After sperm enter the female reproductive tract, the physicochemical and biochemical microenvironment undergoes significant changes. In particular, the large changes in various ions encountered by sperm may alter the physiology of sperm, ultimately compromising capacitation and fertilization. Thus, the rapid response to environmental variations is vital for sperm functions. For example, Calcium, the most crucial ion for sperm functions, enters into sperm via Ca2+ permeable ion channels. The cation channel of sperm (CatSper) is a sperm-specific, pH-sensitive, and Ca2+-permeable ion channel. It is responsible for the predominant Ca2+ entry in mammalian sperm and is involved in nearly every event of sperm to acquire fertilizing capability. In addition, CatSper also serves as a pivotal polymodal chemosensor in mammalian sperm by responding to multiple chemical cues. Physiological chemicals (such as progesterone, prostaglandins, β-defensins, and odorants) provoke Ca2+ entry into sperm by activating CatSper and thus triggering sperm functions.
- CatSper
- chemosensor
- endocrine-disrupting chemicals
- male reproduction
- sperm
1. Introduction
Specifically, the cation channel of sperm (CatSper) is a sperm-specific, pH-sensitive, and Ca
-permeable ion channel [7]. Crucially, this channel is responsible for the predominant Ca
entry in mammalian sperm and is involved in nearly every event by which sperm acquire their fertilizing capability. Additionally, ion channels are capable of transporting ions faster than transporters. This allows sperm to respond quickly to guidance cues within the female reproductive tract. Consequently, CatSper enables the translation of large changes in the microenvironment into changes of [Ca
]
[8]. Although fertilization is at the center of creating new life, it is still a long way from being fully understood. A better understanding of the CatSper channel is important, not only to advance knowledge of the cause of male infertility but also to inspire improvement in the development of male contraceptives. On one hand, the knockout of genes encoding the CatSper channel in male mice, as well as genetic mutations in CatSper genes in humans, lead to male infertility and the inability of sperm to undergo hyperactivation and to penetrate oocytes. On the other hand, CatSper plays a pivotal role in responding to multiple chemical cues, including physiological chemicals (such as progesterone [P4] and prostaglandins [PGs]), and synthetic and natural chemicals (such as medicines and endocrine disrupting chemicals [EDCs]). Therefore, CatSper is also a pivotal polymodal chemosensor in mammalian sperm [9].
2. Overview of CatSper
3. CatSper and Physiological Chemicals
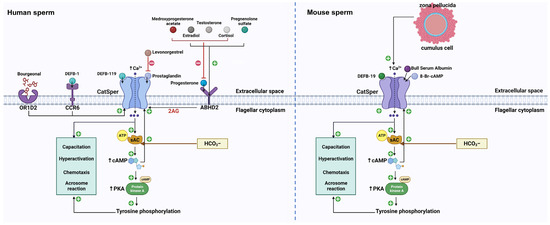
3.1. CatSper and Endogenous Steroids
3.2. CatSper and PGs
3.3. CatSper and cAMP
3.4. CatSper and ZP Glycoproteins
3.5. CatSper and Bovine Serum Albumin (BSA)
3.6. CatSper and DEFBs
3.7. CatSper and Neurotransmitters
3.7. CatSper and Neurotransmitters
In mammals, receptors for many neurotransmitters and neuromodulators (such as acetylcholine, adenosine, adenosine triphosphate, γ-aminobutyric acid, serotonin, norepinephrine, and dopamine) are found in sperm. Therefore, a sperm is regarded as a neuron with a tail [53,54][53][54]. Interestingly, P4 activates CatSper in human sperm via an unconventional endocannabinoid signaling pathway (P4/ABHD2/2-AG/CatSper) [31]. In addition, serotonergic signals enhance hamster sperm hyperactivation via CatSper [55].3.8. CatSper and Odorant Attractants
Sperm chemotaxis guides sperm toward the oocyte and is closely related to sperm capacitation, hyperactivation, the acrosome reaction, and male fertility. In humans, bourgeonal is a typical odorant and chemoattractant that is proposed to activate olfactory receptors (OR1D2) and to open CatSper to increase [Ca2+]i via a G-protein-coupled receptor/olfactory G-protein/cAMP/PKA pathway [9,56,57][9][56][57]. Moreover, men with idiopathic infertility and low sensitivity to bourgeonal have decreased OR1D2 protein expression and bourgeonal-activated CatSper current in their sperm [58]. These findings link odor perception to CatSper and male infertility. This sperm odorant attractant may provide a feasible screening method for CatSper-related male infertility.4. CatSper and Medicines
4.1. CatSper and Traditional Medicine
CatSper is regarded as a primary target for the pharmacological treatment of male infertility and a novel non-hormone target for male contraception. Some traditional medicine has shown promise for improving male infertility through CatSper. Sheng Jing Shan (SJS), a traditional Chinese medicine, has shown efficacy in treating asthenozoospermia. Notably, SJS effectively improved the sperm motility of a rat model of cyclophosphamide (CP)-induced asthenozoospermia by upregulating Catsper1 expression [59]. Trigonelline semen (TS), also known as fenugreek, is a natural herbal substance recognized for its ability to improve sperm count and motility in infertile men [60]. In a rat model of CP-induced male infertility, TS effectively restored sperm count, motility, testosterone levels, and the expression of Catsper1, Catsper2, Catsper3, and Catsper4 [61]. Panax ginseng, a well-known traditional medicine with multiple pharmacological activities, is beneficial in treating various diseases [62]. Regarding male fertility, studies have noted that mice treated with P. ginseng exhibit increased sperm motility and Ca2+ levels [63]. P. ginseng significantly increases the expression of Catsper1, Catsper2, Catsper3, and Catsper4 in mouse sperm [63]. A recent investigation reported that a natural herb called Putranjiva roxburghii could effectively upregulate the expression of CatSper genes in bull sperm and markedly boost sperm motility [64]. In addition, escanbil is a traditional medicine applied to treat abnormal menstruation and menstrual cramps in folk medicine [65]. It improves sperm motility and alters the expression of CatSper genes in aging mice [66]. These results suggest that CatSper may be a potential therapeutic agent for natural medicine treatment of male infertility.4.2. CatSper and Anti-Depressants
