Allergic diseases are a set of chronic inflammatory disorders of lung, skin, and nose epithelium characterized by aberrant IgE and Th2 cytokine-mediated immune responses to exposed allergens. The prevalence of allergic diseases, including asthma, allergic rhinitis, and atopic dermatitis, has increased dramatically worldwide in the past several decades. Evidence suggests that diet and nutrition play a key role in the development and severity of allergic diseases. Dietary components can differentially regulate allergic inflammation pathways through host and gut microbiota-derived metabolites, therefore influencing allergy outcomes in positive or negative ways. A broad range of nutrients and dietary components (vitamins A, D, and E, minerals Zn, Iron, and Se, dietary fiber, fatty acids, and phytochemicals) are found to be effective in the prevention or treatment of allergic diseases through the suppression of type 2 inflammation.
- allergy
- allergic inflammation
- asthma
- allergic rhinitis
- atopic dermatitis
- dietary lipids
- dietary fiber
- dietary flavonoids
1. Introduction
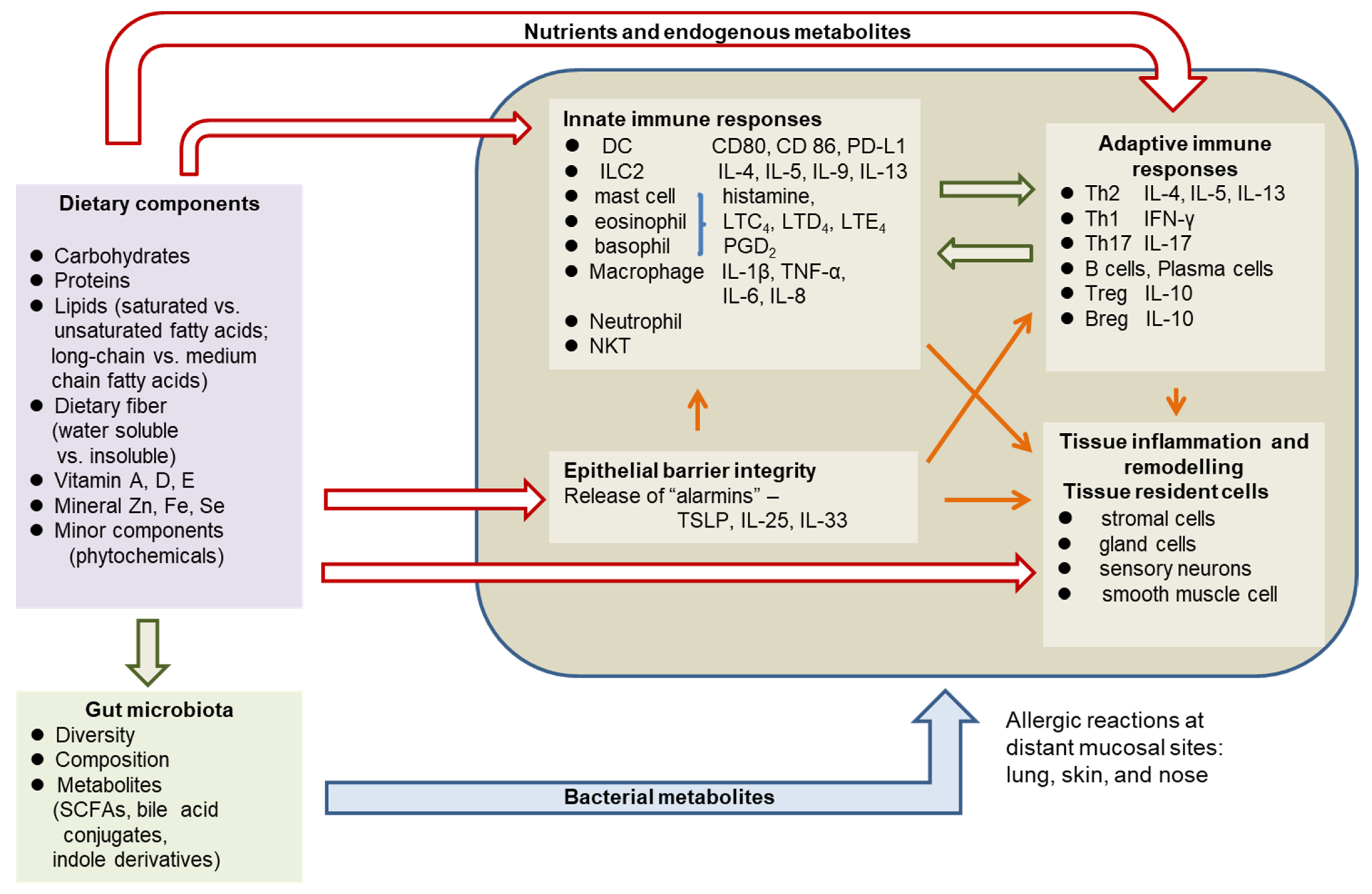
2. Pathophysiology of Allergic Diseases
3. The Role of Diet and Nutritional Status in Allergy
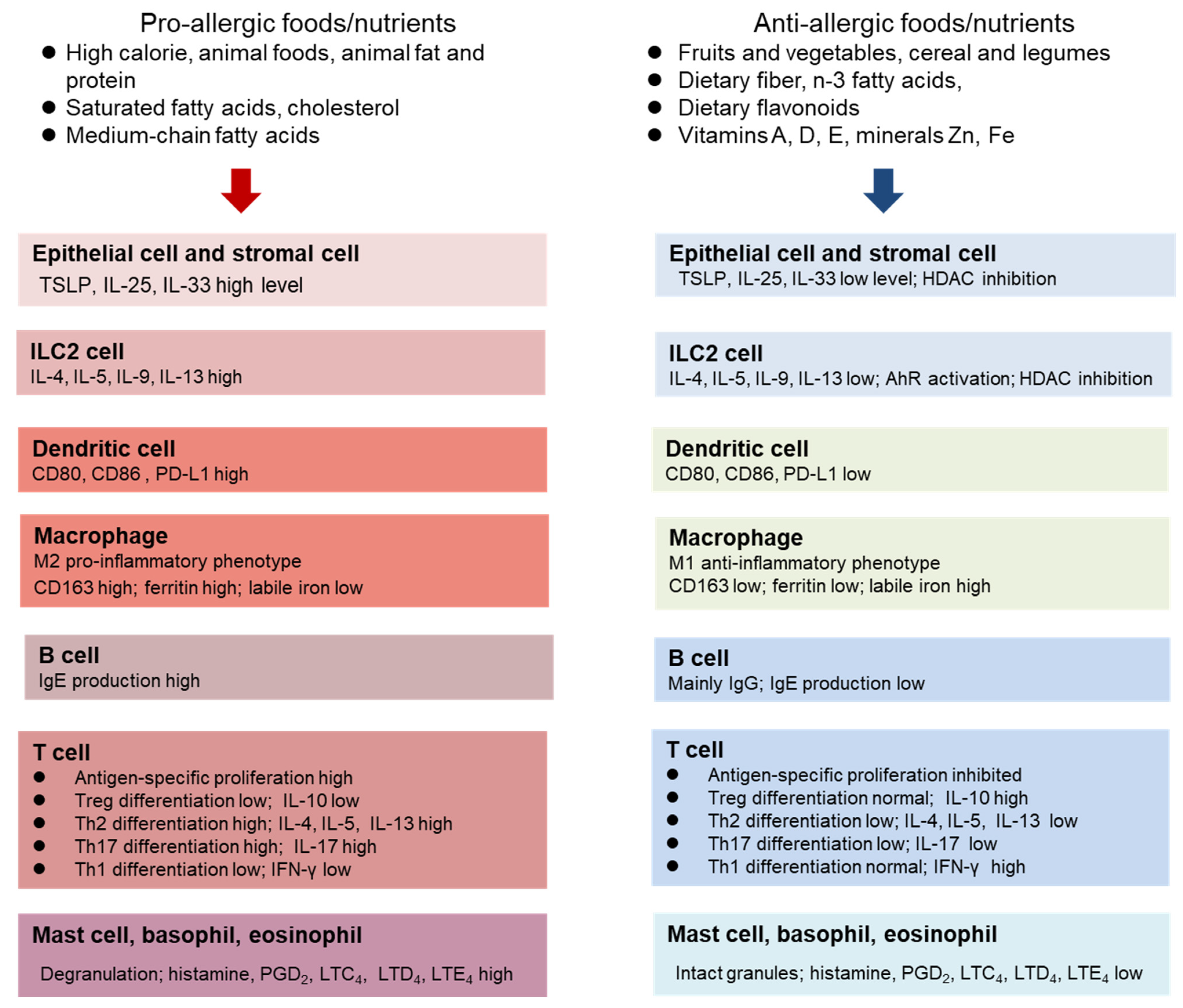
Dietary factors not only affect the development of allergic diseases [5,6,46,50][5][6][26][27] but also influence disease course and severity [50,58][27][28]. Different dietary components are related to differential allergy outcomes. The intake of high energy, high saturated fat, high protein, and low fiber increases the risks of asthma and AR [6,46][6][26]. In contrast, high consumption of vegetables and fruits, olive oil, and fish, characteristic of a Mediterranean diet, has been linked with lower risks of asthma and AR [5,7,8,46,59][5][7][8][26][29]. Recent evidence suggests that higher dietary fiber intake is associated with fewer asthma symptoms [58][28]. Moreover, adequate intake of micronutrients is associated with a lower risk of atopic diseases and reduction of symptoms [50][27]. The identified diet and nutritional risk factors for allergy are listed below:- High energy
- High protein
- High saturated fat, n-6 fatty acids, medium-chain fatty acids, cholesterol
- Low total dietary fiber
- Low vegetables and fruits
- High simple sugar and processed foods
- Low level of Zn, Fe, Vitamins A, D, E
3.1. Dietary protein, Amino Acids, and Energy
3.2. Dietary Lipids
The amount of dietary lipids and type of fatty acids influence allergic inflammation. High total fat, animal fat, saturated fatty acids (SFAs), cholesterol, n-6 polyunsaturated fatty acids (PUFAs), and medium-chain fatty acids (MCFs) are risk factors, whereas monounsaturated fatty acids (MUFAs) and n-3 PUFAs have protective properties. High animal fat and SFAs are associated with allergic rhinitis in human adults while high MUFA intake is associated with a lower risk for asthma [46,59][26][29]. A high-fat diet (60% Kcal from saturated fat) has been shown to increase serum TSLP in C57BL/6 mice and exacerbate dermatitis in mice through upregulation of TSLP in NC/Nga mice that develop AD spontaneously [18][39]. The high-fat diet increased TSLP in dorsal skin, infiltration of inflammatory cells, and epidermal thickening in NC/Nga mice compared with a low-fat diet. Dermatitis score was much lower in high-fat-fed NC-TSLP-KO mice, suggesting TSLP mediates a high-fat-diet-induced increase in dorsal skin inflammation [18][39]. Long-term feeding (10 months since weaning) of a Western diet (21.2% fat, 34% sucrose, and 0.2% cholesterol) also substantially increased spontaneously developed dermatitis in aged C57BL/6 mice, as compared with a control diet (5.2% fat, 12% sucrose, and 0.01% cholesterol) [71][40]. The Western diet-fed mice had increased epidermal thickness in their dorsal skin and much more epidermal hyperplasia in the lesion skin, with hypergranulosis and spongiosis typical of AD [71][40]. The Western diet leads to increased total bile acids, altered bile acid profiles, and elevated bile acid signaling through two bile acid receptors TGR5 (transmembrane G-protein-coupled receptor-5) and S1PR2 (sphingosine-1-phosphate receptor-2) in the lesion skin [71][40]. Lowering serum cholesterol with a bile acid sequestrant cholestyramine reduced epidermal hyperplasia and decreased Th2 and Th17 cytokines [71][40]. Besides saturated fatty acids and cholesterol, medium-chain fatty acids (MCFs) contained in coconut oil or palm oil also prove to be a dietary risk