Injuries to the peripheral nervous system are a common clinical issue, causing dysfunctions of the motor and sensory systems. Surgical interventions such as nerve autografting are necessary to repair damaged nerves. Even with autografting, i.e., the gold standard, malfunctioning and mismatches between the injured and donor nerves often lead to unwanted failure. Thus, there is an urgent need for a new intervention in clinical practice to achieve full functional recovery. Nerve guidance conduits (NGCs), providing physicochemical cues to guide neural regeneration, have great potential for the clinical regeneration of peripheral nerves. Typically, NGCs are tubular structures with various configurations to create a microenvironment that induces the oriented and accelerated growth of axons and promotes neuron cell migration and tissue maturation within the injured tissue. Once the native neural environment is better understood, ideal NGCs should maximally recapitulate those key physiological attributes for better neural regeneration.
- nerve guidance conduits
- regenerative medicine
- biomimetic
- peripheral nerve regeneration
1. Introduction
2. Physicochemical Properties and Regeneration Capacity of Peripheral Nerves
2.1. Structural, Compositional, and Physical Properties of Peripheral Nerves
Structurally, nerves within the PNS are organized in a hierarchal fashion to facilitate signal transduction from the CNS to peripheral tissues and organs. As the functional unit of the nervous system, neurons are specialized cells that transmit electrical and chemical signals to communicate with other cells at the synaptic junction [1]. They have three major components: (1) the cell body, where essential cell functions are performed, (2) the dendrites, which receive signals at the synapse, and (3) the axon, which acts as a cable to transmit electrical signals to the next synapse. The axon is typically covered by a myelin sheath, comprising lipids and proteins, to protect the axon and conduct electrical signals [16]. The myelinated axons, also called nerve fibers, vary in diameter from 0.2 to 20 µm, depending on their function and location [17], and are surrounded by the basal lamina. The basal lamina is a highly specialized ECM secreted by myelinating SCs, and it is primarily composed of type IV collagen and laminin crosslinked with proteoglycans such as heparin sulfonate [18]. The space between individual nerve fibers is called the endoneurium, and it comprises highly aligned type III collagen fibers in the same direction as the axons [19]. Nerve fibers are bundled into fascicles via a connective tissue layer called the perineurium. The size of fascicles differs between nerves and within the same nerve at different locations [20]. Fascicles are, further, bundled with a connective tissue layer called the epineurium to form a larger nerve. In addition to neurons, the PNS contains several glial cells, such as SCs, satellite glial cells (SGCs), and neural stem cells, that maintain the extracellular environment and support the neurons. SCs protect the peripheral axons, regardless of myelination. During the myelination of the peripheral axon, an SC envelops a segment of the axon within its cytoplasmic groove and wraps around the axon to form the myelin sheath [19]. Unmyelinated axons are protected and supported by SCs that envelop and surround multiple unmyelinated axons to form small, shallow grooves in the nerve bundle. However, it requires a series of SCs to enclose an entire axon. SGCs are another functional cell type found in the PNS that envelop the cell bodies of neurons in the peripheral ganglia [19]. While their functions are not fully understood, SGCs are believed to play a critical role in neural homeostasis and have been studied in relation to pain [23][21]. Throughout the connective tissue of the nerves, neural stem cells can be found in the ganglia. These stem cells are typically dormant but can be activated after injury to aid in the regeneration of damaged tissue [17]. Peripheral nerves experience various stresses during body movements such as stretching. As such, the PNS possesses specific mechanical properties crucial to its function and response to forces. Peripheral nerves are viscoelastic, enabling them to deform while dissipating energy. Typically, nerves can elongate 6% to 8% during normal body movement without damaging the tissue. However, straining the nerves by more than 11% but to an extent that is still within the toe region may trigger pain (See Figure 21). Upon exceeding 15%, the start of the elastic region, nerves begin to experience damage [17,18][17][18]. However, due to their viscoelastic nature, the maximal strain that nerves can withstand prior to injury depends on the strain rate [26][22].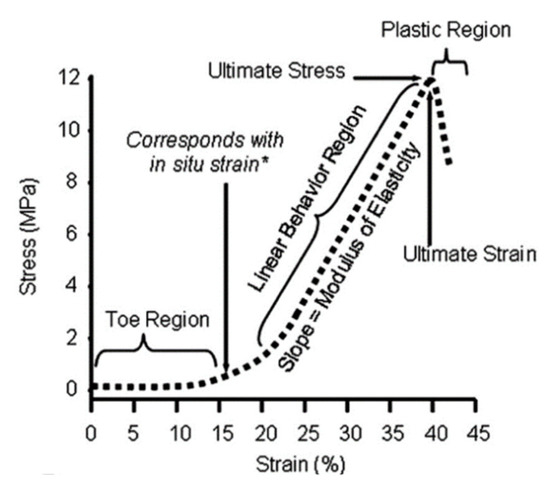
2.2. Regeneration of Peripheral Nerve Injuries
3. Design Considerations for Biomimetic Nerve Guidance Conduits
NGCs have undergone an evolution from simple, hollow-tube designs to multi-channeled structures and from nondegradable materials to bioactive, degradable materials. For example, first-generation conduits were solely meant to act as barriers against connective tissue infiltration while guiding directional nerve regrowth [31][24]. These conduits typically comprise decellularized allogenic or xenogeneic tissue or non-biodegradable materials such as silicone or polytetrafluorethylene (PTFE). However, decellularized grafts rely on scarce and costly sources, and non-biodegradable materials could cause complications, such as fibrotic encapsulation. Thus, the design of second-generation nerve conduits has focused on improving the biocompatibility of the implants via utilizing resorbable materials that are semi-permeable. Still, the simple, hollow tubular design of second-generation NGCs yields subpar functional regeneration compared to autologous nerve grafting. In recognition of the limitations of these designs and the growing body of evidence demonstrating the influence of extracellular features such as material stiffness, hydrophilicity, topography, and chemical properties on neural cell processes [32[25][26],33], emerging efforts are being made to formulate an encouraging microenvironment to improve functional recovery. As such, there is increased interest in designing NGCs that can recapitulate the key features of the native ECM of the PNS [6,30,31][6][24][27]. Diverse attempts have been made to fabricate NGCs that mimic various aspects, such as the composition, structure, and mechanics of nervous tissue.3.1. Compositional Considerations
3.1.1. Material Choice
Nerve conduits can be made from various materials, including synthetic, natural, or hybrid biomaterials [36,37][28][29], and each type of material has its advantages and disadvantages. For example, nerve conduits made from collagen and poly (lactic-co-glycolic) acid (PLGA) have already received approval for the clinical treatment of peripheral nerve injuries because of their degradability and good biocompatibility. However, these grafts only partially meet clinical needs due to their limited effectiveness in promoting nerve repair, as well as the associated costs [36][28]. Compared to natural materials, synthetic polymers offer several benefits, such as ease of modification, minimal risk of pathogenic infection, low toxicity, and, presumably, fewer immune responses [38][30]. However, these materials often fail to provide biological guidance, as they lack the functional motifs found in the native ECM of nervous tissue. Conversely, natural materials are appropriate for cell growth and adhesion but may cause uninvited immune responses and are less reproducible [39][31].Biomimetic Synthetic Materials