Selective serotonin reuptake inhibitors are the most widely used antidepressants in the United States and Europe, but recent research has highlighted their potential to impair male fertility [71][67]. Researchers have demonstrated that sertraline inhibits CatSper currents induced by intracellular alkalinization, voltage changes, P4, and PGs in human sperm [72][68]. Sertraline has a significant inhibitory effect on the acrosome reaction and viscous-medium penetration induced by P4 and PGs [72][68]. These findings suggest that the therapeutic administration of sertraline for depression may impair human reproduction.4.3. CatSper and 5-Alpha Reductase Inhibitors
Finasteride (FS) and dutasteride (DS), two 5-alpha reductase inhibitors, are widely used to treat benign prostate hyperplasia. However, their prolonged use has been shown to adversely affect male semen quality [73][69]. FS activates CatSper, at least partially, via PG binding sites, whereas DS activates CatSper, at least partially, through P4 binding sites in human sperm [74][70]. Thus, they interfere with Ca2+ signaling mediated by PGs and P4. In addition, the exposure of mice to DS affected sperm count and motility and the expression of CatSper genes in caput and caudal epididymal sperm [75][71].4.4. CatSper and Analgesics
Paracetamol is widely used as a mild analgesic to alleviate fever and pain. However, rodent studies have shown that paracetamol may have negative effects on sperm count and motility due to its endocrine effects. Additionally, high concentrations of paracetamol in male urine have been linked to lower sperm motility [76][72]. In human sperm, paracetamol is metabolized to N-arachidonoylphenolamine via fatty acid amide hydrolase expressed in the sperm neck region. N-arachidonoylphenolamine directly activates human CatSper, reduces sperm motility, and affects viscous-medium penetration [77][73].4.5. CatSper and Ca
2+
Channel Blockers
Nifedipine is a Ca2+ channel blocker and is used as an antihypertensive medicine. It exhibits anti-fertility effects in male rats, resulting in a significant reduction in sperm motility and count [78][74]. Nifedipine treatment reduces sperm motility and count and substantially downregulates the expression of CatSper genes in mouse epididymal sperm [75][71]. In addition, RU1968, a steroid-based selective and potent cross-species inhibitor of CatSper, has been demonstrated to suppress the activation of CatSper in human, mouse, and sea urchin sperm [79][75]. Therefore, nifedipine serves as a powerful tool for the investigation of the physiological function of CatSper in human sperm and for the promotion of the development of non-hormonal male contraceptives.4.6. CatSper and Phosphodiesterase (PDE)-Inhibitors
Trequinsin hydrochloride, a PDE-3 inhibitor, has emerged as a promising CatSper agonist. In human sperm, trequinsin hydrochloride exhibits a P4-like agonist profile and significantly potentiates the CatSper current, effectively increasing sperm hyperactivation and viscous-medium penetration [80][76]. Additionally, this CatSper agonist induces a concentration-dependent elevation in Ca2+ levels through cross-desensitization with PGE1 [80][76].4.7. CatSper and Anti-Inflammatory Drugs
Cisplatin is the most widely used drug in oncology treatment. However, cisplatin-based treatment of testicular cancer disrupts spermatogenesis and reduces the sperm motility of patients [81][77]. The indole derivative N′-(4-dimethylaminobenzylidene)-2-1-(4-(methylsulfinyl) benzylidene)-5-fluoro-2-methyl-1H-inden-3-yl) acetohydrazide (MMINA) has significant anti-inflammatory and antioxidant effects and can protect against the testicular toxicity induced by cisplatin [82][78]. Most importantly, MMINA activates CatSper by upregulating the expression of CatSper genes in rat sperm [83][79]. Moreover, MMINA is capable of forming hydrogen bonds with CatSper [83][79].5. CatSper and EDCs
5.1. CatSper and Environmental Estrogens
Initially, EDCs were called xenoestrogens due to their estrogenic, antiestrogenic, androgenic, and antiandrogenic effects [85][80]. Steviol, a natural non-caloric sweetener metabolite, exerts endocrine effects on human sperm by antagonizing P4 and agonizing CatSper, resulting in a rapid influx of Ca2+ [86][81]. Bisphenol A (BPA), a ubiquitous EDC and synthetic organic compound, has been significantly and negatively associated with male fertility [87][82]. BPA binds to estrogen receptors α and β and exhibits estrogenic activity [88][83]. Animal studies have revealed that BPA impairs sperm function by reducing the expression of CatSper genes and the CatSper current [89][84]. In GC-2 cells, a mouse spermatogonia cell line, BPA decreased the growth rate and [Ca2+]i, and downregulated the expression of Catsper1, Catsper2, Catsper3, and Catsper4 through Ten-eleven translocation 1 [90][85]. In humans, bisphenol A diglycidyl ether and bisphenol analogs—but not BPA—activate CatSper [91][86]. Diethylstilbestrol, a well-known, synthetic, non-steroidal estrogen, potentiates CatSper currents, increases the [Ca2+]i, and inhibits P4-induced Ca2+ influx and sperm functions in humans [92][87]. Perfluorooctane acid, an organic pollutant, activates CatSper to elevate the [Ca2+]i in human sperm [93][88]. Like diethylstilbestrol, perfluorooctane acid suppresses the P4-induced CatSper current, Ca2+ influx, and sperm functions [93][88]. In addition, the diversity of EDCs implies that even heavy metals may possess estrogenic activity. Cadmium is considered an EDC with significant toxicity to the reproductive system; it acts as an estrogen mimic and has the ability to bind ERs [94][89]. Cadmium impairs sperm function via a CatSper-mediated mechanism by affecting the expression of CatSper genes in mice [95][90].5.2. CatSper and Pesticides
