Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Matthew Jay Budoff and Version 2 by Camila Xu.
Iodixanol is an iso-osmolar non-ionic dimeric hydrophilic contrast agent with a higher viscosity than the monomeric agents. It is the only Food and Drug Administration (FDA)-approved iso-osmolar agent in the United States, and it is the only contrast agent with an FDA-approved indication for use in cardiac computed tomographic angiography (CCTA), to assist in the diagnostic evaluation of patients with suspected coronary artery disease.
- iodixanol
- cardiac CT
- coronary CT
- CCTA
- contrast media
- intravenous contrast media
1. Introduction
In the 1890s, Haschek and Lidenthal used a suspension of chalk and zinnober, a mercury compound, to image the vessels in an amputated hand. Since then, there has been immense research in this area, to develop less toxic and more efficacious contrast agents with more attenuation of X-rays [1]. The ideal intravascular contrast medium is water-soluble, stable, biologically inert, isotonic to plasma, selectively excreted, and available at a reasonable cost [2][3][2,3].
Water-soluble contrast agents, based on covalently bound iodine, were first made known by Binz and Rath in the 1920s [4][5][4,5]. X-rays are easily attenuated by iodine, as the atomic radius of the covalently bound iodine atoms is 133 picometers, which is in the range of X-ray wavelengths (10 to 10,000 picometers). The covalent bonding to a stable, functional group like benzene also reduces the toxicity of free iodide [6]. Iodinated contrast agents (ICAs) have become quintessential to the practice of radiology and medicine overall. It is approximated that about 75 million doses of ICAs are administered worldwide each year and continue to increase with the use of diagnostic modalities [7].
The ICAs are classified as being ionic or non-ionic (based on the presence or absence of a carboxylate side chain) and monomers or dimers (based on the number of benzene rings). Ionic monomeric agents which dissociate into ions in solution have high osmolality and are hypertonic when compared to plasma. Non-ionic contrast media, which do not dissociate into ions in a solution, have lower osmolality and can be isotonic to plasma [7].
2. Pharmacological Properties
Iodixanol is a water-soluble iso-osmolar non-ionic dimeric iodinated contrast agent for intravascular administration. Chemically, it is 5,5′-[(2-hydroxy-1,3-propanediyl) bis (acetylamino)]bis[N,N′-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzenedicarboxamide] (Figure 1), with a molecular weight of 1550.20 and iodine content of 49.1%. It has six iodine atoms per molecule, no carboxyl groups, and nine hydroxyl groups evenly distributed around the molecule [8].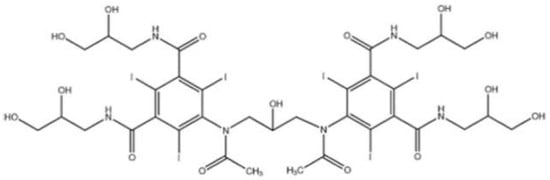
Figure 1.
Chemical structure of iodixanol (Visipaque).
