Umbelliferone (UMB), known as 7-hydroxycoumarin, hydrangine, or skimmetine, is a naturally occurring coumarin in the plant kingdom, mainly from the Umbelliferae family that possesses a wide variety of pharmacological properties. In addition, the use of nanoparticles containing umbelliferone may improve anti-inflammatory or anticancer therapy. Also, its derivatives are endowed with great potential for therapeutic applications due to their broad spectrum of biological activities such as anti-inflammatory, antioxidant, neuroprotective, antipsychotic, antiepileptic, antidiabetic, antimicrobial, antiviral, and antiproliferative effects.
1. Introduction
Phytochemicals constitute a large group of bioactive compounds derived from natural resources, especially those of plant origin. Among them, coumarins containing a 2
H-1-benzopyran-2-one core found in a wide range of plants demonstrate the broad spectrum of pharmacological properties, including anticancer, antimicrobial, antiviral, anticoagulant, antihypertensive, anti-inflammatory, and antioxidant or neuroprotective activities
[1].
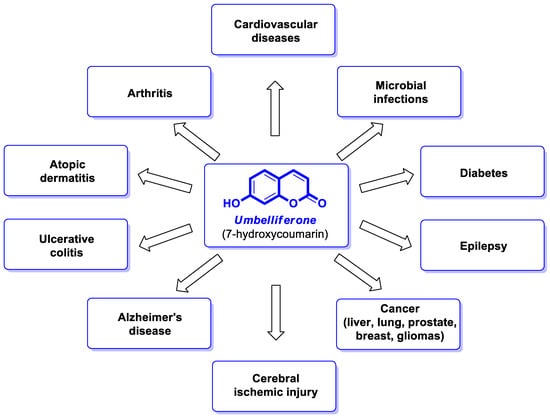
Umbelliferone (UMB) (
Figure 1), also known as 7-hydroxycoumarin, hydrangine, or skimmetine, is one of the most common plant-based coumarins present as a secondary metabolite in the flowers, fruits, and roots of almost all higher plants, mainly from the
Umbelliferae/
Apiaceae family
[2]. The potential therapeutic effects of UMB in diabetes, cardiovascular or neurodegenerative diseases, inflammatory disorders, various cancer types, and microbial infections
[3,4,5][3][4][5] (
Figure 1) have gained increasing interest in the development of its synthetic derivatives with beneficial pharmacological activities.
Figure 1. Therapeutical potential of umbelliferone [3,4,5]. Therapeutical potential of umbelliferone [3][4][5].
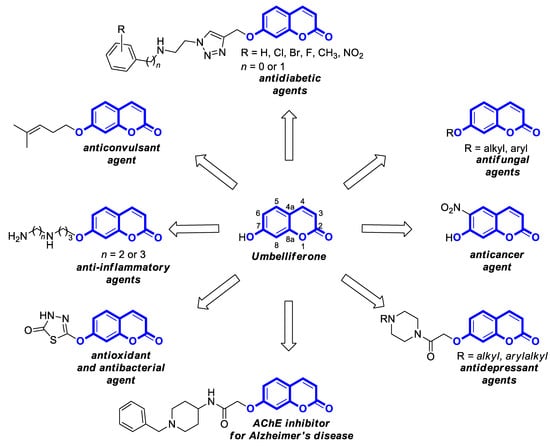
In addition, an accessible scaffold for transformation into various biologically active functionalized 7-hydroxycoumarins (
Figure 2)
[3,4,6,7,8,9,10,11][3][4][6][7][8][9][10][11] along with the lack of oral toxicity within the dose range of 200 mg/kg
[12,13,14][12][13][14] make umbelliferone an attractive platform for the development of bioactive 7-hydroxycoumarin-based compounds in drug design.
Figure 2. Biologically active 7-hydroxycoumarins derived from umbelliferone [3,6,7,8,9,10,11].
2. Anti-Inflammatory Activity
2.1. Anti-Inflammatory Properties of Umbelliferone
Inflammation is part of a complex biological process in the human body caused by various stimuli including pathogenic microorganisms, cell damage, irritants, or immune reactions. Because this process is necessary to protect the body, it should lead to the removal of pathogens and allow the tissue to return to its physiological state. On the other hand, prolonged inflammation is associated with the development of minor-to-major diseases such as rheumatoid arthritis, chronic asthma, multiple sclerosis, inflammatory bowel disease, or psoriasis, as well as cancer [15,16].
