Atopic dermatitis (AD) is a chronically relapsing inflammatory skin disorder characterized by itching and eczematous lesions. It is often associated with a personal or familial history of allergic diseases. Allergic inflammation induced by immunoglobulin E and T-helper type 2 (Th2) cell responses to common environmental agents has been suggested to play an essential role in AD pathogenesis. The standard therapies for AD, including topical or systemic agents, focus on controlling skin inflammation. Monoclonal antibody to interleukin-4 receptor alpha or Janus kinase inhibitors can provide significant clinical improvements in patients with AD by inhibiting Th2 cell-mediated skin inflammation. However, the clinical efficacy of the Th2 cell-targeted therapy is transient and incomplete in patients with AD. Patients with AD are seeking a permanent cure. Regulatory T (Treg) cells play a critical role in the maintenance of immune tolerance and suppress the development of autoimmune and allergic diseases. Therefore, the development of novel immunomodulatory strategies that can improve a long-term clinical outcome and provide a long-term treatment-free clinical remission of AD (disease-modifying therapy) is needed. Regulatory T (Treg) cells play a critical role in the maintenance of immune tolerance and suppress the development of autoimmune and allergic diseases. This entry provides three working hypotheses and perspectives for the treatment of AD by Treg cell activation. (1) A decreased number or function of Treg cells is a critical event that causes the activation of Th2 cells, leading to the development and maintenance of AD. (2) Activation of Treg cells is an effective therapeutic approach for AD. (3) Many different immunomodulatory strategies activating Treg cells can provide a long-term clinical improvement of AD by induction of immune tolerance. The Treg cell-targeted immunomodulatory therapies for AD include allergen immunotherapy, microbiota, vitamin D, polyvalent human immunoglobulin G, monoclonal antibodies to the surface antigens of T cell or antigen-presenting cell, and adoptive transfer of autologous Treg cells or genetically engineered Treg cells expanded in vitro.
- hypersensitivity
- immunomodulation
- allergy and immunology
- immunotherapy
- immune tolerance
- atopic dermatitis
- atopic eczema
- regulatory T cell
1. Introduction
2. Unmet Needs of Patients with AD
Patients with AD are seeking a permanent cure. This fact is supported by the persistence of various complementary and alternative therapies for AD [10], as well as multiple internet and social media contents created by patients with AD who want to share their personal experiences on successful AD self-management. However, physicians are explaining to patients suffering from AD that there is still no cure for AD and that it should be managed by continuous medical treatments [3]. This significant mismatch between patient’s needs and current medical therapies for AD necessitates further development of novel immunomodulatory strategies to achieve an LTCI of AD.3. Hypothesis on the Pathogenesis of AD