3. Image Quality
Higher iodine concentration in the ICA has been attributed to higher mean attenuation in the descending aorta and coronary arteries on CCTA. In a study by Cademitri et al., iodixanol 320 had higher mean attenuation than iohexol (300 mg of iodine/mL) but lower than iohexol (350 mg I/mL), iomeprol (350 mg I/mL), and iomeprol (400 mg I/mL) [9]. Similar results were seen in the study by Honoris et al. [10]. Mean vascular enhancement across all coronary segments was highest in iopamidol (370 mg I/mL), followed by iohexol 350, iodixanol 320, and iodixanol 270, respectively (p < 0.002). Image quality, however, graded on a scale of 1–4, by the consensus of two cardiologists, was highest with iso-osmolar iodixanol 320 and 270 (3.27/4.0) and lowest with low-osmolar iopamidol (3.07), (p = 0.09) [10]. In the IsoCOR trial (Effect of Iso-osmolar Contrast Medium on Coronary Opacification and Heart Rhythm in Coronary CT Angiography), patients undergoing CCTA were randomized to iso-osmolar iodixanol 270 or low-osmolar iopromide 300. There was no statistical difference in coronary enhancement by the two agents (469 HU ± 167 vs. 447 HU ± 166, respectively (p = 0.241)) [11]. The arterial opacification is dependent on the iodine delivery rate, which in turn depends on the rate of contrast injection and concentration of iodine in the contrast medium. To keep a similar level of contrast enhancement (350 HU) during CCTA, iso-osmolar iodixanol 320 needed a higher simulated flow rate (3.90 vs. 3.62 mL/s, p = 0.017) and lower iodine delivery rate (1.34 vs. 1.25 g/s, p = 0.024) when compared with low-osmolar iopromide 370 in the study by Yang et al. [12]. It has also been suggested that the higher viscosity of iodixanol 320 at 37 °C might also result in a more concentrated intravascular flow pattern, which would lead to similar intra-cardiac iodine concentration as that of iohexol [6]. Appropriate pre-heating of iodixanol is of paramount importance to limit the higher viscosity. When adjusted for iodine concentration in the IsoCOR study, the mean bolus size of iodixanol 270 was larger than that of iopromide 300 (76.8 mL ± 11.6 v/s 69.7 mL ± 10.8, respectively; p < 0.001) [13]. Importantly, the voltage used can also affect the attenuation and image quality, with lower kilovolts (<106), demonstrating higher attenuation values than those using 107–120 or higher kilovolts to acquire images. This is true for all contrast types and must be carefully controlled in any comparative study. Tsai et al. investigated the cardiac enhancement by iohexol 350 and iodixanol 320 on multiple detector computed tomography (MDCT). There was no significant statistical difference between the two agents, though iohexol showed slightly higher enhancement (11.2 HU). However, on the delayed scans, iodixanol had better (7.7 HU) and longer enhancement (p < 0.05) in the left ventricle [13]. As dimers have larger molecule sizes than monomers, there is a slower diffusion rate through the capillary pores during the delayed period, leading to longer-lasting attenuation differences between the coronary arteries and myocardial tissue [13]. Less dilution by influx from the extravascular space is expected with decreased osmolarity. This would also be relevant when choosing contrast agents in patients with compromised renal function [14]. Tsai et al. performed a randomized trial of iodixanol 320 and iohexol 350 in 72 patients undergoing MDCT [13]. They showed that enhancement in the right heart, left heart, coronary arteries, and LV myocardium in the arterial phase showed no statistical difference (p > 0.05) between the two groups. They concluded that iodixanol 320 can provide vascular enhancement in cardiac MDCT that is similar to iohexol 350 [15]. In a prospective multi-center register study with 874 patients by Budoff et al. [16], using cardiac outcomes as the endpoint, the sensitivity of iodixanol-enhanced CCTA was 96.1%, 95.8%, and 94.7% at 1, 6, and 12 months, respectively, with a specificity of 84.5%, 86.6%, and 87.0%, and NPV > 99.0%. Prior trials using iodixanol as the ICA for CCTA have also yielded similar diagnostic accuracy for obstructive coronary artery stenosis, as well as 99% negative predictive value at both the patient and segment levels [17][18][17,18]. In a study conducted by Christensen et al., the contrast-to-noise ratio (CNR) was significantly increased for iopamidol 370 versus