Similar to other natural coumarins including scopoletin, visnadin, marmin, daphnethin, or esculetin, umbelliferone also exhibits a favorable anti-inflammatory effect via various inflammatory signaling pathways [3,5,17,18,19].
In allergic conditions, the increase in NO production is associated with the severity of allergic symptoms, and its generation is regulated by inducible nitric oxidase synthase (iNOS) genes [20,21]. In turn, Nrf2 (nuclear factor erythroid 2 (NEF)-related factor 2) is a key signaling pathway involved in the regulation of the endogenous antioxidant system formed by heme oxygenase-1 (HO-1), superoxide dismutase (SOD), catalase (CAT), nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX), as well as thioredoxin and it protects cells from the oxidative stress markers [22].
Biologically active 7-hydroxycoumarins derived from umbelliferone [3][6][7][8][9][10][11].
2. Anti-Inflammatory Activity
2.1. Anti-Inflammatory Properties of Umbelliferone
Inflammation is part of a complex biological process in the human body caused by various stimuli including pathogenic microorganisms, cell damage, irritants, or immune reactions. Because this process is necessary to protect the body, it should lead to the removal of pathogens and allow the tissue to return to its physiological state. On the other hand, prolonged inflammation is associated with the development of minor-to-major diseases such as rheumatoid arthritis, chronic asthma, multiple sclerosis, inflammatory bowel disease, or psoriasis, as well as cancer [15][16].
Similar to other natural coumarins including scopoletin, visnadin, marmin, daphnethin, or esculetin, umbelliferone also exhibits a favorable anti-inflammatory effect via various inflammatory signaling pathways [3][5][17][18][19].
In allergic conditions, the increase in NO production is associated with the severity of allergic symptoms, and its generation is regulated by inducible nitric oxidase synthase (iNOS) genes [20][21]. In turn, Nrf2 (nuclear factor erythroid 2 (NEF)-related factor 2) is a key signaling pathway involved in the regulation of the endogenous antioxidant system formed by heme oxygenase-1 (HO-1), superoxide dismutase (SOD), catalase (CAT), nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX), as well as thioredoxin and it protects cells from the oxidative stress markers [22].
It has been reported that intraperitoneal administration of 1, 10, and 50 mg/kg of umbelliferone in BALB/c mice significantly attenuated both acute histamine- and chronic picryl chloride-induced ear edema reducing the allergic symptoms and the oxidative stress by the induction of the Nrf2 expression on the one hand and downregulation of iNO expression on the other hand [24].It has been reported that intraperitoneal administration of 1, 10, and 50 mg/kg of umbelliferone in BALB/c mice significantly attenuated both acute histamine- and chronic picryl chloride-induced ear edema reducing the allergic symptoms and the oxidative stress by the induction of the Nrf2 expression on the one hand and downregulation of iNO expression on the other hand [23].
2.2. Synthetic 7-Hydroxycoumarin-Based Compounds as Anti-Inflammatory Agents
Given its favorable anti-inflammatory activity, the umbelliferone framework has been used for chemical modification to identify original and effective compounds that can serve as anti-inflammatory agents [17,31].Given its favorable anti-inflammatory activity, the umbelliferone framework has been used for chemical modification to identify original and effective compounds that can serve as anti-inflammatory agents [17][24].
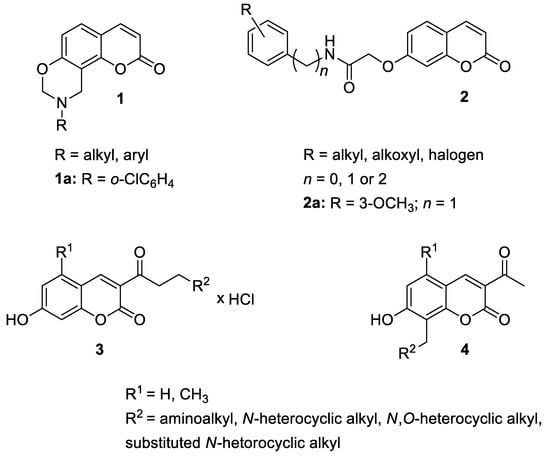
Recently, 9,10-dihydrochromeno[8.7-
e][1,3]oxazin-2(8H)-one derivatives (
1) were designed and synthesized as potential anti-inflammatory agents (
Figure 3) [32].
