Soot formation is an inevitable consequence of the combustion of carbonaceous fuels in environments rich in reducing agents. Efficient management of pollution in various contexts, such as industrial fires, vehicle engines, and similar applications, relies heavily on the subsequent oxidation of soot particles. Among the oxidizing agents employed for this purpose, oxygen, carbon dioxide, water vapor, and nitrogen dioxide have all demonstrated effectiveness.
- soot
- impact
- oxidation
- catalysts
1. Soot Formation and Methodology for Soot Oxidation
A few years ago, diesel engines gained worldwide fame owing to their unique features, such as low fuel consumption, long range, and greater thermal efficiency, compared with other engines [1][2][1,2]. The other side of the picture is also quite harsh given diesel engine exhaust emissions, which create a great threat for us on this planet [3][4][5][3,4,5]. The composition of their exhaust emanations is a combination of different gases, vapors, particulate matter (soot), liquid aerosols, nitrogen, water, CO, NOx, SOx, and polycyclic aromatic hydrocarbons (PAHs), and they make our world alarmingly polluted with air pollution [6][7][8][6,7,8]. The overall composition of diesel exhausts and their threats to human life and the environment are described in Table 1 [9][10][11][12][9,10,11,12].
| Pollutants | Concentration | Threats |
|---|---|---|
| Soot | 20–200 mg/m3 | Eyes problems, cancer, asthma, skin infections, lung damage, heart issues |
| NOx | 30–1000 ppm | Chest pain, respiratory and lungs problems, cough |
| SOx | Proportional to fuel S content | Acid rain, skin problems |
| CO2 | 2–12 vol% | Green house effect, acid rain, lung disease |
| CO | 100–1000 ppm | Hpertension, head pressure, lung disease |
| HC | 50–500 ppm | Eyes irritation, lungs issues, respiratory problems |
| PAH | 0.3 mg/mil | Kindney and liver damage |

2. Effect on Health and the Environment
3. Diversity in Catalysts for Removal of Soot
Recent research efforts have focused on developing platinum group metal-free catalysts for soot oxidation. Moreover, innovative catalyst materials, such as transition metal oxides and non-noble metal catalysts, have shown promising results in catalyzing soot oxidation while addressing cost and sustainability concerns [41][68]. Similarly, nanostructured catalyst materials have emerged as a prominent trend in soot oxidation research [42][69]. Nanostructured catalysts exhibit increased surface area and enhanced catalytic activity, enabling more efficient soot oxidation at lower temperatures. Research has explored various nanoarchitectures, including nanoparticles, nanowires, and nanotubes, to optimize catalytic performance [43][44][70,71]. Advances in catalyst support materials have further improved the durability and effectiveness of catalysts in soot oxidation [45][46][72,73]. Innovative support materials, such as perovskites, zeolites, and mesoporous materials, have been investigated for their potential to enhance catalyst stability and longevity under harsh operating conditions [47][48][49][74,75,76]. However, the identification of ceria-based catalysts as highly effective materials for soot oxidation has only recently been reported [50][51][40,77]. Ceria-based catalysts, known for their oxygen storage and release capabilities, have played a prominent role in reducing the temperature required for soot oxidation and enhancing catalytic activity. PGM (platinum group metal) catalysts are extremely dynamic [52][53][78,79]. PGMs, including platinum, palladium, and rhodium, are commonly used as catalysts for the oxidation of soot in various applications, particularly automotive exhaust systems. The surface environments of the costly metals and catalysts, their surface area, and other surface parameters significantly clean the emissions gas during automobile catalytic conversion. Normally, Rh, Ru, Au, and Pt particles adhere to the base material. However, they are costly, and they are susceptible to greater price increases with increasing demands due to their low richness [54][55][80,81]. Perovskite catalysts have gained significant attention in recent years due to their potential applications in various catalytic processes, including soot oxidation [56][85]. They are normally represented using the formula ABO3, wherein the A and B parts represent two cations of dissimilar dimensions, while O is an anion bridge that interacts with cations [57][58][86,87]. Part A generally belongs to elements of alkaline/alkali earth metals (Sr, Cs, Ca, Ba, Ra) [59][60][61][62][63][88,89,90,91,92] and/or rare earth (La, Ce, Nd, etc.) [64][65][66][93,94,95] with a larger radius of approximately 0.90 Å compared with the transition metal of part B (Ag [67][96], Fe [68][97], Co, Zn, Cu, Ni, Mn, Cr, Ru, Al [65][94]) with an approximate radius of 0.51 Å [69][98]. In a cubic cell, the constituent A-atom occupies the dice corner positions (0, 0, 0), B conquers the position of body center (1/2, 1/2, 1/2), and O occupies the position of face center (1/2, 1/2, 0). In addition, part A, which is normally coordinated to 12 oxygen molecules, forms a dodecahedral site. Part B is occupied by six O-atoms in octahedral coordination [70][71][99,100]. Layered double hydroxides (LDHs) are a class of materials that have been explored for various catalytic applications, including soot oxidation. LDHs are also known as