p,p′-Dichlorodiphenyldichloroethylene, a metabolite of dichloro-diphenyl-trichloroethane commonly found in human reproductive fluids, activates CatSper to induce Ca2+ entry into sperm and disrupts acrosome reaction [96][91]. Pentachlorophenol, a widely used pesticide, suppresses the P4-induced CatSper current, Ca2+ influx, and sperm functions in humans [97][92]. Recently, a study investigated the effect of 53 pesticides and pesticide metabolites on human sperm. The results demonstrated that, although 26 pesticides activated CatSper and interfered with signaling triggered by P4 and PGs, they may interact with the unique binding sites or the P4 and PG binding sites of CatSper [98][93]. Thus, pesticides, either alone or in low-dose mixtures, have the potential to negatively affect sperm function by interfering with normal Ca2+ signaling in human sperm via CatSper.5.3. CatSper and Chemical Ultraviolet (UV) Filters
Chemical UV filters, commonly present in daily-use sunscreens, are among the most potent triggers of Ca2+ signaling. They directly activate CatSper in human sperm and elevate [Ca2+]i [84][94]. A recent study investigated the effect of 31 chemical UV filters approved in the European Union and the United States on human sperm. Although 29 of the 31 chemical UV filters induced Ca2+ signaling in human sperm, only nine of these chemicals could activate CatSper, including 4-Methylbenzylidene camphor, 3-Benzylidene camphor, meradimate, amiloxate, octisalate, benzylidene camphor sulfonic acid, homosalate, benzophenone-3, and octinoxate [99][95]. Of these chemicals, 3-Benzylidene camphor, benzylidene camphor sulfonic acid, and 4-Methylbenzylidene camphor have been found to competitively inhibit P4-induced Ca2+ signaling and target its binding sites in CatSper [84,99][94][95]. These results suggest that some chemical UV filters have the potential to interfere with P4-induced Ca2+ signaling and negatively affect sperm functions.6. CatSper and Drugs of Abuse
Interestingly, some addictive drugs affect sperm functions through CatSper. Methamphetamine (METH) is a highly addictive central nervous system stimulant that has detrimental effects on male reproductive health, including impaired spermatogenesis, testicular damage, and abnormal sperm quality [100][96]. In particular, a novel investigation showed that rats receiving METH resulted in a decrease in testis and epididymis weight [101][97]. Meanwhile, the relative expression levels of Catsper1, Catsper2, Catsper3, and Catsper4, as well as the sperm motility associated gene Mvh, were decreased significantly [101][97]. In addition, the exclusive expression of Catsper1–4 in testes is required for sperm motility and fertility [21,102][21][98]. As a result, the downregulation of these genes induced by METH increases the possibility of male infertility. Therefore, men addicted to METH may encounter potential reproductive problems. Ketamine, a dissociative anesthetic widely used in human and animal medicine, has become a popular recreational drug because it can induce hallucinatory effects. ketamine affects sperm motility, viscous-medium penetration, and the P4-induced acrosome reaction by inhibiting CatSper in human sperm, thus decreasing [Ca2+]i [103][99]. In addition, ketamine is an antagonist of the N-Methyl-D-aspartic acid (NMDA) receptor. The NMDA receptor is expressed in human sperm and involved in the inhibitory effect of ketamine on human sperm functions [104][100]. Specifically, NMDA, the physiological ligand of NMDA, could partly alleviate the motility of human sperm and significantly recover the capacitation and acrosome reaction, as well as [Ca2+]i [104][100]. Therefore, the competitive receptor binding between ketamine and NMDA may provide novel insight for clinical diagnoses of ketamine abusers. Collectively, CatSper-related drugs of abuse have been implicated in impaired sperm function and/or male infertility.7. CatSper and Antioxidants
Oxidative stress occurs when the generation of reactive oxygen species (ROS) exceeds the natural antioxidant defenses of bodies. Thus, the precise balance of ROS and antioxidants within sperm are necessary for capacitation and fertilization. The major effect of oxidative stress compromising sperm function is caused by two principal mechanisms, DNA damage and lipid peroxidation [105][101]. In human sperm, ROS damages DNA directly by the production of 1,N6-ethenoadenosine and 1,N2-ethenoguanosine, resulting in DNA structure instability and leading to single-strand breaks [106][102]. Once the transcription and translation of post-spermiogenesis stop, the DNA repair during developing sperm is terminated [107][103]. Hence, sperm function and pregnancy outcome are strongly impacted. To counteract ROS damage, the human body has developed a variety of antioxidant strategies. For instance, non-enzymatic antioxidants contained within the seminal fluid, like vitamin E and selenium [108][104]. Interestingly, treatment with these two antioxidants upregulates the expression of Catsper in the testes of young adult and aged male mice, which are the genes responsible for sperm motility [109,110][105][106]. Meanwhile, sperm parameters such as viability rate and morphology also show an improvement after treatment [109,110][105][106]. Consequently, these two essential components play a crucial role in the maintenance of male reproduction.References
- Gervasi, M.G.; Visconti, P.E. Molecular changes and signaling events occurring in spermatozoa during epididymal maturation. Andrology 2017, 5, 204–218.
- TMortimer, S. A critical review of the physiological importance and analysis of sperm movement in mammals. Hum. Reprod. 1997, 3, 403–439.
- Perez-Cerezales, S.; Boryshpolets, S.; Eisenbach, M. Behavioral mechanisms of mammalian sperm guidance. Asian J. Androl. 2015, 17, 628–632.
- Costello, S.; Michelangeli, F.; Nash, K.; Lefievre, L.; Morris, J.; Machado-Oliveira, G.; Barratt, C.; Kirkman-Brown, J.; Publicover, S. Ca2+-stores in sperm: Their identities and functions. Reproduction 2009, 138, 425–437.
- Molina, L.C.P.; Luque, G.M.; Balestrini, P.A.; Marín-Briggiler, C.I.; Romarowski, A.; Buffone, M.G. Molecular Basis of Human Sperm Capacitation. Front. Cell Dev. Biol. 2018, 6, 72.
