Nanotechnology is one of the most active research fields in materials science. Metal-organic frameworks (MOFs) have the benefits of having a sizable specific surface area, extremely high porosity, changeable pore size, post-synthesis modification, and extreme thermal stability. Graphene oxide (GO) has attracted significant research interest due to its similar surface area to MOFs. Furthermore, oxygen-containing groups presented in graphene oxide offer the unique processing and handling advantages of amphiphilicity and dispersion in water. MOF-based GO has recently attracted attention due to its resemblance to metal ions and organic binding linkers. It has sparked great interest in the past few years due to its distinct characteristics and higher performance compared to MOFs or GO alone.
- metal-organic frameworks (MOFs)
- nanomaterial
- photocatalysis
- electrocatalysis adsorption
1. Introduction
2. The Properties of Metal-Organic Frameworks/Graphene Oxide (MOFs/GO)
The combination concept of GO material and MOF substrates was suggested to address the respective drawbacks of GO and MOFs. The composite membrane has a bifunctional characteristic that allows it to interact with metal ions in MOFs due to the presence of epoxy and hydroxyl functional groups on either side of the GO lamellae [51][7]. Using 2-methylimidazole as a reasonably priced ligand and water as a solvent, Hong et al. [52][8] successfully synthesized Ni-Co-MOF/GO at a low temperature. Graphene oxide, which has a two-dimensional (2D) structure, and MOF, which has a three-dimensional (3D) structure with hierarchical pores and high stability, synergistically work together to improve properties such as the large internal surface area. At a current density of 1 A g−1, the specific capacities of Ni-Co-MOF and Ni-Co-MOF/GO were 120.0 and 230.9 F g−1, respectively. The addition of graphene oxide to Ni-Co-MOF resulted in the Ni-Co-MOF/GO electrode having excellent electrochemical properties. In a different work, Kumar et al. [53][9] used a grinding approach to synthesize MOF-5@GO nanocomposites in a single-pot process. Due to its instability in water, the MOF-5 has a distinctive issue, and the combination of the MOF-5 and GO is chiefly required to address its flaws. Based on this, the interactions between GO and MOF-5 composites improve the MOF-5 framework’s hydrophobic stability. Consequently, the MOF-5@GO nanocomposite enables interactions between oxygenated functional groups of GO and open sites of the zinc cluster of MOF-5. As a result, the maintenance of crystallinity boosts MOF-5’s conductivity and improves its adsorption characteristics [53][9]. Using a hydrothermal technique, Heu et al. [55][10] generated photocatalytic nanofiltration (NF) membranes with improved flux and antifouling capabilities from a layered in situ nanocomposite of MOFs (UiO-66) and GO (UiO-66_GO) over a polyamide NF membrane. In situ growth of UiO-66 particles on a (2D) GO sheet revealed that the GO oxygen functional groups enhanced UiO-66 formation, while raising the dispersion force between UiO-66 particles and stifling their aggregation, enabling the control of their physicochemical characteristics, such as morphology, size, and structure. By making the membrane surface more hydrophilic and smoother, the UiO-66_GO nanocomposite at a loading of 15 wt% increased pure water flux by 187% compared to the pristine NF membrane flux. However, due to its high flux improvement (169%) and the UiO-66_GO nanocomposite’s good stability on the membrane surface, the loading of 10 wt% of the UiO-66_GO composite was ideal.3. Synthesis Methods of Metal-Organic Frameworks/Graphene Oxide (MOFs/GO)
3.1. Post-Synthesis Method
The first method for modifying MOFs is post-synthetic modification (PSM), which involves grafting organic functionalities and crosslinkers onto the surface of the particles of MOFs to produce functionalization materials [59][11]. This technique was used to prepare MOFs/GO composites. It entails preparing the MOFs beforehand and adding them to the GO, as shown in Figure 51 [60][12].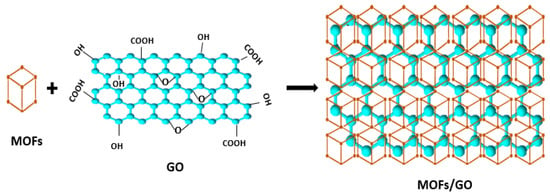
3.2. Hydrothermal and Solvothermal Methods
Two of the most widely applied in situ techniques are the hydrothermal and solvothermal techniques. They are referred to as synthesis methods, where the reaction occurs at pressures greater than 1 bar and temperatures higher than the solvent boiling point. The variables support the development of crystalline products [63][14]. The only difference between the two methods in the synthetic pathways is the kind of solvent employed. It can either be aqueous (hydrothermal) or nonaqueous (solvothermal). Moreover, the researchers prefer this method due to its simplicity, quick processing time, and high composite yield [64][15]. The advancement of renewable energy systems is an intriguing possibility for electrochemical water splitting. Additionally, to reduce their overpotentials and speed up their practical application, it is strongly suggested to fabricate suitable electrocatalysts for water splitting. Ahmed Malik et al. [65][16] effectively produced Ce-MOF and GO@CeMOF using solvothermal techniques, and CeO2 from Ce-MOF and GO@CeMOF using calcination. The methods necessary to fabricate the GO@Ce-MOF utilizing the solvothermal approach are illustrated in Scheme 2Scheme 1. The oxygen evolution reaction (OER) was studied as a potential electrocatalyst using the preceding catalysts, with the lowest overpotentials, tiny Tafel slope, minimum impedance, and highest peak current density, where the electrocatalysts displayed exceptional results. The electrocatalytic activity of the calcined samples was more effective when compared to that of simple MOFs and their composites.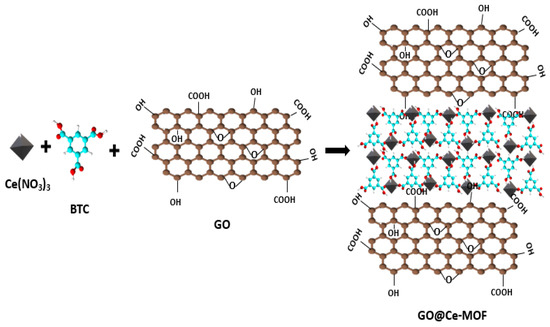
