Lung diseases rank third in terms of mortality and represent a significant economic burden globally. Scientists have been conducting research to better understand respiratory diseases and find treatments for them. An ideal in vitro model must mimic the in vivo organ structure, physiology, and pathology. Organoids are self-organizing, three-dimensional (3D) structures originating from adult stem cells, embryonic lung bud progenitors, embryonic stem cells (ESCs), and induced pluripotent stem cells (iPSCs). These 3D organoid cultures may provide a platform for exploring tissue development, the regulatory mechanisms related to the repair of lung epithelia, pathophysiological and immunomodulatory responses to different respiratory conditions, and screening compounds for new drugs. To create 3D lung organoids in vitro, both co-culture and feeder-free methods have been used. However, there exists substantial heterogeneity in the organoid culture methods, including the sources of type 2 alveolar cells (AT2) cells, media composition, and feeder cell origins.
- organoids
- alveolar type 2 cells
- stem cells
- 3D cultures
- pulmonary diseases
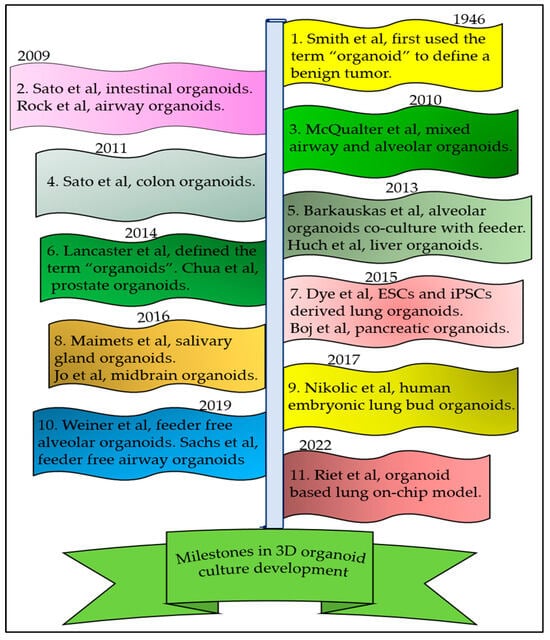
1. Introduction
2. Sources of AT2 Cells
3. AT2 Organoid Culture Methods
Lung AT2 organoids serve as a robust model for studying lung diseases, drug screening, investigating lung development, and designing new therapies (Figure 2). Over past decades, various methods have been employed to cultivate distal lung organoids, depending on the cell sources, growth supplements, and supporting cells. The most utilized approaches include co-culture and feeder-free (AT2-cell-only) procedures.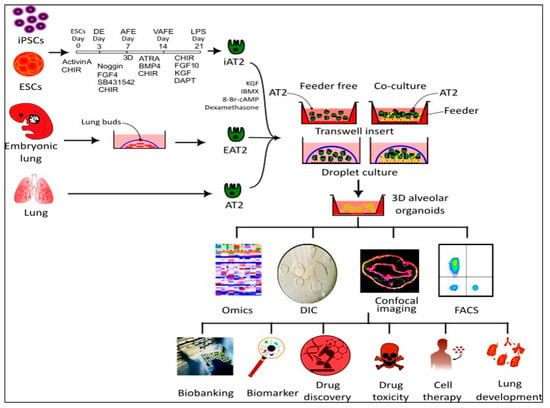
3.1. Co-Culture Method
3.1.1. EpCAM+ AT2 Co-Cultured with Fibroblast Cells
Earlier attempts to grow organoids in Matrigel from EpCAM+ AT2 cells alone failed, but complex epithelial cell colonies were generated when co-cultured with Sca-1+ mesenchymal cells [31]. This study showed that αSMA+ mesenchymal cells tightly surrounded epithelial colonies, suggesting a dependency of lung epithelial cells on feeder (fibroblast)-cell-released FGF-10 and HGF to form colonies in vitro. This observation aligns with in vivo studies demonstrating that FGF-10 and HGF regulate lung development [44][58][59][46,70,71]. Later, McQualter et al. [60][72] showed that CD166− and CD166+ lung stromal cells exhibited different epithelial-supportive capacities. They reported that the in vitro expansion of lung stromal cells resulted in the downregulation of FGF10 expression, reducing their ability to support epithelial colony formation.3.1.2. AT2 Co-Cultured with PDGFRA+ Cells
Barkauskas et al. reported that AT2 cells function as stem cells in the distal lung and form alveolospheres in vitro [4]. They genetically labeled AT2 cells and purified Sftpc-CreER:Rosa-Tm lineage-positive cells using FACS. To validate that PDGFRA+ mesenchyme cells were an integral part of AT2 niches, these AT2 populations were co-cultured with primary PDGFRA+ mesenchyme cells (in a 1:20 ratio) or with neonatal mouse lung fibroblasts. They found that AT2 cells co-cultured with PDGFRA+ mesenchyme cells developed larger and more numerous colonies compared to those co-cultured with immortalized fibroblast cell lines. Furthermore, the same group reproduced their murine alveolar model in humans by growing HTII-280+ AT2 cells co-cultured with the MRC5 cell line to form alveolospheres. Subsequently, several studies adapted this protocol with variations in culture media composition, the AT2-to-fibroblast ratio, and the types of feeder cells used.3.1.3. Human iPSC-Derived AT2