Figure 3. Anti-inflammatory 7-hydroxycoumarin-based compounds
1–
4.
Among the compounds that showed anti-inflammatory activity, analogue
1a
has been found to exert the most potent biological effect, which was determined to be capable of decreasing the concentration of pro-inflammatory cytokines including TNF-α and IL-6 in lipopolysaccharide (LPS)-induced cytokine release in RAW264.7 mouse macrophages. It has been indicated that derivative
1a
can inhibit inflammatory responses by suppressing the MAPK (mitogen-activated protein kinase) and NK-
κB signaling pathways that play a pivotal role in the regulation of inflammatory cytokines [33]. B signaling pathways that play a pivotal role in the regulation of inflammatory cytokines [26].
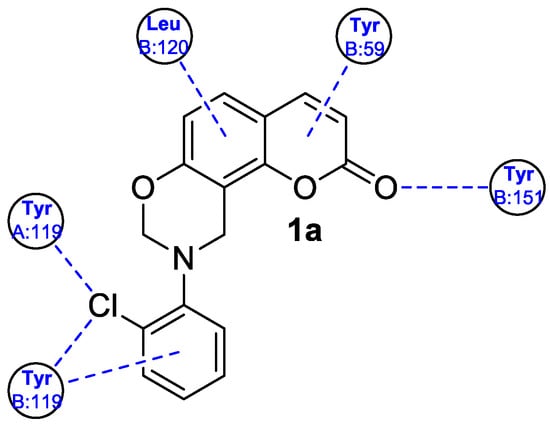
Figure 4. A 2D representation of docked ligand
1a in TNF-α.
The newly synthesized 2-[(2-oxo-2
H-chromen-7-yl)oxy]acetamides of general formula
2 (
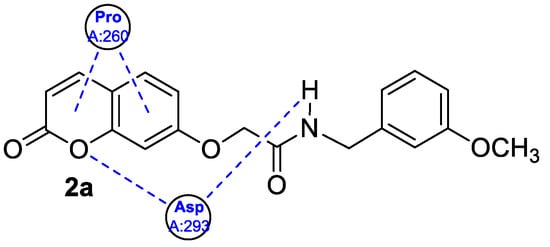
Figure 3) hybridized with substituted aniline or benzylamine moieties were also explored for their potential anti-inflammatory activity against LPS-induced IL-6 and TNF-α release in RAW264.7 cells [34].) hybridized with substituted aniline or benzylamine moieties were also explored for their potential anti-inflammatory activity against LPS-induced IL-6 and TNF-α release in RAW264.7 cells [27].
Figure 5. A 2D model of the interaction between 7-hydroxycoumarin derivative
2a with the active site of NF-κB p65.
Additionally, in 2021, Gao et al. used the Knoevenagel reaction and Pechmann condensation to develop a new series of 3-acetyl-7-hydroxycoumarin Mannich bases (
3) and Betti bases (
4) (
Figure 3) that were explored in vitro for their anti-inflammatory activity [35].) that were explored in vitro for their anti-inflammatory activity [28].
3. Antioxidant Activity
3.1. Antioxidant Properties of Umbelliferone
Oxidative stress is implicated in a number of pathological conditions such as cardiovascular diseases, cancer, neurodegenerative diseases, diabetes mellitus, ischemia/reperfusion injury, or rheumatoid arthritis, as well as in the ageing process through multiple mechanisms, where free radicals contribute to cellular damage [36]. Therefore, there is a growing interest in antioxidant agents with therapeutic potential [37].Oxidative stress is implicated in a number of pathological conditions such as cardiovascular diseases, cancer, neurodegenerative diseases, diabetes mellitus, ischemia/reperfusion injury, or rheumatoid arthritis, as well as in the ageing process through multiple mechanisms, where free radicals contribute to cellular damage [29]. Therefore, there is a growing interest in antioxidant agents with therapeutic potential [30].
In this line, the antioxidant potential of umbelliferone is also worth mentioning. As was nicely elaborated by Mazimba [3] and Lin et al. [5], its antioxidant properties are associated with the ability to scavenge free radicals as well as the inhibition of lipid peroxidation.