factor for allergy [5]. In a mouse model of peanut allergy, compared with n-6 PUFAs from peanut oil, MCFs decreased dietary peanut or OVA antigen absorption into the circulation and increased antigen in the Peyer’s patches, which resulted in a significant increase in activated DC cells [72][41]. The phospholipids isolated from asparagus (Asparagus officinalis L.) are demonstrated to have anti-allergic properties. Oral administration of these phospholipids suppressed serum total IgE and OVA-specific IgE in OVA-challenged mice and ameliorated clinical scores of AD induced by picryl chloride in NC/Nga mice [73][42]. Phospholipid and glycolipid fractions from asparagus also potently inhibited β-hexosaminidase release from cultured RBL-2H3 (rat basophilic leukemia-histamine-releasing cell line) cells, indicating a direct effect on degranulation in allergic responses [73][42]. Although conflicting results are generated from human studies about the effects of long-chain PUFA supplementation on asthma, AR, and AD [74][43], animal studies provide clear evidence of the protection of dietary n-3 PUFA in these allergic conditions. Dietary n-3 fatty acid α-linolenic acid shows beneficial effects in allergic inflammation by improving skin barrier function in AD mice [75][44] and attenuating symptoms in OVA-induced AR in mice, as compared with n-6 fatty acid linoleic acid [76][45]. Dietary linseed oil (enriched with α-linolenic acid) increases EPA-derived metabolite 15-HEPE (hydroxyeicosapentaenoic acid in eosinophils) in eosinophils in the nasal passage, which inhibits mast cell degranulation by binding to PPAR (peroxisome proliferator-activated receptor) γ [76][45].3.3. Dietary Fiber
Recent animal studies show that dietary fiber protects against AD or allergic asthma through its bacterial metabolites short-chain fatty acids, particularly butyrate [14,82,83][14][46][47]. Gut microbiota fermentation of dietary fiber into SCFAs is the key to the gut–skin axis or gut–lung regulation of allergic reactions in the skin and lungs. Consistent with animal studies, dysbiosis characterized by the enrichment of Faecalibacterium prausnitzii and a reduced capacity for butyrate fermentation in the human gut microbiome has been found in patients with AD [84][48]. Gut microbiota-derived butyrate has been found to be inversely associated with mite-specific IgE levels in childhood asthma [85][49]. Short-chain fatty acids, particularly butyrate, regulate type 2 inflammation mainly through the inhibition of HDAC (histone deacetylase) on various immune cells and structural cells. Vancomycin treatment in mice results in dramatic alterations in the gut microbiome characterized by decreased richness, diversity, and decreased abundance of butyrate-producing families, leading to increased susceptibility to allergic inflammation [83][47]. A supplement of SCFA in drinking water attenuated OVA or papain-induced allergic asthma by suppression of DC activation and trafficking, therefore restraining Th2 cell development in Peyer’s patches [83][47]. Butyrate also directly regulates ILC2 cells by suppressing IL-33-induced IL-13 and IL-5 production in cultured ILC2 lung cells from Rag2−/− (recombination-activating gene 2 deficient) mice who lack T cells [24][50]. When administered either through drinking water or through an intranasal route, butyrate ameliorated ILC2 cell-driven lung inflammation. The inhibitory effect of butyrate on ILC2 cell proliferation was due to histone deacetylase (HDAC) inhibition [24][50].3.4. Dietary Flavonoids and Other Phytochemicals
Flavonoids are a major type of phytochemicals in the diet and are naturally occurring phenolic compounds which are commonly found in fruits, vegetables, herbs and spices, legumes, tea, and vinegar [96,97][51][52]. There are six subclasses of dietary flavonoids based on their chemical structures, namely flavanols, flavones, isoflavones, flavanones, flavonols, and anthocyanidin [96,97][51][52]. Accumulating evidence has shown the anti-allergic effect of dietary flavonoids. As a major dietary flavonol-type flavonoid, quercetin is found in many fruits and vegetables including onions, shallots, apples, berries, tea, tomatoes, grapes, nuts, and seeds. The anti-inflammatory effect of quercetin is well documented in various animal models of allergy [98][53]. Quercetin is effective in reducing allergic symptoms by decreasing serum IgE and Th2-related cytokines, reducing eosinophil, neutrophil, and mast cell infiltration into local tissue, reducing epithelial