Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Ahsanullah Unar and Version 2 by Camila Xu.
Disseminated intravascular coagulation (DIC) is a pathological disease that often manifests as a complication in patients with sepsis. Sepsis is a systemic inflammatory response caused by infection and is a major public health concern worldwide.
- sepsis
- disseminated intravascular coagulation
- therapy
- corticosteroids
1. Introduction
Disseminated intravascular coagulation (DIC) is a pathological disease that often manifests as a complication in patients with sepsis. Sepsis is a systemic inflammatory response caused by infection and is a major public health concern worldwide [1]. To understand the evolution of the sepsis concept, Table 1 provides an overview of the differences between the traditional approach based on systemic inflammatory response syndrome (SIRS) and the sepsis-3 definition, which emphasizes organ dysfunction or risk of death [1][2][3][4][5][6][1,2,3,4,5,6]. Coagulation disorders that can lead to the development of DIC are often observed in sepsis. DIC is a disease that results in microvascular coagulation, decreased organ perfusion, organ failure, and an increased risk of death. The incidence rate of DIC is estimated at 2.5 cases per 1000 people, with an 8.7% increase over the two decades [1][3][1,3]. Sepsis disrupts the blood coagulation process and leads to disruption of hemostasis; however, among these, DIC represents the most serious complication. Approximately 50–70% of patients suffer from DIC. In approximately 35% of cases, it manifests itself overtly. The diagnosis of DIC typically involves the assessment of coagulation markers but lacks sufficient specificity. Therefore, it is crucial to distinguish DIC from diseases characterized by platelet count [7][8][7,8]. Unfortunately, several patients who develop thrombocytopenia from a variety of causes are often initially misdiagnosed as having disseminated DIC. This misdiagnosis can result in these patients not receiving the treatment they need. The coagulation process is closely intertwined with the system and is linked to other inflammatory responses [9][10][9,10]. The term immune thrombosis refers to the interaction between coagulation and innate immunity [11]. Traditionally, it has been assumed that coagulation activation is triggered by a tissue factor on monocytes and macrophages that is induced by microorganisms and their components, so-called pathogen-associated molecular patterns (PAMPs) [12].Tissue factor (TF) is a potent initiator of coagulation [13] and induces proinflammatory responses through the activation of protease-activated receptors (PARs) [13][14][13,14]. Phosphatidylserine on the cell membrane has been identified as an important coagulation activator [15]. Apart from these PAMPs, it has also been found that damage-associated molecular patterns (DAMPs) released by injured cells, such as B. cell-free DNA histones and high mobility group box one protein (HMGB1), contribute to the initiation of coagulation [9]. Extracellular neutrophil traps (NETs), composed of DNA fibers, nuclear proteins, and antimicrobial peptides, have been found to enhance thrombogenicity [9].In addition to activation of coagulation, suppression of fibrinolysis is an important feature of sepsis DIC. PAI-1 released from damaged endothelial cells inhibits fibrinolysis and leads to the development of a thrombotic phenotype associated with coagulopathy (Figure 1) [16][17][16,17].
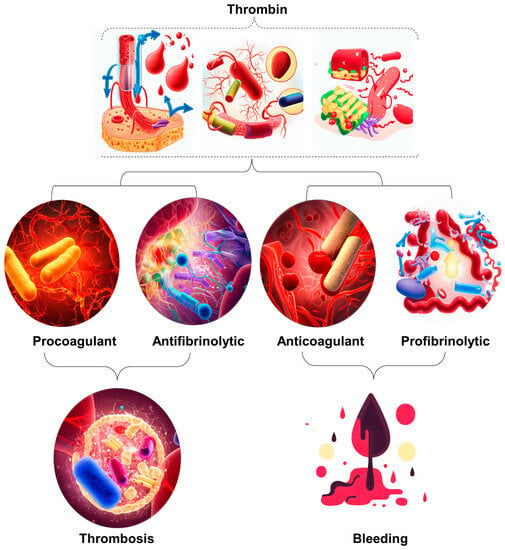
Figure 1. Illustration of the occurrence of excessive thrombin formation in DIC resulting in either bleeding or thrombosis. The specific outcome is determined by the predominant change disrupting the delicate balance between procoagulant and fibrinolytic effects. The dynamic interaction between procoagulant and fibrinolytic mechanisms in DIC plays a crucial role in determining the clinical manifestations of the disease. Therefore, it is imperative to implement timely and targeted therapeutic strategies to maximize patient outcomes.
