Bones are composite materials consisting of organic polymers (mainly type I collagen), carbonated apatite, and water, with volume fractions of 35–45%, 35–45%, and 15–25%, respectively. Carbonated apatite in bone contributes to rigidity, while organic polymers and water contribute to toughness. The inorganic crystal, carbonated apatite, is a plate-shaped crystal with dimensions of 50 nm × 25 nm × 1–4 nm, generating a significant organic–inorganic interface, due to its nanoscale size. This interface is believed to absorb externally applied forces to dissipate mechanical energy to thermal energy. Crystallization of calcium phosphate in a solution or in a dispersion of a polymer attracts interest as a biomimetic synthesis of the bone-like composites.
- bone
- hydroxyapatite
- polysaccharide
- organic–inorganic composites
- organic polymers
- water
1. Introduction
2. General Features of Organic–Inorganic Composites
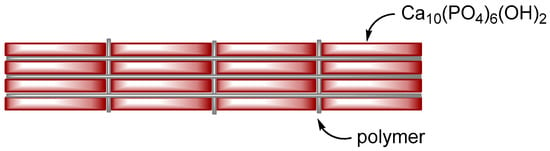
Organic–inorganic composite materials can be broadly classified into two categories: ceramics-reinforced plastics and polymer-reinforced ceramics (Table 1). For the former, the low rigidity of the organic phase is enhanced through the addition of the inorganic phase. Glass fiber-reinforced plastics, clay–polyamide composites [7], and rubber, an important component of automobile tires, for example, are commonly used in daily life. If plastic is too soft to resist external stress, we can add inorganic fillers, such as glass fibers, to increase its stiffness. The major component of the composites of this category is an organic phase, and the minor component is an inorganic phase. For “polymer-reinforced ceramics”, the brittleness of ceramics is improved by incorporating organic polymers, as seen in laminated glass. For laminated glass, due to the sandwiched structure of glass plate–polymer film–glass plate, it would not be broken into pieces if a shock is given, owing to the organic polymer film. The major component of this category is an inorganic phase, and the minor component is an organic phase. Bone is polymer-reinforced ceramics and can be roughly described as nanometer-sized laminated glass with a brick-and-mortar structure (Figure 1). According to the brick-and-mortar model, ceramic in bone is a dispersed phase, and polymer is a continuous phase. When attempting to prepare a bone-like composite, as the weight fraction of ceramics increases and is close to one, the ceramic phase becomes a continuous phase, resulting in fragile materials. Therefore, the fabrication of the brick-and-mortar structure becomes difficult, as the weight fraction of the ceramic phase is close to one.| Ceramic-Reinforced Polymer | Polymer-Reinforced Ceramics | |
|---|---|---|
| Major component | Polymer (matrix, continuous phase) | Ceramics (filler, dispersed phase) |
| Minor component | Ceramics (filler, dispersed phase) | Polymers (matrix, continuous phase) |
| Examples | Glass–fiber reinforced plastics | Laminated glass |
| Polyamide–clay composites | Bones | |
| Tire | Teeth |
3. Preparation of Organic–Inorganic Composites
For the preparation of organic–inorganic composites, the following three protocols have been reported:-
Direct mixing of polymer and inorganic crystals.
-
Polymerization of the monomer in the presence of the inorganic crystal powder.
-
Crystallization of the inorganic phase in the presence of the organic polymer.