thickness in the lung and hyperkeratosis, and suppressing epithelial cell-derived cytokines IL-25, IL-33, and TSLP [98][53]. However, in most in vivo animal studies, quercetin is administered through i.p. injection. As quercetin is a glycone (namely, carbohydrate conjugate), how dietary quercetin is metabolized by the gut microbiota and the subsequent effects on allergic inflammation remain to be explored. In a recent study, oral administration of quercetin was shown to attenuate nasal symptoms of OVA-induced AR in BALB/c (Halsey J Bagg albino mice strain c) mice by suppressing angiogenic factors and proinflammatory cytokines TNF-α, IL-6, and IL-8 in nasal lavage fluids [99][54]. The minimum effective dose for the above in vivo inhibition is similar to the maximum daily recommended dosage for dietary quercetin supplements. Kaempferol, another flavonol-type flavonoid found in many fruits, vegetables, herbs, teas, and medicinal plants, also exhibits anti-inflammatory, antioxidant, and anti-allergic properties. In cultured lung epithelial BEAS-2B (human broncho-epithelial-alveolar stem cell-derived cells) cells, nontoxic kaempferol suppresses LPS (lipopolysaccharide)-induced TGF-β production, TGF-β-induced myofibroblast formation, LPS-induced collagen, and MT1-MMP, suggesting its ability to suppress the epithelial-to-mesenchymal transition and fibrosis. In a mouse model of asthma, orally administered kaempferol not only suppressed eosinophil infiltration and airway inflammation but also inhibited the airway epithelial-to-mesenchymal transition (EMT) and fibrosis [53][55]. As fibrotic airway remodeling is characteristic of asthma, leading to lung function deterioration, and is not treated by current drug therapy, kaempferol may be a potential therapy for asthma-related airway construction and is worthy of further clinical studies. Kaempferol also protects mice against AD by suppressing T cell activation though interaction with MRP-1 [101][56]. Oral administration of naringenin, a flavanone mostly found in citrus peel, was shown to significantly reduce nasal scratching score in rats with OVA-induced AR with improved histology in the nasal epithelium and decreased serum IgE, IL-4, and IL-5 [103][57]. In addition, naringenin inhibited TSLP production in PMA/Ionophore-activated human mast cells (HMC-1 cells) through inhibition of NF-κB and TSLP-induced mRNA expressions of IL-13, TNF-α, IL-17 receptors, and TSLP receptors in these cells [102][58]. The gut microbiota-derived metabolites are critical for the anti-allergic function of some flavonoids. For example, the flavone glycoside diosmin and its aglycone form diometin were shown to diminish DNCB-induced AD symptoms in SKH-1 hairless mice, such as increased trans-epidermal water loss and hydration, epidermal thickness, and infiltration of mast cells [103][57]. Decreased serum IgE and IL-4 in these mice were observed for both diosmin and diometin; however, in cultured RBL-2H3 cells, only diosmetin and not diosmin showed inhibitory effects on IL-4 production. Some dietary phytochemicals other than flavonoids also exhibit strong anti-allergic properties. Licoricidin, a component isolated from licorice (Glycyrrhiza uralensis) root which is a commonly used herb in traditional medicine, shows protection against mouse AD by suppression of T cell activation through regulating PTPN1 activity [105][59]. Resveratrol, the best-studied polyphenol, inhibits mast cell activation and shows potential in treating allergic conditions [107][60].3.5. Vitamins and Minerals
Vitamins and minerals have long been known for their immunomodulatory roles. Vitamins A, D, and E, and trace elements zinc and iron, are particularly important dietary factors, influencing allergic inflammation and the development of allergic diseases. Sufficient intake of Vitamins A, D, and E is required to control asthma [5]. Supplementation with vitamins E and D alone or in combination improves symptom management of AD [116][61]. Serum vitamin D level is a determining factor in remission with standard therapy for AD. A serum level of 1, 25(OH)2VD3 higher than 20 ng/mL plus standard therapy is sufficient to reduce the severity of AD [115][62]. In a randomized, double-blind, placebo-controlled clinical study, an oral supplement of 5000 IU/day vitamin D3 in patients with AD significantly increases the serum level of 1, 25(OH)2VD3 to a much higher level than the placebo group, and this dosage achieved sufficiency in 100% of the patients [115][62]. Vitamin D also