Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Alba Rodríguez Otero and Version 2 by Lindsay Dong.
Rice husks, as a residue from agriculture, had been largely used as a source of power through direct incineration in major rice-producing countries. However, rice husks present an intriguing opportunity as a renewable source of SiO2, offering a low-cost adsorbent with a high surface area and ease of functionalization that can be transformed into diverse mesoporous silica structures or composites, enabling applications in catalysis, drug delivery, water treatment, etc. This dual potential of rice husks can be harnessed by combining bio-oil and syngas production through pyrolysis with the efficient extraction of SiO2, ensuring the comprehensive utilization of the biomass.
- SiO2 nanoparticles
- rice husk
- bio-oil
- circular economy
- biomass
- pyrolysis
1. Introduction
Accessibility to sustainable, low-carbon bio-feedstock is one of the key drivers to achieve a European Union (EU) low-carbon economy by 2050. In this context, the development of biomass conversion technologies is being promoted [1].
Agricultural waste is in the spotlight of sustainable biomass production since an important amount of waste is produced yearly and is usually not fully harnessed. The source of this biomass includes primary agricultural resources produced directly on the field after crop harvesting and secondary plant resources obtained during industrial processing of agricultural crops. The potential estimated for the latter in 2030 in the EU report ranges from 56 to 81 million tons, with France, Austria, Germany and Spain being the countries with the largest contributions [1].
Among secondary plant resources, several residues are included, such as olive pits, fruit pulp, husks, shells, etc. Among the largest residues generated in agriculture are those coming from rice plants (Oryza sativa), especially their husks. Rice husk (RH) is the major by-product in the rice milling industry, which is estimated to be 1.2 billion tons annually worldwide [2][3][4][2,3,4].
Rice Husk as a Source of Bio-Energy and SiO
2
Dried RHs contain about 15–28 wt% silica, depending on the origin and type, and 72–85 wt% lignocellulose (15–22 wt% lignin, 12–29 wt% hemicellulose, 29–36 wt% cellulose and 3–13 wt% traces of metals). As it does not have sufficient protein content (~4 wt% proteins, oils) for agricultural uses, most RHs are either burned or buried in landfills [5][7]. RH is the biomass waste containing the largest amount of silica (~210 g/kg of husk) [6][8]. Even when compared to other husks, such as wheat husk, or other parts of the rice plant, such as the straw, its silica content is the highest. Therefore, as RHs are not easily decomposed by bacteria due to their highly inorganic content, their accumulation in landfills is not a sustainable option. For this reason, RH incineration has been the most common technique for removal since the heating value is around 15 MJ/kg, and so this energy can be harnessed [7][9]. Nevertheless, this method produces a high amount of rice husk ash (RHA) that needs to be disposed of as well, with its most common use currently being as a filler, additive, etc., in the building and construction industry. If RH is not properly pretreated before the incineration, the composition of the ashes results in a low-purity silica composite due to the metal and carbon content present in the husk. Rice plants collect silica from the soil, which is stored as silicic acid inside the cellulose micro-compartments of the plant. The metal content varies according to the soil and manures used, but traces of potassium, calcium, magnesium, iron, sodium, manganese, aluminum, phosphorous and sulfur are found [8][14]. K is the main metal of RH (~2.7 mg/g), followed by Ca (~1.5 mg/g) and Mg and Mn (0.36 mg/g). The presence of K promotes the crystallization of silica into tridymite and cristobalite, a carcinogenic compound featuring a very low surface area (10 m2/g), and leads to the formation of unburnt carbon residues. Then, the silica powder is not white, and the specific surface area is low: from 145 m2/g for calcination at 600 °C to less than 20 m2/g for higher