AD is a multifactorial disorder caused by multiple pathogenic elements, including genetic predisposition, environmental triggers, immune dysfunction, hypersensitivity reaction, chronic skin inflammation, and skin barrier defect [2][3]. Unfortunately, the precise interactions among the multiple pathogenic elements involved in the development and maintenance of AD are not fully understood. Epidemiological and experimental evidence suggests that an impairment of immune tolerance state caused by decreased number and/or function of Treg cells resulting from the exposure to various environmental toxicants is responsible for a rapidly increased prevalence of allergic diseases in the industrialized world [3][11][12][13][14]. In this hypothetical pathogenesis model for AD, a human subject with a genetic predisposition for the development of AD is exposed to environmental toxicants (such as air pollution, volatile organic chemicals, phthalates, and bisphenol A) through the respiratory mucosa, gastrointestinal mucosa, or skin. The exposure to environmental toxicants decreases the number and/or function of Treg cells and impairs immune homeostasis (loss of immune tolerance state) in the subject. The toxicant-induced decreased number and/or function of Treg cells induces hypersensitivity in the human subject through the activation of Th2 cells and the development of an IgE response to common environmental agents. Exposure to sensitized allergens and/or chemical irritants induces a hypersensitivity reaction and chronic inflammation of the skin, which induces and maintains the clinical manifestations of AD (pruritus, dryness, and eczema) (Figure 1).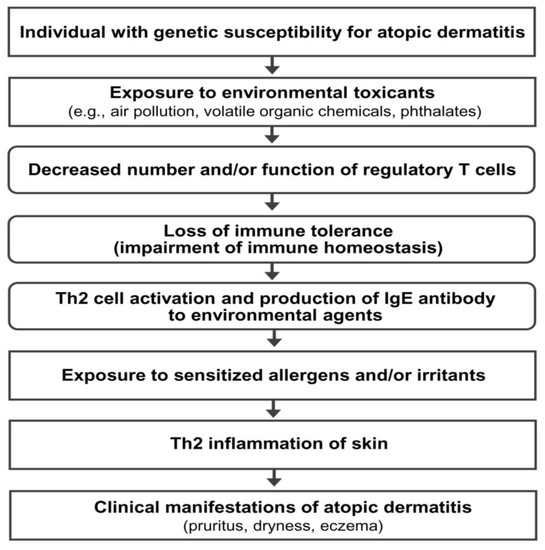
4. Previous Reports on the Long-Term Clinical Improvement of AD
Previous reports have suggested that an LTCI of AD can be achieved through three methods. First, a marked change in the living environment, also known as climatotherapy (relocation of a patient to a different region with a beneficial climate such as a foreign country with favorable weather and clean air), has been reported to induce an LTCI of AD [15][16]. However, the LTCI induced by a marked change in the living environment usually disappears shortly after the patient returns to their previous environment, indicating that change in environmental factors alone cannot normalize the disordered immune system of patients with AD [15][16]. Second, allergen immunotherapy induces an LTCI in some patients with AD [17][18]. Multiple clinical studies have reported an LTCI of AD after allergen immunotherapy [17][18][19][20]. Third, up to 70% of children with AD experience a natural LTCI of AD before puberty [21]. Induction of immune tolerance (recovery of immune homeostasis) is the most probable mechanism of an LTCI observed in children with AD [3], although scientific studies could not directly demonstrate this hypothesis. Mimicking the immunological mechanism responsible for the natural LTCI of AD in children (induction of immune tolerance) may be an ideal therapeutic approach for achieving an LTCI of AD [3].5. Five Questions and Three Hypotheses on the Regulatory T Cell-Targeted Immunomodulatory Strategies to Achieve a Long-Term Clinical Improvement of AD
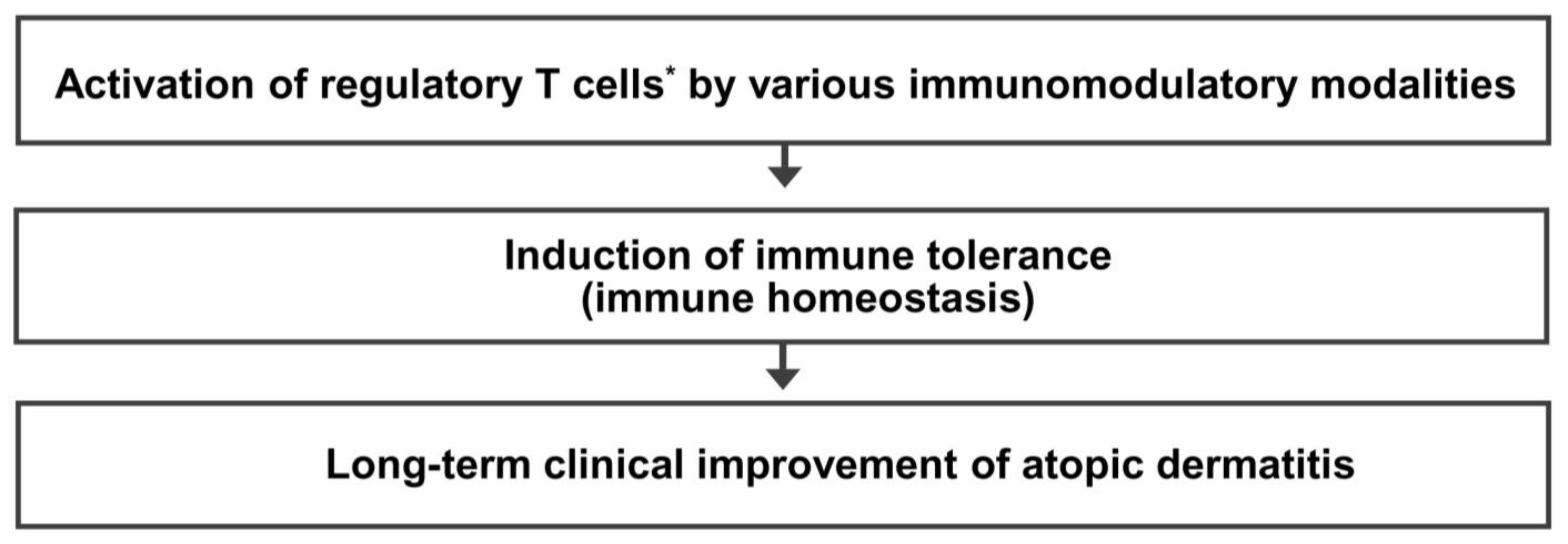
The present author developed five key questions on the immune mechanisms that induce natural LTCI in children with AD and the LTCI observed after allergen immunotherapy in patients with AD (Table 1). Additionally, the present author proposes three working hypotheses and perspectives on immunomodulatory strategies to achieve an LTCI of AD (Table 2). These hypotheses and perspectives suggest that various immunomodulatory strategies activating Treg cells can induce an immune tolerance and achieve an LTCI in patients with AD.|
