechanisms: The Case of Helicobacter pylori and Levodopa
Helicobacter pylori (HP) has been documented to cause inflammation at the intestinal level, delay gastric emptying, and possibly affect the absorption of drugs, such as Levodopa [6265]. In this context, it has been observed that people with Parkinson’s disease who have HP infection show a poor response to Levodopa and experience increased severity of motor symptoms [6366]. However, the elimination of HP has been reported to improve tremor, although it does not change the bioavailability of the drug [6467]. All this information underscores the need for further research to understand how HP influences drug interactions with drugs, such as Levodopa, and other potential drugs.References
- Patel, R.I.; Beckett, R.D. Evaluation of Resources for Analyzing Drug Interactions. J. Med. Libr. Assoc. 2016, 104, 290–295.
- Zeitlinger, M. Drug Interactions. In Clinical Pharmacology: Current Topics and Case Studies; Springer International Publishing: Cham, Switzerland, 2016; pp. 265–292.
- Rai, G.S.; Rozario, C.J. Mechanisms of Drug Interactions II: Pharmacokinetics and Pharmacodynamics. Anaesth. Intensive Care Med. 2023, 24, 217–220.
- Pichini, S.; Di Trana, A.; García-Algar, O.; Busardò, F.P. Editorial: Drug-Drug Interactions in Pharmacology. Front. Pharmacol. 2023, 14, 1155738.
- Hannachi, N.; Camoin-Jau, L. Drug Response Diversity: A Hidden Bacterium? J. Pers. Med. 2021, 11, 345. Heirali, A.; Moossavi, S.; Arrieta, M.C.; Coburn, B. Principles and Terminology for Host–Microbiome–Drug Interactions. Open Forum Infect. Dis. 2023, 10, ofad195.
- Perez, N.B.; Dorsen, C.; Squires, A. Dysbiosis of the Gut Microbiome: A Concept Analysis. J. Holist. Nurs. 2020, 38, 223–232. Attia, H.; ElBanna, S.A.; Khattab, R.A.; Farag, M.A.; Yassin, A.S.; Aziz, R.K. Integrating Microbiome Analysis, Metabolomics, Bioinformatics, and Histopathology to Elucidate the Protective Effects of Pomegranate Juice against Benzo-Alpha-Pyrene-Induced Colon Pathologies. Int. J. Mol. Sci. 2023, 24, 10691.
- Thomson, P.D.; Smith, D.J. What Is Infection? Am. J. Surg. 1994, 167, S7–S11. Aziz, R.K.; Hegazy, S.M.; Yasser, R.; Rizkallah, M.R.; ElRakaiby, M.T. Drug Pharmacomicrobiomics and Toxicomicrobiomics: From Scattered Reports to Systematic Studies of Drug–Microbiome Interactions. Expert. Opin. Drug Metab. Toxicol. 2018, 14, 1043–1055.
- Rizkallah, M.; Saad, R.; Aziz, R. The Human Microbiome Project, Personalized Medicine and the Birth of Pharmacomicrobiomics. Curr. Pharmacogenom. Person. Med. 2010, 8, 182–193. Hannachi, N.; Camoin-Jau, L. Drug Response Diversity: A Hidden Bacterium? J. Pers. Med. 2021, 11, 345.
- Alqahtani, M.S.; Kazi, M.; Alsenaidy, M.A.; Ahmad, M.Z. Advances in Oral Drug Delivery. Front. Pharmacol. 2021, 12, 618411. Perez, N.B.; Dorsen, C.; Squires, A. Dysbiosis of the Gut Microbiome: A Concept Analysis. J. Holist. Nurs. 2020, 38, 223–232.
- Peretti, S.; Torracchi, S.; Russo, E.; Bonomi, F.; Fiorentini, E.; El Aoufy, K.; Bruni, C.; Lepri, G.; Orlandi, M.; Chimenti, M.S.; et al. The Yin-Yang Pharmacomicrobiomics on Treatment Response in Inflammatory Arthritides: A Narrative Review. Genes 2022, 14, 89. Thomson, P.D.; Smith, D.J. What Is Infection? Am. J. Surg. 1994, 167, S7–S11.
- Conti, G.; D’Amico, F.; Fabbrini, M.; Brigidi, P.; Barone, M.; Turroni, S. Pharmacomicrobiomics in Anticancer Therapies: Why the Gut Microbiota Should Be Pointed Out. Genes 2022, 14, 55. Rizkallah, M.; Saad, R.; Aziz, R. The Human Microbiome Project, Personalized Medicine and the Birth of Pharmacomicrobiomics. Curr. Pharmacogenom. Person. Med. 2010, 8, 182–193.