Induced pluripotent stem cells are adult cells genetically reprogrammed to resemble embryonic-like stem cells [61][75]. Patient-specific iPSCs can be a valuable tool for directly studying human diseases and developing personalized medicine using human-derived in vitro models. Several studies have reported the differentiation of iPSCs into NKX2-1+ and SFTPC-expressing AT2 cells [62][63][57,76]. Recently, Jacob et al. reported a modified, robust protocol for the differentiation of iPSC into mature, functional AT2 comparable to primary adult AT2 cells. They found that Wnt signaling, in addition to FGF signaling, together with corticosteroids and cyclic AMP, promotes the maturation of SFTPC+ AT2 from NKX2-1+ precursors in vitro. The feeder-free alveolosphere derived from these AT2 cells displayed classical functional features of mature AT2 cells, including innate immune responsiveness and the processing of surfactant lamellar bodies. This iAT2-derived organoid model could be passaged for up to one year without differentiating into AT1 cells and serving as a source of pure AT2 population [64][77]. In an interesting study, patient-derived iPSCs were differentiated into NKX2-1+ lung epithelial progenitor cells through a 21-day differentiation method. The NKX2-1+ progenitor cells were sorted using Carboxypeptidase M (CPM) as their cell surface marker and cultured alone to grow lung bud organoids. Alternatively, these cells were co-cultured with primary lung fibroblasts to grow alveolar organoids to model HPS-associated interstitial pneumonia (HPSIP) [55][59]. However, this study could not show any significant differences in organoid morphology and size between patient-specific and gene-corrected organoids, likely due to the immaturity of iPSC-derived AT2 cells.3.1.4. Mouse and Human ESC-Derived AT2 Cells
Embryonic stem cells are pluripotent cells with the potential to differentiate into multiple tissue types. Recently, bone-marrow-derived “very small embryonic-like stem cells” (VSELs) have been shown to have the potential to differentiate in vivo into SPC-producing AT2 cells. In a remarkable experiment, VSELs were isolated from SPC-H2B-GFP BAC transgenic mice and administered to a bleomycin-injured lung injury mouse model. After three weeks, VSELs differentiated into GFP+ AT2 cells and regenerated the epithelium in vivo. These VSELs-derived GFP+ AT2 cells were FACS-sorted from mice lungs and used to cultivate organoids in vitro to study their proliferation and differentiation potential. After 21 days of co-culture with MLG cells, AT2 cells from VSEL-transplanted mice developed GFP+ organoids, whereas no GFP+ organoids were observed in the control groups. The lineage of VSEL cells was confirmed via the colocalization of the TTF1 (transcription termination factor 1) marker with GFP+ AT2 cells [65][80]. Nikolić et al. utilized human embryonic lung bud tips progenitor cells to cultivate long-term self-renewing, branching organoids for studying lung development. The embryonic tips, embedded in Matrigel with EGF, FGF7, FGF10, Noggin, RSPO1, CHIR99021, and SB431542, formed spheres within 12 h with a 100% colony-forming efficiency. They were able to passage these organoids for nine passages without changes in morphology, SOX2 and SOX9 expression, or karyotype alterations. This study also showed that the growth conditions for human lung tip organoids do not support the long-term self-renewal of mouse lung tip cells, suggesting species-specific differences during lung development. Interestingly, they observed that mesenchymal cells disappeared after the second passage of organoid cultures, indicating that organoids can be maintained without mesenchymal cells. However, co-culture with canalicular-stage mesenchyme improved alveolar differentiation, leading to the attainment of a bipotent progenitor stage (pro-SFTPC+, HTII-280+, HOPX+ and PDPN+ co-expression, and NKX2-1+) [33][66][33,81].3.2. Feeder-Free Organoid Culture Systems