4. Composite of Hydroxyapatite and Polysaccharide
The use of polysaccharides as an organic component of the composite is an attractive strategy because they are carbon neutral and degrade biologically. There have been several studies on the preparation of the composites of hydroxyapatite and polysaccharides [23][47][55][56][57]. Yao and coworkers crystallized HAP in the presence of polysaccharides and found that the crystal size of HAP decreased in the order of amylose (−OH) > chitosan (−NH2, −NHCOCH3, and −OH) > carrageenan (−OSO3− and −OH) > pectin (−COO− and −OH) [23]. The polar functional groups of these polysaccharides are shown in the parentheses. It seems that the –COO− group is the most effective in suppressing the crystal growth of HAP. We can avoid environmental issues, such as the microplastic pollution of sea water, by employing polysaccharide-based structural materials. The annual production of cellulose is 1.5 × 1012 tons [58], while that of starch is 8 × 108 tons (corn starch) [59]. Considering the large annual production of cellulose, the use of cellulose as an organic phase of the composite seems attractive. Yu and coworkers reported a composite of cellulose nanofibers and TiO2-coated mica. It showed a bending strength of 281 MPa and an elastic modulus of 20 GPa [5].5. Introduction of Hydrophobic Groups for Water-Resistance Enhancement
The surfaces of polysaccharides and hydroxyapatite are hydrophilic and readily hydrated. Thus, when molded composites are immersed in water, they often undergo substantial swelling, resulting in significant changes in mechanical properties. In bone’s apatite, the carboxylate groups of citric acid are adsorbed to the calcium ions on the a and b faces through ionic bonding. Accordingly, the methylene groups of citric acid face outward, contributing to the hydrophobicity of apatite crystals [50]. Bone contains around 10% water, and when it is dried, it becomes more elastic but also more brittle, compromising its toughness [60]. This implies that the water content plays a significant role in bone’s mechanical properties, especially toughness. The synthesized composites of cellulose or starch and hydroxyapatite demonstrated significant water absorption, leading to a near-complete loss of mechanical strength. Therefore, the acylation of the hydroxyl groups of polysaccharides was considered as a means to improve the water resistance of the composites.References
- Elias, H.-G. An Introduction to Polymer Science; VCH: Berlin, Germany, 1996.
- Eerkes-Medrano, D.; Thompson, R.C.; Aldridge, D.C. Microplastics in freshwater systems: A review of the emerging threats, identification of knowledge gaps and prioritisation of research needs. Water Res. 2015, 75, 63–82.
- Stock, S.R. The Mineral-Collagen Interface in Bone. Calcif. Tissue Int. 2015, 97, 262–280.
- Nudelman, F.; Lausch, A.J.; Sommerdijk, N.A.J.M.; Sone, E.D. In vitro models of collagen biomineralization. J. Struct. Biol. 2013, 183, 258–269.
- Guan, Q.; Yang, H.; Han, Z.; Ling, Z.; Yu, S. An all-natural bioinspired structural material for plastic replacement. Nat. Commun. 2020, 11, 5401.
- Curto, M.; Le Gall, M.; Catarino, A.I.; Niu, Z.; Davies, P.; Everaert, G.; Dhakal, H.N. Long-term durability and ecotoxicity of biocomposites in marine environments: A review. RSC Adv. 2021, 11, 32917–32941.
- Okada, A.; Usuki, A. Twenty Years of Polymer-Clay Nanocomposites. Macromol. Mater. Eng. 2006, 291, 1449–1476.
- Bonfield, W.; Grynpas, M.D.; Tully, A.E.; Bowman, J.; Abram, J. Hydroxyapatite reinforced polyethylene—A mechanically compatible implant material for bone replacement. Biomaterials 1981, 2, 185–186.
- Takeoka, Y.; Hayashi, M.; Sugiyama, N.; Yoshizawa-Fujita, M.; Aizawa, M.; Rikukawa, M. In situ preparation of poly(L-lactic acid-co-glycolic acid)/hydroxyapatite composites as artificial bone materials. Polym. J. 2015, 47, 164–170.
- Stupp, S.I.; Ciegler, G.W. Organoapatite: Materials for artificial bone. I. Synthesis and microstructure. J. Biomed. Mater. Res. 1992, 26, 169–183.
- Embery, G.; Rees, S.; Hall, R.; Rose, K.; Waddington, R.; Shellis, P. Calcium- and hydroxyapatite binding properties of glucuronic acid-rich and iduronic acid-rich glycosaminoglycans and proteoglycans. Eur. J. Oral Sci. 1998, 106, 267–273.