- Klünemann, M.; Andrejev, S.; Blasche, S.; Mateus, A.; Phapale, P.; Devendran, S.; Vappiani, J.; Simon, B.; Scott, T.A.; Kafkia, E.; et al. Bioaccumulation of Therapeutic Drugs by Human Gut Bacteria. Nature 2021, 597, 533–538. Alqahtani, M.S.; Kazi, M.; Alsenaidy, M.A.; Ahmad, M.Z. Advances in Oral Drug Delivery. Front. Pharmacol. 2021, 12, 618411.
- Luo, J.-Q.; Ren, H.; Chen, M.-Y.; Zhao, Q.; Yang, N.; Liu, Q.; Gao, Y.-C.; Zhou, H.-H.; Huang, W.-H.; Zhang, W. Hydrochlorothiazide-Induced Glucose Metabolism Disorder Is Mediated by the Gut Microbiota via LPS-TLR4-Related Macrophage Polarization. iScience 2023, 26, 107130. Peretti, S.; Torracchi, S.; Russo, E.; Bonomi, F.; Fiorentini, E.; El Aoufy, K.; Bruni, C.; Lepri, G.; Orlandi, M.; Chimenti, M.S.; et al. The Yin-Yang Pharmacomicrobiomics on Treatment Response in Inflammatory Arthritides: A Narrative Review. Genes 2022, 14, 89.
- de la Cuesta-Zuluaga, J.; Mueller, N.T.; Corrales-Agudelo, V.; Velásquez-Mejía, E.P.; Carmona, J.A.; Abad, J.M.; Escobar, J.S. Metformin Is Associated with Higher Relative Abundance of Mucin-Degrading Akkermansia muciniphila and Several Short-Chain Fatty Acid–Producing Microbiota in the Gut. Diabetes Care 2017, 40, 54–62. Conti, G.; D’Amico, F.; Fabbrini, M.; Brigidi, P.; Barone, M.; Turroni, S. Pharmacomicrobiomics in Anticancer Therapies: Why the Gut Microbiota Should Be Pointed Out. Genes 2022, 14, 55.
- Choi, U.; Lee, C.-R. Distinct Roles of Outer Membrane Porins in Antibiotic Resistance and Membrane Integrity in Escherichia coli. Front. Microbiol. 2019, 10, 952. Klünemann, M.; Andrejev, S.; Blasche, S.; Mateus, A.; Phapale, P.; Devendran, S.; Vappiani, J.; Simon, B.; Scott, T.A.; Kafkia, E.; et al. Bioaccumulation of Therapeutic Drugs by Human Gut Bacteria. Nature 2021, 597, 533–538.
- Scheline, R.R. Drug Metabolism by Intestinal Microorganisms. J. Pharm. Sci. 1968, 57, 2021–2037. Luo, J.-Q.; Ren, H.; Chen, M.-Y.; Zhao, Q.; Yang, N.; Liu, Q.; Gao, Y.-C.; Zhou, H.-H.; Huang, W.-H.; Zhang, W. Hydrochlorothiazide-Induced Glucose Metabolism Disorder Is Mediated by the Gut Microbiota via LPS-TLR4-Related Macrophage Polarization. iScience 2023, 26, 107130.
- Jourova, L.; Anzenbacher, P.; Anzenbacherova, E. Human Gut Microbiota Plays a Role in the Metabolism of Drugs. Biomed. Pap. 2016, 160, 317–326. de la Cuesta-Zuluaga, J.; Mueller, N.T.; Corrales-Agudelo, V.; Velásquez-Mejía, E.P.; Carmona, J.A.; Abad, J.M.; Escobar, J.S. Metformin Is Associated with Higher Relative Abundance of Mucin-Degrading Akkermansia muciniphila and Several Short-Chain Fatty Acid–Producing Microbiota in the Gut. Diabetes Care 2017, 40, 54–62.
- Dempsey, J.L.; Cui, J.Y. Microbiome Is a Functional Modifier of P450 Drug Metabolism. Curr. Pharmacol. Rep. 2019, 5, 481–490. Choi, U.; Lee, C.-R. Distinct Roles of Outer Membrane Porins in Antibiotic Resistance and Membrane Integrity in Escherichia coli. Front. Microbiol. 2019, 10, 952.
- Collins, S.L.; Patterson, A.D. The Gut Microbiome: An Orchestrator of Xenobiotic Metabolism. Acta Pharm. Sin. B 2020, 10, 19–32. Scheline, R.R. Drug Metabolism by Intestinal Microorganisms. J. Pharm. Sci. 1968, 57, 2021–2037.