What is the mechanism of natural induction of a long-term clinical improvement of atopic dermatitis in children?
Is the induction of immune tolerance responsible for a natural long-term clinical improvement of atopic dermatitis in children?
Is the activation of type 1 regulatory T cells a key mechanism inducing a natural long-term clinical improvement of atopic dermatitis?
Is the activation of type 1 regulatory T cells a common mechanism inducing a long-term clinical improvement of atopic dermatitis by allergen immunotherapy and a natural long-term clinical improvement in children with atopic dermatitis?
Can various kinds of immunomodulatory strategies activating regulatory T cells provide a long-term clinical improvement in patients with atopic dermatitis?
|
|
A decreased number or function of regulatory T cells is a critical event that causes the activation of Th2 cells, leading to the development and maintenance of atopic dermatitis.
Activation of regulatory T cells * is an effective therapeutic approach to achieve a long-term clinical improvement of atopic dermatitis.
Many different immunomodulatory strategies activating regulatory T cells can provide a long-term clinical improvement of atopic dermatitis by induction of immune tolerance.
|
* Activation of regulatory T cells means an increase in the number and/or function of regulatory T cells. Th2 cells, T-helper type 2 cells.
6. Mechanism of Immune Tolerance and Rationale of Regulatory T Cell-Targeted Immunomodulatory Therapy for AD
Immune tolerance has been historically defined as a state of unresponsiveness of the immune system to self-antigens and foreign antigens (e.g., proteins and allergens) [22][23]. Prior exposure to a specific antigen has been suggested to induce immune tolerance to the antigen [22][23]. Immune tolerance is crucial for normal physiology, and defects in immune tolerance can lead to autoimmune and allergic diseases [24][25]. However, the present author prefers the term “immune homeostasis” because it is more scientifically appropriate than “immune tolerance” for several reasons. First, “immune tolerance” can be misinterpreted as having no immune response to self or foreign antigens. A well-controlled immune response (immune homeostasis) can maintain a harmoniously controlled immune response in contrast to an exaggerated immune response that can be harmful to the host. A low-grade, well-controlled autoimmune response to a self-antigen is physiological, and this response helps to remove denatured or altered self-antigens [26]. Well-controlled immune responses to microbial organisms also protect the body from severe infections (septicemia and viremia) or hyperactivation of the immune system. Hyperactivation of the immune system induced by viral infection causes uncontrolled systemic inflammation, cytokine release (cytokine storm), multi-organ failure, and death in the coronavirus disease 2019 [27]. Treg cells are a functionally defined subpopulation of T cells that modulate the immune system and play a critical role in immune homeostasis (a well-controlled immune response to self-antigens and foreign antigens), thereby preventing the development