temperatures due to silica crystallization [9][15]. The low economic efficiency of incineration of RH has led to a search for alternative methods to harness this agricultural waste. For instance, pyrolysis of biomass is a hot topic currently since it leads to the production of pyrolysis gas and bio-oil from the decomposition of the lignocellulosic materials, and in the specific case of rice husk, char rich in silica is obtained as a third by-product. So, for these aforementioned reasons, it is considered to be a more efficient way to thermally degrade organic matter [10][11][16,17]. However, it has to be considered that pyrolysis bio-oil has a heating value of around 13–25 MJ/kg, lower than diesel and gasoline [7][9]. Silicon is the second most abundant element in the Earth’s crust after oxygen, which is brought to the soil by chemical and biological processes. Plants are highly involved in such processes as they transform the silicon present in water into water-soluble silicic acid [H4SiO4, or Si(OH)4], which is accumulated in the form of phytoliths, amorphous hydrated silica (SiO2 with 5–10% H2O). In rice plants, these phytoliths accumulate primarily in the husk. The deposition of silicon in this form offers the plant resistance against pathogens, stresses and pests, enhancing growth and development, thus Si availability in the soil where the rice grows is highly important [12][18]. Silica is one of the most important inorganic materials in the industry since it has a wide variety of applications, such as in catalyst production, catalyst supports, drug carriers, construction, glass and ceramics and water filtration, among others. Specifically, nanosized silica is of great interest due to its large surface area, thermal conductivity (insulation), good dispersion performance, ease of functionalization, chemical stability and biocompatibility [13][19]. These properties show an interesting prospect in the development of techniques to harness silica from agricultural waste. To synthesize highly dispersed mesoporous SiO2 NPs, Stöber synthesis is modified by adding surfactants (such as cetyltrimethylammonium bromide or chloride (CTAB or CTAC)) to direct the mesopores formation by micelle-templating. The water content is also increased. For example, one of the best homogeneous, highly dispersed mesoporous SiO2 NPs was achieved by Möller et al. [14][31] by replacing NH4OH with triethanolamine (TEA) in mixtures with a molar composition of 1 TEOS: 0.27 CTAC: 1–4 TEA: 137 H2O: 6.2 EtOH. Mesoporous SiO2 NPs of 50 and 100 nm were obtained at 60 °C for TEA/TEOS = 1 and 4, respectively. Surfactant removal was achieved by a twofold extraction in acidified EtOH or by adding NH4NO3 at 60 °C for 2 h. The most convenient method was acidified EtOH at 25 °C with 30 min ultrasounds. The mesoporous SiO2 NPs feature ordered cylindrical homogenous mesopore of 3.5 nm diameter, specific surface areas of 1000 m2/g and mesopore volume of 0.77 mL/g as MCM-41 materials. Another interesting modification of the Stöber synthesis using CTAB is realized by replacing alcohols with aldehydes [15][16][32,33]. Mixtures with a molar ratio of 1 TEOS: 0.14 CTAB: 1.6–3.4 NH4OH: 100 H2O: 11–22 acetaldehyde at 27 °C give rise to MCM-41-like mesoporous NPs with particle sizes ranging from 40 to 850 nm. The particle size decreases by increasing the pH (adding more NH4OH) or by decreasing the amount of acetaldehyde. Although great results have been achieved in producing porous silica NPs, the mentioned chemical methods imply the use of hazardous chemicals harmful to the environment, such as CTAB, or costly techniques (by the use of TEOS, specifically). For this reason, the interest in developing green processes for producing silica NPs from biomass and agricultural waste is becoming of great interest [17][34], as evidenced by the publication of more than 400 research articles on the topic since 2019 (Figure 13).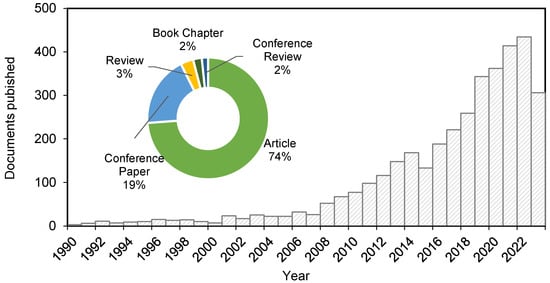
Figure 13. Documents published on silica nanoparticles from rice husk from 1990 to 2023 (Scopus, April 2023).
2. Sustainable Harnessing of SiO
Documents published on silica nanoparticles from rice husk from 1990 to 2023 (Scopus, April 2023).