- Nelson, D.R. Cytochrome P450 Diversity in the Tree of Life. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2018, 1866, 141–154. Jourova, L.; Anzenbacher, P.; Anzenbacherova, E. Human Gut Microbiota Plays a Role in the Metabolism of Drugs. Biomed. Pap. 2016, 160, 317–326.
- Murphy, C.D. Drug Metabolism in Microorganisms. Biotechnol. Lett. 2015, 37, 19–28. Dempsey, J.L.; Cui, J.Y. Microbiome Is a Functional Modifier of P450 Drug Metabolism. Curr. Pharmacol. Rep. 2019, 5, 481–490.
- Jeffreys, L.N.; Poddar, H.; Golovanova, M.; Levy, C.W.; Girvan, H.M.; McLean, K.J.; Voice, M.W.; Leys, D.; Munro, A.W. Novel Insights into P450 BM3 Interactions with FDA-Approved Antifungal Azole Drugs. Sci. Rep. 2019, 9, 1577. Collins, S.L.; Patterson, A.D. The Gut Microbiome: An Orchestrator of Xenobiotic Metabolism. Acta Pharm. Sin. B 2020, 10, 19–32.
- Han, Z.; Cheng, S.; Dai, D.; Kou, Y.; Zhang, X.; Li, F.; Yin, X.; Ji, J.; Zhang, Z.; Wang, X.; et al. The Gut Microbiome Affects Response of Treatments in HER2-negative Advanced Gastric Cancer. Clin. Transl. Med. 2023, 13, e1312. Nelson, D.R. Cytochrome P450 Diversity in the Tree of Life. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2018, 1866, 141–154.
- Hou, X.; Zhang, P.; Du, H.; Chu, W.; Sun, R.; Qin, S.; Tian, Y.; Zhang, Z.; Xu, F. Akkermansia muciniphila Potentiates the Antitumor Efficacy of FOLFOX in Colon Cancer. Front. Pharmacol. 2021, 12, 725583. Murphy, C.D. Drug Metabolism in Microorganisms. Biotechnol. Lett. 2015, 37, 19–28.
- Saqr, A.; Carlson, B.; Staley, C.; Rashidi, A.; Al-Kofahi, M.; Kaiser, T.; Holtan, S.; MacMillan, M.; Young, J.-A.; El Jurdi, N.; et al. Reduced Enterohepatic Recirculation of Mycophenolate and Lower Blood Concentrations Are Associated with the Stool Bacterial Microbiome after Hematopoietic Cell Transplantation. Transplant. Cell Ther. 2022, 28, 372.e1–372.e9. Jeffreys, L.N.; Poddar, H.; Golovanova, M.; Levy, C.W.; Girvan, H.M.; McLean, K.J.; Voice, M.W.; Leys, D.; Munro, A.W. Novel Insights into P450 BM3 Interactions with FDA-Approved Antifungal Azole Drugs. Sci. Rep. 2019, 9, 1577.
- Đanić, M.; Pavlović, N.; Lazarević, S.; Stanimirov, B.; Vukmirović, S.; Al-Salami, H.; Mooranian, A.; Mikov, M. Bioaccumulation and Biotransformation of Simvastatin in Probiotic Bacteria: A Step towards Better Understanding of Drug-Bile Acids-Microbiome Interactions. Front. Pharmacol. 2023, 14, 1111115. Han, Z.; Cheng, S.; Dai, D.; Kou, Y.; Zhang, X.; Li, F.; Yin, X.; Ji, J.; Zhang, Z.; Wang, X.; et al. The Gut Microbiome Affects Response of Treatments in HER2-negative Advanced Gastric Cancer. Clin. Transl. Med. 2023, 13, e1312.
- Wilmanski, T.; Kornilov, S.A.; Diener, C.; Conomos, M.P.; Lovejoy, J.C.; Sebastiani, P.; Orwoll, E.S.; Hood, L.; Price, N.D.; Rappaport, N.; et al. Heterogeneity in Statin Responses Explained by Variation in the Human Gut Microbiome. Med 2022, 3, 388–405.e6. Hou, X.; Zhang, P.; Du, H.; Chu, W.; Sun, R.; Qin, S.; Tian, Y.; Zhang, Z.; Xu, F. Akkermansia muciniphila Potentiates the Antitumor Efficacy of FOLFOX in Colon Cancer. Front. Pharmacol. 2021, 12, 725583.
