Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Soji Morishita and Version 2 by Lindsay Dong.
BCR::ABL1-negative myeloproliferative neoplasms (MPNs) are a group of hematopoietic malignancies in which somatic mutations are acquired in hematopoietic stem/progenitor cells, resulting in an abnormal increase in blood cells in peripheral blood and fibrosis in bone marrow. Mutations in JAK2, MPL, and CALR are frequently found in BCR::ABL1-negative MPNs, and detecting typical mutations in these three genes has become essential for the diagnosis of BCR::ABL1-negative MPNs. Furthermore, comprehensive gene mutation and expression analyses performed using massively parallel sequencing have identified gene mutations associated with the prognosis of BCR::ABL1-negative MPNs such as ASXL1, EZH2, IDH1/2, SRSF2, and U2AF1.
- BCR::ABL1-negative myeloproliferative neoplasms
- gene mutations
- diagnostic marker
- prognosis
1. Introduction
Myeloproliferative neoplasms (MPNs) are characterized as a clonal proliferation of hematopoietic stem/progenitor cells, which cause an increase in one or more mature myeloid lineage cells. MPNs consist of multiple subgroups: chronic myeloid leukemia (CML); polycythemia vera (PV); essential thrombocythemia (ET); prefibrotic primary myelofibrosis (PMF); overt PMF; chronic neutrophilic leukemia (CNL); chronic eosinophilic leukemia (CEL); and unclassifiable MPN, not otherwise specified (Figure 1) [1][2][1,2]. In general, CML involves a typical driver gene alteration, the BCR::ABL1 fusion gene, and more rare diseases, namely CNL, CEL, and unclassifiable MPN, not otherwise specified, have been treated as independent diseases. In contrast, patients with BCR::ABL1-negative MPNs (PV, ET, prefibrotic PMF, and overt PMF) transform to other subgroups with a 10–15% frequency and share common gene mutations, namely JAK2 mutations (V617F and exon 12), MPLW515L/K, and CALR exon 9 frameshift mutations, in a mutually exclusive manner [3][4][5][6][7][8][9][3,4,5,6,7,8,9].
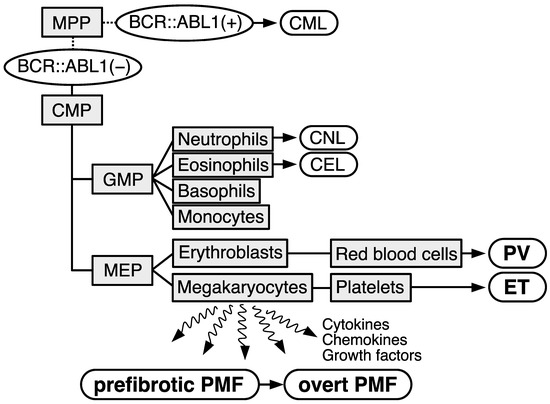
Figure 1. Schematical illustration describing the subtypes of myeloproliferative neoplasms (MPNs). Cell types are depicted as light gray squares. MPP: multipotent progenitor; CMP: common myeloid progenitor; GMP: granulocyte-monocyte progenitor; MEP: megakaryocyte–erythrocyte progenitor. Subtypes of MPNs are depicted as white rounded rectangles. Chronic myeloid leukemia (CML) exhibits BCR::ABL1 gene (BCR::ABL1(+)). Other subtypes are stratified as BCR::ABL1-negative (−) MPNs. CNL: chronic neutrophilic leukemia; CEL: chronic eosinophilic leukemia; PV: polycythemia vera; ET: essential thrombocythemia; PMF: primary myelofibrosis.
