Photodynamic therapy (PDT), largely employed as a clinical treatment for several malignant pathologies, has also gained importance as a promising antimicrobial approach. Antimicrobial PDT (aPDT) relies on the application of a photosensitizer able to produce singlet oxygen (1O2) or other cytotoxic reactive oxygen species (ROS) upon exposure to appropriate light, which leads to cell death after the induced photodamage. Among different types of 2D nanomaterials with antimicrobial properties, phosphorene, the exfoliated form of black phosphorus (bP), has the unique property intrinsic photoactivity exploitable for photothermal therapy (PTT) as well as for PDT against pathogenic bacteria.
- antimicrobial
- phosphorene
- photothermal therapy
- photodynamic therapy
1. Introduction
2. Antimicrobial Phototherapy
In antimicrobial phototherapy (APT), the light is used to treat infections caused by bacteria, viruses, fungi, and other microorganisms. This treatment method harnesses the properties of light, either alone or in combination with suitable photosensitizing agents, to induce a phototoxic or photothermal effect on the targeted microbes. APDT, also called photodynamic inactivation (PDI), and aPTT are antimicrobial approaches that have been shown to effectively inactivate a wide range of pathogens, including microorganisms that are highly resistant to conventional drugs and that form biofilms. Both therapeutic methods exploit the interaction of a photosensitizer with light to produce, in the case of aPDT, either highly reactive oxygen species in the presence of oxygen or, in the case of aPTT, heat and local temperature rise. The synergistic use of aPDT and aPTT can enhance the overall efficacy of antimicrobial phototherapy, leading to significant reductions in bacterial pathogens, eradication of infections, and collapse of biofilms.2.1. Basic Mechanisms
Since many exhaustive reviews have recently described the latest updates on aPDT or PDI [6,7,8,17,18,19,20,21][5][6][7][8][9][10][11][12]. The fundamental mechanism of aPDT is based on the simultaneous presence of three main actors: (1) a photosensitizer (PS), a molecule or a complex that is physiologically harmless and able to absorb light in spectral regions ranging from the visible to near-infrared wavelengths; (2) a light source emitted in the spectral range absorbed by PS; and (3) molecular oxygen, i.e., O2 [22][13] (Figure 1).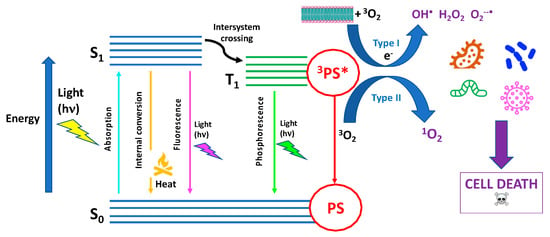
2.2. Antimicrobial PDT Activity
- -
-
Positive charge for high-affinity binding to negatively charged bacterial cell membranes;
- -
-
Low molecular weight or a structure that facilitates penetration through the biofilm matrix;
- -
-
High 1O2 quantum yield;
- -
-
High photostability;
- -
-
No dark toxicity and/or mutagenicity towards host eukaryotic cells in the “therapeutic window” where microorganisms can be killed without damaging the surrounding cells.