Table 1.
][23][22,23]. This suggests that patients excluded by ISTH criteria may suffer from DIC, highlighting the value of JAAM criteria due to their integrative approach. However, the landscape changed with the introduction of the Sepsis-3 definition, which includes the Systemic Inflammatory Response Syndrome (SIRS) score, making the JAAM criteria somewhat less relevant. In response, a new set of criteria called sepsis-induced coagulopathy (SIC) was developed in 2017 to support early DIC diagnosis in sepsis patients. It considers both sepsis and clotting problems, such as a low platelet count. In diagnosing and managing DIC, physicians rely on laboratory findings, including low platelet count, elevated D-dimers, and abnormal clotting times, alongside clinical assessment [24][25][24,25]. These indicators inform the ISTH scoring system for overt DIC diagnosis [2][3][2,3]. Key tests include Complete Blood Count (CBC), Partial Thromboplastin Time (PTT), Prothrombin Time (PT) assay, fibrinogen, and D-dimer assays. D-dimer and Fibrin Degradation Product (FDP) tests offer robust diagnostic value [4]. A comprehensive DIC panel includes D-dimer and FDP for swift diagnosis and antithrombin for severity assessment and prognosis [24][25][26][24,25,26]. Table 2 provides a detailed comparison of the diagnostic criteria used by the ISTH for both open DIC and SIC and the criteria used by the JAAM for DIC. The criteria are divided into low-risk, medium-risk, and high-risk categories, each of which has a specific rating [21][22][23][27][28][29][30][21,22,23,27,28,29,30].
Table 2.
Comparative Evaluation of Diagnostic Criteria Across ISTH Overt DIC, JAAM DIC, and ISTH SIC Scoring Systems.
| Parameter (Units) | Diagnostic Method | Low-Risk Criteria (Score = 1) | Moderate-Risk Criteria (Score = 2) | High-Risk Criteria (Score = 3) | Interpretative Notes |
| Platelet Count (×10⁹ per L) | ISTH Overt DIC | 50–100 | N/A | <80 or 50% drop in 24 h 1 | Lower counts indicate severe clotting issues |
| JAAM DIC | <50 | N/A | N/A | - | |
| ISTH SIC | 100–150 | <100 | N/A | - | |
| Fibrin Degradation Products (FDP)/D-dimer (μg/mL) | ISTH Overt DIC | N/A | Moderate increase 2 | Strong increase 3 | Elevated levels suggest severe clotting issues |
| JAAM DIC | 10–25 | N/A | ≥25 | - | |
| ISTH SIC | N/A | N/A | N/A | - | |
| Prothrombin Time (PT) (seconds or PT-INR) | ISTH Overt DIC | 1.2–1.4 PT-INR | 3–6 s | ≥6 s | Longer times signify clotting dysfunction |
| JAAM DIC | 1.2–1.4 PT-INR | N/A | >1.4 PT-INR | - | |
| ISTH SIC | N/A | N/A | N/A | - | |
| Fibrinogen Levels (g/mL) | ISTH Overt DIC | N/A | N/A | <100 | Low levels indicate severe coagulation issues |
| JAAM DIC | N/A | N/A | N/A | - | |
| ISTH SIC | N/A | N/A | N/A | - | |
| SIRS Score | ISTH Overt DIC | N/A | N/A | N/A | - |
| JAAM DIC | >3 | N/A | N/A | Elevated scores indicate systemic inflammation | |
| ISTH SIC | N/A | N/A | N/A | - | |
| SOFA Score | ISTH Overt DIC | N/A | N/A | N/A | - |
| JAAM DIC | 1 | N/A | N/A | Score assesses multi-organ dysfunction | |
| ISTH SIC | 1 | ≥2 | N/A | - |
1 A reduction of 50% in platelet count within 24 h is indicative of high risk for DIC as per ISTH guidelines. 2 A ‘Moderate increase’ in FDP/D-dimer generally refers to a 10–25% increase from baseline levels. 3 A ‘Strong increase’ in FDP/D-dimer generally refers to an increase greater than 25% from baseline levels. DIC: Disseminated Intravascular Coagulation, a severe disorder causing abnormal blood clotting. SIC: Sepsis-Induced Coagulopathy, a condition where blood clotting is triggered by infection. JAAM: Methodology developed by the Japanese Association for Acute Medicine. ISTH: Methodology established by the International Society on Thrombosis and Haemostasis. PT-INR: Prothrombin Time to International Normalized Ratio, a standardized measure of blood clotting time. Measured in seconds or as a PT to International Normalized Ratio (PT-INR). SIRS: Systemic Inflammatory Response Syndrome, an indicator of systemic inflammation. SOFA: Sequential Organ Failure Assessment, an evaluation of multi-organ functionality. Platelet Count: Measured in ×109 per liter (L); D-dimer Levels: Measured in micrograms per milliliter (μg/mL); Fibrinogen Levels: Measured in grams per milliliter (g/mL); “Score = 1” denotes Low-Risk Criteria, “Score = 2” denotes Moderate-Risk Criteria, and “Score = 3” denotes High-Risk Criteria. ‘N/A’ signifies that the criteria are not applicable under the particular diagnostic method.