2. JAK2 Mutations
The JAK2 mutations considered as driver mutations of BCR::ABL1-negative MPNs are V617F substitution and complex mutations, including missense and in-frame deletions/insertions at exon 12 [3][4][5][6][3,4,5,6]. These mutations concentrate around the JH2 domain, which suppresses the kinase activity of the JH1 domain in JAK2 under a static state. JAK2 mutations decrease the suppression of kinase activity by the JH2 domain, resulting in the constitutive activation of JAK2. The JAK2V617F mutation is a single nucleotide alteration from guanine to thymine at nucleotide position 1849, which causes an amino acid change from V (valine, GTC) to F (phenylalanine, TTC) at codon 617. In addition, the JAK2V617F mutation was initially identified from the three driver gene mutations of BCR::ABL1-negative MPNs [3][4][5][3,4,5] and has been most frequently identified among the patients with BCR::ABL1-negative MPNs, and the positivity is approximately 97% in PV and approximately 50% in ET and PMF. In contrast, JAK2 exon 12 mutations are specific for PV, with 3% positivity, and a variety of mutations have been identified at JAK2 exon 12 (Supplementary Table S1, according to the Catalogue Of Somatic Mutations In Cancer (COSMIC) database, as of June 2023) [10][11][20,21]. Clinically, patients with PV harboring the JAK2V617F mutation exhibit pancytosis, including leukocytosis, thrombocytosis, and erythrocytosis, whereas those harboring the JAK2 exon 12 mutation show only an aggressive increase in red cell mass. As for the relationship between the prognosis and JAK2 mutations, no significant differences between JAK2V617F-mutated and JAK2 exon 12-mutated patients with PV have been observed [12][22]; nevertheless, JAK2V617F is a well-known risk factor of thrombosis [13][14][23,24]. Thrombosis promoted through the increased neutrophil extracellular trap formation was observed in the JAK2V617F-mutated murine model [15][25]. JAK2V617F is also useful for monitoring the efficacy of treatments or predicting the outcome of patients. Pegylated interferon-α, for example, is one of the recently developed drugs against MPNs and decreases the JAK2V617F allele burden in patients [16][17][18][26,27,28].3. MPL Mutations
The majority of MPL mutations involve the substitution of W (tryptophane, c.1542-1544TGG) at codon 515 with other nucleic acids, causing W515L/K/A/R (leucine/lysine/alanine/arginine) mutations [7][19][7,34]. In addition to these W515 mutations, the substitution of S (serine) at codon 505 with N/C (asparagine/cysteine) has been identified [20][21][35,36]. These mutations are located at the membrane-spanning segment of MPL and are considered to involve conformation changes that trigger constitutive activation of the downstream molecules. Although the frequency of MPL mutations in BCR::ABL1-negative MPNs is low (5% at most in ET, <10% in PMF), considering that mutant CALR binds to MPL and activates downstream signals [22][37], signal activation through MPL may play a key role in the pathogenesis of BCR::ABL1-negative MPNs [23][38].4. CALR Exon 9 Frameshift Mutations
The CALR mutation is the most recently discovered among the driver gene mutations in BCR::ABL1-negative MPNs and is found in approximately 20–30% of patients with ET and PMF [8][9][8,9]. This mutation is characterized by the presence of a deletion or insertion at the end of exon 9, the final exon of the CALR gene. Over 100 variations have been found according to the COSMIC database, as of January 2023), all of which cause the same frameshift and produce a common amino acid sequence at the C-terminus when translated into protein. Among them, deletions of 52 bases (type 1, p.L367fs*46) and insertions of 5 bases (type 2, p.K385fs*47) are the major mutations, accounting for approximately 85% of the observed variations. Mutant CALR activates downstream signaling by forming homomultimeric complexes through a new amino acid sequence generated by the mutation, changing the structure of the CALR protein and allowing it to bind with MPL [22][24][25][37,41,42]. Patients with overt PMF harboring a CALR mutation have a better prognosis than those with other driver gene mutations [26][43]. Regarding the CALR mutations in overt PMF, CALR type 1 mutations are dominant, and patients harboring the type 2 mutation exhibit a poorer prognosis than those with the type 1 mutation [27][28][44,45].5. Triple-Negative BCR::ABL1-Negative MPNs
A portion of patients with BCR::ABL1-negative MPNs (none or rare in PV, 10–15% in ET, and ~10% in PMF) have none of the driver mutations, referred to as TN cases. Noncanonical somatic mutations at driver genes of BCR::ABL1-negative MPNs (e.g., JAK2G571S and MPLS204F/P) have been identified in TN cases; however, it should be considered that these mutations do not account for all the remaining cases [29][30][12,46] and no evidence of cytokine-independent cell growth has been reported for these mutations. Whole exome sequencing and analysis of TN-ET have shown that approximately half of the patients exhibited polyclonal cell differentiation, which implies that some cases of thrombocytosis in TN-ET may be caused by nonneoplastic diseases [29][31][12,47].