2.3. Antimicrobial PTT Activity
As previously mentioned, another light-induced therapeutic treatment against bacterial infections is called antimicrobial photothermal therapy (aPTT) [37][27]. It relies on the heating of microbial pathogens at temperatures above 45 °C, at which the viability of most bacteria is impaired, which is achieved by irradiating a suitable photothermal agent. Photothermal antimicrobial agents are nanostructures with significant absorption in the visible and NIR regions that are able to transform the absorbed light into heat. They can be classified according to their chemical structure into plasmonic metals (such as Au, Cu, Pd, and Bi), carbon-based materials (such as carbon dots, GO, and graphene nanosheets), polymers (polydopamine, polyaniline, and polypyrrole), and semiconductors such as bP-NPs.3. Structure and Properties of bP and bP-NPs
At standard temperature and pressure, bP is the most thermodynamically stable phosphorus allotrope. It was obtained for the first time by heating white phosphorus under high pressure (1.2 GPa). Compared to the other two allotropes—the amorphous red phosphorus and the notoriously unstable white phosphorus—bP has a higher density (2.69 g/cm3 vs. 2.05–2.34 g/cm3 and 1.83 g/cm3, respectively) and better thermal stability and can withstand temperatures up to 550 °C in the air without catching fire spontaneously [41][28].bP has historically not attracted much interest from the scientific community until 2014, when it was shown that from its crystal structure, it is possible to obtain a new 2D material known as phosphorene, which is formed by a single layer of phosphorus atoms [42,43][29][30]. Phosphorene has an orthorhombic crystalline structure that contains eight atoms per unit cell. Similar to its precursor (bP), each layer is composed of P atoms held together by a strong covalent bond. However, along the stacking direction, the layers interact through weak van der Waals forces. A monolayer of bP is composed of P atoms with five valence electrons with 3s23p3 configuration, each covalently bonded to the other three neighboring P atoms by their p-orbitals [44,45][31][32].Regarding the single layer, P atoms are not arranged in the same plane: Some are in the upper sublayer, and the others are in the lower sublayer, together constituting the unique puckered honeycomb crystal structure. The distance between the upper and lower sublayers is ~2.1 Å, and the distance between the two layers is about 5 Å [46][33]. bP belongs to the orthorhombic system with reduced symmetry, with the main crystallographic b axis normalized among the layers. In the layer plane, there are two characteristic crystal orientations.3.1. Physical, Mechanical, and Chemical Features of bP and Phosphorene (bP-NPs)
bP is a semiconductor with a direct band gap, high carrier mobility, and thermal stability in vacuum at around 400 °C. It exhibits a tunable bandgap that varies with the number of layers, ranging from 0.3 eV for bulk to 2 eV for the monolayer. This range covers the spectrum between graphene and transition metal dichalcogenides, rendering bP-NPs an extremely appealing option as a 2D semiconductor [52,53,54][34][35][36]. Together with the bandgap, carrier mobility is layer-dependent, and it has been calculated to achieve the values of 10,000–26,000 cm2/Vs for the monolayer [55][37]. The well-known in-plane anisotropy generates peculiar physical and mechanical responses distinguishing between AC and ZZ directions. Anisotropy affects even the optical properties, showing the dichroic behavior of bP-NPs. In optics, dichroic materials absorb light differently based on its polarization. In the case of bP and bP-NPs, only light with a polarization component along the AC direction is absorbed for frequencies close to the bandgap energy [56][38]. Similar results have been recently experimentally obtained by measuring the photoluminescence behavior of bP-NPs [57][39]. bP exhibits unique mechanical properties due to its puckered structure. When stretched along the y-direction, it displays a negative Poisson’s ratio (−0.027) for the z-direction. This implies that when stretched along one direction, the material expands along the transverse direction, which is contrary to what happens with most materials, where stretching along one direction usually results in a reduction of lateral dimension [54,58][36][40]. With reference to their actual and effective employment, it is necessary to point out that due to their peculiar structure, bP and especially bP-NPs are materials that exhibit high reactivity. This makes bP-NPs remarkably responsive to the surrounding environment and potentially suitable to be patterned and/or functionalized for specific performant applications, especially in the optoelectronics fields [46,48,53][33][35][41]. However, phosphorene reactivity under ambient conditions results in physical and structural changes, leading to degradation [60,61,62,63,64,65][42][43][44][45][46][47].3.2. Preparation of bP and bP-NPs