shows potential in managing airway remodeling in asthma, based on a number of in vitro studies showing the inhibitory effects of vitamin D on bronchial smooth muscle cells, human airway smooth muscle cells, human asthmatic bronchial fibroblasts, and human bronchial fibroblasts [54][63]. The trace element zinc is essential for immune function. Zinc deficiency is often linked to allergies. A zinc supplement is shown to be effective in relieving asthma but not beneficial to AD [50,57][27][64]. In an animal asthma model, zinc deficiency is related to greater airway hyper-responsiveness compared with normal zinc intake, whereas zinc supplementation reduces inflammatory cell infiltration and improves clinical symptoms [120][65]. At the cellular level, the beneficial impact of zinc on allergic immune reactions mainly includes T cell differentiation and antigen-specific T cell proliferation. In cultured human PBMCs (peripheral blood mononuclear cells), zinc deficiency increases Th17 differentiation [121][66]. On the other hand, the zinc supplement in the cell culture of allergen-stimulated PBMCs alters the Th1/Th2 ratio and decreases the proportion of Th17 [122][67]. Zinc supplementation also enhances Treg differentiation either in allergen-stimulated PBMCs from atopic patients [37][68] or in TGF-β treated PBMCs and mixed lymphocyte cultures [36][69]. Iron is another trace element that has been linked to the etiology of atopic diseases [123][70]. As the most common nutritional disorder, iron deficiency is associated with half anemia which affects about a third of the world’s population [124][71]. Iron deficiency can be present either as low hemoglobin levels in the blood or with low levels of metabolically active iron despite normal ferritin iron storage in the body [123][70]. While the majority of the iron requirement in the human body is met by recycling from senescent red blood cells by splenic macrophages and redistribution to other cells, dietary intake of iron provides only about one-tenth of the daily requirement [123][70]. Therefore, the macrophage regulation of the iron pool and metabolism is highly important, which determines the activation state of the immune system. Copper is closely related to iron metabolism. The copper-containing ferroxidase ceruloplasmin is involved with iron mobilization during acute inflammation, and its elevation indicates iron deficiency [117,123][70][72]. A recent clinical study in Japan showed that multiple nutritional and gut microbial factors are associated with AR [48][73]. Four nutrients (retinol, vitamin A, cryptoxanthin, and copper) were negatively associated with AR [48][73]. In a cohort study in Poland (n = 80), the plasma level of Cu was found to be associated with AR in children aged 9–12 [125][74]. Selenium is an essential trace element that is very important for optimal immune function. Populations from China, the UK, and Scandinavia generally tend to have reduced Se levels [126][75]. While Se deficiency leads to impaired immune responses, Se supplements boost immune competence. Selenium is an essential component of glutathione peroxidase (GSH-Px), a key antioxidant enzyme that functions to reduce peroxides, therefore protecting against inflammation-induced, excessive oxidative stress-related membrane damage [127][76]. While a lower serum level of selenium is reported to be associated with an increased risk of asthma in human studies [128[77][78],129], an animal study demonstrated that a lower level of selenium is associated with a lower asthma outcome. Although adequate dietary intake of selenium does not protect against the development of allergic asthma in mice, dietary selenium supplements have a synergistic anti-asthma effect with vitamin E in reducing airway inflammation and Th2-related cytokines [118][79].4. Obesity and Allergy
Dietary interventions producing weight loss in obese patients have been shown to be effective in improving asthma control [137][80]. Randomized controlled trials on dietary intervention showed that weight loss through restrictive diets with low energy is effective in improving asthma outcomes [138][81] and reducing airway inflammation in obese patients [139][82]. Even a normal caloric diet with a reduced content of fat, particularly saturated fat, was associated with reduced body weight and improvement of asthma-related quality of life in obese pubertal adolescents [140][83]. Although there are very limited studies, weight loss is associated with improved symptoms in atopic dermatitis.