3. Sustainable Harnessing of SiO
2 from Rice Husks
2.1. Traditional Thermochemical Methods: Calcination
In the 1990s, direct combustion of rice husk without any chemical pretreatment was the most common method [18][19]. The first controlled combustion of RH, where amorphous silica powder was synthesized, was reported by Kapur in 1984. Combustion of rice husk led to a 20 wt% of ashes composed mainly of silica. As no purification pretreatment was applied, no pure silica was obtained [19]. Some acid leaching occurred on the rice husk ash after calcination [20]. However, the specific surface area of the resulting nanoparticles was not reported since the main objective was to determine the diameter of the nanoparticles and the effects of changing the ball milling parameters [20].
The type of rice affects the produced nanoparticles; silica nanoparticles synthesized from brown rice husk had the smallest diameter (10 nm) in comparison with sticky (50 nm) or red (20 nm) rice [21]; thus, the origin of the RH also determines its final structure. The soil quality also affects the elemental composition of the RH. Benassi L. et al. (2015) [22] found a difference in the metal composition of Italian and Indian rice husks, which affected the availability of amorphous silica at the same calcination temperatures. Indian RH from different regions was found to have different metallic content, which also affected the formation of amorphous silica and its purity, pointing out the relevance of K
from Rice Husks
3.1. Traditional Thermochemical Methods: Calcination
In the 1990s, direct combustion of rice husk without any chemical pretreatment was the most common method [35,36]. The first controlled combustion of RH, where amorphous silica powder was synthesized, was reported by Kapur in 1984. Combustion of rice husk led to a 20 wt% of ashes composed mainly of silica. As no purification pretreatment was applied, no pure silica was obtained [36]. Some acid leaching occurred on the rice husk ash after calcination [37]. However, the specific surface area of the resulting nanoparticles was not reported since the main objective was to determine the diameter of the nanoparticles and the effects of changing the ball milling parameters [37].
The type of rice affects the produced nanoparticles; silica nanoparticles synthesized from brown rice husk had the smallest diameter (10 nm) in comparison with sticky (50 nm) or red (20 nm) rice [38]; thus, the origin of the RH also determines its final structure. The soil quality also affects the elemental composition of the RH. Benassi L. et al. (2015) [51] found a difference in the metal composition of Italian and Indian rice husks, which affected the availability of amorphous silica at the same calcination temperatures. Indian RH from different regions was found to have different metallic content, which also affected the formation of amorphous silica and its purity, pointing out the relevance of K
O content in this matter [52].
2.2. Pyrolysis of Rice Husk for Obtaining Bio-Oil and Bio-Silica in the Literature
3.2. Pyrolysis of Rice Husk for Obtaining Bio-Oil and Bio-Silica in the Literature
2.2.1. Bio-Silica Production
3.2.1. Bio-Silica Production
Pyrolysis of acid-leached rice husk in an N2 atmosphere was described by Liou et al. (2004) [24], obtaining carbon/silica powders with a surface area of 261 m
atmosphere was described by Liou et al. (2004) [23], obtaining carbon/silica powders with a surface area of 261 m2
/g at 900 °C. No bio-oil is recovered from this process. In the same study, the acid-leached rice husks were calcined with air, and the surface area obtained was 235 m2
/g.
To our knowledge, the first pyrolysis performed, with the aim of obtaining pure silica nanoparticles, was reported in 2011 by Wang et al. [25]. However, although it is referred to as pyrolysis, it seems more like combustion since heating in the absence of oxygen does not achieve the obtention of pure silica but a composite of carbon and silica [25].
To our knowledge, the first pyrolysis performed, with the aim of obtaining pure silica nanoparticles, was reported in 2011 by Wang et al. [6]. However, although it is referred to as pyrolysis, it seems more like combustion since heating in the absence of oxygen does not achieve the obtention of pure silica but a composite of carbon and silica [6].
Liou et al. (2011) performed pyrolysis on acid-leached (hot HCl) RH at 700 °C for 1 h and extracted SiO2 by NaOH 1.5 M at 100 °C for 1 h to produce sodium silicate [26]. The addition of 1 M HCl to this sodium silicate solution to reach pH 7 at 50 °C allowed for the precipitation of SiO
by NaOH 1.5 M at 100 °C for 1 h to produce sodium silicate [53]. The addition of 1 M HCl to this sodium silicate solution to reach pH 7 at 50 °C allowed for the precipitation of SiO2
NPs of 6–8 nm diameter (350 m2
/g, mesopore diameter 10.4 nm, V = 0.79 mL/g). These NPs are non-porous and agglomerated, and Na content (1.31 mg/g) is superior to the parent RH.