Several synthetic procedures have been developed and tested for the preparation of phosphorene using both bottom-up and top-down approaches. Bottom-up methods directly synthesize bP-NPs from different molecular precursors through chemical reactions. Chemical vapor deposition, i.e., direct vapor deposition of red phosphorus or bulk bP in vacuum or argon, has not produced satisfactory results [50,67,68][48][49][50]. The precursors’ instability and procedural problems constitute a major limitation to the development of massive production of bP-NPs through bottom-up methods. Top-down methods involve separating the stacked layers of bulk bP to obtain single- or few-layered nanosheets by breaking the Van der Waals bonding. For this method, it is essential to begin with high-purity bulk bP, which can be synthesized using established methodologies. Bridgman first synthesized black phosphorus by heating white phosphorus at 200 °C under high pressure (1.2–1.3 GPa). In 2007, it was found that it could be prepared from red phosphorus at low pressure and 873 K by adding small quantities of gold, tin, and tin(IV) iodide [54][36]. All these processes can produce good-quality bP but are expensive and low-yielding. To date, the absence of a safe, high-throughput, and scalable route for producing bP remains one of the main limits to phosphorene uses. To produce bP-NPs, exfoliation is needed. Based on multilevel quantum chemical calculations, the exfoliation energy of bP is around 151 meV per atom (larger than that of graphite, 61 meV), which accounts for the relative difficulty in exfoliating bP. This is associated with a non-negligible electronic density overlap between the layers; indeed, it should be pointed out that there is debate regarding whether the interlayer bonding can be classified as a Van der Waals type. When a solid, layered material is immersed in a liquid, the interfacial tension is significantly high so that the material–solvent interactions are not able to outweigh the interlayer interactions, and spontaneous exfoliation does not occur. It is indeed necessary to apply external energy to win secondary intra-layer interactions and exfoliate the material. Ultrasounds are used to generate microbubbles, the growth and collapse of which are attributed to the cavitation-induced pressure pulses and acoustic waves consisting of alternate regions of compression and rarefaction [70][51]. The choice of the solvent is crucial; it should have a surface tension similar to the surface energy of the 2D material to maximize the exfoliation rate and inhibit the restacking of nanosheets. In the case of bP, solvents with surface tensions of 35–40 mJ/m2 are used, such as dimethylformamide (DMF), dimethyl sulfoxide (DMSO), N-methyl-2-pyrrolidone (NMP), and N-cyclohexyl-2-pyrrolidone (CHP) [50,54][36][48]. Although anhydrous organic solvents may produce high-quality flakes, they have high boiling points, making postprocessing and disposal more difficult, and they are also hazardous to human health and the environment. After the exfoliation process, if successful, solvent molecules surround the nanoparticles through solvation, which stabilizes them. These molecules are challenging to remove, and they remain even after subsequent centrifugation and redispersion steps that are essential for a final collection of the products. Therefore, particularly in biomedical fields, this can cause safety issues. For this reason, water-based solutions have also been studied as possible sonication mediums. Since water has a surface tension of about 73 mJ/m2, and bulk bP is insoluble in water, stabilizing surfactants are needed to produce stable flakes and avoid aggregation. It is also important to use de-oxygenated water to prevent phosphorene from oxidizing.4. Antimicrobial Photoactivity of bP
After its discovery, bP attracted the interest of researchers mainly due to its possible applications in optoelectronics, photonics, and advanced engineering, and only in the last few years has it also emerged as a possible new 2D material for biomedical applications. bP is highly biodegradable, biocompatible, and safe for use. These properties are essential for the use of bP in medicine [46,48,73,74,75,76,77,78,79][33][41][52][53][54][55][56][57][58]. In living organisms, phosphorus is a crucial element that constitutes approximately 1% of the total body weight. When it degrades, it transforms into harmless phosphate, which exhibits high biocompatibility and low cytotoxicity, preventing its in vivo accumulation. As a 2D material, it intrinsically has a large surface area, making it suitable for the absorption of drug molecules and making it easier to control the kinetics of release [80][59]. It also has a high modulus; thus, it can be used to improve the mechanical strength of biomedical implants.