Managing DIC is a multifaceted challenge, and the primary goal is to address the underlying infection. However, there’s limited solid evidence supporting the use of anticoagulant therapy alongside antibiotics and source control. Several large-scale randomized controlled trials (RCTs) involving different anticoagulants have failed to provide conclusive evidence of their effectiveness. Nonetheless, guidelines recommend anticoagulant therapy for patients with severe sepsis-associated DIC, particularly if they have coagulation problems. These recommendations have been validated by the ISTH DIC Scientific Standardization Committee [1][21][27][31][1,21,27,31]. A hypothesis known as the “East Asian paradox” suggests that unique genetic or environmental factors specific to East Asian populations might contribute to the observed differences in DIC diagnosis and management. The choice of anticoagulant therapy for sepsis-induced DIC depends on the condition’s severity and the specific diagnostic criteria in use. Japanese researchers are encouraged to share their clinical research data internationally to improve the understanding of DIC and its management [30]. Overall, there is a major discrepancy between diagnostic and treatment options in Japan and Western countries, and further research is needed to establish an international consensus on DIC as a therapeutic target. Investigations should also focus on the coagulofibrinolytic system in sepsis and the impact of racial characteristics on thrombolytic mechanisms.
3. Can Sepsis-Induced DIC Patients Benefit from Corticosteroids?
Corticosteroids, commonly referred to as CS, continue to be a subject of debate within the community regarding their use in treating sepsis, septic shock, and DIC. There is no consensus due to conflicting results from studies. Gibbison et al. [32] Annane et al. [33] Salluh et al. [34] share perceptions about the possible advantages of CS in addressing sepsis-induced DIC. However, they also highlight the need for research considering the nature of the current evidence. There is obviously a need for more high-quality randomized controlled trials based on these two comprehensive reviews, reflecting the existing uncertainty in this area. In contrast, Rochwerg et al. [35] reported that sepsis patients treated with CS may have reduced mortality. However, they also noted the low reliability of these results, consistent with the urge for more detailed investigation expressed in earlier studies. In addition, according to Gibbison et al. [32], there is quite a positive response to CS on coagulation factors. Despite the possibility of a positive result, the researchers emphasized the experimental nature of their findings and the need for future studies to confirm them. A positive perspective was provided by Gazzaniga et al. [36], who suggested a potential reduction in the risk of SIC with the use of CS. However, like the other studies, they acknowledged the moderate quality of their evidence, further underscoring the need for higher-quality studies. Finally, Ni et al. [37] and Liang et al. [38] found potential benefits of CS in reducing mortality in patients with septic shock and improving outcomes in patients with sepsis-induced coagulopathy. Despite these encouraging results, both studies recommended further investigation and careful interpretation of the results due to possible confounding factors. Considering these diverse studies, it becomes clear that while there are hints of potential benefits associated with CS in sepsis and septic shock treatment, the current evidence is still inconclusive, with questions about the certainty and quality of the findings. The consensus among all authors is the pressing need for further rigorous and high-quality research to substantiate these preliminary findings, assess the potential risks and benefits more robustly, and clarify the role of CS in the treatment of sepsis and sepsis-induced coagulopathy. Moreover, future research should consider the factors that might explain the discrepancies in these studies’ results, such as variations in dosage, timing of administration, patient population, patient endotypes, and study design (Table 3). Until a clear consensus appears from more definitive research, clinicians should make decisions on the use of CS on a case-by-case basis, considering each patient’s individual circumstances, the potential benefits and risks of corticosteroid use, and the existing guidelines.Table 3.