hydrotalcite-like compounds or anionic clays. They are made of positively charged metal hydroxide layers and charge-balancing anions in the interlayer regions [72][73][108,109]. LDHs exhibit anion exchange characteristics due to the weakly carbonate-bonded anions in their interlayer region. Moreover, selection for the anions can be done at the initial precipitation during its preparation. It is important to mention here that during calcination, metal-oxide products can be produced from these LDHs, which are further used for various applications [74][75][114,115]. In addition, the resultant metal oxides possess higher surface areas, which is normally beneficial for catalysis applications. Mixed metal oxides (MMOs) of inner and outer transition metals, alkaline, rare earth, and alkali group metals have potential for various catalytic applications [76][123]. Different reactions have been used for MMO synthesis. For example, alkylation, the Mannich reaction, oxidation, reduction, multicomponent, condensation, cycloaddition, deprotection, hydroxylation, and other reactions can be done successfully in different reaction conditions [77][124]. The mixed metal oxides use an interesting mechanistic approach to convert the NO and soot into their respective components, such as NO2 and CO2 (Figure 411) [75][115].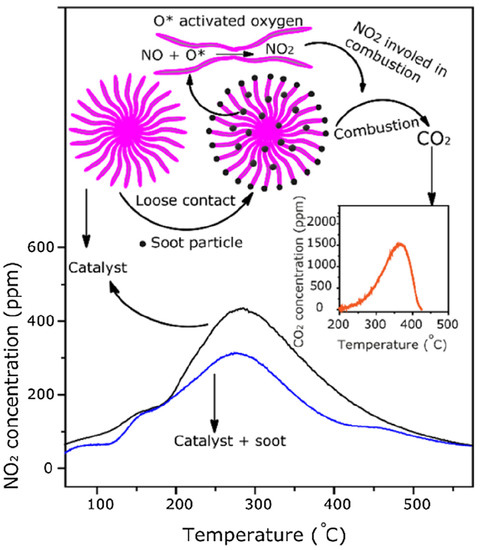
4. Ceria-Based Mixed Metal Oxides
Ceria (CeO2) is of most significant importance as a component of three-way catalysts (TWCs) given its storage capacity (OSC) for oxygen [78][125]. It has attained a significant rank among the metal oxides that have been extensively studied to date [79][80][126,127]. The research direction proposed by Trovarelli has opened a new door for ceria-based catalysts, indicating their potential in theoretical and practical applications as well as providing structural insights for their derived catalysts. Meanwhile, they function to support and boost the catalytic performances of metal catalysts [81][128]. The effects of the nanometric sizes and morphologies of ceria-based catalysts have been studied since the last decade, and various studies have reported on their synthesis pathways, chemical properties, geometries, characteristics, and catalytic performance in the oxidation of CO to date [82][129]. Recently, a correlation has been reported for redox properties between surface properties and the crystal morphology of ceria-based cubes, polyhedrons, and rods. Observations indicated that face reconstruction, size, and nanomorphology influence their performance, selectivity and stability [83][130]. In the ceria cubic structure, the fcc group, which is regarded as a stable surface plane, shows a lower coordination number compared to its bulk with divergent terminating structures on surfaces, including repetitive O-Ce-O interlayers, both elements Ce and O, and a O-Ce-O-Ce echoing unit. However, in thermally controlled systems, stable surfaces are normally generated during crystal growth and finally develop specific nanoshapes [84][131].
Remarkably, every stable plane displays various reduction features. The redox process of Ce4+ to Ce3+ produces vacancies for oxygen that play a vital role in oxygen packing and oxidation reactions. There is no theoretical basis; the growth plans follow the order of reactivity for oxygen vacancy defect formation, providing the basis for experimental work to assess the relationship between the catalytic performance and nanocrystal morphology of ceria [85][133]. The oxygen vacancies and surface chemistry strongly depend on the nanometric size of particles, and these factors are strongly enhanced when the particle size is less than 10 nm. Oxygen vacancy creation modeling investigations focused on size revealed that their energy is governed by the position of the oxygen atom lattice; for nanoparticles (NPs) with a size of 2–4 nm, its value approaches the minimum level [86][134].
The catalytic performance of the nanorods was observed to be associated with loosely bound oxygen. The nanorods’ performances were lower than those of nanowires, regardless of the fact that nanorods and nanowires exhibit predominantly reactive planes; this could be attributed to a higher concentration of surface-active planes [87][137]. Hierarchically, mesoporous ceria is prepared using diatom templates, which have greater Ce3+ content, a high specific surface area (SSA) (78 m2 g1), facile reducibility, a higher number of oxygen vacancies, and enhanced CO oxidation compared with bulk ceria. Moreover, ultrasound synthesis was reported to form nanoflowers, nanospheres, nanorods, and nanoribbons of ceria nanostructures (size ~5 nm) [88][138]. This synthesis was performed in a single step using various kinds of ionic liquids. The shape and structure of the final product depend on how it was heated. For example, under [C4mim][Tf2N], the ionothermal fabrication method produced flower and nanorod shapes, while the ultrasound method produced nanospheres. Nanoshape activity order followed the order of the SSAs; however, this order was not found to be proportional to them, indicating that oxygen vacancies as well as structural defects play crucial roles. Sonochemistry under [C4mim][Tf2N] generates nanospheres with the best performance. This is because the nanospheres have a large SSA, a mesoporous structure, a higher number of surface oxygen vacancies, and small particle size [89][139].