3.3. In Situ Method
3.4. Co-Precipitation Method
In recent years, solution precipitation has been a widely used method for producing inorganic nanomaterials. The “co-precipitation method” is another name for this process because the cations simultaneously precipitate. The co-precipitation technique is easier to control, uses milder experimental conditions than hydrothermal reactions, is simple, and is faster for MOFs’ production and their composites [72][20]. Xu et al. (79) prepared Co-ZIF-adsorbed borate ions with functionalized rGO (ZIF-67/rGO-B), conducive to lowering the fire risk of epoxy resin (EP). Firstly, ZIF-67 was loaded onto the surface of graphene using the co-precipitation method. The authors discovered that it decreased the fire danger of polymer materials and the agglomeration of rGO in composites. After that, boron ions were successfully adsorbed onto the ZIF-67 surface to produce a brand-new hybrid, known as ZIF-67/rGO-B. Moreover, ZIF-67/rGO-B was physically blended into EP, and then the fire risk of EP composites was examined using cone calorimeter tests, the limited oxygen index (LOI), and other methods. According to the combustion results, EP composites produced significantly less heat and smoke. Exceptionally, when compared to pure EP, the peak heat release rate (pHRR), total heat release (THR), and maximum value of smoke density (Ds,max) of the composite with 2 wt% ZIF-67/RGO-B were all decreased, by 65.1%, 41.1%, and 66.0%, respectively [73][21].3.5. Mixing Method
Physical mixing is a quick and practical technique for synthesizing MOFs/GO nanocomposites. To produce the composite, MOFs and GO are first prepared ahead of time and then directly combined [75][22]. To improve the electrochemical anodic performance of the parent pure Ni-based MOF (Ni-MOF), Haroon et al. [76][23] prepared the Ni-MOF@rGO composite using low temperatures, linked to a representative Ni-MOF with rGO via an intermediary physical mixing method. This method was exceptional because it produced close contacts between the Ni-MOF and thermally regenerated rGO, while maintaining the MOFs’ anonymity at low temperatures (300 °C). With a capacity of 385 mAhg−1 (100 mAg−1), which preserved its consistency for 400 charge–discharge cycles, the Ni-MOF-rGO composite, or Ni-BDC/rGO@300, had a significantly improved rate and cycling stability as compared to the pristine MOFs, which degraded to 272 mAg−1 in just 250 cycles. The Ni-BDC/rGO@300 composite displayed a capacity of 205 mAhg−1 at 1 Ag−1, while the pristine Ni-MOF capacity was 113 mAhg−1, demonstrating significant rate improvements at higher currents. Scheme 3Scheme 2 illustrates the general synthetic process [76][23].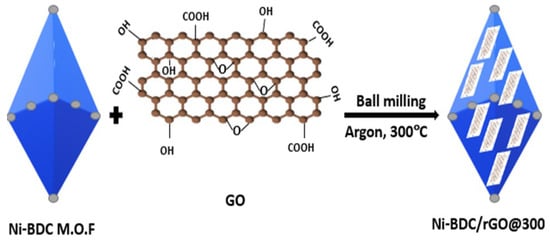
3.6. Ultrasonication Method
3.6. Ultrasonication Method
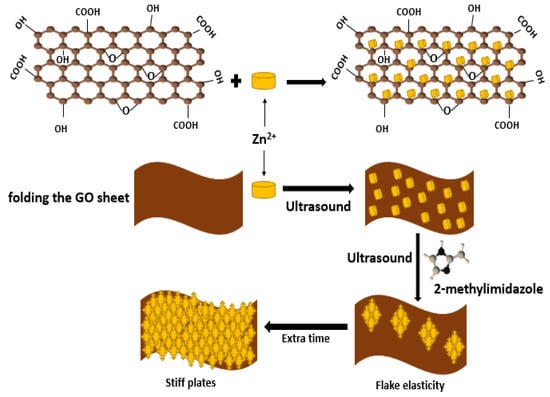
4. Applications of Metal-Organic Frameworks/Graphene Oxide (MOFs/GO)
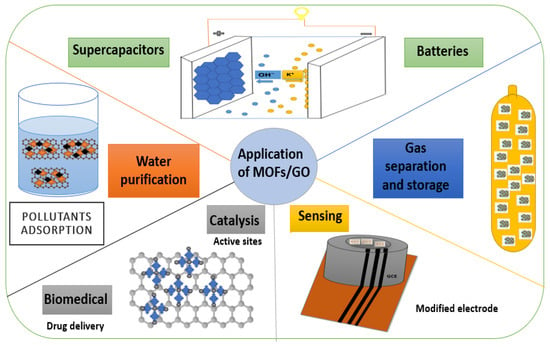
4.1. Supercapacitors
4.2. Gas Separation and Storage
Composite materials based on MOFs are observed to be a promising class of materials because they have the potential to combine the benefits of MOFs and other elements. Additionally, the disadvantages of each component are individually minimized in adsorption and separation systems [103][34]. MOFs are the dominant component of MOFs/GO composites, which produce admixtures rather than composites with covalent bonds. By using the GO substrate as a ruling component and support, nanoscale MOFs will be chemically bonded over the top of the platform [104][35]. MOF materials offer a lot of benefits for CO2 capture. Al (HCOO)3 (ALF), a form of aluminum, is also among the most basic MOFs. To address its cost and scalability, Evans et al. [105][36] produced ALF characteristics from readily available and affordable starting materials, such as formic acid and aluminum hydroxide, as mechanical materials to examine the adsorption [105][36]. To increase the capabilities of MOFs for the adsorption of CO2, Zhao et al. [106][37] produced a composite material of L-arginine-modified MOFs with GO (MOFs/GO-Arg). The bridging of L-arginine-modified GO with the linker MOF (Cu-BTC) was chosen due to its stability, simplicity of synthesis, and affordability. By adjusting the pore shape and chemical environment at the interface between MOFs and GO, the performance of CO2 adsorption can improve.4.3. Water Purification