of autoimmune diseases, allergic diseases, and allograft rejection [28][29]. Animal experiments have suggested that decreased number and/or function (deficiency or dysfunction) of Treg cells serves as a key immune abnormality responsible for the development of autoimmune and allergic diseases [30]. Treg cells are classified as natural Treg cells (nTreg cells) and induced Treg cells (iTreg cells) [31]. nTreg cells express forkhead box P3 (Foxp3), CD4, and CD25 markers [32][33][34]. nTreg cells mediate “central immune tolerance” by deleting autoreactive lymphocyte clones before they develop into fully immunocompetent cells during lymphocyte development in the thymus [35]. Peripheral immune tolerance is mediated by iTreg cells and develops after T cells mature and enter the peripheral tissues and lymph nodes [36]. In peripheral immune tolerance, the immune response to a certain antigen can also be decreased by repeated antigenic exposure or by antigenic exposure in tolerogenic conditions [36]. iTreg cells arise extra-thymically from conventional (or naïve) CD4+ helper T cells in the presence of transforming growth factor-β (TGF-β), retinoic acid, and T cell receptor (TCR)-mediated antigen presentation by antigen-presenting cells (dendritic cells (DCs) or macrophages) in the peripheral tissue or nearby lymphoid tissue [31]. Among the iTreg cells, the IL-10-producing CD4+ Treg cells without Foxp3 expression (type 1 Treg cells: Tr1 cells) play a key role in allergen tolerance and can be induced by allergen immunotherapy in humans [37][38][39][40]. Previous studies have indicated that allergen-specific Tr1 cells are the predominant type of T cell response to allergens in healthy individuals and prevent unwanted hypersensitivity reactions to environmental antigens, such as house dust mites, pollen, and food [41][42]. There are significant differences in the proportions of three different allergen-specific T cell subtypes (T-helper type 1: Th1, Th2, and Tr1 cells) in peripheral blood between healthy non-allergic human subjects and allergic individuals [42]. The imbalance in the ratio of allergen-specific Th1, Th2, and Tr1 cells appears to be critical in the development of allergic diseases, and recovery of balance among allergen-specific Th1, Th2, and Tr1 cells may provide remission of allergic diseases, including AD [42]. Therefore, immunomodulatory approaches activating Tr1 cells with antigen-specific and/or antigen-nonspecific stimulations could induce an LTCI of AD by inducing immune homeostasis (immune tolerance state). The presence of inborn errors of immunity (IEI) caused by genetic mutations that affect the immune system is important scientific evidence supporting the hypothesis that immune dysfunction resulting from a decreased number and/or function of Treg cells is critical in the pathogenesis of allergic diseases, including AD [30][43]. IEI are clinically expressed as increased susceptibility to infections, autoimmunity, allergy, bone marrow failure, and/or malignancy [44]. In human subjects, mutations of the Foxp3 gene (a master control gene of Treg cells) resulting in absent or dysfunctional Treg cells are responsible for immune dysregulation, polyendocrinopathy, enteropathy, X-linked (IPEX) syndrome, which is clinically characterized by neonatal autoimmune enteropathy, diabetes and thyroiditis, food