- Turjeman, S.; Koren, O. Using the Microbiome in Clinical Practice. Microb. Biotechnol. 2022, 15, 129–134. Saqr, A.; Carlson, B.; Staley, C.; Rashidi, A.; Al-Kofahi, M.; Kaiser, T.; Holtan, S.; MacMillan, M.; Young, J.-A.; El Jurdi, N.; et al. Reduced Enterohepatic Recirculation of Mycophenolate and Lower Blood Concentrations Are Associated with the Stool Bacterial Microbiome after Hematopoietic Cell Transplantation. Transplant. Cell Ther. 2022, 28, 372.e1–372.e9.
- Leardini, D.; Venturelli, F.; Baccelli, F.; Cerasi, S.; Muratore, E.; Brigidi, P.; Pession, A.; Prete, A.; Masetti, R. Pharmacomicrobiomics in Pediatric Oncology: The Complex Interplay between Commonly Used Drugs and Gut Microbiome. Int. J. Mol. Sci. 2022, 23, 15387. Đanić, M.; Pavlović, N.; Lazarević, S.; Stanimirov, B.; Vukmirović, S.; Al-Salami, H.; Mooranian, A.; Mikov, M. Bioaccumulation and Biotransformation of Simvastatin in Probiotic Bacteria: A Step towards Better Understanding of Drug-Bile Acids-Microbiome Interactions. Front. Pharmacol. 2023, 14, 1111115.
- Fierer, J.; Looney, D.; Pechère, J.-C. Nature and Pathogenicity of Micro-Organisms. In Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2017; pp. 4–25.e1. Wilmanski, T.; Kornilov, S.A.; Diener, C.; Conomos, M.P.; Lovejoy, J.C.; Sebastiani, P.; Orwoll, E.S.; Hood, L.; Price, N.D.; Rappaport, N.; et al. Heterogeneity in Statin Responses Explained by Variation in the Human Gut Microbiome. Med 2022, 3, 388–405.e6.
- Abdallah, Y.E.H.; Chahal, S.; Jamali, F.; Mahmoud, S.H. Drug-Disease Interaction: Clinical Consequences of Inflammation on Drugs Action and Disposition. J. Pharm. Pharm. Sci. 2023, 26, 11137. Turjeman, S.; Koren, O. Using the Microbiome in Clinical Practice. Microb. Biotechnol. 2022, 15, 129–134.
- Renton, K.W.; Knickle, L.C. Regulation of Hepatic Cytochrome P-450 during Infectious Disease. Can. J. Physiol. Pharmacol. 1990, 68, 777–781. Leardini, D.; Venturelli, F.; Baccelli, F.; Cerasi, S.; Muratore, E.; Brigidi, P.; Pession, A.; Prete, A.; Masetti, R. Pharmacomicrobiomics in Pediatric Oncology: The Complex Interplay between Commonly Used Drugs and Gut Microbiome. Int. J. Mol. Sci. 2022, 23, 15387.
- Lenoir, C.; Rollason, V.; Desmeules, J.A.; Samer, C.F. Influence of Inflammation on Cytochromes P450 Activity in Adults: A Systematic Review of the Literature. Front. Pharmacol. 2021, 12, 733935. Fierer, J.; Looney, D.; Pechère, J.-C. Nature and Pathogenicity of Micro-Organisms. In Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2017; pp. 4–25.e1.
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2018, 9, 7204–7218. Abdallah, Y.E.H.; Chahal, S.; Jamali, F.; Mahmoud, S.H. Drug-Disease Interaction: Clinical Consequences of Inflammation on Drugs Action and Disposition. J. Pharm. Pharm. Sci. 2023, 26, 11137.
- Morgan, E.T. Regulation of Cytochromes P450 During Inflammation and Infection. Drug Metab. Rev. 1997, 29, 1129–1188. Renton, K.W.; Knickle, L.C. Regulation of Hepatic Cytochrome P-450 during Infectious Disease. Can. J. Physiol. Pharmacol. 1990, 68, 777–781.
- de Jong, L.M.; Jiskoot, W.; Swen, J.J.; Manson, M.L. Distinct Effects of Inflammation on Cytochrome P450 Regulation and Drug Metabolism: Lessons from Experimental Models and a Potential Role for Pharmacogenetics. Genes 2020, 11, 1509. Lenoir, C.; Rollason, V.; Desmeules, J.A.; Samer, C.F. Influence of Inflammation on Cytochromes P450 Activity in Adults: A Systematic Review of the Literature. Front. Pharmacol. 2021, 12, 733935.
- Sudsakorn, S.; Bahadduri, P.; Fretland, J.; Lu, C. 2020 FDA Drug-Drug Interaction Guidance: A Comparison Analysis and Action Plan by Pharmaceutical Industrial Scientists. Curr. Drug Metab. 2020, 21, 403–426. Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2018, 9, 7204–7218.