Plant-based diets are effective for weight loss [143,144,145][84][85][86] and can be an effective strategy for weight control, as well as in the treatment of obesity [145][86]. A plant-based vegan diet excludes all animal products, mainly consisting of grains, legumes, and vegetables and fruits; while in comparison, a vegetarian diet does not eliminate all animal products but emphasizes the consumption of fruits, vegetables, and nuts [145][86]. The weight reduction effect of such diets may be attributed to reduced calories and low fat intake [145][86]. Plant protein, as part of a plant-based diet, has recently been shown to be a contributing factor for weight control in overweight individuals [143][84]. An increased intake of protein and a decreased intake of animal protein are associated with a decrease in body fat mass. Plant-based diets are nutritionally adequate if planned well [144][85]. However, nutrient intake in the long term can be a concern, as revealed in a study of the weight-loss effects of a vegan diet in overweight postmenopausal women. The adoption of a low-fat vegan diet for 14 weeks leads to changes in macronutrients such as decreased intake of total fat, saturated fat and cholesterol, protein, and increased carbohydrate and fiber intake [144][85]. In terms of micronutrients, the vegan diet increased intakes of total vitamin A, β-carotene, thiamine, vitamin B6, folic acid, vitamin C, magnesium, and potassium, but decreased intakes of vitamin D, vitamin B12, calcium, phosphorous, selenium, and zinc [144][85]. Fortified food or supplements may help those following a vegan diet to meet the requirements of micronutrient intakes.
5. Conclusions
In conclusion, diet and nutrition play a key role in the development and severity of allergic diseases by regulating tissue and immune homeostasis. Excessive calories, high intake of protein and saturated fatty acids, or lack of dietary fiber and micronutrients can trigger the defense mechanism in the immune system and prime the host for allergic reactions. Therefore, calorie restriction, coupled with sufficient dietary fiber and adequate macronutrient intake, will be essential for maintaining immune tolerance to allergens. The plant-based diets, which emphasize the high consumption of fruits and vegetables, grains, and legumes while avoiding or reducing animal foods, are associated with the reduction of inflammation and weight loss. Further dietary intervention studies are warranted to explore the potential beneficial effects of plant-based diets and the specific nutrients related to such diets on allergic outcomes. As basic research efforts identify more novel dietary components with anti-allergic properties, randomized placebo-controlled trials are also needed to verify their efficacy in human patients. Nutritional therapy holds great promise in reducing allergy symptoms, either as primary therapy and treatment or in support of drug therapy. Assessment of nutritional status and anthropometric characteristics of the patients, and analysis of host and gut microbiota by the multi-omics approach, will be important in future clinical trials to identify novel mechanisms linking nutrition and allergy.References
- Undem, B.J.; Taylor-Clark, T. Mechanisms underlying the neuronal-based symptoms of allergy. J. Allergy Clin. Immunol. 2014, 133, 1521–1534.
- Holgate, S.T.; Wenzel, S.; Postma, D.S.; Weiss, S.T.; Renz, H.; Sly, P.D. Asthma. Nat. Rev. Dis. Primers 2015, 1, 15025.
- Bousquet, J.; Anto, J.M.; Bachert, C.; Baiardini, I.; Bosnic-Anticevich, S.; Canonica, C.W.; Melén, E.; Palomares, O.; Scadding, G.K.; Togias, A.; et al. Allergic rhinitis. Nat. Rev. Dis. Primers 2020, 6, 95.
- Weidinger, S.; Novak, N. Atopic dermatitis. Lancet 2016, 387, 1109–1122.
- Julia, V.; Macia, L.; Dombrowicz, D. The impact of diet on asthma and allergic diseases. Nature 2015, 15, 308–322.
- Lin, Y.P.; Kao, Y.C.; Pan, W.H.; Yang, Y.H.; Chen, Y.C.; Lee, Y.L. Associations between respiratory diseases and dietary patterns derived by factors analysis and reduced rank regression. Ann. Nutr. Metab. 2016, 68, 306–314.
- Netting, M.J.; Middleton, P.F.; Markrides, M. Does maternal diet during pregnancy and lactation affect outcomes in offspring? A systemic review of food-based approaches. Nutrition 2014, 30, 1225–1241.
- Thorburn, A.N.; Macia, L.; Mackay, C.R. Diet, metabolites, and “Western-lifestyle” inflammatory diseases. Immunity 2014, 40, 833–842.
- Pascal, M.; Perez-Gordo, M.; Caballero, T.; Escribese, M.M.; Longo, M.N.L.; Luerigo, O.; Manso, L.; Matheu, V.; Seoane, E.; Zamorano, M.; et al. Microbiome and allergic diseases. Front. Immunol. 2018, 9, 1584.
- McKenzie, C.; Tan, J.; Macia, L.; Mackay, C.R. The nutrition-gut microbiome-physiology axis and allergic diseases. Immunol. Rev. 2017, 278, 277–295.
- Sugihara, K.; Kamada, N. Diet-microbiota interactions in inflammatory bowel disease. Nutrients 2021, 13, 1533.
- Zhang, P. Influence of foods and nutrition on the gut microbiome and implications for intestinal health. Int. J. Mol. Sci. 2022, 23, 9588.
- Arifuzzaman, M.; Won, T.H.; Li, T.T.; Yano, H.; Digumarthi, S.; Heras, A.F.; Zhang, W.; Parkhurst, C.N.; Kashyap, S.; Jin, W.B.; et al. Inulin fiber promotes microbiota-derived bile acids and type 2 inflammation. Nature 2022, 611, 578–584.
- Trompette, A.; Pernot, J.; Perdijk, O.; Alqahtani, R.A.A.; Domingo, J.S.; Camacho-Muñoz, D.; Wong, N.C.; Kendall, A.C.; Wiederkehr, A.; Nicod, L.P.; et al. Gut-derived short-chain fatty acids modulate skin barrier integrity by promoting keratinocyte metabolism and differentiation. Mucosal Immunol. 2022, 15, 908–926.
- Palomares, O.; Akdis, M.; Martin-Frontecha, M.; Akdis, C.A. Mechanisms of immune regulation in allergic diseases: The role of regulatory T and B cells. Immunol. Rev. 2017, 278, 219–236.
- Roan, F.; Obata-Ninomiya, K.; Ziegler, S.F. Epithelial cell-derived cytokines: More than just signaling the alarm. J. Clin. Investig. 2019, 129, 1441–1451.
- Dahlgren, M.W.; Jones, S.W.; Cautivo, K.M.; Dubinin, A.; Oritiz-Carpena, J.F.; Farhat, S.; Yu, K.S.; Lee, K.; Wang, C.Q.; Molofsky, A.V.; et al. Adventitial stromal cells define group 2 innate lymphoid cell tissue niches. Immunity 2019, 50, 702–722.
- Klose, C.S.N.; Artis, D. Innate lymphoid cells control signaling circuits to regulate tissue-specific immunity. Cell Res. 2020, 30, 475–491.