3.2. Synthetic 7-Hydroxycoumarin-Based Compounds as Antioxidant Agents
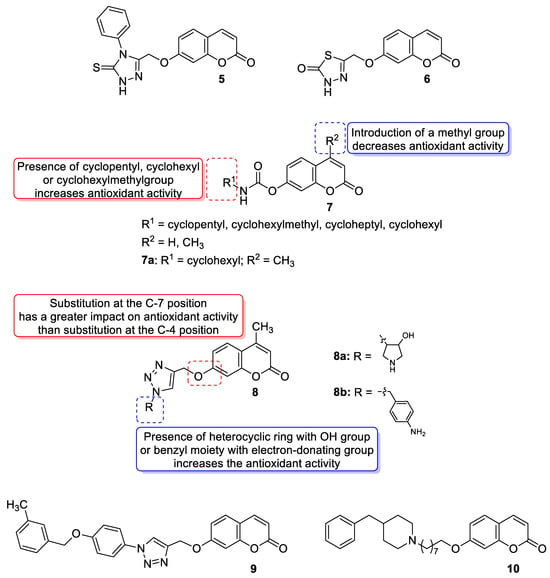
With regards to the antioxidant activity of 7-hydroxycoumarin-based compounds, Al-Majedy et al. designed and synthesized two series of modified 7-hydroxycoumarins and evaluated them for their antioxidant potency [39,40]. Among them, the best radical scavenging properties were shown by 7-[(4-phenyl-5-thioxo-4,5-dihydro-1With regards to the antioxidant activity of 7-hydroxycoumarin-based compounds, Al-Majedy et al. designed and synthesized two series of modified 7-hydroxycoumarins and evaluated them for their antioxidant potency [31][32]. Among them, the best radical scavenging properties were shown by 7-[(4-phenyl-5-thioxo-4,5-dihydro-1 H-1,2,4-triazol-3-yl)methoxy]coumarin (
5) and 5-{[(coumarin-7-yl)oxy]methyl}-1,3,4-thiadiazol-2(3
H)-one (
6) (
Figure 6), which exhibited the inhibition of 91% and 88% of free radicals, respectively, at a concentration of 250 µg/mL in the 2,2′-diphenyl-1-picrylhydrazyl radical assay (DPPH) [40].), which exhibited the inhibition of 91% and 88% of free radicals, respectively, at a concentration of 250 µg/mL in the 2,2′-diphenyl-1-picrylhydrazyl radical assay (DPPH) [32].
Figure 6. Antioxidant 7-hydroxycoumarin-based compounds
5–
10.
In 2018, Kurt et al. evaluated novel coumarin carbamate derivatives (
7) (
Figure 6) for their anticholinesterase, antioxidant, and anti-aflatoxigenic activities [41]. The synthesized compounds exhibited moderate-to-low radical scavenging ability (IC) for their anticholinesterase, antioxidant, and anti-aflatoxigenic activities [33]. The synthesized compounds exhibited moderate-to-low radical scavenging ability (IC 50 = 23.15–>200 µM) in 2,2′-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) analysis compared to
quercetin (IC
50 = 15.49 µM) used as a standard compound.
Regarding the antioxidant activity of 7-hydroxycoumarin-based compounds, some studies have been recently carried out showing the potential of coumarins linked with 1,2,3-triazoles [42,43]. Worth noting are coumarins of general formula Regarding the antioxidant activity of 7-hydroxycoumarin-based compounds, some studies have been recently carried out showing the potential of coumarins linked with 1,2,3-triazoles [34][35]. Worth noting are coumarins of general formula 8 prepared by Joy et al. through the copper catalyzed azide-alkylene cycloaddition reaction (
Figure 6) [42].
Moreover, Kaushik and Chacal synthesized two series of coumarin-1,2,3-triazole hybrid molecules using the click chemistry approach from the coumarin-based terminal alkynes and aromatic azides and tested their antioxidant activity via the DPPH method [43]. However, all compounds displayed lower DPPH-based radical scavenging activity (ICMoreover, Kaushik and Chacal synthesized two series of coumarin-1,2,3-triazole hybrid molecules using the click chemistry approach from the coumarin-based terminal alkynes and aromatic azides and tested their antioxidant activity via the DPPH method [35]. However, all compounds displayed lower DPPH-based radical scavenging activity (IC 50 = 3.33–8.75 μg/mL) compared to the standard ascorbic acid (IC
50 = 1.23 μg/mL), and the presence of the electron-donating groups on the benzyl moiety in the structure of these compounds might contribute to increased antioxidant activity. In addition, the 7-hydroxycoumarin-based compounds generally evidenced higher activity than their 4-hydroxycoumarin-based counterparts. The best result was found for derivative
9 with an IC
50 value of 3.33 μg/mL (
Figure 6) [43].