6. CREB3L1 as a Novel Diagnostic Marker of BCR::ABL1-Negative MPNs
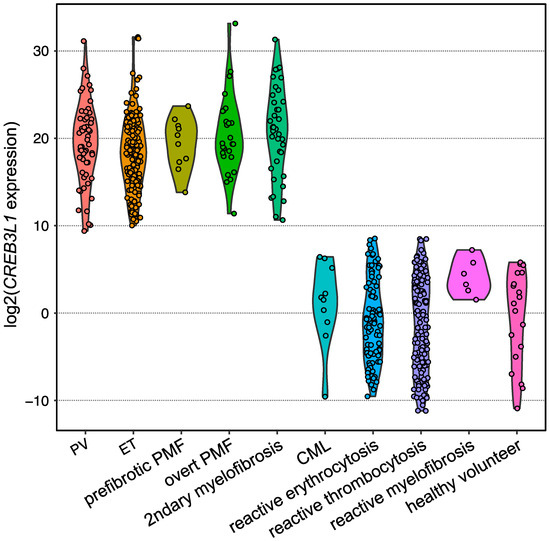
To diagnose TN cases in practice, a histopathological diagnosis of the BM biopsy is required. However, the discrimination of TN from reactive cases is challenging because the pathological diagnosis of BCR::ABL1-negative MPNs is not always reproducible, even for expert hematopathologists. By focusing on the fact that typical clinical presentations (i.e., thrombocytosis) of ET are similar regardless of the presence and type of driver gene mutations, and that the downstream RNA expression may be common and different from that in reactive cases, differential expression analysis utilizing RNA from platelet-rich plasma (PRP) obtained from ET and reactive thrombocytosis patients was conducted. As a result, CREB3L1 was found to be specifically overexpressed among ET patients [32][13]. CREB3L1 is a transcription factor that localizes in the endoplasmic reticulum (ER), migrates into the nucleus in response to ER stress, and induces the expression of various genes [33][49]. Although the role of CREB3L1 in the pathogenesis of BCR::ABL1-negative MPNs remains unclear, the IRE1a/XBP1 pathway, which is an ER stress-responsible pathway other than CREB3L1, is activated by the CALR type 1 mutation and drives BCR::ABL1-negative MPNs [34][50]. Expansion of the testing of CREB3L1 overexpression for other subtypes of BCR::ABL1-negative MPNs harboring one of the driver gene mutations in the validation analysis by employing quantitative PCR revealed that CREB3L1 was overexpressed widely among BCR::ABL1-negative MPNs compared with those of reactive cases and healthy volunteers (Figure 25). The area under the ROC curve showed that the sensitivity and specificity were both 1.0000, indicating that the CREB3L1 overexpression in PRP discriminates driver gene-mutated BCR::ABL1-negative MPNs from reactive cases [32][13]. Further investigations are required to determine whether CREB3L1 expression is a diagnostic marker for BCR::ABL1-negative MPNs, including TN cases.
Figure 25. Violin plot showing the expression levels of CREB3L1 measured using reverse transcription quantitative PCR. Dots represent the CREB3L1 levels of the individuals, which are expressed as the value relative to the mean expression levels among healthy volunteers. B2M was used as an internal control.
7. Nondriver Mutations and Their Association with the Prognosis of BCR::ABL1-Negative MPNs
Comprehensive genome analyses, such as MPS, have identified some mutations relating to epigenetic modification and RNA-splicing among patients with BCR::ABL1-negative MPNs at low frequency [35][36][14,15]. MPS-based comprehensive target resequencing methodology focusing on these nondriver mutations also revealed that the mutations highly accumulated in patients with BCR::ABL1-negative MPNs were correlated with a poor prognosis. Based on these findings, mutation-enhanced prognostic scoring systems based on the positivity of nondriver mutations have been proposed and are widely used to estimate the prognostic risks of patients with BCR::ABL1-negative MPNs.
Similar to the results obtained by other groups, patients with PMF harboring ASXL1, EZH2, and/or SRSF2 mutations exhibited significantly shorter 5-year overall survival, and these gene mutations are also the poor prognostic factors of PMF that were demonstrated in the Japanese cohort [37][16]. Furthermore, regarding ET and PV, the frequencies of ASXL1 and EZH2 mutations increase as the diseases progresses from ET or PV to prefibrotic PMF and overt PMF, whereas the frequencies of DNMT3A and TET2 mutations are unrelated to disease type. This implies that ASXL1 and EZH2 mutations are related to disease progression, whereas DNMT3A and TET2 mutations may trigger the disease. Logistic regression analysis showed that ASXL1 mutation-positive ET/PV patients had a high rate of progression to leukemia and myelofibrosis [38][17].