- Kiss, E.A.; Vonarbourg, C.; Kopfmann, S.; Hobeika, E.; Finke, D.; Esser, C.; Diefenbach, A. Natural aryl hydrocarbon receptor ligands control organogenesis of intestinal lymphoid follicles. Science 2011, 334, 1561–1565.
- Li, S.; Bostick, J.W.; Ye, J.; Qiu, J.; Zhang, B.; Urban, J.F.; Auram, D.; Zhou, L. Aryl hydrocarbon receptor signaling cell intrinsically inhibits intestinal group innate lymphoid cell function. Immunity 2018, 49, 915–928.
- Van der Marel, A.P.J.; Samsom, J.N.; Greuter, M.; van Berkel, L.A.; O’Toole, T.; Kraal, G.; Mebius, R.E. Blockade of IDO inhibits nasal tolerance induction. J. Immunol. 2007, 179, 894–900.
- Ünüvar, S.; Erge, D.; Kiliçarslan, B.; Bağ, H.G.G.; Çatal, F.; Girgin, G.; Baydar, T. Neopterin levels and indoleamine 2,3-dioxygenase activity as biomarkers of immune system activation and childhood allergic diseases. Ann. Lab. Med. 2019, 39, 284–290.
- Bousquet, J.; Khaltaev, N.; Cruz, A.A.; Denburg, J.; Fokkens, W.J.; Togias, A.; Zuberbier, T.; Baena-Cagnani, C.E.; Canonica, G.W.; van Weel, C.; et al. AR and its impact on asthma (ARIA) 2008 update. Allergy 2008, 63 (Suppl. S86), 8–160.
- Leung, D.Y.M.; Berdyshev, E.; Gloeva, E. Cutaneous barrier dysfunction in allergic diseases. J. Allergy Clin. Immnol. 2020, 145, 1485–1497.
- Kraft, M.T.; Prince, B.T. AD is a barrier issue, not an allergy issue. Immunol. Allergy Clin. N. Am. 2019, 39, 507–519.
- Urrutia-Pereira, M.; Mocelin, L.P.; Ellwood, P.; Garcia-Marcos, L.; Simon, L.; Rinelli, P.; Chong-Neto, H.J.; Solé, D. Prevalence of rhinitis and associated factors in adolescents and adults: A global asthma network study. Rev. Paul. Pediatr. 2023, 41, e2021400.
- Peroni, D.G.; Hufnagl, K.; Comberiati, P.; Roth-Walter, F. Lack of iron, zinc, and vitamins as contributor to the etiology of atopic diseases. Front. Nutr. 2023, 9, 1032481.
- Andrianasolo, R.M.; Hercberg, S.; Kesse-Guyot, E.; Druesne-Pecollo, N.; Touvier, M.; Galan, P.; Varraso, R. Association between dietary fiber intake and asthma (symptoms and control) results from the French national e-cohort NutriNet-Santé. Brit. J. Nutr. 2019, 122, 1040–1051.
- Cazzoletti, L.; Zanolin, M.E.; Speita, F.; Bono, R.; Chamitava, L.; Cerveri, I.; Garcia-Larsen, V.; Grosso, A.; Mattioli, V.; Pirina, P.; et al. Dietary fats, olive oil and respiratory diseases in Italian adults: A population-based study. Clin. Exp. Allergy 2019, 49, 799–807.
- Zhou, Y.J.; Li, L.S.; Sun, J.L.; Guan, K.; Wei, J.F. 1H NMR-based metabolomics study of metabolic profiling for pollinosis. World Allergy Org. J. 2019, 12, 100005.
- Ma, G.C.; Wang, T.S.; Wang, J.; Ma, Z.J.; Pu, S.B. Serum metabolomics of patients with AR. Biomed. Chromatogr. 2020, 34, e4739.
- Yoshino, K.; Sakai, K.; Okada, H.; Sakai, T.; Yamamoto, S. IgE responses in mice fed moderate protein deficient and high protein diets. J. Nutr. Sci. Vitaminol. 2003, 49, 172–178.
- Fan, W.Y.; Kouda, K.; Nakamura, H.; Takeuchi, H. Effects of dietary restriction on spontaneous dermatitis in NC/Nga mice. Exp. Biol. Med. 2001, 226, 1045–1050.
- Kositz, C.; Schroecksnadel, K.; Grander, G.; Schennach, H.; Kofler, H.; Fuchs, D. Serum tryptophan concentration in patients predicts outcome of specific immunotherapy with pollen extracts. Int. Arch. Allergy Immunol. 2008, 147, 35–40.
- Licari, A.; Fuchs, D.; Marseglia, G.; Ciprandi, G. Tryptophan metabolic pathway and neopterin in asthmatic children in clinical practice. Ital. J. Pediatr. 2019, 45, 11.
- Luukkainen, A.; Karjalainen, J.; Hurme, M.; Paavonen, T.; Toppila-salmi, S. Relationships of indoleamine 2,3-dioxygenase activity and cofactors with asthma and nasal polyps. Am. J. Rhinil. Allergy 2014, 28, e5–e10.
- Gostner, J.M.; Becker, K.; Kofler, H.; Strasser, B.; Fuchs, D. Tryptophan metabolism in allergic disorders. Int. Arch. Allergy Immunol. 2016, 169, 203–215.
- Cruzat, V.; Rogero, M.M.; Keane, K.N.; Curi, R.; Newshoime, P. Glutamine: Metabolism and immune function, supplementation and clinical transition. Nutrients 2018, 10, 1564.
- Moon, P.D.; Han, N.R.; Kim, H.M.; Jeong, H.J. High-fat diet exacerbates dermatitis through up-regulation of TSLP. J. Investig. Dermatol. 2019, 139, 1198–1201.
- Jena, P.K.; Sheng, L.; McNeil, K.; Chau, T.Q.; Yu, S.; Kiuru, M.; Fung, M.A.; Hwang, S.T.; Wan, Y.J.Y. Long-term western diet intake leads to dysregulated bile acid signaling and dermatitis with Th2 and Th17 pathway features in mice. J. Dermatol. Sci. 2019, 95, 13–20.