4.1. Mechanisms of bP Photoactivity
As previously mentioned, bP has a bandgap dependent on the number of layers, which varies from 0.3 eV to 2 eV, from bulk to monolayer. The 3O2/1O2 redox potentials fall within this range [64][46], and thus, bP-NPs can act as light absorbers, mediating the energy transfer to oxygen molecules in the surrounding environment. Molecular dynamic simulations help to predict the mechanism of singlet oxygen (1O2) generation: First, oxygen molecules interact with P lone pairs, leading to the adsorption of O2, and then, the generation of 1O2 occurs through charge transfer [14][60]. The so-generated 1O2 is very unstable and reacts with target systems in the surroundings. Illumination is needed to excite the transition from the ground state of bP-NPs. The most convenient wavelength range for the photosensitizer activation is between 600 and 800 nm, which is called the “optimal therapeutic window”, since it is therapeutically safe and allows for effective tissue penetration of light while still providing enough energy to allow the transition to the excited singlet state of oxygen [23][14] without compromising biological tissues.4.2. Bare bP-NPs
The antimicrobial activities of exfoliated bP nanosheets have been compared to those of bulk bP and other 2D materials such as graphene and transition metal dichalcogenides such as MoS2, showing a better performance in killing both Gram-negative Escherichia coli and Gram-positive Staphylococcus aureus that cause serious infections. Under 808 nm laser irradiation, a value of 99.2% in bacterial killing percentage against both E. coli and S. aureus was reached mainly by means of photothermal inactivation with a negligible cytotoxicity towards mammal cells even at high bP-NP concentrations [84][61]. A few nanograms of bP nanosheets have been shown to be enough for a strong and broad-spectrum antimicrobial activity toward the bacteria Escherichia coli, Pseudomonas aeruginosa, MRSA, Salmonella typhimurium, and Bacillus cereus as well as the fungal strains Candida albicans, Candida auris, and Cryptococcus neoformans, displaying the effectiveness of bP-NPs as an antibacterial additive in surface coatings, too. High-resolution microscopy and ATR-FTIR studies have revealed that the physical interaction of the bP-NPs with the microbial membranes, together with the oxidative stress, cause important physical and biochemical damages to the phospholipids and to the amide I and II proteins, whereas this results in slight chemical modifications to polysaccharides and nucleic acids of Gram-positive methicillin-resistant Staphylococcus aureus (MRSA) and Gram-negative Pseudomonas aeruginosa and to the fungal species Candida albicans [86][62].4.3. bP-NPs-Based Hybrid Materials
bP has also emerged as a suitable nanomaterial that allows for drug delivery and therapeutics due to its high surface area. As it is negatively charged in water with an interlay distance of ~5.24 Å, the encapsulation of small and positively charged molecular drugs within the interlayer spaces is possible mainly through electrostatic interactions. To date, three major strategies have been explored for developing bP-NPs-based hybrid materials: electrostatic interaction, covalent bonding, and noncovalent bonding (e.g., hydrophobic interactions) [83][63].5. Conclusions
The advancements in biomedical research and the development of nanotechnology can have great potential for fighting multi-drug-resistant microbial pathogens. Among 2D nanomaterials, bP-NP has shown promising properties for various applications, including antimicrobial phototherapy. It has a high specific surface area, which can enhance its interactions with microbial pathogens. The puckered structure of bP-NPs along with their peculiar anisotropy contribute to unique electronic, mechanical, and photoactivity properties that are potentially exploitable in an antimicrobial system. The thickness-dependent band gap (0.3–2 eV) allows it to absorb a wide range of wavelengths from ultraviolet to NIR. Due to its broad absorption band and unique electronic structure, bP-NP possesses an intrinsic photoactivity that makes it an effective phototherapeutic agent in PTT and PDT against pathogenic bacteria. Furthermore, bP nanomaterials have excellent biocompatibility and biodegradability in vivo, minimizing potential adverse effects on living tissues and facilitating their use in antibacterial activity as well as other biomedical applications. Phosphorene-based nanomaterials can be engineered for targeted drug delivery. This is particularly relevant in antimicrobial phototherapy, where the precise delivery of therapeutic agents to the infection site is crucial for maximizing treatment efficacy and minimizing side effects. Along with their many discussed advantages in antimicrobial applications, there are still several challenges facing bP-NPs that need to be addressed, such as the need for efficient and low-cost synthesis strategies to facilitate large-scale production, stability optimization for ensuring efficacy in biomedical applications, and systematic studies correlating structural characteristics with the antimicrobial properties. In fact, although numerous current studies and insights on the structure, chemical, and physical characteristics of bare bP-NPs are present, the research on applications of bP-NPs-based hybrids in aPDT and aPTT often lacks clear reports on the structure/chemical composition of the investigated hybrid systems and on the mechanisms involved in the photoactivation phenomena. The modification and embedding of bP-NPs in different substrates, such as other photosensitizers, drugs, and protective agents, to create hybrid systems can complicate the identification and rationalization of individual components’ contributions to photoactivation processes.
References
- Shaw, Z.L.; Kuriakose, S.; Cheeseman, S.; Dickey, M.D.; Genzer, J.; Christofferson, A.J.; Crawford, R.J.; McConville, C.F.; Chapman, J.; Truong, V.K.; et al. Antipathogenic Properties and Applications of Low-Dimensional Materials. Nat. Commun. 2021, 12, 3897.
- Jayakumar, A.; Mathew, S.; Radoor, S.; Kim, J.T.; Rhim, J.-W.; Siengchin, S. Recent Advances in Two-Dimensional Nanomaterials: Properties, Antimicrobial, and Drug Delivery Application of Nanocomposites. Mater. Today Chem. 2023, 30, 101492.