allergies, and skin eczema [43][45]. AD is a frequent clinical manifestation observed in IPEX syndrome [43][45]. Another example is Wiskott–Aldrich syndrome (WAS) [46][47]. WAS is a rare X-linked recessive disease characterized by eczema, thrombocytopenia, immune deficiency, and bloody diarrhea [43]. WAS is caused by the genetic defects producing defective proteins (WAS proteins) that have a central role in actin polymerization and cytoskeletal rearrangement. Both nTreg cells and iTreg cells are defective in WAS [46]. In the WAS, the eczematous eruption is indistinguishable from AD when diagnostic criteria for AD are used and clears dramatically after a successful transplantation of bone marrow (hematopoietic stem cells) from a healthy donor [47]. This evidence from IEI demonstrates that Treg cell dysfunction is critically involved in the pathogenesis of the AD [30][43].7. Immunomodulatory Strategies Activating Regulatory T Cells for AD: In Vivo Activation
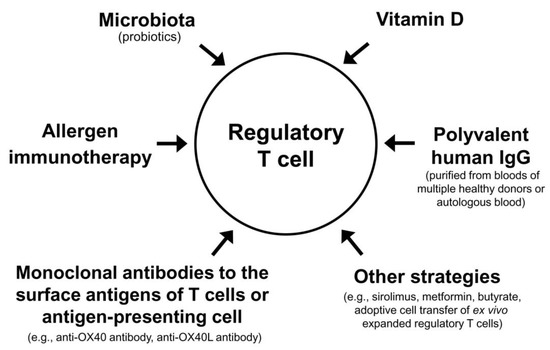
The Treg cell-targeted immunomodulatory therapies for AD include allergen immunotherapy, microbiota, vitamin D, polyvalent human immunoglobulin G (IgG), monoclonal antibodies to the surface antigens of T cell or antigen-presenting cell, and adoptive transfer of autologous Treg cells or genetically engineered Treg cells expanded in vitro (Table 3).|
Strategies with proven clinical efficacy by at least one randomized clinical trial |
|
Allergen immunotherapy
Microbial therapy (probiotics)
Vitamin D
Subcutaneous or intramuscular injection of polyvalent human IgG from multiple healthy blood donors
Intramuscular injection of autologous total IgG
Monoclonal antibody to antigen on the surface of T cells (anti-OX40 antibody)
|
|
Strategies without proven clinical efficacy in patients with atopic dermatitis by a clinical trial |
Metformin
|
IgG, immunoglobulin G.
8. Combinations of Different Modalities Activating Regulatory T Cells
9. Conclusions
References
- Wollenberg, A.; Christen-Zach, S.; Taieb, A.; Paul, C.; Thyssen, J.P.; de Bruin-Weller, M.; Vestergaard, C.; Seneschal, J.; Werfel, T.; Cork, M.J.; et al. ETFAD/EADV Eczema task force 2020 position paper on diagnosis and treatment of atopic dermatitis in adults and children. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 2717–2744.
- Ratchataswan, T.; Banzon, T.M.; Thyssen, J.P.; Weidinger, S.; Guttman-Yassky, E.; Phipatanakul, W. Biologics for Treatment of Atopic Dermatitis: Current Status and Future Prospect. J. Allergy Clin. Immunol. Pract. 2021, 9, 1053–1065.
- Nahm, D.H. Personalized Immunomodulatory Therapy for Atopic Dermatitis: An Allergist’s View. Ann. Dermatol. 2015, 27, 355–363.
- Wang, F.P.; Tang, X.J.; Wei, C.Q.; Xu, L.R.; Mao, H.; Luo, F.M. Dupilumab treatment in moderate-to-severe atopic dermatitis: A systematic review and meta-analysis. J. Dermatol. Sci. 2018, 90, 190–198.
- Simpson, E.L.; Bieber, T.; Guttman-Yassky, E.; Beck, L.A.; Blauvelt, A.; Cork, M.J.; Silverberg, J.I.; Deleuran, M.; Kataoka, Y.; Lacour, J.P.; et al. Two Phase 3 Trials of Dupilumab versus Placebo in Atopic Dermatitis. N. Engl. J. Med. 2016, 375, 2335–2348.
- Li, C.; Sun, X.; Zhao, K.; Meng, F.; Li, L.; Mu, Z.; Han, X. Efficacy and Safety of Janus Kinase Inhibitors for the Treatment of Atopic Dermatitis: A Systematic Review and Meta-Analysis. Dermatology 2022, 238, 725–735.