To effectively remove different pollutants from polluted water and wastewater streams, MOF materials, a hybrid class of substances with a metallic center and organic linkers, are incredibly effective due to their porous nature [108][38]. Pharmaceuticals, particularly antibiotics, are removed from contaminated water using MOF-based adsorbents and catalysts [109][39]. Sewage treatment employs a variety of methods to remove environmental pollutants. Separation, absorption, photocatalytic degradation, and membrane filtration are the typical techniques [110][40]. The development of GO/MOFs, which have higher hydrophilicity, fouling resistance, and selectivity, has sparked interest in the water industry [111][41]. Textile wastewater is one of the most challenging for wastewater treatment due to the highly contaminating solid pollutants, such as dyes. In another study, Jafarian et al. [112][42] synthesized a novel NF membrane to remove DIRECT RED 16 (DR16) dyes and humic acid from synthetic wastewater. To form this membrane, they deposited a thin layer of GO and a Zn-based metal-organic framework (ZIF-7) on a chitosan (CTS)-coated polyethersulfone (PES) substrate. The surface membrane was made rougher and more hydrophilic by adding GO-ZIF-7. According to the data, the 5GO-ZIF membrane (~94%) had the highest dye clearance rate. Additionally, the highly hydrophilic surface of the GO-ZIF layer and the biocidal activities of GO and zinc introduced by the GO-ZIF-7 nanocomposite improved the antifouling and anti-biofouling capabilities of the membrane. It was observed that overlaying the CTS membrane with the 5GO-ZIF nanocomposite layer lowered the pure water flux (11.4%), which may be a drawback for actual industrial applications. The increase in mass transfer resistance caused by the addition of layers comprising the GO-ZIF nanocomposite can be used to explain the decrease in pure water flux [112][42].4.4. Sensors
MOFs are combined with some conducting materials, such as carbon and conducting polymers, to produce materials for electrochemical sensors with good analytical performance. Graphene is an example of a well-known 2D substance that has gained interest due to its excellent electrical conductivity and surface area. By contrast, MOFs and graphene produced new physical and chemical properties [114][43]. Due to their substantial specific surface area, high electrical conductivity, and superior chemical stability, graphene and its analogs are widely employed to develop electrochemical sensors. The MOF conductivity effectively increased by combining graphene and its analogs with MOFs [115][44]. Environmental monitoring would greatly benefit from constructing a sensitive voltametric platform to examine the excessively toxic organic pollutant p-chloronitrobenzene (p-CNB). A composite made of nickel-based MOF (Ni-MOF) and GO was successfully synthesized by Gao et al. [116][45] using simple and affordable chemical precipitation. The Ni-MOF/GO composite was modified to produce an electrochemical sensor for p-CNB. With the help of electrochemical impedance spectroscopy, distinguishing pulse voltammetry and cyclic voltammetry were used to examine the electrochemical characteristics of as-prepared sensors. Thus, the detection limit of the composite sensor was 8.0 nM (S/N = 3), and it demonstrated superior electrocatalytic performance towards the oxidation of p-CNB in the concentration range of 0.10–300.0 μM. Subsequent research revealed the production sensing platform to have good long-lasting stability, strong selectivity, and reproducibility [116][45].4.5. Catalysis
In many diverse industries, particularly the chemical sector, catalysts (homogeneous and heterogeneous) play a significant role. The industrial use of homogeneous catalysts is restricted because they frequently have a higher catalytic activity and are challenging to recover from the reaction solution. Contrarily, heterogeneous catalysts (supported catalysts) are easily removed from the reaction solution and repeatedly used. These catalysts have received significant attention when used in different reaction systems [118,119][46][47]. Catalytic processes are necessary for over 90% of chemical industrial processes and over 20% of all chemical products. Catalyst production is a crucial process that requires extensive research to produce the high-performance catalysts currently in use [120][48]. The MOFs-GO have become a new class of catalytic composite materials in the nanoarchitecture field because of their high surface area, unique electrical properties, magnificent conductivity, and hydrophilic nature. Nano-structuring porous catalysts would be a crucial way to expose accessible active sites to achieve high catalytic activity [121][49]. MOFs-GO is a hybrid material that combines the distinct benefits of MOFs and GO and is ideal for immobilizing nanoparticles (NPs) [122][50].4.5.1. Electrocatalysts
The study of environmentally friendly energy has received significant attention from researchers due to an evident trend of rising energy consumption. The hydrogen evolution reaction (HER), the oxygen evolution reaction (OER), and the oxygen reduction reaction (ORR) are consequential electrochemical reactions used in fuel cells and batteries for energy storage. Graphene-based materials are frequently used in composite MOFs to create efficient electrocatalysts because of their low cost and high carrier conductivity [125,126,127][51][52][53]. Gopi and colleagues [128][54] produced the catalyst V-Ni 0.06 Fe0.06 MOF/GO using an in situ technique. They performed excellent bifunctional electrocatalysis for the hydrogen evolution reaction (HER) and the anodic oxygen evolution reaction (OER) and showed high durability in both acidic and alkaline mediums. The choice of V, Ni, and Fe redox metal nodes in combination with a highly porous MOF/GO composite for the water splitting reaction is motivated by the abundance of free carriers at the Fermi level of the V atom and the more active edge sites of the Fe atom. Moreover, the MOF-to-graphene MOF conducting mechanism was improved by 2D-graphene-combined MOF composites.4.5.2. Photocatalysts