- Kirkham, J.; Brookes, S.J.; Shore, R.C.; Wood, S.R.; Smith, D.A.; Zhang, J.; Chen, H.; Robinson, C. Physico-chemical properties of crystal surfaces in matrix-mineral interactions during mammalian biomineralisation. Current Opin. Colloid Interface Sci. 2002, 7, 124–132.
- Bertoni, E.; Bigi, A.; Falini, G.; Panzavolta, S.; Roveri, N. Hydroxyapatite/polyacrylic acid nanocrystals, J. Mater. Chem. 1999, 9, 779–782.
- Song, J.; Saiz, E.; Bertozzi, C.R. A New Approach to Mineralization of Biocompatible Hydrogel Scaffolds: An Efficient Process toward 3-Dimensional Bonelike Composites. J. Am. Chem. Soc. 2003, 125, 1236–1243.
- Kikuchi, M.; Ikoma, T.; Itoh, S.; Matsumoto, H.N.; Koyama, Y.; Takakuda, K.; Shinomiya, K.; Tanaka, J. Biomimetic synthesis of bone-like nanocomposites using the self-organization mechanism of hydroxyapatite and collagen. Compos. Sci. Technol. 2004, 64, 819–825.
- Pramanik, N.; Biswas, S.K.; Pramanik, P. Synthesis and Characterization of Hydroxyapatite/Poly(Vinyl Alcohol Phosphate) Nanocomposite Biomaterials. Int. J. Appl. Ceram. Technol. 2008, 5, 20–28.
- Meldrum, F.C.; Coelfen, H. Controlling Mineral Morphologies and Structures in Biological and Synthetic Systems. Chem. Rev. 2008, 108, 4332–4432.
- Fujisawa, R.; Tamura, M. Acidic bone matrix proteins and their roles in calcification. Front. Biosci. 2012, 17, 1891–1903.
- Coleman, R.J.; Jack, K.S.; Perrier, S.; Grondahl, L. Hydroxyapatite Mineralization in the Presence of Anionic Polymers. Cryst. Growth Des. 2013, 13, 4252–4259.
- Veis, A.; Dorvee, J.R. Biomineralization Mechanisms: A New Paradigm for Crystal Nucleation in Organic Matrices. Calcif. Tissue Int. 2013, 93, 307–315.
- Zhou, Y.; Liu, Q.; Zhao, Z.; Wang, W.; Zheng, L.; Li, Z. Preparation and Characterization of Phosphorylated Collagen and Hydroxyapatite Composite as a Potential Bone Substitute. Chem. Lett. 2013, 42, 83–85.
- Morimune-Moriya, S.; Kondo, S.; Sugawara-Narutaki, A.; Nishimura, T.; Kato, T.; Ohtsuki, C. Hydroxyapatite formation on oxidized cellulose nanofibers in a solution mimicking body fluid. Polym. J. 2015, 47, 158–163.
- Fang, W.; Zhang, H.; Yin, J.; Yang, B.; Zhang, Y.; Li, J.; Yao, F. Hydroxyapatite Crystal Formation in the Presence of Polysaccharide. Cryst. Growth Des. 2016, 16, 1247–1255.
- Kusakabe, A.; Hirota, K.; Mizutani, T. Crystallisation of hydroxyapatite in phosphorylated poly(vinyl alcohol) as a synthetic route to tough mechanical hybrid materials. Mater. Sci. Eng. C 2017, 70, 487–493.
- Diegmueller, J.J.; Cheng, X.; Akkus, O. Modulation of Hydroxyapatite Nanocrystal Size and Shape by Polyelectrolytic Peptides. Cryst. Growth Desig. 2009, 9, 5220–5226.
- Barthelat, F.; Yin, Z.; Buehler, M.J. Structure and mechanics of interfaces in biological materials. Nat. Rev. Mater. 2016, 1, 16007.
- Mayer, G. Rigid Biological Systems as Models for Synthetic Composites. Science 2005, 310, 1144–1147.
- Schweizer, S.; Taubert, A. Polymer-Controlled, Bio-Inspired Calcium Phosphate Mineralization from Aqueous Solution. Macromol. Biosci. 2007, 7, 1085–1099.