- Guttman-Yassky, E.; Teixeira, H.D.; Simpson, E.L.; Papp, K.A.; Pangan, A.L.; Blauvelt, A.; Thaci, D.; Chu, C.Y.; Hong, H.C.; Katoh, N.; et al. Once-daily upadacitinib versus placebo in adolescents and adults with moderate-to-severe atopic dermatitis (Measure Up 1 and Measure Up 2): Results from two replicate double-blind, randomised controlled phase 3 trials. Lancet 2021, 397, 2151–2168.
- Verhagen, J.; Akdis, M.; Traidl-Hoffmann, C.; Schmid-Grendelmeier, P.; Hijnen, D.; Knol, E.F.; Behrendt, H.; Blaser, K.; Akdis, C.A. Absence of T-regulatory cell expression and function in atopic dermatitis skin. J. Allergy Clin. Immunol. 2006, 117, 176–183.
- Fyhrquist, N.; Lehtimaki, S.; Lahl, K.; Savinko, T.; Lappetelainen, A.M.; Sparwasser, T.; Wolff, H.; Lauerma, A.; Alenius, H. Foxp3+ cells control Th2 responses in a murine model of atopic dermatitis. J. Investig. Dermatol. 2012, 132, 1672–1680.
- Vieira, B.L.; Lim, N.R.; Lohman, M.E.; Lio, P.A. Complementary and Alternative Medicine for Atopic Dermatitis: An Evidence-Based Review. Am. J. Clin. Dermatol. 2016, 17, 557–581.
- Genuis, S.J. Sensitivity-related illness: The escalating pandemic of allergy, food intolerance and chemical sensitivity. Sci. Total Env. 2010, 408, 6047–6061.
- Lee, J.H.; Lee, H.S.; Park, M.R.; Lee, S.W.; Kim, E.H.; Cho, J.B.; Kim, J.; Han, Y.; Jung, K.; Cheong, H.K. Relationship between indoor air pollutant levels and residential environment in children with atopic dermatitis. Allergy Asthma Immunol. Res. 2014, 6, 517–524.
- Ahn, K. The role of air pollutants in atopic dermatitis. J. Allergy Clin. Immunol. 2014, 134, 993–999; discussion 1000.
- Kim, E.H.; Jeon, B.H.; Kim, J.; Kim, Y.M.; Han, Y.; Ahn, K.; Cheong, H.K. Exposure to phthalates and bisphenol A are associated with atopic dermatitis symptoms in children: A time-series analysis. Environ. Health 2017, 16, 24.
- Byremo, G.; Rod, G.; Carlsen, K.H. Effect of climatic change in children with atopic eczema. Allergy 2006, 61, 1403–1410.
- Vahavihu, K.; Ylianttila, L.; Salmelin, R.; Lamberg-Allardt, C.; Viljakainen, H.; Tuohimaa, P.; Reunala, T.; Snellman, E. Heliotherapy improves vitamin D balance and atopic dermatitis. Br. J. Dermatol. 2008, 158, 1323–1328.
- Lee, J.; Lee, H.; Noh, S.; Bae, B.G.; Shin, J.U.; Park, C.O.; Lee, K.H. Retrospective Analysis on the Effects of House Dust Mite Specific Immunotherapy for More Than 3 Years in Atopic Dermatitis. Yonsei Med. J. 2016, 57, 393–398.
- Chait, I.; Allkins, V. Remission of life-long atopic dermatitis after hyposensitisation to house dust mite. Practitioner 1985, 229, 609–612.
- Leroy, B.P.; Lachapelle, J.-M.; Somville, M.; Jacquemin, M.; Saint-Remy, J. Injection of allergen-antibody complexes is an effective treatment of atopic dermatitis. Dermatology 1991, 182, 98–106.
- Tuft, L. Studies in atopic dermatitis. V. Problems in inhalant hyposensitization and results of treatment. J. Allergy 1960, 31, 1–11.
- Hua, T.C.; Hwang, C.Y.; Chen, Y.J.; Chu, S.Y.; Chen, C.C.; Lee, D.D.; Chang, Y.T.; Wang, W.J.; Liu, H.N. The natural course of early-onset atopic dermatitis in Taiwan: A population-based cohort study. Br. J. Dermatol. 2014, 170, 130–135.