Most recently, a new 7-hydroxycoumarin derivative
10 (
Figure 6) was invented as a potential antioxidant agent [44]. Although the antioxidant activity of ) was invented as a potential antioxidant agent [36]. Although the antioxidant activity of 10 was lower than the standard BHT (
p < 0.05), it was found that all used concentrations (0.03125–1 mg/mL) owed its ability to scavenge radicals in the DPPH assay. The experimental antioxidant properties of coumarin
10 were also supported by molecular docking analysis that revealed the possible interactions of derivative
10 with the active binding site of CYP450.
3.3. Metal Complexes with 7-Hydroxycoumarin-Based Compounds as Antioxidant Agents
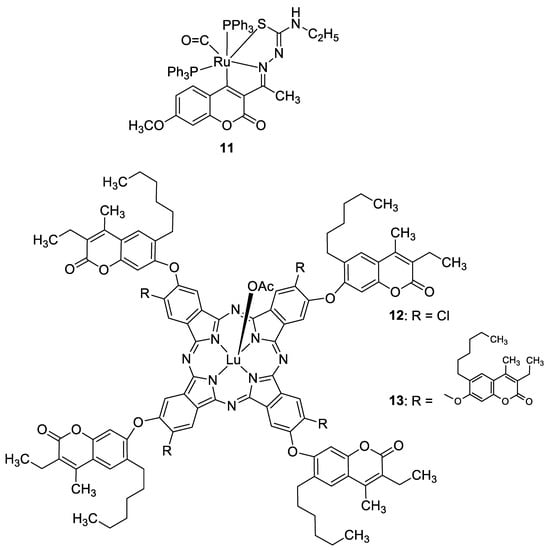
Recently, it was demonstrated that the radical scavenging ability of novel 3-acetyl-7-methoxy-4
N-substituted thiosemicarbazones may be increased by ruthenium chelation [47]. The best radical scavenging properties have been shown by Ru(II) complex
-substituted thiosemicarbazones may be increased by ruthenium chelation [37]. The best radical scavenging properties have been shown by Ru(II) complex
11
(
Figure 7
), which displayed an antioxidant potency with about a fifteen-fold lower IC
50
value than standard vitamin C in the DPPH model (IC
50
= 5.28 µM vs. IC
50
= 98.72 µM).
Figure 7. Antioxidant 7-hydroxycoumarin-based metal complexes
11–
13.
In 2020, Özdemir et al. synthesized a series of 7-oxy-3-ethyl-6-hexyl-4-methylcoumarin-substituted lutetium(III) phthalocyanine compounds, whose antioxidant properties were evaluated [48]. Complexes In 2020, Özdemir et al. synthesized a series of 7-oxy-3-ethyl-6-hexyl-4-methylcoumarin-substituted lutetium(III) phthalocyanine compounds, whose antioxidant properties were evaluated [38]. Complexes 12 and
13 (
Figure 7) displayed much better 2,2′-azino-bis-3-ethylbenzthiazoline-6-sulphonic acid (ABTS)-based radical cation scavenging activity compared with standard butylated hydroxyanisole (BHA), 120.344 mM troloxy/mg and 188.733 mM troloxy/mg vs. 52.63 mM troloxy/mg. On the other hand, the FRAP (Ferric Reducing Antioxidant Power) and CUPRAC (Cupric Reducing Antioxidant Capacity) analyses evidenced their lesser potency compared to BHT and vitamin C used as standards [48].
) displayed much better 2,2′-azino-bis-3-ethylbenzthiazoline-6-sulphonic acid (ABTS)-based radical cation scavenging activity compared with standard butylated hydroxyanisole (BHA), 120.344 mM troloxy/mg and 188.733 mM troloxy/mg vs. 52.63 mM troloxy/mg. On the other hand, the FRAP (Ferric Reducing Antioxidant Power) and CUPRAC (Cupric Reducing Antioxidant Capacity) analyses evidenced their lesser potency compared to BHT and vitamin C used as standards [38].