- Palmer, L.C.; Newcomb, C.J.; Kaltz, S.R.; Spoerke, E.D.; Stupp, S.I. Biomimetic Systems for Hydroxyapatite Mineralization Inspired by Bone and Enamel. Chem. Rev. 2008, 108, 4754–4783.
- Munch, E.; Launey, M.E.; Alsem, D.H.; Saiz, E.; Tomsia, A.P.; Ritchie, R.O. Tough, Bio-Inspired Hybrid Materials. Science 2008, 322, 1516–1520.
- Roeder, R.K.; Converse, G.L.; Kane, R.J.; Yue, W. Hydroxyapatite-Reinforced Polymer Biocomposites for Synthetic Bone Substitutes. JOM 2008, 60, 38–45.
- Li, B.; Wang, X.; Ma, J.; Huang, L. Preparation of phosphorylated chitosan/chitosan/hydroxyapatite composites by co-precipitation method. Adv. Mater. Res. 2009, 79–82, 401–404.
- Nudelman, F.; Sommerdijk, N.A.J.M. Biomineralization as an inspiration for materials chemistry. Angew. Chem. Int. Ed. 2012, 51, 6582–6596.
- Zhang, C.Y.; Chen, J.; Zhuang, Z.; Zhang, T.; Wang, X.P.; Fang, Q.F. In situ hybridization and characterization of fibrous hydroxyapatite/chitosan nanocomposite. J. Appl. Polym. Sci. 2012, 124, 397–402.
- Onoki, T.; Nakahira, A.; Tago, T.; Hasegawa, Y.; Kuno, T. Novel low temperature processing techniques for apatite ceramics and chitosan polymer composite bulk materials and its mechanical properties. Appl. Surf. Sci. 2012, 262, 263–266.
- Yang, L.; Ning, X.; Bai, Y.; Jia, W. A scalable synthesis of non-agglomerated and low-aspect ratio hydroxyapatite nanocrystals using gelatinized starch matrix. Mater. Lett. 2013, 113, 142–145.
- Bleek, K.; Taubert, A. New developments in polymer controlled, bioinspired calcium phosphate mineralization from aqueous solution. Acta Biomater. 2013, 9, 6283–6321.
- Ma, J.; Wang, J.; Ai, X.; Zhang, S. Biomimetic self-assembly of apatite hybrid materials: From a single molecular template to bi-/multi-molecular templates. Biotechnol. Adv. 2014, 32, 744–760.
- Won, J.-E.; El-Fiqi, A.; Jegal, S.-H.; Han, C.-M.; Lee, E.-J.; Knowles, J.C.; Kim, H.-W. Gelatin-apatite bone mimetic co-precipitates incorporated within biopolymer matrix to improve mechanical and biological properties useful for hard tissue repair. J. Biomater. Appl. 2014, 28, 1213–1225.
- Li, J.; Baker, B.A.; Mou, X.; Ren, N.; Qiu, J.; Boughton, R.I.; Liu, H. Biopolymer/Calcium Phosphate Scaffolds for Bone Tissue Engineering. Adv. Healthc. Mater. 2014, 3, 469–484.
- Shu, Y.; Yin, P.; Liang, B.; Wang, H.; Guo, L. Bioinspired Design and Assembly of Layered Double Hydroxide/Poly(vinyl alcohol) Film with High Mechanical Performance. ACS Appl. Mater. Interfaces 2014, 6, 15154–15161.
- Cheng, Q.; Jiang, L.; Tang, Z. Bioinspired Layered Materials with Superior Mechanical Performance. Acc. Chem. Res. 2014, 47, 1256–1266.
- Wegst, U.G.K.; Bai, H.; Saiz, E.; Tomsia, A.P.; Ritchie, R.O. Bioinspired structural materials. Nat. Mater. 2015, 14, 23–36.
- Le, H.; Natesan, K.; Pranti-Haran, S. Mechanical property and biocompatibility of co-precipitated nano hydroxyapatite-gelatine composites. J. Adv. Ceram. 2015, 4, 237–243.