According to researchers, MOFs are highly efficient in the photocatalytic degradation of pollutants and organic dyes when used as catalysts in photocatalytic processes. The retrieval and separation of photocatalyst MOFs from the reaction mixture can be facilitated and improved through modifications. Thus, the performance of photocatalysis can be enhanced by GO [130,131][55][56]. To improve the catalytic performance of MOFs, Jin et al. [132][57] intensively hybridized them with GO; however, it is still unclear how the pore structure of MOFs affects the activity of GO/MOF hybrids. To investigate the connection between the pore structure of MIL-125 and the activity of the hybrid, they created a variety of GO/MIL-125 hybrids using the sonication process. The obtained GO/MIL-125(H) has an electron mediator, a significant surface area, displays an assistant photothermal effect, and has hierarchical pores that aid in the adsorption and photodegradation of toluene. The photocatalytic activities of UiO-66 and GO (UiO-66_GO) nanocomposites for the degradation of carbamazepine (CBZ) were examined in a different study by Heu et al. [133][58] using a one-step hydrothermal method. Under a range of circumstances, including GO loading, catalyst dosage, initial pollutant concentration, and solution pH, the nanocomposite for the degradation of CBZ was investigated. Increased surface area and porosity, a smaller bandgap, and better light absorption are all factors that were found to be responsible for GO’s enhanced photocatalytic activity. Additionally, the composite demonstrated notable stability and recyclability over five successive cycles of photocatalytic degradation. Modifying semiconductors with GO as an electron acceptor is a successful method for enhancing photocatalytic activity. Additionally, it serves as a great source of inspiration for the growth of additional GO-based composite photocatalysts and the use of UiO-66 in water or wastewater treatment methods. The UiO-66_GO nanocomposites demonstrated a high overall removal efficiency (>90%) in 2 h and a photocatalytic rate constant of up to 0.0136 min−1. The results of the experiments supported the idea of a photocatalytic mechanism for enhanced CBZ photodegradation by showing that O2• and OH• are the responsible radicals for photocatalytic degradation. Further, using this composite material as a solar-base catalyst is possible due to its ability to absorb light in the visible light spectrum (400–700 nm) [133][58].4.6. Batteries
Fuel cells and large-capacity, inexpensive, and sustainable rechargeable batteries have drawn a lot of attention due to the new energy industry’s rapid development [134][59]. Batteries are known to store relatively large amounts of energy compared to supercapacitors but have relatively low power delivery or uptake, a short life cycle, and thermal management issues [135][60]. Due to their unique properties of the adaptable structure, high porosity, and abundance of active sites, MOFs have received extensive research as a typical inorganic–organic nanomaterial in the growth of battery electrodes [136][61]. Li et al. [140][62] successfully produced Ni(OH)2-GO electrode materials using a two-step synthesis process. The numerous functional groups on GO nanosheets aid in the nucleation and growth of Ni-MOFs. The subsequent hydrolysis of Ni-MOF makes it possible to successfully prepare Ni(OH)2-GO for its promising use in supercapacitors. Within Ni(OH)2-GO, the GO content decreases to 7.41 wt%. The electron transfer between Ni(OH)2 and GO readily occurs during the electrochemical reaction, drastically increasing the electrochemical activity of Ni(OH)2.4.7. Other Applications (Biomedical)
Numerous MOFs and MOF-derived nanomaterials have been developed and used in biomedicine for antibacterial mechanisms [142][63]. In different studies, MOF composites have also been examined for bio-applications such as drug delivery, bio-imaging, and cancer treatment [143][64]. There is not a thorough review report available that explains how to functionalize GO with MOFs for biomedical applications or how to use natural chemotherapeutic agents for cancer therapeutics [144][65]. The amount of GO’s active surface available for interacting with bioactive molecules was constrained by its intense agglomeration. Combining it with other NPs, such as MOFs, is an appealing strategy for overcoming this restriction and enhancing GO’s efficiency in the biomedical sector [145][66]. To compare their effectiveness as a vehicle for the anticancer drug carriers 5-Fu, Pooresmaeil and colleagues [146][67] created three different types of chitosan-based microspheres: CS, chitosan-coated zinc-based MOF (CS/Zn-MOF), and a chitosan-coated hybrid of ZnMOF with GO (CS/Zn-MOF@GO). The ternary of hybrid CS/Zn-MOF@GO microspheres was found to have the highest amount of 5-fluorouracil (5-Fu) loading, at about 45%. The rough-surfaced, 5-Fu-loaded CS microspheres (5-Fu@CS/Zn-MOF@GO microspheres) displayed a pH-sensitive and sustained release pattern for the 5-Fu that was loaded. Therefore, the total amount of drug released over time at pH 5.0 was roughly twice that at pH 7.4. Ultimately, CS/Zn-MOF@GO microspheres demonstrated acceptable enzymatic biodegradability and good biocompatibility with the epithelial human breast cancer cell line MDA-MB 231. The ability of 5-Fu-loaded CS/Zn-MOF@GO microspheres to treat tumor cells was demonstrated by the cell viability of 41.2% following 48 treatments with 5-Fu@CS/Zn-MOF@GO microspheres [146][67].5. The Advantages and Challenges of Metal-Organic Frameworks/Graphene Oxide (MOFs/GO)