Although AT2 organoid co-cultures have been used to model lung diseases, they have several drawbacks that render them an undesirable system. One major drawback is the separation of AT2 cells from feeder cells for subsequent transplantation, as transplanting fibroblast cells can induce fibrosis following injury [67][68][83,84]. The variations in the type of feeder cells and the composition and concentration of growth factors secreted by the lung mesenchyme make co-culture disadvantageous for modeling cellular therapies. The co-culture system’s niche induces the differentiation of AT2 to AT1 cells. Recent studies have revealed that, in feeder-free organoid cultures, both AT2 cells and iAT2 cells do not spontaneously differentiate into AT1 cells. Instead, their differentiation into AT1 cells necessitates the addition of specific AT1 differentiation induction factors [69][85]. The feeder-free organoid cultures of AT2 cells in defined medium conditions (AT2 maintenance medium) maintain AT2 cells in a proliferative state and can be scaled up and passaged multiple times to yield a large number of AT2 cells. Feeder-free, serum-free cultures maintain the clonal expansion of EpCAM+ LysoTracker+ AT2 cells for up to 180 days [70][56]. These AT2 cells can be stored in liquid nitrogen for extended periods, and the same passage can be used for multiple experiments. This approach increases the reproducibility of results and reduces the number of animals used in the research for isolating AT2 cells, as the cells isolated from one mouse can be used for multiple experiments or studies.3.3. D Matrix Alternatives to Matrigel for Organoid Cultures
Organoid growth not only depends on growth factors and cell–cell interactions but also on signals from the extracellular matrix (ECM), which is an intriguing aspect of stem cell niches. Accumulated evidence has suggested that both physiological and mechanical cues from the extracellular matrix (ECM) may contribute to the maturation and differentiation of type 2 alveolar epithelial cells (AT2) [69][85]. In co-culture experiments involving iAT2 and fibroblasts, it was observed that fibroblasts express ECM genes and growth factors when AT2 cells are undergoing a transition or transdifferentiation into AT1 cells [52][54]. To date, both co-cultures and feeder-free 3D cultures of AT2 organoids have primarily relied on Matrigel [4][9][52][4,9,54]. Matrigel is the most commonly used medium to support 3D organoid growth. It is an ECM derived from cancerous mouse tissue and is commonly utilized for stem and cancer cell proliferation. While organoids grown in Matrigel mimic tissue structure, organ physiology, and function, it is derived from animal sources and presents a significant challenge in defining culture conditions due to lot-to-lot variability. The Matrigel system also produces heterogeneous organoids in terms of shape, size, and composition [71][79]. A chemically defined matrix system would provide more reliable data that could be clinically translated. Hoffman et al. conducted a study on a hydrogel derived from the extracellular matrix (ECM) of human alveolar cells (referred to as aECM hydrogel) for 3D organoid culture [52][54]. This hydrogel was obtained by processing alveolar-enriched fractions of decellularized human lungs. Their proteomics analysis revealed, that while the hydrogel did not encompass the complete spectrum of native ECM proteins, it exhibited enrichment in key proteins like COL1, COL3, and FBN1. The study demonstrated that the aECM hydrogel not only supported the proliferation of iAT2 (type 