- Finkelstein, M.; Etkovitz, N.; Breitbart, H. Ca(2+) signaling in mammalian spermatozoa. Mol. Cell. Endocrinol. 2020, 516, 110953.
- Singh, A.P.; Rajender, S. CatSper channel, sperm function and male fertility. Reprod. Biomed. Online 2015, 30, 28–38.
- Jeschke, J.K.; Biagioni, C.; Schierling, T.; Wagner, I.V.; Börgel, F.; Schepmann, D.; Schüring, A.; Kulle, A.E.; Holterhus, P.M.; von Wolff, M.; et al. The Action of Reproductive Fluids and Contained Steroids, Prostaglandins, and Zn(2+) on CatSper Ca(2+) Channels in Human Sperm. Front. Cell Dev. Biol. 2021, 9, 699554.
- Brenker, C.; Goodwin, N.; Weyand, I.; Kashikar, N.D.; Naruse, M.; Krähling, M.; Müller, A.; Kaupp, U.B.; Strünker, T. The CatSper channel: A polymodal chemosensor in human sperm. EMBO J. 2012, 31, 1654–1665.
- Huang, X.; Miyata, H.; Wang, H.; Mori, G.; Iida-Norita, R.; Ikawa, M.; Percudani, R.; Chung, J.J. A CUG-initiated CATSPERθ functions in the CatSper channel assembly and serves as a checkpoint for flagellar trafficking. bioRxiv 2023.
- Yang, F.; Gracia Gervasi, M.; Orta, G.; Tourzani, D.A.; De la Vega-Beltran, J.L.; Ruthel, G.; Darszon, A.; Visconti, P.E.; Wang, P.J. C2CD6 regulates targeting and organization of the CatSper calcium channel complex in sperm flagella. Development 2022, 149, dev199988.
- Lin, S.; Ke, M.; Zhang, Y.; Yan, Z.; Wu, J. Structure of a mammalian sperm cation channel complex. Nature 2021, 595, 746–750.
- Zhao, Y.; Wang, H.; Wiesehoefer, C.; Shah, N.B.; Reetz, E.; Hwang, J.Y.; Huang, X.; Wang, T.E.; Lishko, P.V.; Davies, K.M.; et al. 3D structure and in situ arrangements of CatSper channel in the sperm flagellum. Nat. Commun. 2022, 13, 3439.
- Avenarius, M.R.; Hildebrand, M.S.; Zhang, Y.; Meyer, N.C.; Smith, L.L.; Kahrizi, K.; Najmabadi, H.; Smith, R.J. Human male infertility caused by mutations in the CATSPER1 channel protein. Am. J. Hum. Genet. 2009, 84, 505–510.
- Luo, T.; Chen, H.Y.; Zou, Q.X.; Wang, T.; Cheng, Y.M.; Wang, H.F.; Wang, F.; Jin, Z.L.; Chen, Y.; Weng, S.Q.; et al. A novel copy number variation in CATSPER2 causes idiopathic male infertility with normal semen parameters. Hum. Reprod. 2019, 34, 414–423.
- Wang, J.; Tang, H.; Zou, Q.; Zheng, A.; Li, H.; Yang, S.; Xiang, J. Patient with CATSPER3 mutations-related failure of sperm acrosome reaction with successful pregnancy outcome from intracytoplasmic sperm injection (ICSI). Mol. Genet. Genom. Med. 2021, 9, e1579.
- Chung, J.J.; Navarro, B.; Krapivinsky, G.; Krapivinsky, L.; Clapham, D.E. A novel gene required for male fertility and functional CATSPER channel formation in spermatozoa. Nat. Commun. 2011, 2, 153.
- Carlson, A.E.; Quill, T.A.; Westenbroek, R.E.; Schuh, S.M.; Hille, B.; Babcock, D.F. Identical phenotypes of CatSper1 and CatSper2 null sperm. J. Biol. Chem. 2005, 280, 32238–32244.
- Jin, J.; Jin, N.; Zheng, H.; Ro, S.; Tafolla, D.; Sanders, K.M.; Yan, W. Catsper3 and Catsper4 are essential for sperm hyperactivated motility and male fertility in the mouse. Biol. Reprod. 2007, 77, 37–44.
- Qi, H.; Moran, M.M.; Navarro, B.; Chong, J.A.; Krapivinsky, G.; Krapivinsky, L.; Kirichok, Y.; Ramsey, I.S.; Quill, T.A.; Clapham, D.E. All four CatSper ion channel protein are required for male fertility and sperm cell hyperactivated motility. Proc. Natl. Acad. Sci. USA 2007, 104, 1219–1223.
- Avidan, N.; Tamary, H.; Dgany, O.; Cattan, D.; Pariente, A.; Thulliez, M.; Borot, N.; Moati, L.; Barthelme, A.; Shalmon, L.; et al. CATSPER2, a human autosomal nonsyndromic male infertility gene. Eur. J. Hum. Genet. 2003, 11, 497–502.
- Williams, H.L.; Mansell, S.; Alasmari, W.; Brown, S.G.; Wilson, S.M.; Sutton, K.A.; Miller, M.R.; Lishko, P.V.; Barratt, C.L.R.; Publicover, S.J.; et al. Specific loss of CatSper function is sufficient to compromise fertilizing capacity of human spermatozoa. Hum. Reprod. 2015, 30, 28–38.
- Brown, S.G.; Miller, M.R.; Lishko, P.V.; Lester, D.H.; Publicover, S.J.; Barratt, C.L.R.; Da Silva, S.M. Homozygous in-frame deletion in CATSPERE in a man producing spermatozoa with loss of CatSper function and compromised fertilizing capacity. Hum. Reprod. 2018, 33, 1812–1816.
- Wang, H.; McGoldrick, L.L.; Chung, J.-J. Sperm ion channels and transporters in male fertility and infertility. Nat. Rev. Urol. 2021, 18, 46–66.