- Shakir, M.; Jolly, R.; Khan, M.S.; e Iram, N.; Khan, H.M. Nano-hydroxyapatite/chitosan starch nanocomposite as a novel bone construct: Synthesis and in vitro studies. Int. J. Biol. Macromol. 2015, 80, 282–292.
- Zakharov, N.A.; Demina, L.I.; Aliev, A.D.; Kiselev, M.R.; Matveev, V.V.; Orlov, M.A.; Zakharova, T.V.; Kuznetsov, N.T. Synthesis and Properties of Calcium Hydroxyapatite/Silk Fibroin Organomineral Composites. Inorg. Mater. 2017, 53, 333–342.
- Miculescu, F.; Maidaniuc, A.; Voicu, S.I.; Thakur, V.K.; Stan, G.E.; Ciocan, L.T. Progress in Hydroxyapatite. Starch Based Sustainable Biomaterials for Biomedical Bone Substitution Applications. ACS Sustain. Chem. Eng. 2017, 5, 8491–8512.
- Yang, R.L.; Zhu, Y.J.; Chen, F.F.; Qin, D.D.; Xiong, Z.C. Bioinspired Macroscopic Ribbon Fibers with a Nacre-Mimetic Architecture Based on Highly Ordered Alignment of Ultralong Hydroxyapatite Nanowires. ACS Nano 2018, 12, 12284–12295.
- Zima, A. Hydroxyapatite-chitosan based bioactive hybrid biomaterials with improved mechanical strength. Spectrochim. Acta A 2018, 193, 175–184.
- Hu, Y.-Y.; Rawal, A.; Schmidt-Rohr, K. Strongly bound citrate stabilizes the apatite nanocrystals in bone. Proc. Natl. Acad. Sci. USA 2010, 52, 22425–22429.
- Kawasaki, T.; Takahashi, S.; Ikeda, K. Hydroxyapatite high-performance liquid chromatography: Column performance for proteins. Eur. J. Biochem. 1985, 152, 361–371.
- Dorozhkin, S.V.; Epple, M. Biological and Medical Significance of Calcium Phosphates. Angew. Chem. Int. Ed. 2002, 41, 3130–3146.
- Bouville, F.; Maire, E.; Meille, S.; Van de Moortele, B.; Stevenson, A.J.; Deville, S. Strong, tough and stiff bioinspired ceramics from brittle constituents. Nat. Mater. 2014, 13, 508–514.
- Sakhavand, N.; Shahsavari, R. Universal composition-structure property maps for natural and biomimetic platelet-matrix composites and stacked heterostructures. Nat. Commun. 2015, 6, 6523.
- Kim, M.; Yeo, M.; Kim, M.; Kim, G. Biomimetic cellulose/calcium-deficient hydroxyapatite composite scaffolds fabricated using an electric field for bone tissue engineering. RSC Adv. 2018, 8, 20637–20647.
- Malkaj, P.; Pierri, E.; Dalas, E. The crystallization of hydroxyapatite in the presence of sodium alginate. J. Mater. Sci. Mater. Med. 2005, 16, 733–737.
- Ingole, V.H.; Vuherer, T.; Maver, U.; Vinchurkar, A.; Ghule, A.V.; Kokol, V. Mechanical Properties and Cytotoxicity of Differently Structured Nanocellulose-hydroxyapatite Based Composites for Bone Regeneration Application. Nanomaterials 2020, 10, 25.
- Klemm, D.; Heublein, B.; Fink, H.; Bohn, A. Cellulose: Fascinating Biopolymer and Sustainable Raw Material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393.
- Schierbaum, F. The Current World Corn Situation: Production, Uses and Ending Stocks; Revised Figures for Fiscal Years 2006/07, 2007/08, Current Figures 2008/09, Production Profiles and Outlook. Starch—Stärke 2010, 62, 272–276.
- Nyman, J.S.; Roy, A.; Shen, X.; Acuna, R.L.; Tyler, J.H.; Wang, X. The influence of water removal on the strength and toughness of cortical bone. J. Biomech. 2006, 39, 931–938.