- Zhang, C.; Wang, Y.; Ma, J.; Zhang, Q.; Wang, F.; Liu, X.; Xia, T. Black Phosphorus for Fighting Antibiotic-Resistant Bacteria: What Is Known and What Is Missing. Sci. Total Environ. 2020, 721, 137740.
- Favron, A.; Gaufrès, E.; Fossard, F.; Phaneuf-L’Heureux, A.-L.; Tang, N.Y.-W.; Lévesque, P.L.; Loiseau, A.; Leonelli, R.; Francoeur, S.; Martel, R. Photooxidation and Quantum Confinement Effects in Exfoliated Black Phosphorus. Nat. Mater. 2015, 14, 826–832.
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial Photodynamic Therapy—What We Know and What We Don’t. Crit. Rev. Microbiol. 2018, 44, 571–589.
- Maisch, T. Photoantimicrobials—An Update. Transl. Biophotonics 2020, 2, e201900033.
- Correia, J.H.; Rodrigues, J.A.; Pimenta, S.; Dong, T.; Yang, Z. Photodynamic Therapy Review: Principles, Photosensitizers, Applications, and Future Directions. Pharmaceutics 2021, 13, 1332.
- Kashef, N.; Huang, Y.-Y.; Hamblin, M.R. Advances in Antimicrobial Photodynamic Inactivation at the Nanoscale. Nanophotonics 2017, 6, 853–879.
- Cieplik, F.; Tabenski, L.; Buchalla, W.; Maisch, T. Antimicrobial Photodynamic Therapy for Inactivation of Biofilms Formed by Oral Key Pathogens. Front. Microbiol. 2014, 5, 405.
- Hu, X.; Huang, Y.-Y.; Wang, Y.; Wang, X.; Hamblin, M.R. Antimicrobial Photodynamic Therapy to Control Clinically Relevant Biofilm Infections. Front. Microbiol. 2018, 9, 1299.
- Delcanale, P.; Abbruzzetti, S.; Viappiani, C. Photodynamic Treatment of Pathogens. Riv. Nuovo Cim. 2022, 45, 407–459.
- Pérez-Laguna, V.; Gilaberte, Y.; Millán-Lou, M.I.; Agut, M.; Nonell, S.; Rezusta, A.; Hamblin, M.R. A Combination of Photodynamic Therapy and Antimicrobial Compounds to Treat Skin and Mucosal Infections: A Systematic Review. Photochem. Photobiol. Sci. 2019, 18, 1020–1029.
- Nonell, S. Oxygen (and Lack Thereof) in Photodynamic Therapy. Photodiagnosis Photodyn. Ther. 2023, 41, 103440.
- Wong, W.F.; Melendez, A.J. INTRODUCTION. Clin. Exp. Pharma Physio 2006, 33, 480–481.
- Zhang, Y.; Zhu, Y.; Chen, J.; Wang, Y.; Sherwood, M.E.; Murray, C.K.; Vrahas, M.S.; Hooper, D.C.; Hamblin, M.R.; Dai, T. Antimicrobial Blue Light Inactivation of Candida Albicans: In Vitro and In Vivo Studies. Virulence 2016, 7, 536–545.
- Wang, Y.; Wu, X.; Chen, J.; Amin, R.; Lu, M.; Bhayana, B.; Zhao, J.; Murray, C.K.; Hamblin, M.R.; Hooper, D.C.; et al. Antimicrobial Blue Light Inactivation of Gram-Negative Pathogens in Biofilms: In Vitro and In Vivo Studies. J. Infect. Dis. 2016, 213, 1380–1387.
- Dai, T.; Gupta, A.; Huang, Y.-Y.; Yin, R.; Murray, C.K.; Vrahas, M.S.; Sherwood, M.E.; Tegos, G.P.; Hamblin, M.R. Blue Light Rescues Mice from Potentially Fatal Pseudomonas Aeruginosa Burn Infection: Efficacy, Safety, and Mechanism of Action. Antimicrob. Agents Chemother. 2013, 57, 1238–1245.