iodixanol 320 (aortic root, p = 0.021; left main, p = 0.032; left anterior descending, p = 0.033; left circumflex, p = 0.039; and right, p = 0.009) [18]. Choi et al. performed a double-blinded, randomized, and parallel study on 300 consecutive outpatients who underwent outpatient CCTAs to evaluate CAD. The subjects were assigned to receive either iodixanol 320 or iohexol 350. The image quality of each group was not significantly different from each other. The iodixanol group, however, had a higher CNR (p = 0.04) but showed no difference in image noise (p = 0.63) and signal-to-noise ratio (SNR) (p = 0.07) [15]. In a randomized, double-blinded parallel study with 200 patients undergoing elective cardiac angiography, receiving either iodixanol or iohexol, no statistical difference was noted in the angiographic quality between the two groups (p = 0.885) [19]. Similar results were noted in another cardiac angiography study comparing iodixanol and ioxaglate [20].4. Tolerability and Safety
ICAs differ in their tolerability profiles. Iso-osmolarity and lower iodine content have been proven to be safer and more tolerable in humans [10]. The toxicity of poly-iodinated contrast media is a function of the molecule’s physicochemical properties: notably ionicity, higher osmolality and viscosity, and the chemical interaction of reactive iodine anions with biological sites [12].4.1. Pain and Discomfort
Pain and discomfort following contrast administration is not only inconvenient for the patient but can also compromise image quality due to motion artifacts produced by the patient’s body movements during imaging. If the motion artifacts are significant enough to render the study non-evaluable, repeating the study would lead to additional contrast and radiation exposure to the patient and increased cost to the healthcare system. In earlier studies by Klow et al. and Hill et al. in patients undergoing invasive cardiac angiography, the patients receiving iodixanol had a significantly lower incidence of moderate to severe discomfort than those receiving iohexol [19][21][19,21]. In a more recent study with patients receiving iodixanol or iopamidol for CCTA, there was no difference in the number of patients having moderate to severe symptoms. However, a subgroup analysis of patients > 55 years of age showed lower rates of moderate to severe symptoms in the iodixanol group (8.5% vs. 24.6%, p = 0.01) [22]. In another study, while 27% of patients receiving iodixanol 320 reported pain or warmth, the rate was 13.1% for iodixanol 270, 21.3% for iopamidol, and 16.7% for iohexol (p < 0.05) [10]. When compared to a hyperosmolar agent, an iso-osmolar agent has greater vascular stability in arterioles that supply the skeletal muscles and skin over the extremities. Hence, there is no activation of vascular endothelium and no vasodilation followed by vasoconstriction. The activation of nociceptors in nerves supplying the neurovascular bundles is also less by iso-osmolar agents. This vascular stability and attenuated nociceptor activation may result in less pain and discomfort when iodixanol is used [12]. A study by Murphy et al. showed that diatrizoate, a high-osmolar agent, had higher pain and discomfort rates than low-osmolar iohexol and ioxaglate, thus proving iodine content was not responsible for adverse effects [23]. Hence, it can be hypothesized that the osmolarity of the ICA is the determinant of pain and discomfort after contrast injection and not viscosity or iodine content.4.2. Flushing
Flushing is the most common side effect after intravenous contrast injection. Iodixanol 270 had the lowest incidence of flushing (46%; 95% CI 36–56%) in 513 patients undergoing CCTA when compared to iopamidol (78%; 95% CI 59–87%) and iohexol (72%; 95% CI 55–85) (p < 0.001)] [10]. Patients receiving iodixanol had a lower incidence of moderate to severe flushing (3.0% vs. 12.8%, p = 0.005) than iopamidol in another study. The difference was more profound in patients over 55 years of age (8.5% for iodixanol vs. 24.6% iopamidol, p = 0.01) [22]. Other adverse effects reported, but very infrequently with iodixanol, include chest or leg pain, visual disturbance, dizziness, hypotension, vasovagal reaction, headache, nasal symptoms, taste disturbances, coughing, dyspnea, skin reactions, nausea, vomiting, and shivering [3].4.3. Heart Rate Variability