- Sykes, M. Immune tolerance: Mechanisms and application in clinical transplantation. J. Intern. Med. 2007, 262, 288–310.
- Sakaguchi, S.; Yamaguchi, T.; Nomura, T.; Ono, M. Regulatory T cells and immune tolerance. Cell 2008, 133, 775–787.
- Sharkey, P.; Thomas, R. Immune tolerance therapies for autoimmune diseases: Shifting the goalpost to cure. Curr. Opin. Pharmacol. 2022, 65, 102242.
- Akdis, M.; Akdis, C.A. Therapeutic manipulation of immune tolerance in allergic disease. Nat. Rev. Drug Discov. 2009, 8, 645–660.
- Ray, S.K.; Putterman, C.; Diamond, B. Pathogenic autoantibodies are routinely generated during the response to foreign antigen: A paradigm for autoimmune disease. Proc. Natl. Acad. Sci. USA 1996, 93, 2019–2024.
- Mohamed Khosroshahi, L.; Rezaei, N. Dysregulation of the immune response in coronavirus disease 2019. Cell Biol. Int. 2021, 45, 702–707.
- O’Garra, A.; Vieira, P. Regulatory T cells and mechanisms of immune system control. Nat. Med. 2004, 10, 801–805.
- Miyara, M.; Sakaguchi, S. Natural regulatory T cells: Mechanisms of suppression. Trends Mol. Med. 2007, 13, 108–116.
- Miyara, M.; Wing, K.; Sakaguchi, S. Therapeutic approaches to allergy and autoimmunity based on FoxP3+ regulatory T-cell activation and expansion. J. Allergy Clin. Immunol. 2009, 123, 749–755; quiz 756–757.
- Schmitt, E.G.; Williams, C.B. Generation and function of induced regulatory T cells. Front. Immunol. 2013, 4, 152.
- Sakaguchi, S.; Mikami, N.; Wing, J.B.; Tanaka, A.; Ichiyama, K.; Ohkura, N. Regulatory T Cells and Human Disease. Annu. Rev. Immunol. 2020, 38, 541–566.
- Ferreira, L.M.R.; Muller, Y.D.; Bluestone, J.A.; Tang, Q. Next-generation regulatory T cell therapy. Nat. Rev. Drug Discov. 2019, 18, 749–769.
- Fontenot, J.D.; Gavin, M.A.; Rudensky, A.Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 2003, 4, 330–336.
- Riemann, M.; Andreas, N.; Fedoseeva, M.; Meier, E.; Weih, D.; Freytag, H.; Schmidt-Ullrich, R.; Klein, U.; Wang, Z.-Q.; Weih, F. Central immune tolerance depends on crosstalk between the classical and alternative NF-κB pathways in medullary thymic epithelial cells. J. Autoimmun. 2017, 81, 56–67.
- Xing, Y.; Hogquist, K.A. T-cell tolerance: Central and peripheral. Cold Spring Harb. Perspect. Biol. 2012, 4, a006957.
- Akdis, C.A.; Akdis, M.; Blesken, T.; Wymann, D.; Alkan, S.S.; Muller, U.; Blaser, K. Epitope-specific T cell tolerance to phospholipase A2 in bee venom immunotherapy and recovery by IL-2 and IL-15 in vitro. J. Clin. Investig. 1996, 98, 1676–1683.
- Akdis, C.A.; Blesken, T.; Akdis, M.; Wuthrich, B.; Blaser, K. Role of interleukin 10 in specific immunotherapy. J. Clin. Investig. 1998, 102, 98–106.
- Francis, J.N.; Till, S.J.; Durham, S.R. Induction of IL-10+CD4+CD25+ T cells by grass pollen immunotherapy. J. Allergy Clin. Immunol. 2003, 111, 1255–1261.