Typically, synthetic materials are chosen for their ability to mimic the mechanical or conductive properties of native nerves [64][32]. To accommodate the mechanical complexity of native nerves, efforts have been made to combine various synthetic polymers, such as polylactic acid (PLA), polyglycolic acid (PGA), poly(lactic-co-glycolic acid) (PLGA), and polycaprolactone (PCL), to create multilayered NGCs with varying mechanical strength and degradation profiles [65][33]. Considering the conductive nature of nerves for electrical signals, synthetic conductive polymers have received particular attention for their use in fabricating NGCs. Emerging evidence has shown that incorporating conductive polymers into NGCs promotes axonal outgrowth [68,69][34][35], aids in establishing connections with neuronal circuits, and enhances nerve impulse conduction to encourage nerve regeneration [70][36]. Among several representative conductive synthetic materials, such as polypyrrole (PPy), polyaniline (PANI), poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS), and reduced graphene oxide (rGO), PPy has been demonstrated to be a popular choice for bioelectric applications such as electrode-cell interfaces in bionics [41][37]. The characteristics of PPy, such as its biocompatibility, conductivity, dynamic nature, and ease of polymerization, make it an ideal candidate for creating NGCs. PANI is another material that is commonly employed to create conductive NGCs. Studies have shown that PANI is beneficial in promoting cell attachment, proliferation, and differentiation [44][38], and PANI-coated intraluminal microtubes within NGCs favor nerve regeneration [43][39]. Lately, PEDOT:PSS has also gained substantial recognition and growing interest in biomedical applications for its unique properties [71][40]. As a polyelectrolyte complex, PEDOT:PSS has excellent conductivity and can disperse in an aqueous solution to form hydrogels, allowing for easy processing and application [45][41]. In an effort to further demonstrate the importance of conductivity, NGCs coated with carbon nanotubes (CNTs) or PEDOT:PSS were used to regenerate the recurrent laryngeal nerve defects in a rabbit model, and it was found that the conductive NGCs could help improve vocal cord mobility and reduce thyroarytenoid muscle atrophy [46][42].Biomimetic Natural Materials
To take advantage of the inherent bioactivity of natural polymers, many materials, such as chitosan, collagen, silk fibroin, gelatin, HA, and fibrin, have been used to fabricate NGCs. These materials can create an environment that facilitates and promotes cellular activities and supports cell-material interactions that are favorable for nerve regeneration [35][43]. Among various natural polymers, ECM-derived biomaterials, such as collagen, gelatin (hydrolyzed collagen), and fibrin, are commonly used in particular consideration of their presence in native nerve ECM. As the primary component of nervous ECM, collagen has been extensively explored to fabricate NGCs. A recent study showed that NGCs comprising collagen and chondroitin-6-sulfate and incorporated with fibronectin and laminin biomolecules enhanced the proliferation of SCs in vitro and regenerated the injured rat sciatic nerve in a similar capacity to autografts [49][44]. Despite the noticeable advantages, ECM-derived materials often face challenges, such as high manufacturing costs and large source-dependent variabilities. In response, there is a continuous motivation to seek alternative ECM-like materials, such as silk fibroin (SF) and chitosan (CS). With a degradation rate close to the rate of nerve tissue ingrowth [63][45], SF-based NGCs displayed a lower risk of inflammation and rejection [72][46].Biomimetic Hybrid Materials
Hybrid biomaterials, a subset of composites combining the biological properties of natural materials with the mechanical and functional properties of synthetic materials, offer a wide range of benefits, including tunable mechanical and degradation properties, scalable production, ease of processing, and biological activity [75][47]. There are many variations of hybrid materials, but recent interest in mimicking both the composition and function of nerves has led to the development of hybrid NGCs that contain two to three types of materials: (1) natural materials to improve cell adhesion and migration, (2) conductive synthetic materials to mimic the function of nerves, and (3) an optional synthetic material to provide mechanical strength. In an effort to fabricate hybrid conductive conduits, PEDOT:PSS has often been included together with other materials, given that PEDOT:PSS can be made into various forms, is mechanically robust, and is highly elastic. For example, a recent publication demonstrated that a gelatin–PEDOT:PSS hydrogel was conductive and biocompatible, and was able to prolong astrocyte growth [76][48]. While this combination has not been used in NGCs, the encouraging results in supporting the cells of the CNS also imply its potential for PNI regeneration. rGO is another popular choice for creating hybrid conductive NGCs due to its high conductivity, mechanical properties, and ability to be coated onto or dispersed into other materials. For example, NGCs fabricated via coating rGO on M. Menelaus butterfly wings effectively promoted the axonal regrowth and functional regeneration of nerve and muscle tissue compared to non-conductive controls [68][34]. To better tailor the mechanical properties, additional materials can also be included in the mixture. For example, in a recent study on the creation of a hollow NGC, PLCL with good strength and toughness was added to SF to improve the flexibility and strength of the electrospun nanofibers of a conduit, which was coated with PPy [46][42]. This hybrid conductive NGC improved the early proliferation of SCs post-surgery and enhanced myelin formation, and most importantly, it was demonstrated to be effective in restoring sciatic function, similar to autologous graft, 12 weeks post-surgery.3.1.2. Cellular and Biomolecular Choices
Cellular Choices