The patient’s heart rate and its variation play a vital role in cardiac CT imaging due to the limitations in the temporal resolution [24][25][26][24,25,26]. Besides mean heart rate, significant variations in heart rate per minute (due to significant R-R interval variations) also cause misregistration of ECG-gated data, hence causing artifacts in the reconstructed image. These if severe may render the study non-evaluable. Heart rate variability might also potentially increase overall radiation dose, due to longer scan times. Giesler et al. [27] and Hoffmann et al. [28] showed that a multitude of quality measures of CCTA including sensitivity, image quality, and evaluable coronary artery segments was inversely proportional to heart rate. In a CCTA study by Choi et al., median HR change was higher with iohexol when compared to iodixanol (p < 0.001) [15][27][28][15,27,28]. While iodixanol showed more HR variation < 3 beats/min (86% v/s 72%, p = 0.03), HR variability > 4 beats per min was noted more with iohexol (p = 0.003) [15]. In another study, HR variability was lowest for CCTA subjects with iodixanol 320 (2.5 ± 3.0), followed by iodixanol 270 (3.4 ± 5.2), and was highest with iopamidol 370 (4.9 ± 5.8) (p < 0.001) [10]. Studies have shown no difference in heart rate variability between iodixanol and iopidamol in patients undergoing CCTA (p = 0.56) [22], as well as non-gated chest CT angiography (p = 0.72) [29]. No significant difference was seen in the mean deviation of HR in patients undergoing CCTA, receiving iodixanol versus iomeprol (1.4 beats/min v/s 4.4 beats/min, p value not significant). However, the latter group showed a greater number of arrhythmic heartbeats during the scan (p < 0.001) [30]. Ioxaglate has been shown to induce a greater increase in mean heart rate than iodixanol in patients undergoing cardiac angiography and ventriculography (p < 0.05). Both ioxaglate and iodixanol caused significant QT interval prolongation (p < 0.005), but the changes were more marked with ioxaglate [31]. When compared with iomeprol, no significant differences were seen in terms of mean changes in HR during left coronary arteriography (p = 0.8), right coronary arteriography (p = 0.9), or left ventriculography (p = 0.8) by Schmid et al. [32]. The contrast injection during cardiac catheterization could presumably imply more susceptibility to reflex tachycardia, secondary to direct and nondiluted contrast agent injection, than during left ventriculography. Other factors such as the volume of contrast media used, image acquisition time, and route of administration vary during these procedures which might influence the ICA’s effect on cardiac parameters [33]. This could perhaps explain the results from studies where there was no difference in HR variability during left ventriculography between groups who received iodixanol and other ICAs [20][34][35][20,34,35] (Table 1).Table 1.
Major trials of tolerability.
| Authors | Randomized | Number of Patients | Mean Age | Contrast | Endpoints | Results |
|---|---|---|---|---|---|---|
| Lily Honoris et al., 2017 [10] | Yes | N = 513 | 57 ± 11 | Iodixanol Iohexol Iopamidol |
Flushing | Iodixanol superior |
Table 2.
Major trials of MACE (major adverse cardiac events).
| Study | Number of Pts | Mean Age | Contrast | Endpoint | Results | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Qian et al., 2017 [37] | N = 90 | 62 ± 13 | Iodixanol Iopromide |
90-Day cardiac event Acute heart failure |
Iodixanol superior No difference |
||||||
| Xiao et al., 2016 [36] | Yes | N = 2000 | |||||||||
| Bertrand et al., 2000 [38] | N = 1411 | 61.6 ± 10.6 | 52 ± 13.3 | IodixanolIodixanol 320 Iohexol | IoxaglateUrticaria and nausea |
Acute renal failureIodixanol superior | |||||
| No difference | Carlartrand-Lefebvre et al., 2011 [29] | Yes | N = 130 | 52 ± 16 | Iopamido Iodixanol 320 |
||||||
| Davidson et al., 2000 [39] | N = 810 | 61 ± 12 | Iodixanol Ioxaglate | HR | During hospital stay | No difference | |||||
| 30-Day cardiac event | Iodixanol superior | No difference | Jared D. Christensen et al., 2011 [7] | Yes | N = 60 | 53.5 ± 15.1 | Iodixanol 320 Iopamidol 370 |
HR | No difference | ||
| Harrison et al., 2003 [43] | N = 1276 | - | Iodixanol |
Ryo Nakazato, et al., 2016 [22] | Yes | N = 266 | 57.2 ± 11.7 | Iodixanol Iopamidol |
HR Flushing |
No difference No difference |
|
| Choi et al., 2012 [15] | Yes | N = 300 | 62 ± 11 | Iodixanol 320 Iohexol 350 |
HR | Iodixanol superior |