Summary of Therapies for Sepsis-Related DIC.
| Therapy | Mechanism of Action | Dosage and Administration | Efficacy | Adverse Effects | References |
|---|---|---|---|---|---|
| Unfractionated Heparin (UFH) | Anticoagulant | Dosage: Based on weight, typically 80 units/kg bolus followed by 18 units/kg/hr infusion | Limited high-quality evidence for use in sepsis-related DIC. Small trials show potential benefits in early-stage sepsis patients but not necessarily in sepsis DIC patients | Bleeding risk | [19][39][40][41][19,39,40,41] |
| Recombinant Soluble TM (rsTM) | Alleviates DIC and reduces mortality | Dosage: Varies, typically administered intravenously | More effective than UFH in alleviating DIC and reducing mortality in infectious DIC patients | NS * | [39][40][41][42][43][39,40,41,42,43] |
| Activated Protein C (APC) | Anticoagulant and anti-inflammatory agent; degrades extracellular histones | Dosage: Varies, typically administered intravenously | No significant difference in response rates compared to UFH for DIC; reduces bleeding risk and mortality | Bleeding risk | [44][45][46][47][48][49][44,45,46,47,48,49] |
| High-dose Antithrombin (AT) | Reduces mortality in DIC patients without significant bleeding events | Dosage: Varies, typically administered intravenously | No reduction in mortality in sepsis patients; increases bleeding risk | Increased bleeding risk | [44][45][49][50][44,45,49,50] |
| Corticosteroids | Unclear mechanism; potential benefits in sepsis-induced DIC | Dosage: Varies depending on the specific corticosteroid used and patient condition | Contrasting findings, inconclusive evidence. Some studies suggest potential benefits, while others show no significant impact or potential harm | Potential adverse effects: increased risk of infection, metabolic disturbances | [32][33][34][35][36][38][51][32,33,34,35,36,38,51] |
| Thrombomodulin alfa (rTM) | Binds to thrombin, activates protein C, downregulates coagulation | Dosage: Varies, typically administered intravenously | Reduction in overall mortality rates, minimized bleeding complications | NS * | [8][52][53][8,52,53] |
| Vitamin C | Potential antioxidant, anti-inflammatory, and anticoagulant properties | Dosage: Varies, typically administered intravenously | Inconclusive evidence. Some studies show potential benefits in certain parameters, while others show no significant impact or potential harm | NS * | [54][55][56[60][61][54,55][57][58],56[,5759,58],59,60,61] |
| Fibrinolytic Therapy | Reduces clot formation, improves organ perfusion | Dosage: Varies depending on the specific fibrinolytic agent used | Impact on clinical outcomes inconclusive; some studies show improvements in coagulation parameters, while others show no significant effect | Bleeding risk | [62][63][64][65][66][67][62,63,64,65,66,67] |
| Platelet Transfusion | Controversial; potential benefits in severe thrombocytopenia or active bleeding | Dosage: Varies depending on the patient’s platelet count and clinical condition | Evidence supporting efficacy is sparse; conflicting recommendations | Potential adverse effects: bleeding complications | [68][69][70][71][72][73][68,69,70,71,72,73] |
| Granulocyte Colony-Stimulating Factor (G-CSF) | Stimulates production and mobilization of neutrophils | Dosage: Varies, typically administered subcutaneously or intravenously | Potential benefits in improving coagulation parameters | NS * | [74][75][76][77][74,75,76,77] |
| Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) | Acts on neutrophils and monocytes/macrophages | Dosage: Varies, typically administered subcutaneously or intravenously | Impact on sepsis-induced DIC not yet clearly defined | NS | [74][75][74,75] |