In recognition of the roles of SCs and neural stem cells in supporting, promoting, and guiding axonal sprouting following PNIs, patient-derived cells have been incorporated into NGCs to minimize the risk of immune rejection [82][49], promote axonal regrowth, and provide neurotrophic support [73][50]. For example, Liu et al. demonstrated that collagen-based conduits loaded with neural stem cells returned functional levels in the regeneration of 3-mm rat spinal cord injury models [83][51]. This is very promising, given that spinal cord injuries are more difficult to regenerate than PNIs. However, neural stem cell transplants alone cannot regenerate PNIs, as evidenced by the outperformance of neural stem-cell-seeded NGCs over the cell transplant-only group in a mouse sciatic injury model [84][52]. Despite the encouraging results from the encapsulation of SCs and neural stem cells in NGCs, it remains challenging to harvest and expand such autologous cells for clinical use. As such, Schwann-like cells derived from mesenchymal stem cells (MSCs) have been considered a promising alternative [11,86][11][53]. Results demonstrate that MSCs enhance the expression of neurotrophic and angiogenic factors that can better emulate the native environment for nerve regeneration [86][53].Biomolecular Choices
Given that the primary purpose of encapsulating cells in NGCs is their ability to secrete growth factors and ECM proteins, oftentimes, reaching the desired therapeutic concentration is challenging. To this end, incorporating biomolecules directly into NGCs would allow for more control of the concentration needed, along with reductions in cost, time, and the risk of immune rejection associated with cellularized NGCs. The typical biomolecules involved in nerve regeneration are neurotrophic factors (NTFs), such as NGF, BDNF, or glial-derived neurotrophic factor (GDNF), which promote neural cell survival and guide axonal growth [91][54]. Since NTFs are present at different stages and varying concentrations during nerve regeneration, the regeneration capacity and rate are closely regulated by the initial concentration, dosage, and release kinetics of NTFs [92][55].3.2. Structural Considerations
3.2.1. Biomimetic Architectures
To better guide axonal growth and support nerve regeneration, it would be desirable for NGCs to capture the microstructural features of native nerves [99][56]. In recognition of the drawbacks of non-porous, hollow NGCs, such as the limited capacity of bridging nerve gaps over 1 cm and poor permeability to nutrients and growth factors [100][57], secondary structures such as grooves, pores, channels, and fillers have been added to NGCs [101][58]. The creation of aligned grooves or channels in NGCs could effectively mimic the fibrin network and Büngner bands for axonal regeneration, while the presence of fillers and pores supports cell infiltration, neovascularization, and nutrient diffusion to the regenerating nerves [102][59] (see Figure 52).
Biomimetic Topographical Features of NGCs
Biomimetic Fillers for NGCs
Hollow NGCs, regardless of their added topographical features, still fail to capture the key structural attributes of native nerves, that is, containing multiple fascicles that each house numerous axons. In this regard, a growing interest has shifted to filling the luminal space of NGCs with various fillers, especially the aligned anisotropic ones, closely mimicking the conditions of the endoneurium surrounding individual axons to improve the regenerative capacity of nerves. There is growing interest in utilizing natural polymers as NGC fillers in the forms of hydrogels, filaments, or porous sponges. Due to their soft properties and biocompatibility, these materials can serve effectively as regenerative guides for repairing PNIs and supporting the functionality of nerve conduits during the repair process [117][71]. Compared to hydrogels, anisotropic aligned fillers are believed to better induce axonal organization than random fillers by encouraging axons to extend through the conduit while facilitating nutrient exchange. For instance, CS fibers or chitin-containing composite fibers are employed as fillers. These fibers guide axonal growth through their oriented structure to establish electrical connectivity between injured nerve terminals [119][72]. Fillers can also be functionalized with biomolecules or conductive polymers to achieve high biomimicry. The introduction of pores can aid in nerve regeneration and nutrient exchange; however, pores larger than 30 µm may lead to the infiltration of fibrous tissue, thereby blocking axonal regeneration. To this end, coating the fillers with fibrin and HA can help alleviate this issue [104][61].Multi-Channeled NGCs
While filled NGCs are able to facilitate neuronal cell adhesion, promote cell differentiation, and guide neurite outgrowth [123[73][74][75],124,125], fillers, occupying the luminal space, would limit the number and size of axons growing through the conduit [126][76]. As such, multi-channeled NGCs have been proposed to enhance nerve regeneration within large nerve defects, as they can provide better guidance for axonal ingrowth than the pores of anisotropic fillers [93,126][76][77]. These channels also partially mimic the unique morphology and structure of peripheral nerve fascicles, where myelinated and unmyelinated axons are surrounded by a collagen-based endoneurium, to promote neurogenesis and nerve regeneration [62][78]. The primary difficulty in mimicking fascicles is the large variation in fascicle size and axon number, which are closely related to the location and function of the peripheral nerve [127][79]. Typically, the diameter of axons ranges between 4 and 16 μm [128][80], and fascicles can contain a few to several hundred axons [129][81]; thus, it remains highly challenging to identify an ideal channel size that effectively mimics the fascicle anatomy. Through experimentation, researchers have noticed that the channel diameter of ~125 μm seemed more favorable to encouraging functional regeneration [85][82]. Apart from the channel diameter, the number of channels in an NGC also impacts nerve regeneration. As expected, a higher density of channels could effectively reduce the dispersion of axons, therefore promoting more directed and organized axonal growth [2].3.2.2. Fabrication of Biomimetic Features
Many fabrication technologies, such as electrospinning, 3D printing, molding, casting, freeze-drying, and micropatterning, are available to deliver some of the biomimetic features to the above-discussed NGCs [2,135][2][83]. Depending on the fabrication method, there is a good possibility of controlling the porosity, channel size, density, and alignment, but each method has its limitations. Inkjet printing and micropatterning are less utilized to create NGCs than electrospinning, but both techniques enable control over the local topography. Inkjet printing, a subtype of 3D printing, uses heat or piezoelectric pulses to jet bio-inks in a drop-by-drop fashion. This method can create spatial and concentration gradients of various biomolecules, print conductive patterns, and localize cells onto NGC surfaces [15]. Due to the ready incorporation of growth factors, cells, or conductive nanoparticles into inks for inkjet printing, it is very likely to achieve the bioactive topographical modifications of NGCs and specifically promote neurite outgrowth [14,140,141][14][84][85]. On the other hand, micropatterning is a template-based method that uses photolithography, microcontact, or microfluidic stamping to create a variety of micropatterns, including lines, dots, and grids, to guide axonal alignment and promote neurite outgrowth [142,143,144][86][87][88]. However, similar to electrospinning, inkjet printing and micropatterning are also done on 2D surfaces, which generally require the rolling step to create conduit structures. Given that a pore size between 10 and 40 µm and a porosity of roughly 80% are typically needed to recapitulate the native nerve environment and encourage PNI regeneration [120][89], efforts have been geared toward the creation of NGCs using freeze-drying or solvent casting/salt leaching. Freeze-drying is often employed to create porous conduits by sublimating the solidified solvent (e.g., ice) between polymer molecules and leaving the interstitial space empty [104][61]. The porosity of the NGC can be easily customized by adjusting the polymer concentration, and the pore morphology can be tailored by varying the freezing speed and methods. Although its ability to create interconnected anisotropic pores within NGCs has been recognized [49][44], the freeze-drying method has limited control over the size and uniformity of pores. Three-dimensional printing and molding have been exploited to create multichannel NGCs. Three-dimensional printing, particularly stereolithography (SLA) and fused deposition modeling (FDM), offers a platform to fabricate complex structures with the opportunity to encapsulate living cells and biomolecules [94,96][90][91]. For example, the SLA-enabled fabrication of NGCs from gelatin–methacrylate (GelMA) demonstrated the possibility of creating multi-channeled NGCs with varying diameters and densities [57][92]. However, constrained by the printing resolution and time consumption, molding becomes a primary choice for fabricating multichanneled NGCs. To overcome the limitations of individual fabrication methods, it would preferably combine various methods or use hybrid technologies to fabricate biomimetic NGCs. For example, Liu et al. used near-field electrowriting, dip-coating, and electrospinning to create a tri-layer conduit with structural characteristics resembling the human nerve [56][93].3.2.3. Mechanical Properties of Biomimetic NGCs
In addition to composition and design, the mechanical properties of the NGCs should, preferably, match the stiffness, flexibility, and elongation of native nerves. Evidently, the NGCs that closely resemble the morphological, physical, and mechanical properties of native nervous ECM tend to achieve more effective therapeutic outcomes [143][87]. Since the elastic modulus of human peripheral nerves is approximately 0.5–13 MPa [154][94], NGCs made from soft polymers with an elastic modulus in this range or slightly higher have immense potential to heal PNIs. During normal body movement, the nerves can be stretched as much as 6 to 8%; thus, NGCs should be able to tolerate such a stretch under repetitive stresses [18].4. Conclusions
While peripheral nerves can regenerate, large gaps (>1 cm) typically result in unsuccessful healing and a loss of function. In view of the shortage of, and comorbidities associated with, autografting, NGCs serve as a promising alternative to bridge the gap of the severely damaged nerve and guide axonal growth. To maximize the regenerative capacity of NGCs and restore lost nerve function following severe PNIs, it is preferred for these conduits to deliver a native PNS-like environment via the optimal selection of composition, structural configurations, and functionality close to the native nerves. With respect to composition, hybrid biomaterials combining both the biological attributes of natural materials and the mechanical/conductive properties of synthetic materials are the most attractive ones to achieve the desirable physical performance (e.g., flexibility, degradation, and conductivity) while inducing the required cellular responses (e.g., the recruitment of neuronal cells, the migration of SCs, and improved myelination). NGCs can further benefit from the incorporation of cells such as SC or neural stem cells and biomolecules, such as fibronectin, laminin, NGFs, and/or SC-secreted exosomes, to better mimic the composition of native nerves and promote cell adhesion, SC migration, vascular infiltration, axonal growth, and consequent functional recovery. To mimic the structural hierarchy of PNS, where each nerve houses the fascicles and each fascicle holds the nerve fibers to facilitate signal transduction from the CNS to peripheral tissues, biomimetic NGCs are also designed to include additional features, such as grooves, pores, channels, and fillers. Parallel grooves and channels should present nano- and micro-scale topographical cues similar to the fibrin network. Additionally, NGCs should contain fillers and pores (10–40 µm in size with 80% porosity) to support cell infiltration, neovascularization, and nutrient diffusion to facilitate nerve regeneration. Finally, conduits should have multiple channels whose size and density maximally mimic the fascicles at the injury site. To implement the structural complexity of biomimetic NGCs, various fabrication technologies, such as electrospinning, 3D printing, molding, casting, freeze-drying, and micropatterning, can be adopted as either single fabrication platforms or combined ones to create those biomimetic features. Advances in high-resolution fabrication, such as e-beam lithography, would further enhance the capability of creating NGCs with delicate structures.References
- Lopes, B.; Sousa, P.; Alvites, R.; Branquinho, M.; Sousa, A.C.; Mendonça, C.; Atayde, L.M.; Luís, A.L.; Varejão, A.S.P.; Maurício, A.C. Peripheral Nerve Injury Treatments and Advances: One Health Perspective. Int. J. Mol. Sci. 2022, 23, 918.
- Yan, Y.; Yao, R.; Zhao, J.; Chen, K.; Duan, L.; Wang, T.; Zhang, S.; Guan, J.; Zheng, Z.; Wang, X.; et al. Implantable nerve guidance conduits: Material combinations, multi-functional strategies and advanced engineering innovations. Bioact. Mater. 2022, 11, 57–76.
- Bhandari, P.S. Management of peripheral nerve injury. J. Clin. Orthop. Trauma 2019, 10, 862–866.
- Zhang, F.; Fischer, K.A. End-to-side neurorrhaphy. Microsurgery 2002, 22, 122–127.
- Yan, Z.; Qian, Y.; Fan, C. Biomimicry in 3D printing design: Implications for peripheral nerve regeneration. Regen. Med. 2021, 16, 683–701.
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Qasim, M.; Zafar, S.; Aziz, N.; Razzaq, A.; Hussain, R.; de Aguilar, J.L.G.; et al. Current status of therapeutic approaches against peripheral nerve injuries: A detailed story from injury to recovery. Int. J. Biol. Sci. 2020, 16, 116–134.
- Faroni, A.; Mobasseri, S.A.; Kingham, P.J.; Reid, A.J. Peripheral nerve regeneration: Experimental strategies and future perspectives. Adv. Drug Deliv. Rev. 2015, 82, 160–167.
- Behtaj, S.; Ekberg, J.A.K.; St John, J.A. Advances in Electrospun Nerve Guidance Conduits for Engineering Neural Regeneration. Pharmaceutics 2022, 14, 219.
- Joung, D.; Lavoie, N.S.; Guo, S.Z.; Park, S.H.; Parr, A.M.; McAlpine, M.C. 3D Printed Neural Regeneration Devices. Adv. Funct. Mater. 2020, 30, 1906237.
- Wofford, K.L.; Shultz, R.B.; Burrell, J.C.; Cullen, D.K. Neuroimmune interactions and immunoengineering strategies in peripheral nerve repair. Prog. Neurobiol. 2022, 208, 102172.