- Fischer, L.; Lucendo-Villarin, B.; Hay, D.C.; O’Farrelly, C. Human PSC-Derived Hepatocytes Express Low Levels of Viral Pathogen Recognition Receptors, but Are Capable of Mounting an Effective Innate Immune Response. Int. J. Mol. Sci. 2020, 21, 3831. Morgan, E.T. Regulation of Cytochromes P450 During Inflammation and Infection. Drug Metab. Rev. 1997, 29, 1129–1188.
- Lenoir, C.; Terrier, J.; Gloor, Y.; Curtin, F.; Rollason, V.; Desmeules, J.A.; Daali, Y.; Reny, J.; Samer, C.F. Impact of SARS-CoV-2 Infection (COVID-19) on Cytochromes P450 Activity Assessed by the Geneva Cocktail. Clin. Pharmacol. Ther. 2021, 110, 1358–1367. de Jong, L.M.; Jiskoot, W.; Swen, J.J.; Manson, M.L. Distinct Effects of Inflammation on Cytochrome P450 Regulation and Drug Metabolism: Lessons from Experimental Models and a Potential Role for Pharmacogenetics. Genes 2020, 11, 1509.
- Morgan, E. Impact of Infectious and Inflammatory Disease on Cytochrome P450–Mediated Drug Metabolism and Pharmacokinetics. Clin. Pharmacol. Ther. 2009, 85, 434–438. Sudsakorn, S.; Bahadduri, P.; Fretland, J.; Lu, C. 2020 FDA Drug-Drug Interaction Guidance: A Comparison Analysis and Action Plan by Pharmaceutical Industrial Scientists. Curr. Drug Metab. 2020, 21, 403–426.
- Zoulikha, M.; Huang, F.; Wu, Z.; He, W. COVID-19 Inflammation and Implications in Drug Delivery. J. Control. Release 2022, 346, 260–274. Fischer, L.; Lucendo-Villarin, B.; Hay, D.C.; O’Farrelly, C. Human PSC-Derived Hepatocytes Express Low Levels of Viral Pathogen Recognition Receptors, but Are Capable of Mounting an Effective Innate Immune Response. Int. J. Mol. Sci. 2020, 21, 3831.
- Kumar, D.; Trivedi, N. Disease-Drug and Drug-Drug Interaction in COVID-19: Risk and Assessment. Biomed. Pharmacother. 2021, 139, 111642. Lenoir, C.; Terrier, J.; Gloor, Y.; Curtin, F.; Rollason, V.; Desmeules, J.A.; Daali, Y.; Reny, J.; Samer, C.F. Impact of SARS-CoV-2 Infection (COVID-19) on Cytochromes P450 Activity Assessed by the Geneva Cocktail. Clin. Pharmacol. Ther. 2021, 110, 1358–1367.
- Deshpande, K.; Lange, K.R.; Stone, W.B.; Yohn, C.; Schlesinger, N.; Kagan, L.; Auguste, A.J.; Firestein, B.L.; Brunetti, L. The Influence of SARS-CoV-2 Infection on Expression of Drug-Metabolizing Enzymes and Transporters in a HACE2 Murine Model. Pharmacol. Res. Perspect. 2023, 11, e01071. Morgan, E. Impact of Infectious and Inflammatory Disease on Cytochrome P450–Mediated Drug Metabolism and Pharmacokinetics. Clin. Pharmacol. Ther. 2009, 85, 434–438.
- Le Carpentier, E.C.; Canet, E.; Masson, D.; Martin, M.; Deslandes, G.; Gaultier, A.; Dailly, É.; Bellouard, R.; Gregoire, M. Impact of Inflammation on Midazolam Metabolism in Severe COVID-19 Patients. Clin. Pharmacol. Ther. 2022, 112, 1033–1039. Zoulikha, M.; Huang, F.; Wu, Z.; He, W. COVID-19 Inflammation and Implications in Drug Delivery. J. Control. Release 2022, 346, 260–274.
- Lê, M.P.; Jaquet, P.; Patrier, J.; Wicky, P.-H.; Le Hingrat, Q.; Veyrier, M.; Kauv, J.; Sonneville, R.; Visseaux, B.; Laouénan, C.; et al. Pharmacokinetics of Lopinavir/Ritonavir Oral Solution to Treat COVID-19 in Mechanically Ventilated ICU Patients. J. Antimicrob. Chemother. 2020, 75, 2657–2660. Kumar, D.; Trivedi, N. Disease-Drug and Drug-Drug Interaction in COVID-19: Risk and Assessment. Biomed. Pharmacother. 2021, 139, 111642.