- Navarro, B.; Kirichok, Y.; Clapham, D.E. KSper, a pH-sensitive K+ current that controls sperm membrane potential. Proc. Natl. Acad. Sci. USA 2007, 104, 7688–7692.
- Chavez, J.C.; Ferreira, J.J.; Butler, A.; De La Vega Beltran, J.L.; Trevino, C.L.; Darszon, A.; Salkoff, L.; Santi, C.M. SLO3K+ channels control calcium entry through CATSPER channels in sperm. J. Biol. Chem. 2014, 289, 32266–32275.
- Hwang, J.Y.; Chung, J.J. CatSper Calcium Channels: 20 Years On. Physiology 2023, 38, 125–140.
- Brown, S.G.; Costello, S.; Kelly, M.C.; Ramalingam, M.; Drew, E.; Publicover, S.J.; Barratt, C.L.R.; Da Silva, S.M. Complex CatSper-dependent and independent i signalling in human spermatozoa induced by follicular fluid. Hum. Reprod. 2017, 32, 1995–2006.
- Sumigama, S.; Mansell, S.; Miller, M.; Lishko, P.V.; Cherr, G.N.; Meyers, S.A.; Tollner, T. Progesterone Accelerates the Completion of Sperm Capacitation and Activates CatSper Channel in Spermatozoa from the Rhesus Macaque. Biol. Reprod. 2015, 93, 130.
- Lishko, P.V.; Botchkina, I.L.; Kirichok, Y. Progesterone activates the principal Ca2+ channel of human sperm. Nature 2011, 471, 387–391.
- Miller, M.R.; Mannowetz, N.; Iavarone, A.T.; Safavi, R.; Gracheva, E.O.; Smith, J.F.; Hill, R.Z.; Bautista, D.M.; Kirichok, Y.; Lishko, P.V. Unconventional endocannabinoid signaling governs sperm activation via the sex hormone progesterone. Science 2016, 352, 555–559.
- Uhler, M.L.; Leungt, A.; Chan, S.Y.W.; Wang, C. Direct effects of progesterone and antiprogesterone on human sperm hyperactivated motility and acrosome reaction. Fertil. Steril. 1992, 58, 1191–1198.
- Mannowetz, N.; Miller, M.R.; Lishko, P.V. Regulation of the sperm calcium channel CatSper by endogenous steroids and plant triterpenoids. Proc. Natl. Acad. Sci. USA 2017, 114, 5743–5748.
- Brenker, C.; Schiffer, C.; Wagner, I.V.; Tüttelmann, F.; Röpke, A.; Rennhack, A.; Kaupp, U.B.; Strünker, T. Action of steroids and plant triterpenoids on CatSper Ca(2+) channels in human sperm. Proc. Natl. Acad. Sci. USA 2018, 115, E344–E346.
- Rehfeld, A. Revisiting the action of steroids and triterpenoids on the human sperm Ca2+ channel CatSper. Mol. Hum. Reprod. 2020, 26, 816–824.
- Sánchez González, S.R.; Mata Martínez, E.; Torres Juárez, J.A.; Arias, R.J.; De Blas, G.A.; Sánchez Tusie, A.A. Cortisol modulates Ca(2+) signaling and acrosome reaction in human sperm. Andrology 2023, 11, 134–142.
- Carlson, E.J.; Georg, G.I.; Hawkinson, J.E. Steroidal Antagonists of Progesterone- and Prostaglandin E(1)-Induced Activation of the Cation Channel of Sperm. Mol. Pharmacol. 2022, 101, 56–67.
- Shimizu, Y.; Yorimitsu, A.; Maruyama, Y.; Kubota, T.; Aso, T.; ABronson, R. Prostaglandins induce calcium influx in human spermatozoa. Mol. Hum. Reprod. 1998, 4, 555–561.
- Strünker, T.; Goodwin, N.; Brenker, C.; Kashikar, N.D.; Weyand, I.; Seifert, R.; Kaupp, U.B. The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature 2011, 471, 382–386.
- Xia, J.; Reigada, D.; Mitchell, C.H.; Ren, D. CATSPER channel-mediated Ca2+ entry into mouse sperm triggers a tail-to-head propagation. Biol. Reprod. 2007, 77, 551–559.
- Kobori, H.; Miyazaki, S.; Kuwabara, Y. Characterization of intracellular Ca(2+) increase in response to progesterone and cyclic nucleotides in mouse spermatozoa. Biol. Reprod. 2000, 63, 113–120.
- Hereng, T.H.; Elgstoen, K.B.; Eide, L.; Rosendal, K.R.; Skalhegg, B.S. Serum albumin and HCO3− regulate separate pools of ATP in human spermatozoa. Hum. Reprod. 2014, 29, 918–930.
- Boisen, I.M.; Rehfeld, A.; Mos, I.; Poulsen, N.N.; Nielsen, J.E.; Schwarz, P.; Rejnmark, L.; Dissing, S.; Bach-Mortensen, P.; Juul, A.; et al. The Calcium-Sensing Receptor Is Essential for Calcium and Bicarbonate Sensitivity in Human Spermatozoa. J. Clin. Endocrinol. Metab. 2021, 106, e1775–e1792.
- Ferreira, J.J.; Lybaert, P.; Puga-Molina, L.C.; Santi, C.M. Conserved Mechanism of Bicarbonate-Induced Sensitization of CatSper Channels in Human and Mouse Sperm. Front. Cell Dev. Biol. 2021, 9, 733653.
- Xia, J.; Ren, D. Egg coat proteins activate calcium entry into mouse sperm via CATSPER channels. Biol. Reprod. 2009, 80, 1092–1098.
- Balbach, M.; Hamzeh, H.; Jikeli, J.F.; Brenker, C.; Schiffer, C.; Hansen, J.N.; Neugebauer, P.; Trötschel, C.; Jovine, L.; Han, L.; et al. Molecular Mechanism Underlying the Action of Zona-pellucida Glycoproteins on Mouse Sperm. Front. Cell Dev. Biol. 2020, 8, 572735.