- Rodríguez-Sánchez, D.N.; Pinto, G.B.A.; Cartarozzi, L.P.; de Oliveira, A.L.R.; Bovolato, A.L.C.; de Carvalho, M.; da Silva, J.V.L.; de Dernowsek, J.A.; Golim, M.; Barraviera, B.; et al. 3D-printed nerve guidance conduits multi-functionalized with canine multipotent mesenchymal stromal cells promote neuroregeneration after sciatic nerve injury in rats. Stem Cell Res. Ther. 2021, 12, 303.
- Sabongi, R.G.; Fernandes, M.; Dos Gomes Santos, J.B. Peripheral nerve regeneration with conduits: Use of vein tubes. Neural Regen. Res. 2015, 10, 529–533.
- Zargar Kharazi, A.; Dini, G.; Naser, R. Fabrication and evaluation of a nerve guidance conduit capable of Ca2+ ion release to accelerate axon extension in peripheral nerve regeneration. J. Biomed. Mater. Res. Part A 2018, 106, 2181–2189.
- Qian, Y.; Zhao, X.; Han, Q.; Chen, W.; Li, H.; Yuan, W. An integrated multi-layer 3D-fabrication of PDA/RGD coated graphene loaded PCL nanoscaffold for peripheral nerve restoration. Nat. Commun. 2018, 9, 323.
- Kang, N.U.; Lee, S.J.; Gwak, S.J. Fabrication Techniques of Nerve Guidance Conduits for Nerve Regeneration. Yonsei Med. J. 2022, 63, 114–123.
- Liu, Y.; Zhang, X.; Xiao, C.; Liu, B. Engineered hydrogels for peripheral nerve repair. Mater. Today Bio 2023, 20, 100668.
- Nadeau, S.; Filali, M.; Zhang, J.; Kerr, B.J.; Rivest, S.; Soulet, D.; Iwakura, Y.; de Vaccari, J.P.R.; Keane, R.W.; Lacroix, S. Functional recovery after peripheral nerve injury is dependent on the pro-inflammatory cytokines IL-1β and TNF: Implications for neuropathic pain. J. Neurosci. 2011, 31, 12533–12542.
- Topp, K.S.; Boyd, B.S. Structure and biomechanics of peripheral nerves: Nerve responses to physical stresses and implications for physical therapist practice. Phys. Ther. 2006, 86, 92–109.
- Dalton, P.D.; O’Neill, K.L.; Pêgo, A.P.; Plant, G.W.; Nisbet, D.R.; Oudega, M.; Brook, G.A.; Harvey, A.R. Tissue engineering of the nervous system. In Tissue Engineering; Elsevier: Amsterdam, The Netherlands, 2023; pp. 585–627.
- Dubový, P.; Jančálek, R.; Kubek, T. Role of Inflammation and Cytokines in Peripheral Nerve Regeneration. Int. Rev. Neurobiol. 2013, 108, 173–206.
- Martini, F.H.; Tallitsch, R.B.; Judi, L.; Nath, P.D. Human Anatomy; Pearson: London, UK, 2016; ISBN 9780134320762.
- Estrada, J.B.; Cramer, H.C.; Scimone, M.T.; Buyukozturk, S.; Franck, C. Neural cell injury pathology due to high-rate mechanical loading. Brain Multiphysics 2021, 2, 100034.
- Kwan, M.K.; Wall, E.J.; Massie, J.; Garfin, S.R. Strain, stress and stretch of peripheral nerve rabbit experiments in vitro and in vivo. Acta Orthop. 1992, 63, 267–272.
- Gaudin, R.; Knipfer, C.; Henningsen, A.; Smeets, R.; Heiland, M.; Hadlock, T. Approaches to peripheral nerve repair: Generations of biomaterial conduits yielding to replacing autologous nerve grafts in craniomaxillofacial surgery. Biomed Res. Int. 2016, 2016, 3856262.
- Kaplan, B.; Levenberg, S. The Role of Biomaterials in Peripheral Nerve and Spinal Cord Injury: A Review. Int. J. Mol. Sci. 2022, 23, 1244.
- Ma, Y.; Gao, H.; Wang, H.; Cao, X. Engineering topography: Effects on nerve cell behaviors and applications in peripheral nerve repair. J. Mater. Chem. B 2021, 9, 6310–6325.
- Du, J.; Liu, J.; Yao, S.; Mao, H.; Peng, J.; Sun, X.; Cao, Z.; Yang, Y.; Xiao, B.; Wang, Y.; et al. Prompt peripheral nerve regeneration induced by a hierarchically aligned fibrin nanofiber hydrogel. Acta Biomater. 2017, 55, 296–309.
- Ciardelli, G.; Chiono, V. Materials for peripheral nerve regeneration. Macromol. Biosci. 2006, 6, 13–26.
- Gan, L.; Zhao, L.; Zhao, Y.; Li, K.; Tong, Z.; Yi, L.; Wang, X.; Li, Y.; Tian, W.; He, X.; et al. Cellulose/soy protein composite-based nerve guidance conduits with designed microstructure for peripheral nerve regeneration. J. Neural Eng. 2016, 13, 056019.
- Xie, H.; Yang, W.; Chen, J.; Zhang, J.; Lu, X.; Zhao, X.; Huang, K.; Li, H.; Chang, P.; Wang, Z.; et al. A Silk Sericin/Silicone Nerve Guidance Conduit Promotes Regeneration of a Transected Sciatic Nerve. Adv. Healthc. Mater. 2015, 4, 2195–2205.
- Parker, B.J.; Rhodes, D.I.; O’Brien, C.M.; Rodda, A.E.; Cameron, N.R. Nerve guidance conduit development for primary treatment of peripheral nerve transection injuries: A commercial perspective. Acta Biomater. 2021, 135, 64–86.
- Suhar, R.A.; Marquardt, L.M.; Song, S.; Buabbas, H.; Doulames, V.M.; Johansson, P.K.; Klett, K.C.; Dewi, R.E.; Enejder, A.M.K.; Plant, G.W.; et al. Elastin-like Proteins to Support Peripheral Nerve Regeneration in Guidance Conduits. ACS Biomater. Sci. Eng. 2021, 7, 4209–4220.
- Farokhi, M.; Mottaghitalab, F.; Shokrgozar, M.A.; Kaplan, D.L.; Kim, H.W.; Kundu, S.C. Prospects of peripheral nerve tissue engineering using nerve guide conduits based on silk fibroin protein and other biopolymers. Int. Mater. Rev. 2017, 62, 367–391.