- Gregoire, M.; Le Turnier, P.; Gaborit, B.J.; Veyrac, G.; Lecomte, R.; Boutoille, D.; Canet, E.; Imbert, B.-M.; Bellouard, R.; Raffi, F. Lopinavir Pharmacokinetics in COVID-19 Patients. J. Antimicrob. Chemother. 2020, 75, 2702–2704. Deshpande, K.; Lange, K.R.; Stone, W.B.; Yohn, C.; Schlesinger, N.; Kagan, L.; Auguste, A.J.; Firestein, B.L.; Brunetti, L. The Influence of SARS-CoV-2 Infection on Expression of Drug-Metabolizing Enzymes and Transporters in a HACE2 Murine Model. Pharmacol. Res. Perspect. 2023, 11, e01071.
- Knöchel, C.; Hefner, G.; Stiehl, T.; Schmidbauer, W. Elevated Clozapine Blood Concentrations After Second COVID-19 Vaccination with Spikevax (COVID-19 Vaccine Moderna). J. Clin. Psychopharmacol. 2022, 42, 317–320. Le Carpentier, E.C.; Canet, E.; Masson, D.; Martin, M.; Deslandes, G.; Gaultier, A.; Dailly, É.; Bellouard, R.; Gregoire, M. Impact of Inflammation on Midazolam Metabolism in Severe COVID-19 Patients. Clin. Pharmacol. Ther. 2022, 112, 1033–1039.
- Thompson, D.; Delorme, C.M.; White, R.F.; Honer, W.G. Elevated Clozapine Levels and Toxic Effects after SARS-CoV-2 Vaccination. J. Psychiatry Neurosci. 2021, 46, E210–E211. Lê, M.P.; Jaquet, P.; Patrier, J.; Wicky, P.-H.; Le Hingrat, Q.; Veyrier, M.; Kauv, J.; Sonneville, R.; Visseaux, B.; Laouénan, C.; et al. Pharmacokinetics of Lopinavir/Ritonavir Oral Solution to Treat COVID-19 in Mechanically Ventilated ICU Patients. J. Antimicrob. Chemother. 2020, 75, 2657–2660.
- Jones, A.E.; Brown, K.C.; Werner, R.E.; Gotzkowsky, K.; Gaedigk, A.; Blake, M.; Hein, D.W.; van der Horst, C.; Kashuba, A.D.M. Variability in Drug Metabolizing Enzyme Activity in HIV-Infected Patients. Eur. J. Clin. Pharmacol. 2010, 66, 475–485. Gregoire, M.; Le Turnier, P.; Gaborit, B.J.; Veyrac, G.; Lecomte, R.; Boutoille, D.; Canet, E.; Imbert, B.-M.; Bellouard, R.; Raffi, F. Lopinavir Pharmacokinetics in COVID-19 Patients. J. Antimicrob. Chemother. 2020, 75, 2702–2704.
- Jetter, A.; Fätkenheuer, G.; Frank, D.; Klaassen, T.; Seeringer, A.; Doroshyenko, O.; Kirchheiner, J.; Hein, W.; Schömig, E.; Fuhr, U.; et al. Do Activities of Cytochrome P450 (CYP)3A, CYP2D6 and P-Glycoprotein Differ between Healthy Volunteers and HIV-Infected Patients? Antivir. Ther. 2010, 15, 975–983. Knöchel, C.; Hefner, G.; Stiehl, T.; Schmidbauer, W. Elevated Clozapine Blood Concentrations After Second COVID-19 Vaccination with Spikevax (COVID-19 Vaccine Moderna). J. Clin. Psychopharmacol. 2022, 42, 317–320.
- Obradovic, B.; Roberts, O.; Owen, A.; Milosevic, I.; Milic, N.; Ranin, J.; Dragovic, G. Expression of CYP2B6 Enzyme in Human Liver Tissue of HIV and HCV Patients. Medicina 2023, 59, 1207. Thompson, D.; Delorme, C.M.; White, R.F.; Honer, W.G. Elevated Clozapine Levels and Toxic Effects after SARS-CoV-2 Vaccination. J. Psychiatry Neurosci. 2021, 46, E210–E211.
- Venuto, C.S.; Lim, J.; Messing, S.; Hunt, P.W.; McComsey, G.A.; Morse, G.D. Inflammation Investigated as a Source of Pharmacokinetic Variability of Atazanavir in AIDS Clinical Trials Group Protocol A5224s. Antivir. Ther. 2018, 23, 345–351. Jones, A.E.; Brown, K.C.; Werner, R.E.; Gotzkowsky, K.; Gaedigk, A.; Blake, M.; Hein, D.W.; van der Horst, C.; Kashuba, A.D.M. Variability in Drug Metabolizing Enzyme Activity in HIV-Infected Patients. Eur. J. Clin. Pharmacol. 2010, 66, 475–485.