There are many applications for MOFs and materials based on graphene, especially in electrochemistry. The presence of GO during the production of MOFs has several benefits, which include the capacity to tune the particle size, quicker electron mobility, and chemical and thermal stability. The best performance under specific environmental conditions (pH, temperature, humidity, etc.) is necessary for MOF and graphene-based composites to operate in harsh environments, thus constraining their ability to develop. In this regard, MOF/graphene-based materials still have room for improvement [148][68]. Additionally, the combination of GO and MOFs results in the creation of tiny holes, which leads to an increase in the dispersive strength of the MOFs, suppressed MOF aggregation, strong specific adsorption, and a high rate of CO2 storage [149][69]. GO can resolve the weak coordination bonds between metal nodes and organic ligands, guide MOF development, and lessen poor conductivity [150][70]. During the production of MOF derivatives, the merge of graphene-based materials can stop high-temperature etching, break down the MOF structure, and perform other processes that would otherwise lower the specific surface area and active sites of MOFs [151][71]. The composite MOFs/GO have hierarchical pore structures that provide an ideal space for oxygen atoms in GO, enhancing its stability [152][72]. GO combined with MOFs decreases toxicity and exhibits exceptional electrochemical, mechanical, thermal, and electrical properties. Due to the diversity of MOF compounds and complex MOF–GO interactions, MOFs’ growth and structure orientations are still challenging to predict [153][73].References
- Gautam, S.; Agrawal, H.; Thakur, M.; Akbari, A.; Sharda, H.; Kaur, R.; Amini, M. Metal oxides and metal organic frameworks for the photocatalytic degradation: A review. J. Environ. Chem. Eng. 2020, 8, 103726.
- Čitaković, N.M. Physical properties of nanomaterials. Vojnoteh. Glas. Mil. Tech. Cour. 2019, 67, 159–171.
- Zakrzewski, W.; Dobrzyński, M.; Zawadzka-Knefel, A.; Lubojański, A.; Dobrzyński, W.; Janecki, M.; Kurek, K.; Szymonowicz, M.; Wiglusz, R.J.; Rybak, Z. Nanomaterials application in endodontics. Materials 2021, 14, 5296.
- Zhao, D.; Zhang, W.; Wu, Z.H.; Xu, H. Nanoscale metal− organic frameworks and their nanomedicine applications. Front. Chem. 2022, 9, 1243.
- Sajid, M. Nanomaterials: Types, properties, recent advances, and toxicity concerns. Curr. Opin. Environ. Sci. Health 2022, 25, 100319.
- Naghdi, S.; Shahrestani, M.M.; Zendehbad, M.; Djahaniani, H.; Kazemian, H.; Eder, D. Recent advances in application of metal-organic frameworks (MOFs) as adsorbent and catalyst in removal of persistent organic pollutants (POPs). J. Hazard. Mater. 2023, 442, 130127.
- Qu, H.J.; Huang, L.J.; Han, Z.Y.; Wang, Y.X.; Zhang, Z.J.; Wang, Y.; Chang, Q.R.; Wei, N.; Kipper, M.J.; Tang, J.G. A review of graphene-oxide/metal–organic framework composites materials: Characteristics, preparation and applications. J. Porous Mater. 2021, 28, 1837–1865.
- Hong, J.; Park, S.J.; Kim, S. Synthesis and electrochemical characterization of nanostructured Ni-Co-MOF/graphene oxide composites as capacitor electrodes. Electrochim. Acta 2019, 311, 62–71.
- Kumar, G.; Masram, D.T. Sustainable synthesis of MOF-5@ GO nanocomposites for efficient removal of rhodamine B from water. ACS Omega 2021, 6, 9587–9599.
- Heu, R.; Ateia, M.; Yoshimura, C. Photocatalytic nanofiltration membrane using Zr-MOF/GO nanocomposite with high-flux and anti-fouling properties. Catalysts 2020, 10, 711.
- Muthukumaraswamy Rangaraj, V.; Wahab, M.A.; Reddy, K.S.; Kakosimos, G.; Abdalla, O.; Favvas, E.P.; Reinalda, D.; Geuzebroek, F.; Abdala, A.; Karanikolos, G.N. Metal organic framework—based mixed matrix membranes for carbon dioxide separation: Recent advances and future directions. Front. Chem. 2020, 8, 534.
- Haeri, Z.; Ramezanzadeh, B.; Ramezanzadeh, M. Recent progress on the metal-organic frameworks decorated graphene oxide (MOFs-GO) nano-building application for epoxy coating mechanical-thermal/flame-retardant and anti-corrosion features improvement. Prog. Org. Coat. 2022, 163, 106645.
- Karamipour, M.; Fathi, S.; Safari, M. Removal of phenol from aqueous solution using MOF/GO: Synthesis, characteristic, adsorption performance and mechanism. Int. J. Environ. Anal. Chem. 2021, 6, 1–2.
- Mokhtar, N.A.; Zawawi, R.M.; Khairul, W.M.; Yusof, N.A. Electrochemical and optical sensors made of composites of metal–organic frameworks and carbon-based materials. A review. Environ. Chem. Lett. 2022, 20, 3099–3131.
- Nandihalli, N.; Gregory, D.H.; Mori, T. Energy-Saving Pathways for Thermoelectric Nanomaterial Synthesis: Hydrother-mal/Solvothermal, Microwave-Assisted, Solution-Based, and Powder Processing. Adv. Sci. 2022, 9, 2106052.