- Hu, Y.; Chen, Z.; Wang, H.; Guo, J.; Cai, J.; Chen, X.; Wei, H.; Qi, J.; Wang, Q.; Liu, H.; et al. Conductive Nerve Guidance Conduits Based on Morpho Butterfly Wings for Peripheral Nerve Repair. ACS Nano 2022, 16, 1868–1879.
- Farzan, A.; Borandeh, S.; Seppälä, J. Conductive polyurethane/PEGylated graphene oxide composite for 3D-printed nerve guidance conduits. Eur. Polym. J. 2022, 167, 111068.
- Fabbro, A.; Prato, M.; Ballerini, L. Carbon nanotubes in neuroregeneration and repair. Adv. Drug Deliv. Rev. 2013, 65, 2034–2044.
- Pan, X.; Sun, B.; Mo, X. Electrospun polypyrrole-coated polycaprolactone nanoyarn nerve guidance conduits for nerve tissue engineering. Front. Mater. Sci. 2018, 12, 438–446.
- Xu, B.; Bai, T.; Sinclair, A.; Wang, W.; Wu, Q.; Gao, F.; Jia, H.; Jiang, S.; Liu, W. Directed neural stem cell differentiation on polyaniline-coated high strength hydrogels. Mater. Today Chem. 2016, 1–2, 15–22.
- Wang, G.; Wu, W.; Yang, H.; Zhang, P.; Wang, J.Y. Intact polyaniline coating as a conductive guidance is beneficial to repairing sciatic nerve injury. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 128–142.
- Wang, Y.; Zhu, C.; Pfattner, R.; Yan, H.; Jin, L.; Chen, S.; Molina-Lopez, F.; Lissel, F.; Liu, J.; Rabiah, N.I.; et al. A highly stretchable, transparent, and conductive polymer. Sci. Adv. 2017, 3, e1602076.
- Kayser, L.V.; Lipomi, D.J. Stretchable Conductive Polymers and Composites Based on PEDOT and PEDOT:PSS. Adv. Mater. 2019, 31, 1806133.
- Choi, J.-S.; Kim, H.; An, H.-Y.; Shim, B.S.; Lim, J.-Y. Regeneration of Recurrent Laryngeal Nerve using Polycaprolactone (PCL) Nerve Guide Conduit Coated with Conductive Materials. J. Korean Thyroid Assoc. 2015, 8, 88.
- Du, J.; Chen, H.; Qing, L.; Yang, X.; Jia, X. Biomimetic neural scaffolds: A crucial step towards optimal peripheral nerve regeneration. Biomater. Sci. 2018, 6, 1299–1311.
- Hibbitts, A.J.; Kočí, Z.; Kneafsey, S.; Matsiko, A.; Žilić, L.; Dervan, A.; Hinton, P.; Chen, G.; Cavanagh, B.; Dowling, J.K.; et al. Multi-factorial nerve guidance conduit engineering improves outcomes in inflammation, angiogenesis and large defect nerve repair. Matrix Biol. 2022, 106, 34–57.
- Yang, Y.; Ding, F.; Wu, J.; Hu, W.; Liu, W.; Liu, J.; Gu, X. Development and evaluation of silk fibroin-based nerve grafts used for peripheral nerve regeneration. Biomaterials 2007, 28, 5526–5535.
- Nazeer, M.A.; Yilgor, E.; Yilgor, I. Electrospun polycaprolactone/silk fibroin nanofibrous bioactive scaffolds for tissue engineering applications. Polymer 2019, 168, 86–94.
- Cunha, C.; Panseri, S.; Antonini, S. Emerging nanotechnology approaches in tissue engineering for peripheral nerve regeneration. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 50–59.
- Furlani, F.; Montanari, M.; Sangiorgi, N.; Saracino, E.; Campodoni, E.; Sanson, A.; Benfenati, V.; Tampieri, A.; Panseri, S.; Sandri, M. Electroconductive and injectable hydrogels based on gelatin and PEDOT:PSS for a minimally invasive approach in nervous tissue regeneration. Biomater. Sci. 2022, 10, 2040–2053.
- Vallejo, F.A.; Diaz, A.; Errante, E.L.; Smartz, T.; Khan, A.; Silvera, R.; Brooks, A.E.; Lee, Y.S.; Burks, S.S.; Levi, A.D. Systematic review of the therapeutic use of Schwann cells in the repair of peripheral nerve injuries: Advancements from animal studies to clinical trials. Front. Cell. Neurosci. 2022, 16, 929593.
- Zhou, G.; Chen, Y.; Dai, F.; Yu, X. Chitosan-based nerve guidance conduit with microchannels and nanofibers promotes schwann cells migration and neurite growth. Colloids Surf. B Biointerfaces 2023, 221, 112929.
- Liu, S.; Xie, Y.Y.; Di Wang, L.; Tai, C.X.; Chen, D.; Mu, D.; Cui, Y.Y.; Wang, B. A multi-channel collagen scaffold loaded with neural stem cells for the repair of spinal cord injury. Neural Regen. Res. 2021, 16, 2284–2292.
- Ye, J.; Chen, Y.; Yang, H.; Chen, Q.; Huang, Y.; Zhao, J. Human IL12p80 and Neural Stem Cells Enhance Sciatic Nerve Regeneration. Lancent Pschch 2020, 23, 7002.
- Shalaby, S.M.; El-Shal, A.S.; Ahmed, F.E.; Shaban, S.F.; Wahdan, R.A.; Kandel, W.A.; Senger, M.S. Combined Wharton’s jelly derived mesenchymal stem cells and nerve guidance conduit: A potential promising therapy for peripheral nerve injuries. Int. J. Biochem. Cell Biol. 2017, 86, 67–76.
- Lu, Q.; Zhang, F.; Cheng, W.; Gao, X.; Ding, Z.; Zhang, X.; Lu, Q.; Kaplan, D.L. Nerve Guidance Conduits with Hierarchical Anisotropic Architecture for Peripheral Nerve Regeneration. Adv. Healthc. Mater. 2021, 10, 2100427.
- Ramburun, P.; Kumar, P.; Choonara, Y.E.; Bijukumar, D.; du Toit, L.C.; Pilay, V. A Review of Bioactive Release from Nerve Conduits as a Neurotherapeutic Strategy for Neuronal Growth in Peripheral Nerve Injury. BioMed Res. Int. 2014, 2014, 132350.