- Parsons, R.L. Drug Absorption in Gastrointestinal Disease with Particular Reference to Malabsorption Syndromes. Clin. Pharmacokinet. 1977, 2, 45–60. Jetter, A.; Fätkenheuer, G.; Frank, D.; Klaassen, T.; Seeringer, A.; Doroshyenko, O.; Kirchheiner, J.; Hein, W.; Schömig, E.; Fuhr, U.; et al. Do Activities of Cytochrome P450 (CYP)3A, CYP2D6 and P-Glycoprotein Differ between Healthy Volunteers and HIV-Infected Patients? Antivir. Ther. 2010, 15, 975–983.
- Hatton, G.B.; Madla, C.M.; Rabbie, S.C.; Basit, A.W. All Disease Begins in the Gut: Influence of Gastrointestinal Disorders and Surgery on Oral Drug Performance. Int. J. Pharm. 2018, 548, 408–422. Obradovic, B.; Roberts, O.; Owen, A.; Milosevic, I.; Milic, N.; Ranin, J.; Dragovic, G. Expression of CYP2B6 Enzyme in Human Liver Tissue of HIV and HCV Patients. Medicina 2023, 59, 1207.
- Hoque, K.M.; Chakraborty, S.; Sheikh, I.A.; Woodward, O.M. New Advances in the Pathophysiology of Intestinal Ion Transport and Barrier Function in Diarrhea and the Impact on Therapy. Expert. Rev. Anti-Infect. Ther. 2012, 10, 687–699. Venuto, C.S.; Lim, J.; Messing, S.; Hunt, P.W.; McComsey, G.A.; Morse, G.D. Inflammation Investigated as a Source of Pharmacokinetic Variability of Atazanavir in AIDS Clinical Trials Group Protocol A5224s. Antivir. Ther. 2018, 23, 345–351.
- Lamberti, L.M.; Fischer Walker, C.L.; Black, R.E. Systematic Review of Diarrhea Duration and Severity in Children and Adults in Low- and Middle-Income Countries. BMC Public Health 2012, 12, 276. Parsons, R.L. Drug Absorption in Gastrointestinal Disease with Particular Reference to Malabsorption Syndromes. Clin. Pharmacokinet. 1977, 2, 45–60.
- Sapuła, M.; Suchacz, M.; Kozłowska, J.; Cybula, A.; Siwak, E.; Krankowska, D.; Wiercińska-Drapało, A. Adenovirus 36 Infection in People Living with HIV—An Epidemiological Study of Seroprevalence and Associations with Cardiovascular Risk Factors. Viruses 2022, 14, 1639. Hatton, G.B.; Madla, C.M.; Rabbie, S.C.; Basit, A.W. All Disease Begins in the Gut: Influence of Gastrointestinal Disorders and Surgery on Oral Drug Performance. Int. J. Pharm. 2018, 548, 408–422.
- Márquez, V.; Ballesteros, G.; Dobner, T.; González, R.A. Adipocyte Commitment of 3T3-L1 Cells Is Required to Support Human Adenovirus 36 Productive Replication Concurrent with Altered Lipid and Glucose Metabolism. Front. Cell Infect. Microbiol. 2022, 12, 1016200. Hoque, K.M.; Chakraborty, S.; Sheikh, I.A.; Woodward, O.M. New Advances in the Pathophysiology of Intestinal Ion Transport and Barrier Function in Diarrhea and the Impact on Therapy. Expert. Rev. Anti-Infect. Ther. 2012, 10, 687–699.
- Arnold, S.V.; Inzucchi, S.E.; Echouffo-Tcheugui, J.B.; Tang, F.; Lam, C.S.P.; Sperling, L.S.; Kosiborod, M. Understanding Contemporary Use of Thiazolidinediones. Circ. Heart Fail. 2019, 12, e005855. Lamberti, L.M.; Fischer Walker, C.L.; Black, R.E. Systematic Review of Diarrhea Duration and Severity in Children and Adults in Low- and Middle-Income Countries. BMC Public Health 2012, 12, 276.