- Jutel, M.; Akdis, M.; Budak, F.; Aebischer-Casaulta, C.; Wrzyszcz, M.; Blaser, K.; Akdis, C.A. IL-10 and TGF-beta cooperate in the regulatory T cell response to mucosal allergens in normal immunity and specific immunotherapy. Eur. J. Immunol. 2003, 33, 1205–1214.
- Akdis, M. Healthy immune response to allergens: T regulatory cells and more. Curr. Opin. Immunol. 2006, 18, 738–744.
- Akdis, M.; Verhagen, J.; Taylor, A.; Karamloo, F.; Karagiannidis, C.; Crameri, R.; Thunberg, S.; Deniz, G.; Valenta, R.; Fiebig, H. Immune responses in healthy and allergic individuals are characterized by a fine balance between allergen-specific T regulatory 1 and T helper 2 cells. J. Exp. Med. 2004, 199, 1567–1575.
- Vaseghi-Shanjani, M.; Smith, K.L.; Sara, R.J.; Modi, B.P.; Branch, A.; Sharma, M.; Lu, H.Y.; James, E.L.; Hildebrand, K.J.; Biggs, C.M.; et al. Inborn errors of immunity manifesting as atopic disorders. J. Allergy Clin. Immunol. 2021, 148, 1130–1139.
- Tangye, S.G.; Al-Herz, W.; Bousfiha, A.; Cunningham-Rundles, C.; Franco, J.L.; Holland, S.M.; Klein, C.; Morio, T.; Oksenhendler, E.; Picard, C.; et al. Human Inborn Errors of Immunity: 2022 Update on the Classification from the International Union of Immunological Societies Expert Committee. J. Clin. Immunol. 2022, 42, 1473–1507.
- Halabi-Tawil, M.; Ruemmele, F.M.; Fraitag, S.; Rieux-Laucat, F.; Neven, B.; Brousse, N.; De Prost, Y.; Fischer, A.; Goulet, O.; Bodemer, C. Cutaneous manifestations of immune dysregulation, polyendocrinopathy, enteropathy, X-linked (IPEX) syndrome. Br. J. Dermatol. 2009, 160, 645–651.
- Adriani, M.; Jones, K.; Kirby, M.; Anderson, S.; Silvin, C.; Wincowitch, S.; Candotti, F. Defects of Regulatory T Cell function In The Wiskott-Aldrich Syndrome. (49.25). J. Immunol. 2010, 184, 49.25.
- Saurat, J.H. Eczema in primary immune-deficiencies. Clues to the pathogenesis of atopic dermatitis with special reference to the Wiskott-Aldrich syndrome. Acta Derm. -Venereol. Suppl. 1985, 114, 125–128.
- Hesse, L.; van Ieperen, N.; Petersen, A.H.; Elberink, J.; van Oosterhout, A.J.M.; Nawijn, M.C. High dose vitamin D(3) empowers effects of subcutaneous immunotherapy in a grass pollen-driven mouse model of asthma. Sci. Rep. 2020, 10, 20876.
- Jerzynska, J.; Stelmach, W.; Rychlik, B.; Lechanska, J.; Podlecka, D.; Stelmach, I. The clinical effect of vitamin D supplementation combined with grass-specific sublingual immunotherapy in children with allergic rhinitis. Allergy Asthma Proc. 2016, 37, 105–114.
- Chiewchalermsri, C.; Sangkanjanavanich, S.; Pradubpongsa, P.; Mitthamsiri, W.; Jaisupa, N.; Jindarat, S.; Buranapraditkun, S.; Jacquet, A.; Sangasapaviliya, A.; Boonpiyathad, T. Randomized, Double-Blind, Placebo-Controlled Trial of Vitamin D Supplementation in the Build-up Phase of House Dust Mite-Specific Immunotherapy. Allergy Asthma Immunol. Res. 2023, 15, 336–347.
- Leroy, B.P.; Boden, G.; Lachapelle, J.M.; Jacquemin, M.G.; Saint-Remy, J.M. A novel therapy for atopic dermatitis with allergen-antibody complexes: A double-blind, placebo-controlled study. J. Am. Acad. Dermatol. 1993, 28, 232–239.