- Yan, L.; Yao, Z.; Lin, T.; Zhu, Q.; Qi, J.; Gu, L.; Fang, J.; Zhou, X.; Liu, X. The role of precisely matching fascicles in the quick recovery of nerve function in long peripheral nerve defects. Neuroreport 2017, 28, 1008–1015.
- Ni, H.C.; Tseng, T.C.; Chen, J.R.; Hsu, S.H.; Chiu, I.M. Fabrication of bioactive conduits containing the fibroblast growth factor 1 and neural stem cells for peripheral nerve regeneration across a 15 mm critical gap. Biofabrication 2013, 5, 035010.
- Vijayavenkataraman, S.; Zhang, S.; Thaharah, S.; Sriram, G.; Lu, W.F.; Fuh, J.Y.H. Electrohydrodynamic Jet 3D Printed Nerve Guide Conduits (NGCs) for peripheral Nerve Injury Repair. Polymers 2018, 10, 753.
- Carvalho, C.R.; Oliveira, J.M.; Reis, R.L. Modern Trends for Peripheral Nerve Repair and Regeneration: Beyond the Hollow Nerve Guidance Conduit. Front. Bioeng. Biotechnol. 2019, 7, 337.
- Song, S.; Wang, X.; Wang, T.; Yu, Q.; Hou, Z.; Zhu, Z.; Li, R. Additive Manufacturing of Nerve Guidance Conduits for Regeneration of Injured Peripheral Nerves. Front. Bioeng. Biotechnol. 2020, 8, 590596.
- Bhatnagar, D.; Bushman, J.S.; Murthy, N.S.; Merolli, A.; Kaplan, H.M.; Kohn, J. Fibrin glue as a stabilization strategy in peripheral nerve repair when using porous nerve guidance conduits. J. Mater. Sci. Mater. Med. 2017, 28, 79.
- Sun, B.; Zhou, Z.; Wu, T.; Chen, W.; Li, D.; Zheng, H.; El-Hamshary, H.; Al-Deyab, S.S.; Mo, X.; Yu, Y. Development of Nanofiber Sponges-Containing Nerve Guidance Conduit for Peripheral Nerve Regeneration in Vivo. ACS Appl. Mater. Interfaces 2017, 9, 26684–26696.
- Li, X.; Yang, W.; Xie, H.; Wang, J.; Zhang, L.; Wang, Z.; Wang, L. CNT/Sericin Conductive Nerve Guidance Conduit Promotes Functional Recovery of Transected Peripheral Nerve Injury in a Rat Model. ACS Appl. Mater. Interfaces 2020, 12, 36860–36872.
- Mohan, S.; Hernández, I.C.; Wang, W.; Yin, K.; Sundback, C.A.; Wegst, U.G.K.; Jowett, N. Fluorescent Reporter Mice for Nerve Guidance Conduit Assessment: A High-Throughput in vivo Model. Laryngoscope 2018, 128, E386–E392.
- Jenkins, P.M.; Laughter, M.R.; Lee, D.J.; Lee, Y.M.; Freed, C.R.; Park, D. A nerve guidance conduit with topographical and biochemical cues: Potential application using human neural stem cells. Nanoscale Res. Lett. 2015, 10, 264.
- Singh, A.; Asikainen, S.; Teotia, A.K.; Shiekh, P.A.; Huotilainen, E.; Kumar, A. Biomimetic Photocurable Three-Dimensional Printed Nerve Guidance Channels with Aligned Cryomatrix Lumen for Peripheral Nerve Regeneration. ACS Appl. Mater. Interfaces 2018, 10, 43327–43342.
- Pawar, K.; Welzel, G.; Haynl, C.; Schuster, S.; Scheibel, T. Recombinant Spider Silk and Collagen-Based Nerve Guidance Conduits Support Neuronal Cell Differentiation and Functionality in Vitro. ACS Appl. Bio Mater. 2019, 2, 4872–4880.
- Shah, M.B.; Chang, W.; Zhou, G.; Glavy, J.S.; Cattabiani, T.M.; Yu, X. Novel spiral structured nerve guidance conduits with multichannels and inner longitudinally aligned nano fi bers for peripheral nerve regeneration. J. Biomed. Mater. Res. 2018, 107, 1410–1419.
- Zhao, J.; Zhang, S.; Duan, L.; Yao, R.; Yan, Y.; Wang, T.; Wang, J.; Zheng, Z.; Wang, X.; Li, G. Preparation and mechanical optimization of a two-layer silk/magnesium wires braided porous artificial nerve guidance conduit. J. Biomed. Mater. Res. Part A 2022, 110, 1801–1812.
- Kim, S.M.; Lee, M.S.; Jeon, J.; Lee, D.H.; Yang, K.; Cho, S.W.; Han, I.; Yang, H.S. Biodegradable Nerve Guidance Conduit with Microporous and Micropatterned Poly(lactic-co-glycolic acid)-Accelerated Sciatic Nerve Regeneration. Macromol. Biosci. 2018, 18, 1800290.
- Stocco, E.; Barbon, S.; Emmi, A.; Tiengo, C.; Macchi, V.; De Caro, R.; Porzionato, A. Bridging Gaps in Peripheral Nerves: From Current Strategies to Future Perspectives in Conduit Design. Int. J. Mol. Sci. 2023, 24, 9170.
- Tagandurdyyeva, N.A.; Trube, M.A.; Shemyakin, I.O.; Solomitskiy, D.N.; Medvedev, G.V.; Dresvyanina, E.N.; Nashchekina, Y.A.; Ivan’kova, E.M.; Dobrovol’skaya, I.P.; Kamalov, A.M.; et al. Properties of Resorbable Conduits Based on Poly(L-Lactide) Nanofibers and Chitosan Fibers for Peripheral Nerve Regeneration. Polymers 2023, 15, 3323.
- Aigner, T.B.; Haynl, C.; Salehi, S.; O’Connor, A.; Scheibel, T. Nerve guidance conduit design based on self-rolling tubes. Mater. Today Biol. 2020, 5, 100042.
- Teuschl, A.H.; Schuh, C.; Halbweis, R.; Pajer, K.; Márton, G.; Hopf, R.; Mosia, S.; Rünzler, D.; Redl, H.; Nógrádi, A.; et al. A New Preparation Method for Anisotropic Silk Fibroin Nerve Guidance Conduits and Its Evaluation in Vitro and in a Rat Sciatic Nerve Defect Model. Tissue Eng. Part C Methods 2015, 21, 945–957.