- Ren, D.; Navarro, B.; Perez, G.; Jackson, A.C.; Hsu, S.; Shi, Q.; Tilly, J.L.; Clapham, D.E. A sperm ion channel required for sperm motility and male fertility. Nature 2001, 413, 603–609.
- Xia, J.; Ren, D. The BSA-induced Ca2+ influx during sperm capacitation is CATSPER channel-dependent. Reprod. Biol. Endocrinol. 2009, 7, 119.
- Pazgier, M.; Hoover, D.M.; Yang, D.; Lu, W.; Lubkowski, J. Human beta-defensins. Cell. Mol. Life Sci. 2006, 63, 1294–1313.
- Diao, R.; Fok, K.L.; Chen, H.; Mei Kuen, Y.; Duan, Y.; Chung, C.M.; Li, Z.; Wu, H.; Li, Z.; Zhang, H.; et al. Deficient human b-defensin 1 underlies male infertility associated with poor sperm motility and genital tract infection. Sci. Transl. Med. 2014, 6, 249ra108.
- Diao, R.; Wang, T.; Fok, K.L.; Li, X.; Ruan, Y.; Yu, M.K.; Cheng, Y.; Chen, Y.; Chen, H.; Mou, L.; et al. CCR6 is required for ligand-induced CatSper activation in human sperm. Oncotarget 2017, 8, 91445–91458.
- Li, X.; Yuan, C.; Shi, J.; Kang, H.; Chen, Y.; Duan, Y.; Jin, J.; Cheung, L.P.; Li, T.C.; Liu, Y.; et al. β-Defensin 19/119 mediates sperm chemotaxis and is associated with idiopathic infertility. Cell Rep. Med. 2022, 3, 100825.
- Meizel, S. The sperm, a neuron with a tail: ‘Neuronal’ receptors in mammalian sperm. Biol. Rev. Camb. Philos. Soc. 2004, 79, 713–732.
- Ramirez-Reveco, A.; Villarroel-Espindola, F.; Rodriguez-Gil, J.E.; Concha, I.I. Neuronal signaling repertoire in the mammalian sperm functionality. Biol. Reprod. 2017, 96, 505–524.
- Sakamoto, C.; Fujinoki, M.; Kitazawa, M.; Obayashi, S. Serotonergic signals enhanced hamster sperm hyperactivation. J. Reprod. Dev. 2021, 67, 241–250.
- Yoshida, M.; Yoshida, K. Sperm chemotaxis and regulation of flagellar movement by Ca2+. Mol. Hum. Reprod. 2011, 17, 457–465.
- Cheng, Y.-M.; Luo, T.; Peng, Z.; Chen, H.-Y.; Zhang, J.; Zeng, X.-H. OR1D2 receptor mediates bourgeonal-induced human CatSper activation in a G-protein dependent manner. bioRxiv 2019, 757880.
- Sinding, C.; Kemper, E.; Spornraft-Ragaller, P.; Hummel, T. Decreased Perception of Bourgeonal May Be Linked to Male Idiopathic Infertility. Chem. Senses 2013, 38, 439–445.
- Wang, Y.N.; Wang, B.; Liang, M.; Han, C.Y.; Zhang, B.; Cai, J.; Sun, W.; Xing, G.G. Down-regulation of CatSper1 channel in epididymal spermatozoa contributes to the pathogenesis of asthenozoospermia, whereas up-regulation of the channel by Sheng-Jing-San treatment improves the sperm motility of asthenozoospermia in rats. Fertil. Steril. 2013, 99, 579–587.
- Sadogh, A.; Gorji, N.; Moeini, R. Herbal foodstuffs in Avicenna’s recommended diet to improve sperm quality and increase male fertility; an evidence-based approach. J. Complement. Integr. Med. 2021, 19, 47–70.
- Kim, D.R.; Kim, H.Y.; Kim, H.Y.; Chang, M.S.; Park, S.K. Trigonellae Semen Enhances Sperm Motility and the Expression of the Cation Sperm Channel Proteins in Mouse Testes. Evid.-Based Complement. Altern. Med. 2015, 2015, 817324.
- Mancuso, C.; Santangelo, R. Panax ginseng and Panax quinquefolius: From pharmacology to toxicology. Food Chem. Toxicol. 2017, 107, 362–372.
- Park, E.H.; Kim, D.R.; Kim, H.Y.; Park, S.K.; Chang, M.S. Panax ginseng induces the expression of CatSper genes and sperm hyperactivation. Asian J. Androl. 2014, 16, 845–851.
- Italiya, J.M.; Patel, M.R.; Golaviya, A.V.; Patel, S.S.; Thakkar, B.K.; Jakhesara, S.J.; Joshi, C.G.; Koringa, P.G. RNA-sequencing attest increased sperm motility in bovine spermatozoa treated with ethanolic extract of Putranjiva roxburghii. 3 Biotech 2023, 13, 33.
- Kiani, K.; Rudzitis-Auth, J.; Scheuer, C.; Movahedin, M.; Sadati Lamardi, S.N.; Malekafzali Ardakani, H.; Becker, V.; Moini, A.; Aflatoonian, R.; Ostad, S.N.; et al. Calligonum comosum (Escanbil) extract exerts anti-angiogenic, anti-proliferative and anti-inflammatory effects on endometriotic lesions. J. Ethnopharmacol. 2019, 239, 111918.
- Askari Jahromi, M.; Movahedin, M.; Mazaheri, Z.; Amanlu, M.; Mowla, S.J.; Batooli, H. Evaluating the effects of Escanbil (Calligonum) extract on the expression level of Catsper gene variants and sperm motility in aging male mice. Iran. J. Reprod. Med. 2014, 12, 459–466.