- Ahmed Malik, W.M.; Afaq, S.; Mahmood, A.; Niu, L.; Yousaf ur Rehman, M.; Ibrahim, M.; Mohyuddin, A.; Qureshi, A.M.; Ashiq, M.N.; Chughtai, A.H. A facile synthesis of CeO2 from the GO@ Ce-MOF precursor and its efficient performance in the oxygen evolution reaction. Front. Chem. 2022, 10, 996560.
- Fallatah, A.M.; Shah, H.U.; Ahmad, K.; Ashfaq, M.; Rauf, A.; Muneer, M.; Ibrahim, M.M.; El-Bahy, Z.M.; Shahzad, A.; Babras, A. Rational synthesis and characterization of highly water stable MOF@ GO composite for efficient removal of mercury (Hg2+) from water. Heliyon 2022, 8, e10936.
- Liu, K.G.; Sharifzadeh, Z.; Rouhani, F.; Ghorbanloo, M.; Morsali, A. Metal-organic framework composites as green/sustainable catalysts. Coord. Chem. Rev. 2021, 436, 213827.
- Li, H.K.; Ye, H.L.; Zhao, X.X.; Sun, X.L.; Zhu, Q.Q.; Han, Z.Y.; Yuan, R.; He, H. Artful union of a zirconium-porphyrin MOF/GO composite for fabricating an aptamer-based electrochemical sensor with superb detecting performance. Chin. Chem. Lett. 2021, 32, 2851–2855.
- Zaman, N.; Iqbal, N.; Noor, T. Advances and challenges of MOF derived carbon-based electrocatalysts and photocatalyst for water splitting: A review. Arab. J. Chem. 2022, 14, 103906.
- Xu, W.; Wang, X.; Wu, Y.; Li, W.; Chen, C. Functionalized graphene with Co-ZIF adsorbed borate ions as an effective flame retardant and smoke suppression agent for epoxy resin. J. Hazard. Mater. 2019, 363, 138–151.
- Wang, K.; Hui, K.N.; San Hui, K.; Peng, S.; Xu, Y. Recent progress in metal–organic framework/graphene-derived materials for energy storage and conversion: Design, preparation, and application. Chem. Sci. 2021, 12, 5737–5766.
- Haroon, H.; Wahid, M.; Majid, K. Structure-Activity Relationships of a Ni-MOF, a Ni-MOF-rGO, and pyrolyzed Ni/C@rGO Structures for Sodium-ion Batteries. ChemistrySelect 2022, 7, e202202011.
- Arul, P.; Gowthaman, N.S.; John, S.A.; Lim, H.N. Ultrasonic assisted synthesis of size-controlled Cu-metal–organic framework decorated graphene oxide composite: Sustainable electrocatalyst for the trace-level determination of nitrite in environmental water samples. ACS Omega 2020, 5, 14242–14253.
- Kasula, M.; Le, T.; Thomsen, A.; Esfahani, M.R. Silver metal organic frameworks and copper metal organic frameworks immobilized on graphene oxide for enhanced adsorption in water treatment. Chem. Eng. J. 2022, 439, 135542.
- Cui, H.; Cui, S.; Tian, Q.; Zhang, S.; Wang, M.; Zhang, P.; Liu, Y.; Zhang, J.; Li, X. Electrochemical Sensor for the Detection of 1-Hydroxypyrene Based on Composites of PAMAM-Regulated Chromium-Centered Metal–Organic Framework Nanoparticles and Graphene Oxide. ACS Omega 2021, 6, 31184–31195.
- Yang, K.; Dai, Y.; Zheng, W.; Ruan, X.; Li, H.; He, G. ZIFs-modified GO plates for enhanced CO2 separation performance of ethyl cellulose based mixed matrix membranesf. Sep. Purif. Technol. 2019, 214, 87–94.
- Zhu, L.; Meng, L.; Shi, J.; Li, J.; Zhang, X.; Feng, M. Metal-organic frameworks/carbon-based materials for environmental remediation: A state-of-the-art mini-review. J. Environ. Manag. 2019, 232, 964–977.
- Yang, W.; Li, X.; Li, Y.; Zhu, R.; Pang, H. Applications of metal–organic-framework-derived carbon materials. Adv. Mater. 2019, 31, 1804740.
- Wang, D.G.; Liang, Z.; Gao, S.; Qu, C.; Zou, R. Metal-organic framework-based materials for hybrid supercapacitor application. Coord. Chem. Rev. 2020, 404, 213093.
- Huang, S.; Shi, X.R.; Sun, C.; Duan, Z.; Ma, P.; Xu, S. The application of metal–organic frameworks and their derivatives for supercapacitors. Nanomaterials 2020, 10, 2268.
- Li, S.; Shi, C.; Pan, Y.; Wang, Y. 2D/2D NiCo-MOFs/GO hybrid nanosheets for high-performance asymmetrical supercapacitor. Diam. Relat. Mater. 2021, 115, 108358.
- Chen, T.; Yang, A.; Zhang, W.; Nie, J.; Wang, T.; Gong, J.; Wang, Y.; Ji, Y. Architecting Nanostructured Co-BTC@ GO Composites for Supercapacitor Electrode Application. Nanomaterials 2022, 12, 3234.
- Roohollahi, H.; Zeinalzadeh, H.; Kazemian, H. Recent Advances in Adsorption and Separation of Methane and Carbon Dioxide Greenhouse Gases Using Metal–Organic Framework-Based Composites. Ind. Eng. Chem. Res. 2022, 61, 10555–10586.