| Interferon-gamma (IFN-γ) | Improves coagulation abnormalities, shows a trend toward decreased mortality in sepsis-induced coagulopathy patients | Dosage: Varies, typically administered intravenously | Improved coagulation abnormalities, reduced DIC duration, potential decrease in mortality | NS | [53] |
| Mesenchymal Stem Cells (MSCs) | Immunomodulatory effects through cytokine secretion | Dosage: Varies, typically administered intravenously | Promising results in preclinical studies, potential to improve outcomes in sepsis-induced DIC | NS * | [78][79][80][81][82][83][84]85][86][87][78,79,80,81[ |
| Feature | Previous Sepsis Definitions (SIRS-Based) | Sepsis 3 Definition |
|---|---|---|
| Definition | Sepsis is SIRS + confirmed or presumed infections * | Sepsis is life-threatening organ dysfunction due to a dysregulated host response to infection |
| Organ Dysfunction Criteria | Based on individual clinical criteria (e.g., temperature, heart rate, respiratory rate, WBC count) | Organ dysfunction defined as an increase of 2 or more points in the Sequential Organ Failure Assessment (SOFA) score |
| Clinical Criteria | Relatively simple criteria (e.g., T > 38 C or <36 C, p > 90/min, RR > 20/min or PaCO2 < 32 mmHg, WBC > 12 or >10% immature band forms) | qSOFA (HAT) **: Hypotension (SBP ≤ 100 mmHg), Altered mental status (any GCS < 15), Tachypnea (RR ≥ 22) |
| Classification of Severity | Sepsis, Severe Sepsis, Septic Shock | Sepsis, Septic Shock (Severe Sepsis no longer exists) |
| Diagnostic Accuracy | Lack of sensitivity and specificity for diagnosing severe sepsis | Improved predictive validity and accuracy in diagnosing sepsis |
| Use in ICU Patients | SIRS criteria lacked sensitivity for defining sepsis in ICU patients | SOFA score superior to SIRS in predicting mortality in ICU patients |
| Use in Non-ICU Patients | Less accurate in predicting hospital mortality outside the ICU | Similar predictive performance in non-ICU patients |
| Global Applicability | Used globally, but lacks standardization and content validity | Development and validation conducted in high-income countries |
| Prognostic Value | Limited ability to predict patient outcomes and mortality | Enhanced ability to prognosticate patient outcomes and mortality risk |
| Emphasis on Infection Trigger | Inclusion of infection as a crucial component in sepsis diagnosis | Maintains the importance of infection in defining sepsis |
| Endorsement by Professional Orgs. | Various organizations endorsed previous definitions | Not universally endorsed by all organizations |
T > Temperature, p > Pulse Rate, RR > Respiratory Rate, Pa-CO2 > Partial Pressure of Carbon Dioxide (Pa-CO2), WBC > White Blood Cell Count. qSOFA > quick Sequential Organ Failure Assessment, “HAT” represents the three components of qSOFA: H-Hypotension, A-Altered Mental Status. T–Tachypnea. * Sepsis is characterized by Systemic Inflammatory Response Syndrome (SIRS) accompanied by confirmed or presumed infections. ** qSOFA is a simplified bedside tool that aids healthcare providers in quickly assessing patients with suspected infection for signs of organ dysfunction. If a patient presents with two or more of the qSOFA criteria, it indicates a higher risk of sepsis-related complications and may prompt further evaluation and early intervention to improve patient outcomes. However, it is important to note that qSOFA is not intended to diagnose sepsis definitively but serves as a screening tool to identify patients who require closer monitoring and additional evaluation for possible sepsis.