2.2.2. Combined Bio-Silica and Bio-Oil Production
3.2.2. Combined Bio-Silica and Bio-Oil Production
Research has been made on the production of bio-oil from pyrolyzed rice husks, studying its composition and its feasibility as a biofuel.Lu Q et al. (2008) [10] analyzed the properties of bio-oil extracted from rice husk, which were similar to other bio-oils in terms of chemical composition. Acids, alcohols, ketones, sugars, etc., were compounds found in rice husk bio-oil similarly as for other bio-oils from different biomasses. However, they found high concentrations of nitrogen, inorganic elements and many low-boiling compounds, but significantly less heavy components, as well as high contents of water, solids and ash, and the presence of alkali metals, which is undesirable as well. The rice husks were not previously leached with acid, which explains the presence of these metals. Their main conclusion was that there is a need to upgrade the bio-oil produced from rice husk before it can be used as biofuel. In this case, the rice husk was not previously treated, and the bio-oil yield was 50 wt%.
Lu Q et al. (2008) [16] analyzed the properties of bio-oil extracted from rice husk, which were similar to other bio-oils in terms of chemical composition. Acids, alcohols, ketones, sugars, etc., were compounds found in rice husk bio-oil similarly as for other bio-oils from different biomasses. However, they found high concentrations of nitrogen, inorganic elements and many low-boiling compounds, but significantly less heavy components, as well as high contents of water, solids and ash, and the presence of alkali metals, which is undesirable as well. The rice husks were not previously leached with acid, which explains the presence of these metals. Their main conclusion was that there is a need to upgrade the bio-oil produced from rice husk before it can be used as biofuel. In this case, the rice husk was not previously treated, and the bio-oil yield was 50 wt%.
Hsu et al. (2015) [27] studied the pyrolysis of RH in a fluidized bed pyrolizer with the aim of producing syngas and bio-oil, but it does not focus on the production of biochar or silica. The yields obtained with a N
Hsu et al. (2015) [59] studied the pyrolysis of RH in a fluidized bed pyrolizer with the aim of producing syngas and bio-oil, but it does not focus on the production of biochar or silica. The yields obtained with a N2
gas flow of 40 L/min and feeding rate of 10 g/min were the following: syngas: 38.52%; bio-oil: 29.44%; and char: 32.04%. The composition of the bio-oil was analyzed, which showed that it was mainly composed of aromatic compounds.
3. Tailoring of SiO
4. Tailoring of SiO
2
from Rice Husk to Enhance Porosity
3.1. Mesoporous RH-SiO
4.1. Mesoporous RH-SiO
2
Nanoparticles
One of the main goals of the optimization of the extraction process of silica from rice husk is achieving a large surface area since the surface area is linked to the value of the produced material. Efforts to adapt acid-leaching, grinding and calcination parameters to enhance the porosity of silica from rice husk have been reported. The effect of calcination temperature was studied by Manasa et al. (2021). Five silica materials were synthesized, with different calcination conditions in the oven after acid leaching. Temperatures of 550, 600 and 650 °C for 3 h combined with air or N2
flow were studied as parameters for the extraction of SiO2
. The highest BET surface area (SA) (376 m2
/g) was achieved with RH-SIO2
-600A calcined at 600 °C. RH-SiO2
-600A had a pore size of 4.5 nm and a pore volume of 0.38 mL/g. The same RH subjected to pyrolysis with only N2
at 600 °C was a composite C/SiO2
, named RH-SiO2
-600N, which presented a lower SA of 267 m2/g, a smaller pore diameter (2.7 nm) and a smaller pore volume (0.17 mL/g) [28]. With higher temperatures than 600 °C, the SA decreases, and the pore size increases, possibly due to nanoparticles sintering.
/g, a smaller pore diameter (2.7 nm) and a smaller pore volume (0.17 mL/g) [62]. With higher temperatures than 600 °C, the SA decreases, and the pore size increases, possibly due to nanoparticles sintering.
