Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Eleonora Russo and Version 2 by Sirius Huang.
The potential of perinatal tissues to provide cellular populations to be used in different applications of regenerative medicine is well established. Efforts of researchers are being addressed regarding the evaluation of cell products (secreted molecules or extracellular vesicles, EVs) to be used as an alternative to cellular infusion. The data regarding the effective recapitulation of most perinatal cells’ properties by their secreted complement point in this direction. EVs secreted from perinatal cells exhibit key therapeutic effects such as tissue repair and regeneration, the suppression of inflammatory responses, immune system modulation, and a variety of other functions.
- mesenchymal stromal cells
- human perinatal tissues
- extracellular vesicles
1. Introduction
In the last decade, significant advances have been made to fully assess the biology of mesenchymal stromal cells (MSCs) derived from perinatal tissues. The term “perinatal” defines birth-associated tissues and other fetal annexes (e.g., amnion, chorion, and the umbilical cord), obtained immediately after birth. To date, many applications of perinatal MSCs for clinical purposes have been studied with promising results, mainly due to their unique immune and differentiative properties [1]. Despite this, perinatal MSC-based therapy still presents problems related to different aspects, such as the difficulty of developing specific methods of isolation and characterization, as well as the identification of an optimal protocol for ex vivo expansion and the often observed efficacy post transplant [2][3][2,3]. Nevertheless, it has been suggested that the perinatal MSCs’ therapeutic efficacy may be fully recapitulated by their secretome or conditioned medium (e.g., soluble proteins, lipids, and extracellular vesicles) [4]. Thus, a new definition was introduced by Silini et al. namely “perinatal derivatives (PnD)” which includes all birth-associated tissues, the cells they are composed of, and all the biomolecules secreted [5]. Therapeutic approaches based on MSC secretomes may have different advantages over the use of transplanted MSCs, due to the minimal effect on immunogenicity, the higher yields of bioactive molecules, and easier application. Perinatal MSC secretomes seem to exhibit the same anti-inflammatory, immunomodulatory, and regenerative properties as parental MSCs [6][7][8][6,7,8], and appear to be effective in treating the side effects of ischemia–reperfusion injury [9][10][9,10]. Furthermore, the use of secretomes provides a suitable strategy to enhance MSCs’ therapeutic potential and standardize the production of MSC-derived products intended for clinical use [11]. Therefore, the utilization of secretomes could contribute to developing a novel, cell-free therapeutic approach.
The secretome is enriched with extracellular vesicles (EVs), secreted by cells through the development of multivesicular bodies or by cell membrane shedding [12][13][12,13]. They are classified into different types, based on their size, mechanism of biogenesis, functions, and tissue of origin, into: exosomes, microvesicles, ectosomes, and oncosomes [14]. Importantly, the cells (including the perinatal MSCs) are able to release various types of EVs [15]. EVs are present in almost every type of body fluid (e.g., plasma, urine, saliva, amniotic fluid, and others) and are therefore easily accessible. Once in the extracellular space, EVs facilitate cell-to-cell communication, between neighboring and distant cells, through transferring their molecular cargo (enriched in proteins, RNAs, and DNAs), both in physiological and pathological conditions [16][17][18][16,17,18]. The ISEV (International Society for Extracellular Vesicles) suggested using “extracellular vesicle” as the common term for all the vesicles released from cells to avert uncertainty within this complex field [19].
The increasing interest in the therapeutic benefits obtained by using the EVs released by parental MSCs in various diseases reinforces the hypothesis of the usefulness of MSC secretomes as a new therapeutic strategy.
2. Properties of Human Perinatal Tissue and Their Possible Therapeutic Application as Sources of EVs
Perinatal tissues represent a plentiful source of promising cell types. Among the organs that may be used as a source of perinatal cells, the placenta is of key importance for its role in fetal nutrition and development, acting as a functional interface between the mother and fetus, either via the exchange of nutrients or via its immune and endocrine roles [20]. The formation of the placenta is the result of a series of processes that start in the moment of embryo implantation at the end of the first week of development. Then, the organ assists in the development of the embryo and then the fetus until delivery [21]. At delivery, the placenta features a diameter of 20–22 cm, with a thickness of up to 2.5 cm [22]. Structurally, the human placenta is a highly specialized essential organ with a distinction between the maternal-uterine part and the fetal part. Research related to perinatal MSCs began over a decade ago and has grown exponentially with the isolation and characterization of cells from different perinatal tissues [23][24][25][23,24,25] (Figure 1).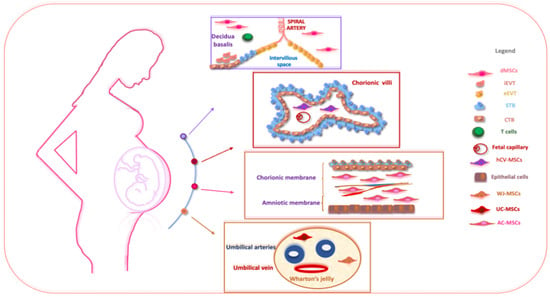
Figure 1. The schematic structure of the placenta and a description of perinatal MSCs. The placenta is an organ in which interactions between maternal and fetal cells, necessary for the development of the fetus, take place. Specifically, the placenta is made up of structures of fetal origin, such as the placental disc, the fetal membranes, divided into amniotic and chorionic membranes, and the umbilical cord, as well as a membrane of maternal origin called the decidua which originates from the endometrium. The chorionic villosity that forms the boundary between maternal and fetal blood during pregnancy represents the functional unit of the placenta. The different structures of the placenta are enriched by MSCs particularly useful in regenerative medicine and for the treatment of various pathologies. Abbreviations: dMSCs: decidua mesenchymal stromal cells; iEVT: inner extravillous trophoblast; eEVT: external extravillous trophoblast; STB: syncytiotrophoblast; CTB: cytotrophoblast; hCV-MSCs: human chorionic villi mesenchymal stromal cells; WJ-MSCs: Wharton’s jelly mesenchymal stromal cells; UC-MSCs: umbilical cord mesenchymal stromal cells; AC-MSCs: amnio-chorion mesenchymal stromal cells.