2. Comparative Analysis of DIC Diagnosis and Treatment: Eastern vs. Western Approaches
The diagnosis and management of DIC manifest distinct variations between Japan and Western countries (Figure 2). These variations are shaped by multiple factors, including differing understandings of thrombolytic mechanisms and the types of evidence deemed valid for therapeutic decision-making. In Japan, clinicians adopt a holistic approach, integrating a wide array of research methodologies, ranging from clinical trials and subgroup analyses to observational studies, to inform treatment protocols [19][20][19,20]. Conversely, Western medical practice primarily relies on large-scale studies that focus on sepsis, often employing randomized controlled trials (RCTs) as the research design [19]. This section will shed light on these distinctions and their implications and as well as highlight the primary commonalities and distinctions in the clinical guidelines for managing DIC as laid out by BCSH (British Committee for Standards in Haematology), JSTH (Japanese Society of Thrombosis and Hemostasis), and SISET (Italian Society for Thrombosis and Hemostasis (Figure 3) [19][20][21][19,20,21]. The International Society on Thrombosis and Haemostasis (ISTH) has established specific criteria for the diagnosis of overt DIC, which include parameters such as low platelet count and prolonged prothrombin time. In contrast, Japan introduced an alternative approach in 2006 called the Japanese Society of Acute Medicine (JAAM) criteria, which emphasizes laboratory tests and clinical data for an accurate diagnosis.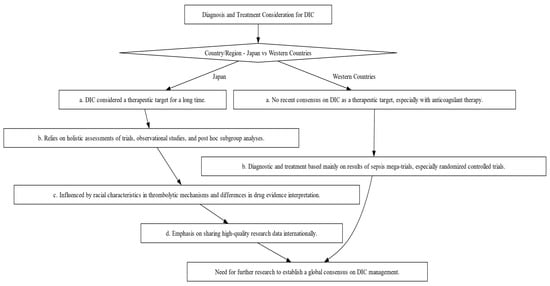
Figure 2. Decision-making Flowchart Depicting the Contrasts in Diagnosis and Treatment Approaches for DIC between Japan and Western Countries. This flowchart illustrates the divergent philosophies and methods for DIC diagnosis and treatment, emphasizing the influence of regional factors such as evidence interpretation and trial designs.
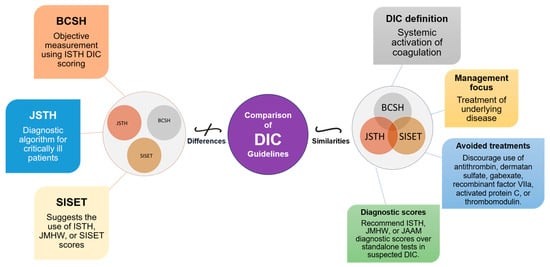

Figure 3. Comparative Overview of DIC Guidelines: Commonalities and Distinctions. This figure illustrates the commonalities and distinctions between DIC guidelines from BCSH (British Committee for Standards in Haematology), JSTH (Japanese Society of Thrombosis and Hemostasis), and SISET (Italian Society for Thrombosis and Haemostasis). Shared principles encompass recognizing DIC as a systemic coagulation activation syndrome with microvascular thrombosis and organ dysfunction, prioritizing treatment of the underlying trigger, and discouraging specific interventions. In suspected DIC cases, all guidelines favor established diagnostic scores (International Society on Thrombosis and Haemostasis (ISTH), the Japanese Ministry of Health and Welfare (JMHW), and the Japanese Association for Acute Medicine (JAAM)). Differences include variations in treatment recommendations, the ISTH’s simple scoring system for overt DIC, JAAM’s focus on critically ill patients, SISET’s endorsement of diagnostic scores, and BCSH’s objective measurement using ISTH DIC scoring system, which is closely linked to clinical outcomes.
A comparative study by Gando et al. found that the JAAM criteria have higher sensitivity compared to the ISTH criteria. Sensitivity here means that JAAM criteria are better able to correctly identify DIC cases. In their study, the JAAM criteria diagnosed DIC in 46.8% of cases, while the ISTH criteria identified it in only 18.1%. It is important that all cases identified according to ISTH criteria were also recorded according to JAAM criteria. When looking at 28-day mortality rates, both criteria showed similar results, with 31.8% for JAAM and 30.1% for ISTH [22
| , |
| 82 |
| , |
| 83 |
| , |
| 84 |
| , |
| 85 |
| , |
| 86 |
| , |
| 87 |
| ] |
* NS stands for Not Specified.