- Li, J.; Wang, Y.; Tang, L.; de Villiers, W.J.S.; Cohen, D.; Woodward, J.; Finkelman, F.D.; Eckhardt, E.R.M. Dietary medium-chain triglycerides promote oral allergic sensitization and orally induced anaphylaxis to peanut protein in mice. J. Allergy Clin. Immunol. 2013, 131, 442–450.
- Iwamoto, A.; Hamajima, H.; Tsuge, K.; Tsuruta, Y.; Nagata, Y.; Yotsumoto, H.; Yanagita, T. Inhibitory effects of green asparagus extract, especially phospholipids, on allergic responses in vitro and in vivo. J. Agric. Food Chem. 2020, 68, 15199–15207.
- Radzikowska, U.; Rinaldi, A.O.; Sözener, Z.Ç.; Karaguzel, D.; Wojcik, M.; Cypryk, K.; Akdis, M.; Akdis, C.A.; Sokolowska, M. The influence of dietary fatty acids on immune responses. Nutrients 2019, 11, 2990.
- Fujii, M.; Nakashima, J.; Tomozawa, J.; Shimazaki, Y.; Ohyanagi, N.; Kawaguchi, S.; Ohya, S.; Kohno, S.; Nabe, T. Deficiency of n-6 polyunsaturated fatty acids is mainly responsible for AD-like pruritic skin inflammation in special diet-fed hairless mice. Exp. Dermatol. 2013, 22, 272–277.
- Sawane, K.; Nagatake, T.; Hosomi, K.; Hirata, S.; Adachi, J.; Abe, Y.; Isoyama, J.; Suzuki, H.; Matsunaga, A.; Kunisawa, J.; et al. Dietary omega-3 fatty acid dampens AR via eosinophilic production of the anti-allergic lipid mediator 15-hydroxyeicosapentaenoic acid in mice. Nutrients 2019, 11, 2868.
- Trompette, A.; Gollwitzer, E.S.; Yadava, K.; Sichelstiel, A.K.; Sprenger, N.; Ngom-Bru, C.; Blanchard, C.; Junt, T.; Harris, N.L.; Marsland, B.J.; et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat. Med. 2014, 20, 159–166.
- Cait, A.; Huges, M.R.; Antignano, F.; Cait, T.; Dimitriu, P.A.; Maas, K.R.; Reynolds, L.A.; Hacker, L.; Mohr, J.; Finlay, B.B.; et al. Microbiome-driven allergic lung inflammation is ameliorated by short-chain fatty acids. Mucosal Immunol. 2018, 11, 785–795.
- Song, H.; Yoo, Y.; Hwang, J.; Na, Y.C.; Kim, H.S. Faecalibacterium prausnitzii subspecies-level dysbiosis in the human gut microbiome underlying AD. J. Allergy Clin. Immunol. 2016, 137, 852–860.
- Chiu, C.Y.; Cheng, M.L.; Chiang, M.H.; Kuo, Y.L.; Tsai, M.H.; Chiu, C.C.; Lin, G. gut microbial-derived butyrate is inversely associated with IgE responses to allergens in childhood asthma. Pediatr. Allergy Immunol. 2019, 30, 689–697.
- Thio, C.L.P.; Chi, P.Y.; Lai, A.C.Y.; Chang, Y.J. Regulation of type 2 innate lymphoid cell-dependent airway hyperreactivity by butyrate. J. Allergy Clin. Immunol. 2018, 142, 1867–1883.
- Rakha, A.; Umar, N.; Rabail, R.; Butt, M.S.; Kieliszek, M.; Hassoun, A.; Aadil, R.M. Anti-inflammatory and anti-allergic potential of dietary flavonoids. Biomed. Pharmacother. 2022, 156, 113945.
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124.
- Jafarinia, M.; Hosseini, M.S.; Kasiri, N.; Fazel, N.; Fathi, F.; Hakemi, M.G.; Eskandari, N. Quercetin with the potential on allergic diseases. Allergy Asthma Clin. Immunol. 2020, 16, 36.
- Okumo, T.; Furuta, A.; Kimura, T.; Yusa, K.; Asano, K.; Sunagawa, M. Inhibition of angiogenic factor productions by quercetin in vitro and in vivo. Medicines 2021, 8, 22.
- Gong, J.H.; Cho, I.H.; Shin, D.; Han, S.Y.; Park, S.H.; Kang, Y.H. Inhibition of airway epithelial-to-mesenchymal transition and fibrosis in endotoxin-induced epithelial cells and ovalbumin-sensitized mice. Lab. Investig. 2014, 94, 297–308.
- Lee, H.S.; Jeong, G.S. Therapeutic effect of kaempferol on AD by attenuation of T cell activity via interaction with multidrug-associated protein. Br. J. Pharmacol. 2021, 178, 1772–1788.
- Park, S.; Bong, S.K.; Lee, J.W.; Park, N.J.; Choi, Y.; Kim, S.M.; Yang, M.H.; Kim, Y.K.; Kim, S.N. Diosmetin and its glycoside, diosmin, improves AD-like lesions in 2,4-dinitrochlorobenzene-induced murine models. Biomol. Ther. 2020, 28, 542–548.
- Sahin, A.; Sakat, M.S.; Kilic, K.; Aktan, B.; Yildirim, S.; Kandemir, F.M.; Dortbudak, M.B.; Kucukler, S. The protective effect of naringenin against ovalbumin-induced AR in rats. Eur. Arch. Oto-Rhino-Laryngol. 2021, 278, 4839–4846.
- Lee, H.S.; Kim, J.; Choi, H.G.; Kim, E.K.; Jun, C.D. Licoricidin abrogates T-cell activation by modulating PTPN1 activity and attenuates AD in vivo. J. Investig. Dermatol. 2021, 141, 2490–2498.
- Civelek, M.; Bilotta, S.; Lorentz, A. Resveratrol attenuates mast cell mediated allergic reactions: Potential for use as a nutraceutical in allergic diseases. Mol. Nutr. Food Res. 2022, 66, 2200170.
- Reynolds, K.A.; Juhasz, M.L.W.; Mesinkovska, N.A. The role of oral vitamins and supplements in the management of AD: A systematic review. Int. J. Dermatol. 2019, 58, 1371–1376.
- Sanchez-Armendariz, K.; Garcia-Gil, A.; Romero, C.A.; Contreras-Ruiz, J.; Karam-Orante, M.; Balcazar-Antonio, D.; Dominguez-Cherit, J. Oral vitamin D3 5000 IU/day as an adjuvant in the treatment of AD: A randomized control trial. Int. J. Dermatol. 2018, 57, 1516–1520.