- Ventura, K.; Arrieta, R.A.; Marcos-Hernández, M.; Jabbari, V.; Powell, C.D.; Turley, R.; Lounsbury, A.W.; Zimmerman, J.B.; Gardea-Torresdey, J.; Wong, M.S.; et al. Superparamagnetic MOF@ GO Ni and Co based hybrid nanocomposites as efficient water pollutant adsorbents. Sci. Total Environ. 2020, 738, 139213.
- Evans, H.A.; Mullangi, D.; Deng, Z.; Wang, Y.; Peh, S.B.; Wei, F.; Wang, J.; Brown, C.M.; Zhao, D.; Canepa, P.; et al. Aluminum formate, Al (HCOO)3: An earth-abundant, scalable, and highly selective material for CO2 capture. Sci. Adv. 2022, 8, eade1473.
- Zhao, Y.; Ge, H.; Miao, Y.; Chen, J.; Cai, W. CO2 capture ability of Cu-based metal-organic frameworks synergized with amino acid-functionalized layered materials. Catal. Today 2020, 356, 604–612.
- Hooriabad Saboor, F.; Nasirpour, N.; Shahsavari, S.; Kazemian, H. The effectiveness of MOFs for the removal of pharmaceuticals from aquatic environments: A review focused on antibiotics removal. Chem. Asian J. 2022, 17, e202101105.
- Chen, J.Q.; Sharifzadeh, Z.; Bigdeli, F.; Gholizadeh, S.; Li, Z.; Hu, M.L.; Morsali, A. MOF Composites as high Potential Materials for Hazardous Organic Contaminants Removal in Aqueous Environments. J. Environ. Chem. Eng. 2023, 11, 109469.
- Yang, F.; Du, M.; Yin, K.; Qiu, Z.; Zhao, J.; Liu, C.; Zhang, G.; Gao, Y.; Pang, H. Applications of Metal-Organic Frameworks in Water Treatment: A Review. Small 2022, 18, 2105715.
- Jun, B.M.; Al-Hamadani, Y.A.; Son, A.; Park, C.M.; Jang, M.; Jang, A.; Kim, N.C.; Yoon, Y. Applications of metal-organic framework based membranes in water purification: A review. Sep. Purif. Technol. 2020, 247, 116947.
- Jafarian, H.; Firouzjaei, M.D.; Aktij, S.A.; Aghaei, A.; Khomami, M.P.; Elliott, M.; Wujcik, E.K.; Sadrzadeh, M.; Rahimpour, A. Synthesis of heterogeneous metal organic Framework-Graphene oxide nanocomposite membranes for water treatment. Chem. Eng. J. 2023, 455, 140851.
- Venkadesh, A.; Mathiyarasu, J.; Radhakrishnan, S. Voltammetric Sensing of Caffeine in Food Sample Using Cu-MOF and Graphene. Electroanalysis 2021, 33, 1007–1013.
- Chang, Y.; Lou, J.; Yang, L.; Liu, M.; Xia, N.; Liu, L. Design and Application of Electrochemical Sensors with Metal–Organic Frameworks as the Electrode Materials or Signal Tags. Nanomaterials 2022, 12, 3248.
- Gao, J.; He, P.; Yang, T.; Wang, X.; Zhou, L.; He, Q.; Jia, L.; Deng, H.; Zhang, H.; Jia, B.; et al. Short rod-like Ni-MOF anchored on graphene oxide nanosheets: A promising voltammetric platform for highly sensitive determination of p-chloronitrobenzene. J. Electroanal. Chem. 2020, 861, 113954.
- Zhou, M.; Liu, M.; Jiang, H.; Chen, R. Controllable synthesis of Pd-ZIF-L-GO: The role of drying temperature. Ind. Eng. Chem. Res. 2021, 60, 4847–4859.
- Miceli, M.; Frontera, P.; Macario, A.; Malara, A. Recovery/reuse of heterogeneous supported spent catalysts. Catalysts 2021, 11, 591.
- Andrade, M.A.; Martins, L.M. Sustainability in catalytic cyclohexane oxidation: The contribution of porous support materials. Catalysts 2019, 10, 2.
- Alamgholiloo, H.; Rostamnia, S.; Zhang, K.; Lee, T.H.; Lee, Y.S.; Varma, R.S.; Jang, H.W.; Shokouhimehr, M. Boosting aerobic oxidation of alcohols via synergistic effect between TEMPO and a composite Fe3O4/Cu-BDC/GO nanocatalyst. ACS Omega 2020, 5, 5182–5191.
- Su, C.; Wang, B.; Li, S.; Wie, Y.; Wang, Q.; Li, D. Fabrication of Pd@ ZnNi-MOF/GO Nanocomposite and Its Application for H2O2 Detection and Catalytic Degradation of Methylene Blue Dyes. ChemistrySelect 2021, 6, 8480–8489.
- Chen, Y.; Huang, N.; Liang, Y. Preparation of CeO2/Cu-MOF/GO composite for efficient electrocatalytic oxygen evolution reaction. Ionics 2021, 27, 4347–4360.
- Tang, B.; Wang, S.; Li, R.; Gou, X.; Long, J. Urea treated metal organic frameworks-graphene oxide composites derived N-doped Co-based materials as efficient catalyst for enhanced oxygen reduction. J. Power Sources 2019, 425, 76–86.
- Radwan, A.; Jin, H.; He, D.; Mu, S. Design engineering, synthesis protocols, and energy applications of MOF-derived electrocatalysts. Nano-Micro Lett. 2021, 13, 1–32.