- Hamblin, M.R.; Viveiros, J.; Yang, C.; Ahmadi, A.; Ganz, R.A.; Tolkoff, M.J. Helicobacter Pylori Accumulates Photoactive Porphyrins and Is Killed by Visible Light. Antimicrob. Agents Chemother. 2005, 49, 2822–2827.
- Morici, P.; Battisti, A.; Tortora, G.; Menciassi, A.; Checcucci, G.; Ghetti, F.; Sgarbossa, A. The In Vitro Photoinactivation of Helicobacter Pylori by a Novel LED-Based Device. Front. Microbiol. 2020, 11, 283.
- Battisti, A.; Morici, P.; Sgarbossa, A. Fluorescence Lifetime Imaging Microscopy of Porphyrins in Helicobacter Pylori Biofilms. Pharmaceutics 2021, 13, 1674.
- Schmid, J.; Hoenes, K.; Vatter, P.; Hessling, M. Antimicrobial Effect of Visible Light—Photoinactivation of Legionella Rubrilucens by Irradiation at 450, 470, and 620 Nm. Antibiotics 2019, 8, 187.
- Wang, Y.; Ferrer-Espada, R.; Baglo, Y.; Goh, X.S.; Held, K.D.; Grad, Y.H.; Gu, Y.; Gelfand, J.A.; Dai, T. Photoinactivation of Neisseria Gonorrhoeae: A Paradigm-Changing Approach for Combating Antibiotic-Resistant Gonococcal Infection. J. Infect. Dis. 2019, 220, 873–881.
- Fyrestam, J.; Bjurshammar, N.; Paulsson, E.; Johannsen, A.; Östman, C. Determination of Porphyrins in Oral Bacteria by Liquid Chromatography Electrospray Ionization Tandem Mass Spectrometry. Anal. Bioanal. Chem. 2015, 407, 7013–7023.
- Yoshida, A.; Sasaki, H.; Toyama, T.; Araki, M.; Fujioka, J.; Tsukiyama, K.; Hamada, N.; Yoshino, F. Antimicrobial Effect of Blue Light Using Porphyromonas Gingivalis Pigment. Sci. Rep. 2017, 7, 5225.
- Biener, G.; Masson-Meyers, D.S.; Bumah, V.V.; Hussey, G.; Stoneman, M.R.; Enwemeka, C.S.; Raicu, V. Blue/Violet Laser Inactivates Methicillin-Resistant Staphylococcus Aureus by Altering Its Transmembrane Potential. J. Photochem. Photobiol. B Biol. 2017, 170, 118–124.
- Alves, E.; Faustino, M.A.; Neves, M.G.; Cunha, A.; Tome, J.; Almeida, A. An Insight on Bacterial Cellular Targets of Photodynamic Inactivation. Future Med. Chem. 2014, 6, 141–164.
- Kaur, K.; Reddy, S.; Barathe, P.; Shriram, V.; Anand, U.; Proćków, J.; Kumar, V. Combating Drug-Resistant Bacteria Using Photothermally Active Nanomaterials: A Perspective Review. Front. Microbiol. 2021, 12, 747019.
- Ahamed, M.I.; Shakeel, N.; Anwar, N. Structure and Fundamental Properties of Black Phosphorus; Inamuddin, Boddula, R., Asiri, A.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; pp. 139–156.
- Fukuoka, S.; Taen, T.; Osada, T. Electronic Structure and the Properties of Phosphorene and Few-Layer Black Phosphorus. J. Phys. Soc. Jpn. 2015, 84, 121004.
- Liu, H.; Neal, A.T.; Zhu, Z.; Luo, Z.; Xu, X.; Tománek, D.; Ye, P.D. Phosphorene: An Unexplored 2D Semiconductor with a High Hole Mobility. ACS Nano 2014, 8, 4033–4041.
- Du, H.; Lin, X.; Xu, Z.; Chu, D. Recent Developments in Black Phosphorus Transistors. J. Mater. Chem. C 2015, 3, 8760–8775.
- Kou, L.; Chen, C.; Smith, S.C. Phosphorene: Fabrication, Properties, and Applications. J. Phys. Chem. Lett. 2015, 6, 2794–2805.
- Pica, M.; D’Amato, R. Chemistry of Phosphorene: Synthesis, Functionalization and Biomedical Applications in an Update Review. Inorganics 2020, 8, 29.