4. Umbelliferone and 7-Hydroxycoumarin-Based Compounds Acting in the Central Nervous System (CNS)
4.1. Neurodegenerative Disorders
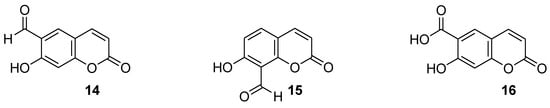
Umbelliferone and its simple derivatives—6-formylumbelliferone (
14) from the plant
Angelica decursiva, its isomeric analogue 8-formylumbelliferone (
15), and umbelliferone 6-carboxylic acid (
16) (
Figure 8)—exhibit potent inhibitory activities towards acetylcholinesterase (AChE), butyrylcholinesterase (BuChE), and aspartic protease β-secretase 1 (BACE1)
[52,53,54][39][40][41]. However, it should be noted that the data regarding the inhibitory activity of umbelliferone towards AChE and BuChE are contradictory.
Numerous diseases can be caused by a defect of more than one biological target—an enzyme or receptor. Thus, such disorders cannot be adequately addressed by the classical ‘one target, one molecule’ approach
[57][42]. A promising strategy to tackle multifactorial diseases, e.g., AD, consists in the design of multifunctional agents, known as ‘hybrid’ molecules. These complex molecules display stable chemical combinations of two drug moieties or pharmacophores acting at different targets. Such ‘dual-acting compounds’ combine two distinct chemical entities
[58,59][43][44]. According to this, Hirbod et al. designed a 7-hydroxycoumarin hybrid bearing a heterocyclic framework—8-hydroxyquinoline
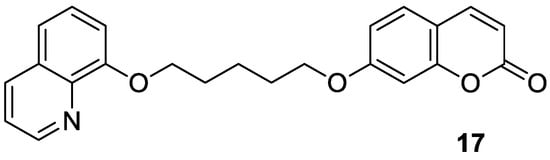
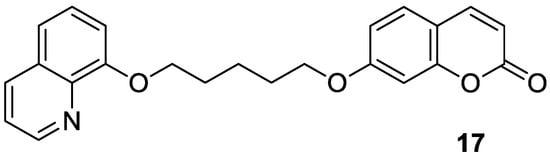
17 (
Figure 9)—as a novel cholinesterase inhibitor
[60][45].
Figure 9. Chemical structure of 7-hydroxycoumarin-based compound
17 as an AChE and BuChE inhibitor.
Numerous diseases can be caused by a defect of more than one biological target—an enzyme or receptor. Thus, such disorders cannot be adequately addressed by the classical ‘one target, one molecule’ approach [57]. A promising strategy to tackle multifactorial diseases, e.g., AD, consists in the design of multifunctional agents, known as ‘hybrid’ molecules. These complex molecules display stable chemical combinations of two drug moieties or pharmacophores acting at different targets. Such ‘dual-acting compounds’ combine two distinct chemical entities [58,59]. According to this, Hirbod et al. designed a 7-hydroxycoumarin hybrid bearing a heterocyclic framework—8-hydroxyquinoline 17 (Figure 9)—as a novel cholinesterase inhibitor [60].
Figure 9. Chemical structure of 7-hydroxycoumarin-based compound 17 as an AChE and BuChE inhibitor.
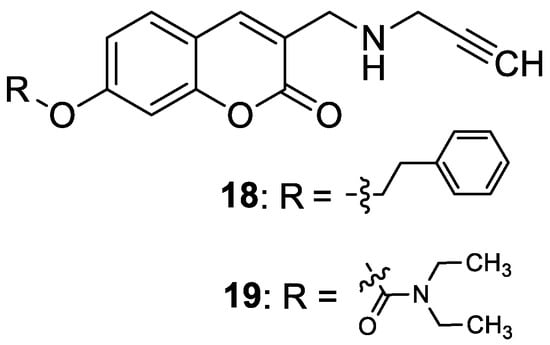
Recently, Mzezewa et al. have described 3-substituted 7-hydroxycoumarin derivatives
18 and
19 as multifunctional anti-Alzheimer’s disease agents (
Figure 10)
[63][46].
Figure 10. Chemical structures of 7-hydroxycoumarin-based compounds
18 and
19 as multifunctional anti-Alzheimer’s disease agents.
In addition, these compounds offer significant neuroprotective effects towards MPP
+-compromised SH-SY5V neuroblastoma cells with no inherent cytotoxicity at 10 µM. Consequently, compounds
18 and
19 have been proposed for further studies to explore their neuroprotective potential in AD and related neurodegenerative diseases such as Parkinson’s disease. Although 7-hydroxycoumarins
18 and
19 exhibited weak cholinesterase inhibitory activity when compared with the reference
denezepril (AChE and BuChE IC
50 = >100 µM vs. AChE IC
50 = 0.007 µM and BuChE IC
50 = 4.40 µM), the tested compounds demonstrated selectivity towards MAO-B with IC
50 values of 0.029 µM and 0.101 µM, respectively.