2 alveolar epithelial) cells and the formation of alveolospheres but also played a role in promoting the morphological differentiation of a subset of iAT2 cells into structures resembling human AT1-like cells. Additionally, the researchers investigated the influence of matrix stiffness by using different concentrations of aECM. Intriguingly, the higher stiffness of the aECM hydrogel led to a decrease in the SFTPC expression in iAT2 cells compared to hydrogels with less stiffness. Moreover, the bulk RNA sequencing of iAT2 cells cultured in the aECM hydrogel revealed changes in the expression of genes associated with iAT2 maturation, transitional cell states, and AT1-associated markers. These findings suggest that ECM stiffness may play a significant role in directing cellular differentiation processes [52][54].4. Applications
Lung cancer is the leading cause of cancer-related deaths globally. The absence of appropriate ex vivo models of the human alveolar epithelium has hindered our understanding of lung cancer pathogenesis and related therapy development. Early-stage diagnosis and treatment are essential for preventing cancer relapse and saving lives. Recently, Dost et al. [72][68] used both mouse AT2 and human iPSCAT2 organoid models to uncover the early consequences of oncogenic KRAS expression in vivo. Their work has provided novel tools for extensive data collection and studying the transcriptional and proteomic changes that distinguish normal epithelial progenitor cells from early-stage lung cancer. Their study revealed that reductions in AT2 lineage marker gene expression are an early consequence of oncogenic KRAS. Multiomics studies demonstrated that SPC-high cells in Kras activation and p53 loss (KP) lung tumor organoids exhibit higher tumorigenic capacity in the lung microenvironment compared to Hmga2-high cells [73][93].
HESC-derived organoids have been used to model human lung development. These organoids contained early-stage proximal and distal airway epithelial cells, including early-staged alveolar type 2 (AT2) cells (SPC+/SOX9+) and immature alveolar type 1 (AT1) cells (HOPX+/SOX9+) in vitro. However, when transplanted in vivo for the short term, these organoids differentiated into only a few distal progenitor epithelial cells (NKX2.1+, SOX9+, and P63+). In contrast, the long-term transplantation of these organoids resulted in the differentiation of lung distal bipotent progenitor cells (PDPN+/SPC+/SOX9+), AT2 cells (SPC+ SPB+), and immature AT1 cells (PDPN+, AQP5−). These long-term transplanted organoids also contained other cell types present in lung tissues, such as mesenchymal cells, vasculature, neuroendocrine-like cells, and nerve fiber structures [74][94].
In a recent study, mouse primary AT2 and human iPSCs-derived AT2 organoids were used to investigate the early stages of lung adenocarcinoma (LUAD) driven by KRAS mutation. The data from the alveolar organoids model may be useful in screening novel drug targets and developing new drug molecules to prevent lung cancer growth at the early stage.
Feeder-free organoid-derived AT2 cells have shown great potential for cellular therapy in lung regeneration. In a recent study, mesenchyme-free AT2 organoids were transplanted into the lungs of mice injured by influenza. The transplanted organoids retained their AT2 fate; however, in some cases, they adopted a dysplastic fate. These dysplastic organoids did not appear to improve the oxygen-exchange capability of the injured lungs in recipient mice. Further investigations have been requested to understand the molecular changes that occur in AT2 organoids after transplantation in the influenza-injured microenvironment in order to optimize organoid transplants [34].