- Cai, Y.; Zhang, G.; Zhang, Y.-W. Electronic Properties of Phosphorene/Graphene and Phosphorene/Hexagonal Boron Nitride Heterostructures. J. Phys. Chem. C 2015, 119, 13929–13936.
- Peruzzini, M.; Bini, R.; Bolognesi, M.; Caporali, M.; Ceppatelli, M.; Cicogna, F.; Coiai, S.; Heun, S.; Ienco, A.; Benito, I.I.; et al. A Perspective on Recent Advances in Phosphorene Functionalization and Its Applications in Devices. Eur. J. Inorg. Chem. 2019, 2019, 1476–1494.
- Carvalho, A.; Wang, M.; Zhu, X.; Rodin, A.S.; Su, H.; Castro Neto, A.H. Phosphorene: From Theory to Applications. Nat. Rev. Mater. 2016, 1, 16061.
- Zhang, Y.; Wang, J.; Liu, Q.; Gu, S.; Sun, Z.; Chu, P.K.; Yu, X. The Electrical, Thermal, and Thermoelectric Properties of Black Phosphorus. APL Mater. 2020, 8, 120903.
- Torbatian, Z.; Asgari, R. Optical Absorption Properties of Few-Layer Phosphorene. Phys. Rev. B 2018, 98, 205407.
- Molas, M.R.; Macewicz, Ł.; Wieloszyńska, A.; Jakóbczyk, P.; Wysmołek, A.; Bogdanowicz, R.; Jasinski, J.B. Photoluminescence as a Probe of Phosphorene Properties. npj 2d Mater. Appl. 2021, 5, 83.
- Jiang, J.-W.; Park, H.S. Negative Poisson’s Ratio in Single-Layer Black Phosphorus. Nat. Commun. 2014, 5, 4727.
- Chaudhary, V.; Neugebauer, P.; Mounkachi, O.; Lahbabi, S.; El Fatimy, A. Phosphorene—An Emerging Two-Dimensional Material: Recent Advances in Synthesis, Functionalization, and Applications. 2D Mater. 2022, 9, 032001.
- Van Druenen, M.; Davitt, F.; Collins, T.; Glynn, C.; O’Dwyer, C.; Holmes, J.D.; Collins, G. Evaluating the Surface Chemistry of Black Phosphorus during Ambient Degradation. Langmuir 2019, 35, 2172–2178.
- Van Druenen, M. Degradation of Black Phosphorus and Strategies to Enhance Its Ambient Lifetime. Adv. Mater. Inter. 2020, 7, 2001102.
- Abellán, G.; Wild, S.; Lloret, V.; Scheuschner, N.; Gillen, R.; Mundloch, U.; Maultzsch, J.; Varela, M.; Hauke, F.; Hirsch, A. Fundamental Insights into the Degradation and Stabilization of Thin Layer Black Phosphorus. J. Am. Chem. Soc. 2017, 139, 10432–10440.
- Zhang, T.; Wan, Y.; Xie, H.; Mu, Y.; Du, P.; Wang, D.; Wu, X.; Ji, H.; Wan, L. Degradation Chemistry and Stabilization of Exfoliated Few-Layer Black Phosphorus in Water. J. Am. Chem. Soc. 2018, 140, 7561–7567.
- Zhou, Q.; Chen, Q.; Tong, Y.; Wang, J. Light-Induced Ambient Degradation of Few-Layer Black Phosphorus: Mechanism and Protection. Angew. Chem. Int. Ed. 2016, 55, 11437–11441.
- Walia, S.; Sabri, Y.; Ahmed, T.; Field, M.R.; Ramanathan, R.; Arash, A.; Bhargava, S.K.; Sriram, S.; Bhaskaran, M.; Bansal, V.; et al. Defining the Role of Humidity in the Ambient Degradation of Few-Layer Black Phosphorus. 2D Mater. 2016, 4, 015025.
- Wu, S.; Hui, K.S.; Hui, K.N. 2D Black Phosphorus: From Preparation to Applications for Electrochemical Energy Storage. Adv. Sci. 2018, 5, 1700491.
- Smith, J.B.; Hagaman, D.; Ji, H.-F. Growth of 2D Black Phosphorus Film from Chemical Vapor Deposition. Nanotechnology 2016, 27, 215602.
- El Hammoumi, M.; Chaudhary, V.; Neugebauer, P.; El Fatimy, A. Chemical Vapor Deposition: A Potential Tool for Wafer Scale Growth of Two-Dimensional Layered Materials. J. Phys. D Appl. Phys. 2022, 55, 473001.