- Kumar, V.S.; Sharma, V.L.; Tiwari, P.; Singh, D.; Maikhuri, J.P.; Gupta, G.; Singh, M.M. The spermicidal and antitrichomonas activities of SSRI antidepressants. Bioorg. Med. Chem. Lett. 2006, 16, 2509–2512.
- Rahban, R.; Rehfeld, A.; Schiffer, C.; Brenker, C.; Egeberg Palme, D.L.; Wang, T.; Lorenz, J.; Almstrup, K.; Skakkebaek, N.E.; Strünker, T.; et al. The antidepressant Sertraline inhibits CatSper Ca2+ channels in human sperm. Hum. Reprod. 2021, 36, 2638–2648.
- Amory, J.K.; Wang, C.; Swerdloff, R.S.; Anawalt, B.D.; Matsumoto, A.M.; Bremner, W.J.; Walker, S.E.; Haberer, L.J.; Clark, R.V. The effect of 5alpha-reductase inhibition with dutasteride and finasteride on semen parameters and serum hormones in healthy men. J. Clin. Endocrinol. Metab. 2007, 92, 1659–1665.
- Birch, M.R.; Dissing, S.; Skakkebæk, N.E.; Rehfeld, A. Finasteride interferes with prostaglandin-induced CatSper signalling in human sperm. Reproduction 2021, 161, 561–572.
- Srivastav, A.; Changkija, B.; Sharan, K.; Nagar, G.K.; Bansode, F.W. Influence of antifertility agents Dutasteride and Nifedipine on CatSper gene level in epididymis during sperm maturation in BALB/c mice. Reproduction 2018, 155, 347–359.
- Smarr, M.M.; Kannan, K.; Chen, Z.; Kim, S.; Buck Louis, G.M. Male urinary paracetamol and semen quality. Andrology 2017, 5, 1082–1088.
- Rehfeld, A.; Frederiksen, H.; Rasmussen, R.H.; David, A.; Chaker, J.; Nielsen, B.S.; Nielsen, J.E.; Juul, A.; Skakkebæk, N.E.; Kristensen, D.M. Human sperm cells can form paracetamol metabolite AM404 that directly interferes with sperm calcium signalling and function through a CatSper-dependent mechanism. Hum. Reprod. 2022, 37, 922–935.
- Morakinyo, A.O.; Iranloye, B.O.; Adegoke, O.A. Antireproductive effect of calcium channel blockers on male rats. Reprod. Med. Biol. 2009, 8, 97–102.
- Rennhack, A.; Schiffer, C.; Brenker, C.; Fridman, D.; Nitao, E.T.; Cheng, Y.M.; Tamburrino, L.; Balbach, M.; Stolting, G.; Berger, T.K.; et al. A novel cross-species inhibitor to study the function of CatSper Ca(2+) channels in sperm. Br. J. Pharmacol. 2018, 175, 3144–3161.
- McBrinn, R.C.; Fraser, J.; Hope, A.G.; Gray, D.W.; Barratt, C.L.R.; da Silva, S.J.M.; Brown, S.G. Novel pharmacological actions of trequinsin hydrochloride improve human sperm cell motility and function. Br. J. Pharmacol. 2019, 176, 4521–4536.
- Howell, S.J.; Shalet, S.M. Spermatogenesis after cancer treatment: Damage and recovery. J. Natl. Cancer Inst. Monogr. 2005, 2005, 12–17.
- Razak, S.; Afsar, T.; Bibi, N.; Abulmeaty, M.; Qamar, W.; Almajwal, A.; Inam, A.; AI Disi, D.; Shabbir, M.; Bhat, M.A. Molecular docking, pharmacokinetic studies, and in vivo pharmacological study of indole derivative 2-(5-methoxy-2-methyl-1H-indole-3-yl)-N’- acetohydrazide as a promising chemoprotective agent against cisplatin induced organ damage. Sci. Rep. 2021, 11, 6245.
- Afsar, T.; Razak, S.; Trembley, J.H.; Khan, K.; Shabbir, M.; Almajwal, A.; Alruwaili, N.W.; Ijaz, M.U. Prevention of Testicular Damage by Indole Derivative MMINA via Upregulated StAR and CatSper Channels with Coincident Suppression of Oxidative Stress and Inflammation: In Silico and In Vivo Validation. Antioxidants 2022, 11, 2063.
- Yilmaz, B.; Terekeci, H.; Sandal, S.; Kelestimur, F. Endocrine disrupting chemicals: Exposure, effects on human health, mechanism of action, models for testing and strategies for prevention. Rev. Endocr. Metab. Disord. 2020, 21, 127–147.
- Shannon, M.; Rehfeld, A.; Frizzell, C.; Livingstone, C.; McGonagle, C.; Skakkebaek, N.E.; Wielogórska, E.; Connolly, L. In vitro bioassay investigations of the endocrine disrupting potential of steviol glycosides and their metabolite steviol, components of the natural sweetener Stevia. Mol. Cell. Endocrinol. 2016, 427, 65–72.
- Zhou, Y.; Xu, W.; Yuan, Y.; Luo, T. What is the Impact of Bisphenol A on Sperm Function and Related Signaling Pathways: A Mini-review? Curr. Pharm. Des. 2020, 26, 4822–4828.
- Le Fol, V.; Ait-Aissa, S.; Sonavane, M.; Porcher, J.M.; Balaguer, P.; Cravedi, J.P.; Zalko, D.; Brion, F. In vitro and in vivo estrogenic activity of BPA, BPF and BPS in zebrafish-specific assays. Ecotoxicol. Environ. Saf. 2017, 142, 150–156.
- Wang, H.F.; Liu, M.; Li, N.; Luo, T.; Zheng, L.P.; Zeng, X.H. Bisphenol A Impairs Mature Sperm Functions by a CatSper-Relevant Mechanism. Toxicol. Sci. 2016, 152, 145–154.