- The role of vitamin D supplementation on airway remodeling in asthma: A systemic review. Nutrients 2023, 15, 2477.
- Khan, A.; Adalsteinsson, J.; Whitaker-Worth, D.L. AD and nutrition. Clin. Dermatol. 2022, 40, 135–144.
- Truong-Tran, A.Q.; Ruffin, R.E.; Foster, P.S.; Koskinen, A.M.; Coyle, P.; Philox, J.C.; Rofe, A.M.; Zalewski, P.D. Altered zinc homeostasis and caspase-3 activity in murine allergic airway inflammation. Am. J. Respir. Cell Mol. Biol. 2002, 27, 286–296.
- Kulik, L.; Maywald, M.; Kloubert, V.; Wessels, I.; Rink, L. Zinc deficiency drives Th17 polarization and promotes loss of Treg cell function. J. Nutr. Biochem. 2019, 63, 11–18.
- Tsai, Y.L.; Ko, W.S.; Hsino, J.L.; Pan, H.H.; Chiou, Y.L. zinc sulfate improved the unbalanced T cell profiles in Der p-allergic asthma: An ex vivo study. Clin. Respir. J. 2018, 12, 563–571.
- Rosenkranz, E.; Hilgers, R.D.; Uciechowski, P.; Petersen, A.; Plümäkers, B.; Rink, L. Zinc enhances the number of regulatory T cells in allergen-stimulated cells from atopic subjects. Eur. J. Nutr. 2017, 56, 557–567.
- Maywald, M.; Meurer, S.K.; Weiskirchen, R.; Rink, L. Zinc supplementation augments TGF-β1-depedent regulatory T cell induction. Mol. Nutr. Food Res. 2017, 61, 1600493.
- Roth-Walter, F. Iron-deficiency in atopic diseases: Innate immune priming by allergens and siderophores. Front. Allergy 2022, 3, 859922.
- Camaschella, C. Iron deficiency. Blood 2019, 133, 30–39.
- Petje, L.M.; Jensen, S.A.; Szikora, S.; Sulzbacher, M.; Bartosik, T.; Pjevac, P.; Hausmann, B.; Hufnagi, K.; Untersmayr, E.; Fischer, L.; et al. Functional iron-deficiency in women with AR is associated with symptoms after nasal provocation and lack of iron-sequestering microbes. Allergy 2021, 76, 2882–2923.
- Sahoyama, Y.; Hamazato, F.; Shiozawa, M.; Nakagawa, T.; Suda, W.; Ogata, Y.; Hachiya, T.; Kawakami, E.; Hattori, M. Multiple nutritional and gut microbial factors associated with AR: The Hitachi Health Study. Sci. Rep. 2022, 12, 3359.
- Podlecka, D.; Jerzynska, J.; Sanad, K.; Polanska, K.; Bobrow-Korzeniowska, M.; Stelmach, I.; Brzozowska, A. Micronutrients and the risks of allergic diseases in school children. Intl. J. Environ. Res. Public Health 2022, 19, 12187.
- Norton, R.L.; Hoffmann, P.R. Selenium and Asthma. Mol. Asp. Med. 2012, 33, 98–106.
- Gozzi-Silva, S.C.; Teixeira, F.M.E.; Duarte, A.J.S.; Sato, M.N.; de Oliveira, L.M. Immunomodulatory role of nutrients: How can pulmonary dysfunctions improve? Front. Nutr. 2021, 8, 674258.
- Chen, M.; Sun, Y.; Wu, Y.L. Lower circulating zinc and selenium levels are associated with an increased risk of asthma: Evidence from a meta-analysis. Public Health Nutr. 2019, 23, 1555–1562.
- Kuti, B.P.; Kuti, D.K.K.; Smith, O.S. Serum zinc, selenium and total antioxidant contents of Nigerian children with asthma: Association with disease severity and symptoms control. J. Trop. Pediatr. 2020, 66, 395–402.
- Jiang, J.; Nasab, E.M.; Athari, S.M.; Athari, S.S. Effects of vitamin E and selenium on AR and asthma pathophysiology. Respir. Physiol. Neurobiol. 2021, 286, 103614.
- Alwarith, J.; Kahleova, H.; Crosby, L.; Brooks, A.; Brandon, L.; Levin, S.M.; Barnard, N.D. The role of nutrition in asthma prevention and treatment. Nutr. Rev. 2020, 78, 928–938.
- Jensen, M.E.; Gibson, P.G.; Collins, C.E.; Hilton, J.M.; Wood, L.G. Diet-induced weight loss in obese children with asthma: A randomized controlled trial. Clin. Exp. Allergy 2013, 43, 775–784.
- Scott, H.A.; Gibson, P.G.; Garg, M.L.; Pretto, J.J.; Morgan, P.J.; Callister, R.; Wood, L.G. Dietary restriction and exercise improve airway inflammation and clinical outcomes in overweight and obese asthma: A randomized trial. Clin. Exp. Allergy 2013, 43, 36–49.
- Luna-Pech, J.A.; Torres-Mendoza, B.M.; Luna-Pech, J.A.; Garcia-Cobas, C.Y.; Navarrete-Navarro, S.; Elizalde-Lozano, A.M. Normocaloric diet improves asthma-related quality of life in obese pubertal adolescents. Int. Arch. Allergy Immunol. 2014, 163, 252–258.
- Kahleova, H.; Dort, S.; Holubkov, R.; Barnard, N.D. A Plant-Based High-Carbohydrate, Low-Fat Diet in Overweight Individuals in a 16-Week Randomized Clinical Trial: The Role of Carbohydrates. Nutrients 2018, 8, 58.
- Turner-McGrievy, G.; Barnard, N.D.; Scialli, A.R.; Lanou, A.J. Effects of a low-fat vegan diet and a Step II diet on macro- and micronutrient intakes in overweight postmenopausal women. Nutrition 2004, 20, 738–746.
- Ivanova, S.; Delattre, C.; Karcheva-Bahchevanska, D.; Benbasat, N.; Nalbantova, V.; Ivanov, K. Plant-Based Diet as a Strategy for Weight Control. Foods 2021, 10, 3052.