- Gopi, S.; Panda, A.; Ramu, A.G.; Theerthagiri, J.; Kim, H.; Yun, K. Bifunctional electrocatalysts for water splitting from a bimetallic (V doped-NixFey) Metal–Organic framework MOF@ Graphene oxide composite. Int. J. Hydrogen Energy 2022, 47, 42122–42135.
- Bagherzadeh, S.B.; Kazemeini, M.; Mahmoodi, N.M. Preparation of novel and highly active magnetic ternary structures (metal-organic framework/cobalt ferrite/graphene oxide) for effective visible-light-driven photocatalytic and photo-Fenton-like degradation of organic contaminants. J. Colloid Interface Sci. 2021, 602, 73–94.
- Qian, Y.; Zhang, F.; Pang, H. A review of MOFs and their composites-based photocatalysts: Synthesis and applications. Adv. Funct. Mater. 2021, 31, 2104231.
- Jin, J.; Kim, J.P.; Wan, S.; Kim, K.H.; Choi, Y.; Li, P.; Kang, J.; Ma, Z.; Lee, J.H.; Kwon, O.; et al. Hierarchical pore enhanced adsorption and photocatalytic performance of graphene oxide/Ti-based metal-organic framework hybrid for toluene removal. Appl. Catal. B Environ. 2022, 317, 121751.
- Heu, R.; Ateia, M.; Awfa, D.; Punyapalakul, P.; Yoshimura, C. Photocatalytic degradation of organic micropollutants in water by Zr-MOF/GO composites. J. Compos. Sci. 2020, 4, 54.
- Xiao, X.; Zou, L.; Pang, H.; Xu, Q. Synthesis of micro/nanoscaled metal–organic frameworks and their direct electrochemical applications. Chem. Soc. Rev. 2020, 49, 301–331.
- Teffu, D.M.; Ramoroka, M.E.; Makhafola, M.D.; Makgopa, K.; Maponya, T.C.; Seerane, O.A.; Hato, M.J.; Iwuoha, E.I.; Modibane, K.D. High-performance supercabattery based on reduced graphene oxide/metal organic framework nanocomposite decorated with palladium nanoparticles. Electrochim. Acta 2022, 412, 140136.
- Zhang, L.; Liu, W.; Shi, W.; Xu, X.; Mao, J.; Li, P.; Ye, C.; Yin, R.; Ye, S.; Liu, X.; et al. Boosting lithium storage properties of MOF derivatives through a wet-spinning assembled fiber strategy. Chem. A Eur. J. 2018, 24, 13792–13799.
- Li, Z.; Ma, Q.; Zhang, H.; Zhang, Q.; Zhang, K.; Mei, H.; Xu, B.; Sun, D. Self-Assembly of Metal–Organic Frameworks on Graphene Oxide Nanosheets and In Situ Conversion into a Nickel Hydroxide/Graphene Oxide Battery-Type Electrode. Inorg. Chem. 2022, 61, 12129–12137.
- Liu, J.; Wu, D.; Zhu, N.; Wu, Y.; Li, G. Antibacterial mechanisms and applications of metal-organic frameworks and their derived nanomaterials. Trends Food Sci. Technol. 2021, 109, 413–434.
- Ge, X.; Wong, R.; Anisa, A.; Ma, S. Recent development of metal-organic framework nanocomposites for biomedical applications. Biomaterials 2022, 281, 121322.
- Sontakke, A.D.; Tiwari, S.; Purkait, M.K. A comprehensive review on graphene oxide-based nanocarriers: Synthesis, functionalization and biomedical applications. FlatChem 2023, 38, 100484.
- Asl, E.A.; Pooresmaeil, M.; Namazi, H. Chitosan coated MOF/GO nanohybrid as a co-anticancer drug delivery vehicle: Synthesis, characterization, and drug delivery application. Mater. Chem. Phys. 2023, 293, 126933.
- Pooresmaeil, M.; Asl, E.A.; Namazi, H. A new pH-sensitive CS/Zn-MOF@ GO ternary hybrid compound as a biofriendly and implantable platform for prolonged 5-Fluorouracil delivery to human breast cancer cells. J. Alloys Compd. 2021, 885, 160992.
- Zhang, X.; Zhang, S.; Tang, Y.; Huang, X.; Pang, H. Recent advances and challenges of metal–organic framework/graphene-based composites. Compos. Part B Eng. 2022, 230, 109532.
- Wang, S.; Ye, B.; An, C.; Wang, J.; Li, Q.; Guo, H.; Zhang, J. Exploring the coordination effect of GO@ MOF-5 as catalyst on thermal decomposition of ammonium perchlorate. Nanoscale Res. Lett. 2019, 14, 1–11.
- Wang, D.; Zhao, Y. Single-atom engineering of metal-organic frameworks toward healthcare. Chem 2021, 7, 2635–2671.
- Li, C.; Ji, Y.; Wang, Y.; Liu, C.; Chen, Z.; Tang, J.; Hong, Y.; Li, X.; Zheng, T.; Jiang, Q.; et al. Applications of Metal–Organic Frameworks and Their Derivatives in Electrochemical CO2 Reduction. Nano-Micro Lett. 2023, 15, 113.
- Zadehahmadi, F.; Eden, N.T.; Mahdavi, H.; Konstas, K.; Mardel, J.I.; Shaibani, M.; Banerjee, P.C.; Hill, M.R. Removal of metals from water using MOF-based composite adsorbents. Environ. Sci. Water Res. Technol. 2023, 9, 1305–1330.
- Mazlan, N.A.; Butt, F.S.; Lewis, A.; Yang, Y.; Yang, S.; Huang, Y. The Growth of Metal–Organic Frameworks in the Presence of Graphene Oxide: A Mini Review. Membranes 2022, 12, 501.