4.2. Neuropsychiatric Diseases
4.2.1. Synthetic 7-Hydroxycoumarin-Based Compounds Targeting Monoamine Oxidase (MAO) and D-Amino Acid Oxidase (DAAO)
Recently, Seong et al. reported 6-formylumbelliferone derivative 14 and its isomeric analogue 15, presented in Figure 8, as highly selective hMAO-A inhibitors [65][47]. The higher selectivity and inhibitory activity towards hMAO-A exhibited 7-hydroxy-2-oxo-2H-chromene-6-carbaldehyde (14) with an IC50 value of 3.23 μM for hMAO-A and an IC50 value of 15.31 μM for hMAO-B. Enzyme kinetic studies revealed that both 6-formylumbelliferone 14 and 8-formylumbelliferone 15 are competitive hMAO inhibitors. These investigations were supported by molecular docking studies. Data revealed that compounds 14 and 15 dock well into the active sites of recombinant human monoamine oxidase A and B. The formyl group of 14 interacts strongly with substrate binding site (SBS) residues Tyr444 and Tyr197 of hMAO-A via water-mediated hydrogen bonds, whereas Phe352 and Tyr407 residues are involved in hydrophobic noncovalent π-π T-shaped (perpendicular T-shaped) and π-π stacking interactions. Hydroxycoumarin derivatives 14 and 15 demonstrated a neuroprotective effect due to their antilipid peroxidation and anti-Aβ25–35 (amyloid β self-assembly) aggregation activity in rat brain tissue.
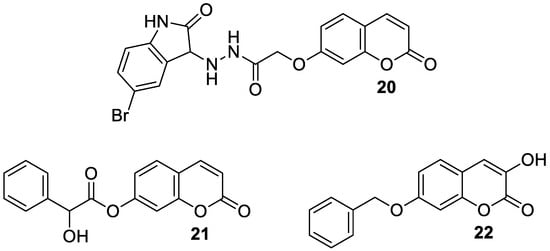
In a study in 2018, Dhirman et al. investigated monoamine oxidase’s inhibitory effects on a series of umbelliferone-based compounds
[66][48]. By substituting the coumarin scaffold at the C-7 position, MAO’s inhibitory potential was significantly increased. MAO inhibition studies have shown that hybrid compounds containing the 5-bromoisatin moiety
20 (
Figure 11) exhibited a pronounced
hMAO-A activity (IC
50 = 7.47 μM), whereas incorporation of the 2-hydroxy-2-phenylacetate moiety into umbelliferone derivative
21 (
Figure 11) resulted in significant
hMAO-B blocking (IC
50 = 10.32 μM). In the same studies, umbelliferone turned out to be less active than the tested compounds (
hMAO-A IC
50 = 18.08 μM and
hMAO-B IC
50 = 12.98 μM)
[66][48].
Figure 11. Chemical structures of 7-hydroxycoumarin-based compounds
20–
22 as MAO and DAAO inhibitors.
In 2022, Bester et al., as a result of their investigations, described the synthesis of 3-hydroxy-7-benzyloxy-2
H-chromen-2-one (
22) starting from 2,4-dihydroxybenzaldehyde,
N-acetylglycine, and acetic anhydride (
Figure 11)
[69][49]. Compound
22 was identified as a potent, selective inhibitor of MAO-B (IC
50 = 0.012 μM) and DAAO (IC
50 = 1.86 μM). The results obtained were an improvement or comparable to those of the reference inhibitors:
coumarin (MAO-B IC
50 = 2.56 μM),
isatin (MAO-B IC
50 = 3.90 μM), and 3-methylpyrazole-5-carboxylic acid (DAAO IC
50 = 1.88 μM).