- Pasarica, M.; Mashtalir, N.; McAllister, E.J.; Kilroy, G.E.; Koska, J.; Permana, P.; de Courten, B.; Yu, M.; Ravussin, E.; Gimble, J.M.; et al. Adipogenic Human Adenovirus Ad-36 Induces Commitment, Differentiation, and Lipid Accumulation in Human Adipose-Derived Stem Cells. Stem Cells 2008, 26, 969–978. Sapuła, M.; Suchacz, M.; Kozłowska, J.; Cybula, A.; Siwak, E.; Krankowska, D.; Wiercińska-Drapało, A. Adenovirus 36 Infection in People Living with HIV—An Epidemiological Study of Seroprevalence and Associations with Cardiovascular Risk Factors. Viruses 2022, 14, 1639.
- Tapia-Rivera, J.C.; Mendoza-Jaramillo, H.E.; González-Villaseñor, C.O.; Ramirez-Flores, M.; Aguilar-Velazquez, J.A.; López-Quintero, A.; Pérez-Guerrero, E.E.; Vargas-Rodriguez, M.d.l.Á.; Gutiérrez-Hurtado, I.A.; Martínez-López, E. Effect of Human Adenovirus 36 on Response to Metformin Monotherapy in Obese Mexican Patients with Type 2 Diabetes: A Prospective Cohort Study. Viruses 2023, 15, 1514. Márquez, V.; Ballesteros, G.; Dobner, T.; González, R.A. Adipocyte Commitment of 3T3-L1 Cells Is Required to Support Human Adenovirus 36 Productive Replication Concurrent with Altered Lipid and Glucose Metabolism. Front. Cell Infect. Microbiol. 2022, 12, 1016200.
- Leta, V.; Klingelhoefer, L.; Longardner, K.; Campagnolo, M.; Levent, H.Ç.; Aureli, F.; Metta, V.; Bhidayasiri, R.; Chung-Faye, G.; Falup-Pecurariu, C.; et al. Gastrointestinal Barriers to Levodopa Transport and Absorption in Parkinson’s Disease. Eur. J. Neurol. 2023, 30, 1465–1480. Arnold, S.V.; Inzucchi, S.E.; Echouffo-Tcheugui, J.B.; Tang, F.; Lam, C.S.P.; Sperling, L.S.; Kosiborod, M. Understanding Contemporary Use of Thiazolidinediones. Circ. Heart Fail. 2019, 12, e005855.
- Zhong, R.; Chen, Q.; Zhang, X.; Li, M.; Lin, W. Helicobacter Pylori Infection Is Associated with a Poor Response to Levodopa in Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis. J. Neurol. 2022, 269, 703–711. Pasarica, M.; Mashtalir, N.; McAllister, E.J.; Kilroy, G.E.; Koska, J.; Permana, P.; de Courten, B.; Yu, M.; Ravussin, E.; Gimble, J.M.; et al. Adipogenic Human Adenovirus Ad-36 Induces Commitment, Differentiation, and Lipid Accumulation in Human Adipose-Derived Stem Cells. Stem Cells 2008, 26, 969–978.
- Lolekha, P.; Sriphanom, T.; Vilaichone, R.-K. Helicobacter Pylori Eradication Improves Motor Fluctuations in Advanced Parkinson’s Disease Patients: A Prospective Cohort Study (HP-PD Trial). PLoS ONE 2021, 16, e0251042. Tapia-Rivera, J.C.; Mendoza-Jaramillo, H.E.; González-Villaseñor, C.O.; Ramirez-Flores, M.; Aguilar-Velazquez, J.A.; López-Quintero, A.; Pérez-Guerrero, E.E.; Vargas-Rodriguez, M.d.l.Á.; Gutiérrez-Hurtado, I.A.; Martínez-López, E. Effect of Human Adenovirus 36 on Response to Metformin Monotherapy in Obese Mexican Patients with Type 2 Diabetes: A Prospective Cohort Study. Viruses 2023, 15, 1514.
- Leta, V.; Klingelhoefer, L.; Longardner, K.; Campagnolo, M.; Levent, H.Ç.; Aureli, F.; Metta, V.; Bhidayasiri, R.; Chung-Faye, G.; Falup-Pecurariu, C.; et al. Gastrointestinal Barriers to Levodopa Transport and Absorption in Parkinson’s Disease. Eur. J. Neurol. 2023, 30, 1465–1480.
- Zhong, R.; Chen, Q.; Zhang, X.; Li, M.; Lin, W. Helicobacter Pylori Infection Is Associated with a Poor Response to Levodopa in Patients with Parkinson’s Disease: A Systematic Review and Meta-Analysis. J. Neurol. 2022, 269, 703–711.
- Lolekha, P.; Sriphanom, T.; Vilaichone, R.-K. Helicobacter Pylori Eradication Improves Motor Fluctuations in Advanced Parkinson’s Disease Patients: A Prospective Cohort Study (HP-PD Trial). PLoS ONE 2021, 16, e0251042.