- Mukhopadhyay, T.K.; Datta, A. Disentangling the Liquid Phase Exfoliation of Two-Dimensional Materials: An “In Silico ” Perspective. Phys. Chem. Chem. Phys. 2020, 22, 22157–22179.
- Zhu, Y.; Xie, Z.; Li, J.; Liu, Y.; Li, C.; Liang, W.; Huang, W.; Kang, J.; Cheng, F.; Kang, L.; et al. From Phosphorus to Phosphorene: Applications in Disease Theranostics. Coord. Chem. Rev. 2021, 446, 214110.
- Qiu, M.; Ren, W.X.; Jeong, T.; Won, M.; Park, G.Y.; Sang, D.K.; Liu, L.-P.; Zhang, H.; Kim, J.S. Omnipotent Phosphorene: A next-Generation, Two-Dimensional Nanoplatform for Multidisciplinary Biomedical Applications. Chem. Soc. Rev. 2018, 47, 5588–5601.
- Tatullo, M.; Genovese, F.; Aiello, E.; Amantea, M.; Makeeva, I.; Zavan, B.; Rengo, S.; Fortunato, L. Phosphorene Is the New Graphene in Biomedical Applications. Materials 2019, 12, 2301.
- Akhtar, M.; Anderson, G.; Zhao, R.; Alruqi, A.; Mroczkowska, J.E.; Sumanasekera, G.; Jasinski, J.B. Recent Advances in Synthesis, Properties, and Applications of Phosphorene. npj 2d Mater. Appl. 2017, 1, 5.
- Liu, X.; Gaihre, B.; George, M.N.; Li, Y.; Tilton, M.; Yaszemski, M.J.; Lu, L. 2D Phosphorene Nanosheets, Quantum Dots, Nanoribbons: Synthesis and Biomedical Applications. Biomater. Sci. 2021, 9, 2768–2803.
- Kim, J.; Sando, S.; Cui, T. Biosensor Based on Layer by Layer Deposited Phosphorene Nanoparticles for Liver Cancer Detection. In Proceedings of the ASME 2017 International Mechanical Engineering Congress and Exposition, Tampa, FL, USA, 3–9 November 2017.
- Liu, H.; Mei, Y.; Zhao, Q.; Zhang, A.; Tang, L.; Gao, H.; Wang, W. Black Phosphorus, an Emerging Versatile Nanoplatform for Cancer Immunotherapy. Pharmaceutics 2021, 13, 1344.
- Wang, Z.; Liu, Z.; Su, C.; Yang, B.; Fei, X.; Li, Y.; Hou, Y.; Zhao, H.; Guo, Y.; Zhuang, Z.; et al. Biodegradable Black Phosphorus-Based Nanomaterials in Biomedicine: Theranostic Applications. CMC 2019, 26, 1788–1805.
- Wang, H.; Yang, X.; Shao, W.; Chen, S.; Xie, J.; Zhang, X.; Wang, J.; Xie, Y. Ultrathin Black Phosphorus Nanosheets for Efficient Singlet Oxygen Generation. J. Am. Chem. Soc. 2015, 137, 11376–11382.
- Sun, Z.; Zhang, Y.; Yu, H.; Yan, C.; Liu, Y.; Hong, S.; Tao, H.; Robertson, A.W.; Wang, Z.; Pádua, A.A.H. New Solvent-Stabilized Few-Layer Black Phosphorus for Antibacterial Applications. Nanoscale 2018, 10, 12543–12553.
- Shaw, Z.L.; Cheeseman, S.; Huang, L.Z.Y.; Penman, R.; Ahmed, T.; Bryant, S.J.; Bryant, G.; Christofferson, A.J.; Orrell-Trigg, R.; Dekiwadia, C.; et al. Illuminating the Biochemical Interaction of Antimicrobial Few-Layer Black Phosphorus with Microbial Cells Using Synchrotron Macro-ATR-FTIR. J. Mater. Chem. B 2022, 10, 7527–7539.
- Liu, W.; Dong, A.; Wang, B.; Zhang, H. Current Advances in Black Phosphorus-Based Drug Delivery Systems for Cancer Therapy. Adv. Sci. 2021, 8, 2003033.