4.2.2. Synthetic 7-Hydroxycoumarin-Based Compounds Targeting Serotonin Receptors
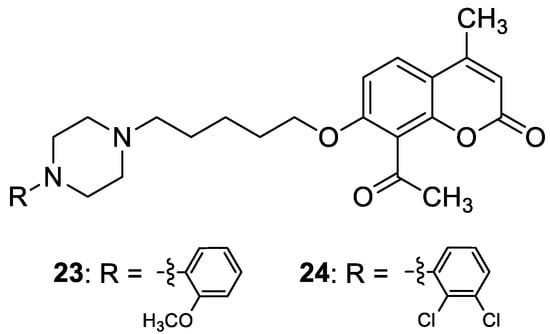
Recent studies have clearly demonstrated that umbelliferone-based compounds may interact with serotonin receptors. In 2021, among a series of 7-hydroxycoumarins bearing a piperazine moiety, 7-hydroxycoumarin derivatives
23 and
24 (
Figure 12) showed high antagonistic activity against serotonin receptors
[70][50].
Figure 12. Chemical structures of 7-hydroxycoumarin-based compounds
23 and
24 targeting 5-HT receptors.
In the paper, the authors claimed that the substitution pattern dictates the selectivity and affinity of tested compounds for 5-HT receptors. The structure–activity analysis showed that the presence of a five-carbon atom linker and 2-methoxyphenyl group attached to the piperazine moiety (compound
23) was the most beneficial for 5-HT
1A antagonistic activity, whereas the (2,2-dichloro)piperazin-1-yl moiety is associated with a higher inhibition of the 5-HT
2A receptor (compound
24).
4.3. Antiepileptic Agents
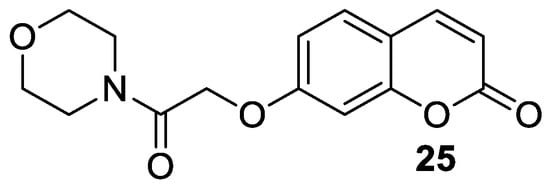
The antiepileptic effects of 7-hydroxycoumarin derivatives may be associated with the synergistic effect on γ-aminobutyric acid ionotropic receptors (GABA
A). In the reaction of 7-hydroxycoumarin and 2-chloro-1-morpholinoethan-1-one, Yakovleva and collaborators synthesized umbelliferone derivative
25 (
Figure 143) containing a morpholine-acetamide group at position C-7
[73][51]. Compound
25 showed pronounced antiepileptic activity in the
corazole-GABA
A receptor antagonist convulsion test. The effectiveness of 7-hydroxycoumarin derivative
25 is associated with the morpholine ring, which has an optimal lipophilic-hydrophilic profile. The antiepileptic effect of derivative
25 at a dose of 200 mg/kg was comparable to that of reference valproic acid at the same dose. A further increase in the dose to 300 mg/kg led to an increase in the anticonvulsant activity of
25 [73][51].
Figure 143. Chemical structure of 7-hydroxycoumarin-based compound
25 as an antiepileptic agent.
5. Umbelliferone and 7-Hydroxycoumarin-Based Compounds as Antidiabetic Agents
Extracts of widely cultivated plants, such as
Musa species (banana flower ethanolic extracts) containing umbelliferone, were identified as potential antidiabetic herbal remedies in the management of diabetes and associated complications. Isolated umbelliferone increased the activity of crucial enzymes involved in glucose utilization and the glycolytic activity of the liver in alloxan-induced diabetic rats [74].
Umbelliferone was also reported to be effective in diabetic cardiomyopathy (DCM) by suppressing Janus kinase2 (JAK2) and the signal transducer and activator of the transcription signaling pathway (STAT3) [77]. Moreover, umbelliferone in a type 2 diabetic rat model at doses of 10 and 30 mg/kg decreased levels of glucose, glycated hemoglobin (HbA1c), tumor necrosis factor (TNF-α), and interleukin-6 (IL-6).
In this context, it should be mentioned that 6-formylumbelliferone ( species (banana flower ethanolic extracts) containing umbelliferone, were identified as potential antidiabetic herbal remedies in the management of diabetes and associated complications. Isolated umbelliferone increased the activity of crucial enzymes involved in glucose utilization and the glycolytic activity of the liver in alloxan-induced diabetic rats [52].
Umbelliferone was also reported to be effective in diabetic cardiomyopathy (DCM) by suppressing Janus kinase2 (JAK2) and the signal transducer and activator of the transcription signaling pathway (STAT3) [53]. Moreover, umbelliferone in a type 2 diabetic rat model at doses of 10 and 30 mg/kg decreased levels of glucose, glycated hemoglobin (HbA1c), tumor necrosis factor (TNF-α), and interleukin-6 (IL-6).
In this context, it should be mentioned that 6-formylumbelliferone (