- Yuan, W.B.; Chen, H.Q.; Li, J.Z.; Zhou, S.M.; Zeng, Y.; Fan, J.; Zhang, Z.; Liu, J.Y.; Cao, J.; Liu, W.B. TET1 mediated male reproductive toxicity induced by Bisphenol A through Catsper-Ca(2+) signaling pathway. Environ. Pollut. 2022, 296, 118739.
- Rehfeld, A.; Andersson, A.M.; Skakkebaek, N.E. Bisphenol A Diglycidyl Ether (BADGE) and Bisphenol Analogs, but Not Bisphenol A (BPA), Activate the CatSper Ca(2+) Channel in Human Sperm. Front. Endocrinol. 2020, 11, 324.
- Zou, Q.X.; Peng, Z.; Zhao, Q.; Chen, H.Y.; Cheng, Y.M.; Liu, Q.; He, Y.Q.; Weng, S.Q.; Wang, H.F.; Wang, T.; et al. Diethylstilbestrol activates CatSper and disturbs progesterone actions in human spermatozoa. Hum. Reprod. 2017, 32, 290–298.
- Yuan, Y.; Ding, X.; Cheng, Y.; Kang, H.; Luo, T.; Zhang, X.; Kuang, H.; Chen, Y.; Zeng, X.; Zhang, D. PFOA evokes extracellular Ca(2+) influx and compromises progesterone-induced response in human sperm. Chemosphere 2020, 241, 125074.
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocr. Rev. 2009, 30, 293–342.
- Wang, H.F.; Chang, M.; Peng, T.T.; Yang, Y.; Li, N.; Luo, T.; Zhou, M.Z.; Zeng, X.H.; Zheng, L.P. Exposure to Cadmium Impairs Sperm Functions by Reducing CatSper in Mice. Cell. Physiol. Biochem. 2017, 42, 44–54.
- Tavares, R.S.; Mansell, S.; Barratt, C.L.; Wilson, S.M.; Publicover, S.J.; Ramalho-Santos, J. p,p′-DDE activates CatSper and compromises human sperm function at environmentally relevant concentrations. Hum. Reprod. 2013, 28, 3167–3177.
- Zhang, X.; Kang, H.; Peng, L.; Song, D.; Jiang, X.; Li, Y.; Chen, H.; Zeng, X. Pentachlorophenol inhibits CatSper function to compromise progesterone’s action on human sperm. Chemosphere 2020, 259, 127493.
- Birch, M.R.; Johansen, M.; Skakkebaek, N.E.; Andersson, A.M.; Rehfeld, A. In vitro investigation of endocrine disrupting effects of pesticides on Ca(2+)-signaling in human sperm cells through actions on the sperm-specific and steroid-activated CatSper Ca(2+)-channel. Environ. Int. 2022, 167, 107399.
- Schiffer, C.; Müller, A.; Egeberg, D.L.; Alvarez, L.; Brenker, C.; Rehfeld, A.; Frederiksen, H.; Wäschle, B.; Kaupp, U.B.; Balbach, M.; et al. Direct action of endocrine disrupting chemicals on human sperm. EMBO Rep. 2014, 15, 758–765.
- Rehfeld, A.; Dissing, S.; Skakkebaek, N.E. Chemical UV Filters Mimic the Effect of Progesterone on Ca(2+) Signaling in Human Sperm Cells. Endocrinology 2016, 157, 4297–4308.
- Alavi, S.H.; Taghavi, M.M.; Moallem, S.A. Evaluation of effects of methamphetamine repeated dosing on proliferation and apoptosis of rat germ cells. Syst. Biol. Reprod. Med. 2008, 54, 85–91.
- Allaeian Jahromi, Z.; Meshkibaf, M.H.; Naghdi, M.; Vahdati, A.; Makoolati, Z. Methamphetamine Downregulates the Sperm-Specific Calcium Channels Involved in Sperm Motility in Rats. ACS Omega 2022, 7, 5190–5196.
- Wang, H.; Liu, J.; Cho, K.H.; Ren, D. A novel, single, transmembrane protein CATSPERG is associated with CATSPER1 channel protein. Biol. Reprod. 2009, 81, 539–544.
- He, Y.; Zou, Q.; Li, B.; Chen, H.; Du, X.; Weng, S.; Luo, T.; Zeng, X. Ketamine inhibits human sperm function by Ca(2+)-related mechanism. Biochem. Biophys. Res. Commun. 2016, 478, 501–506.
- Chen, Y.; Xu, W.; Yuan, Y.; Chen, H.; Zheng, S.; He, Y.; Luo, T. N-Methyl-d-aspartic Acid (NMDA) Receptor Is Involved in the Inhibitory Effect of Ketamine on Human Sperm Functions. Int. J. Mol. Sci. 2021, 22, 12370.
- Wright, C.; Milne, S.; Leeson, H. Sperm DNA damage caused by oxidative stress: Modifiable clinical, lifestyle and nutritional factors in male infertility. Reprod. Biomed. Online 2014, 28, 684–703.
- Badouard, C.; Ménézo, Y.; Panteix, G.; Ravanat, J.L.; Douki, T.; Cadet, J.; Favier, A. Determination of new types of DNA lesions in human sperm. Zygote 2008, 16, 9–13.
- Gonzalez-Marin, C.; Gosalvez, J.; Roy, R. Types, causes, detection and repair of DNA fragmentation in animal and human sperm cells. Int. J. Mol. Sci. 2012, 13, 14026–14052.
- Tremellen, K. Oxidative stress and male infertility—A clinical perspective. Hum. Reprod. Update 2008, 14, 243–258.
- Mohammadi, S.; Jalali, M.; Nikravesh, M.R.; Fazel, A.; Ebrahimzadeh, A.; Gholamin, M.; Sankian, M. Effects of Vitamin-E treatment on CatSper genes expression and sperm quality in the testis of the aging mouse. Iran. J. Reprod. Med. 2013, 11, 989–998.
- Mohammadi, S.; Movahedin, M.; Mowla, S.J. Up-regulation of CatSper genes family by selenium. Reprod. Biol. Endocrinol. 2009, 7, 126.