- Lee, Y.B.; Polio, S.; Lee, W.; Dai, G.; Menon, L.; Carroll, R.S.; Yoo, S.S. Bio-printing of collagen and VEGF-releasing fibrin gel scaffolds for neural stem cell culture. Exp. Neurol. 2010, 223, 645–652.
- Zhao, X.; Fan, C.; Wang, J.; Xiong, H.; Zhu, T.; Liu, Y.; Pan, H.; Weijia Lu, W. Bioinspired multichannel nerve guidance conduit based on shape memory nanofibers for potential application in peripheral nerve repair. ACS Nano 2020, 14, 12579–12595.
- Chang, Y.C.; Chen, M.H.; Liao, S.Y.; Wu, H.C.; Kuan, C.H.; Sun, J.S.; Wang, T.W. Multichanneled Nerve Guidance Conduit with Spatial Gradients of Neurotrophic Factors and Oriented Nanotopography for Repairing the Peripheral Nervous System. ACS Appl. Mater. Interfaces 2017, 9, 37623–37636.
- Wang, J.; Cheng, Y.; Wang, H.; Wang, Y.; Zhang, K.; Fan, C.; Wang, H.; Mo, X. Biomimetic and hierarchical nerve conduits from multifunctional nanofibers for guided peripheral nerve regeneration. Acta Biomater. 2020, 117, 180–191.
- Belanger, K.; Schlatter, G.; Hébraud, A.; Marin, F.; Testelin, S.; Dakpé, S.; Devauchelle, B.; Egles, C. A multi-layered nerve guidance conduit design adapted to facilitate surgical implantation. Health Sci. Rep. 2018, 1, e86.
- Grinberg, Y.; Schiefer, M.A.; Tyler, D.J.; Gustafson, K.J. Fascicular perineurium thickness, size, and position affect model predictions of neural excitation. IEEE Trans. Neural Syst. Rehabil. Eng. 2008, 16, 572–581.
- Stewart, J.D. Peripheral nerve fascicles: Anatomy and clinical relevance. Muscle Nerve 2003, 28, 525–541.
- Li, Y.; Men, Y.; Wang, B.; Chen, X.; Yu, Z. Co-transplantation of Schwann cells and neural stem cells in the laminin-chitosan-PLGA nerve conduit to repair the injured recurrent laryngeal nerve in SD rats. J. Mater. Sci. Mater. Med. 2020, 31, 99.
- Anastasiou, E.; Lorentz, K.O.; Stein, G.J.; Mitchell, P.D. Prehistoric schistosomiasis parasite found in the Middle East. Lancet Infect. Dis. 2014, 14, 553–554.
- Das, S.R.; Lentner, M.T.; Hondred, J.A. Electrical Differentiation of Mesenchymal Stem Cells into Schwann-Cell-Like Phenotypes Using Inkjet-Printed Graphene Circuits Electrical Differentiation of Mesenchymal Stem Cells into Schwann-Cell-Like Phenotypes Using Inkjet-Printed Graphene. Adv. Healthc. Mater. 2017, 6, 1601087.
- Ilkhanizadeh, S.; Teixeira, A.I.; Hermanson, O. Inkjet printing of macromolecules on hydrogels to steer neural stem cell differentiation. Biomaterials 2007, 28, 3936–3943.
- Huang, W.C.; Lin, C.C.; Chiu, T.W.; Chen, S.Y. 3D Gradient and Linearly Aligned Magnetic Microcapsules in Nerve Guidance Conduits with Remotely Spatiotemporally Controlled Release to Enhance Peripheral Nerve Repair. ACS Appl. Mater. Interfaces 2022, 14, 46188–46200.
- Yucel, D.; Kose, G.T.; Hasirci, V. Polyester based nerve guidance conduit design. Biomaterials 2010, 31, 1596–1603.
- Zhang, D.; Yao, Y.; Duan, Y.; Yu, X.; Shi, H.; Nakkala, J.R.; Zuo, X.; Hong, L.; Mao, Z.; Gao, C. Surface-Anchored Graphene Oxide Nanosheets on Cell-Scale Micropatterned Poly(d, l-lactide-co-caprolactone) Conduits Promote Peripheral Nerve Regeneration. ACS Appl. Mater. Interfaces 2020, 12, 7915–7930.
- Kokai, L.E.; Lin, Y.C.; Oyster, N.M.; Marra, K.G. Diffusion of soluble factors through degradable polymer nerve guides: Controlling manufacturing parameters. Acta Biomater. 2009, 5, 2540–2550.
- Chen, Y.S.; Ng, H.Y.; Chen, Y.W.; Cho, D.Y.; Ho, C.C.; Chen, C.Y.; Chiu, S.C.; Jhong, Y.R.; Shie, M.Y. Additive manufacturing of Schwann cell-laden collagen/alginate nerve guidance conduits by freeform reversible embedding regulate neurogenesis via exosomes secretion towards peripheral nerve regeneration. Biomater. Adv. 2023, 146, 213276.
- Namini, M.S.; Ebrahimi-Barough, S.; Ai, J.; Jahromi, H.K.; Mikaeiliagah, E.; Azami, M.; Bahrami, N.; Lotfibakhshaiesh, N.; Saremi, J.; Shirian, S. Tissue-Engineered Core-Shell Silk-Fibroin/Poly-l-Lactic Acid Nerve Guidance Conduit Containing Encapsulated Exosomes of Human Endometrial Stem Cells Promotes Peripheral Nerve Regeneration. ACS Biomater. Sci. Eng. 2023, 9, 3496–3511.
- Ye, W.; Li, H.; Yu, K.; Xie, C.; Wang, P.; Zheng, Y.; Zhang, P.; Xiu, J.; Yang, Y.; Zhang, F.; et al. 3D printing of gelatin methacrylate-based nerve guidance conduits with multiple channels. Mater. Des. 2020, 192, 108757.
- Liu, S.; Sun, L.; Zhang, H.; Hu, Q.; Wang, Y.; Ramalingam, M. High-resolution combinatorial 3D printing of gelatin-based biomimetic triple-layered conduits for nerve tissue engineering. Int. J. Biol. Macromol. 2021, 166, 1280–1291.
- Zhu, W.; Tringale, K.R.; Woller, S.A.; You, S.; Johnson, S.; Shen, H.; Schimelman, J.; Whitney, M.; Steinauer, J.; Xu, W.; et al. Rapid continuous 3D printing of customizable peripheral nerve guidance conduits. Mater. Today 2018, 21, 951–959.
