Solid oxide fuel cells (SOFCs) are amongst the most widely used renewable alternative energy systems with near-zero carbon emission, high efficiency, and environment-friendly features. However, the high operating temperature of SOFCs is still considered a major challenge due to several issues regarding the materials’ corrosion, unwanted reactions between layers, etc. Thus, low-temperature SOFCs (LT-SOFCs) have gained significant interest during the past decades. Despite the numerous advantages of LT-SOFCs, material selection for each layer is of great importance as the common materials have not shown a desirable performance so far. In addition to the selection of the materials, fabrication techniques have a great influence on the properties of the SOFCs. As SOFCs with thinner layers showed lower polarisation resistance, especially in the electrolyte layer, different thin-film fabrication methods have been employed, and their effect on the overall performance of SOFCs has been evaluated.
- solid oxide fuel cells
- low-temperature SOFCs
- materials selection
- thin film
1. Introduction
Fuel cells are electrochemical devices that convert chemical energy into electricity with low emissions [1]. Since fuel cells do not have the limitations of internal combustion engines (Carnot cycle), they can generate electricity with higher efficiency [2]. In addition, the progressive increase in utilising hydrogen as a clean energy carrier has caused a great interest in developing more efficient fuel cells as a prospective power source by both research and manufacturing communities. In this regard, fuel cells have already shown great potential in providing electricity for rural areas with no or limited access to the public grid, removing the huge cost of wiring and electricity transfer to these areas [3][4].
According to the choice of fuel and electrolyte, fuel cells are categorised into six major groups: (1) alkaline fuel cell (AFC) [5], (2) phosphoric acid fuel cell (PAFC) [6], (3) solid oxide fuel cell (SOFC) [7], (4) molten carbonate fuel cell (MCFC) [8], (5) proton exchange membrane fuel cell (PEMFC) [9], and (6) direct methanol fuel cell (DMFC) [10]. SOFCs show promising properties compared to other types. SOFCs are among the most promising fuel cells in providing efficient electric power generation and substantial environmental benefits in case of a high level of fuel flexibility. It is this fuel flexibility that has made it possible to operate SOFCs on today’s conventional hydrocarbon fuels, such as methane, methanol, and ethanol [11]. It should be mentioned that anode, cathode, and electrolyte materials differ from one fuel cell technology to another. For example, polybenzimidazole (PBI) and Nafion are the most common membrane materials in PEMFCs, yttria-stabilised zirconia (YSZ), gadolinium-doped ceria (GDC), and lanthanum strontium gallium magnesium oxide (LSGM) are the most common electrolyte materials in SOFCs, molten potassium and lithium carbonate are the most common electrolyte materials in MCFCs, etc. Several anode and cathode materials can also be mentioned for each fuel cell technology, including Ni/GDC, LSCF, lithium metatitanate, porous Ni, and Pt [9][12][13]. Nonetheless, further development is required to overcome some existing limitations surrounding common fuel cell technologies. Reducing cost, improving durability, and further optimising performance have been the focus of most fuel cell research at the individual cell level, stack level, and general system level [14].
2. Ceramic Processing Techniques
2.1. Thin-Film Fabrication Methods
Since the ohmic resistance is inversely related to the thickness of the electrolyte layer, an important approach for increasing the performance of the cell at LT would be to decrease the thickness of this layer [42]. A membrane thickness of 1 μm for conventional YSZ could decrease its ohmic resistance to a point that it would be possible to ensure a reasonable power output for a cell operating at 500 °C [37][43]. Thus, a great deal of focus has been placed on the development of thin-film electrolytes, as a practical method for moving towards lower operating temperatures. Advanced thin-film fabrication techniques, such as chemical vapor deposition (CVD), physical vapor deposition (PVD), pulsed laser deposition (PLD), aerosol deposition (AD), tape casting, screen printing, and spin coating, have allowed the production of ultra-thin electrolyte layers with thicknesses as low as 10 μm [43]. Some of these techniques are simple and robust, such as screen printing and tape casting, while others offer a higher uniformity and control over the deposited thin layer, such as PVD and CVD. However, neither provides altogether a high deposition rate, low capital cost, smooth and dense layers, durability, and low process temperatures. Considering the targeted objectives of developing LT-SOFCs, having a broader choice of material, and using metallic parts, avoiding high-temperature fabrication techniques is of great importance.2.1.1. Chemical Vapour Deposition (CVD) Process
CVD has shown growing potential as a nanoscale fabrication technique for SOFCs. In vapor deposition coating processes, vaporised materials are transferred on a substrate in a vapour form. In the CVD method, a chemical reaction is involved in the conversion of vapours to solids. In vapour deposition coating processes, vaporised materials are transferred on a substrate in a vapour form [44][45]. The CVD process can be divided into several categories, including atmospheric pressure CVD (APCVD), low-pressure CVD (LPCVD), ultrahigh-vacuum CVD (UHVCVD), aerosol-assisted CVD (AACVD), metal–organic chemical vapour deposition (MOCVD), and plasma-enhanced CVD (PECVD) [46][47]. In the CVD method, a chemical reaction is involved in the conversion of vapours to solids. During the CVD process, precursors are able to reach the surface of complex structures, providing a relatively uniform layer over the entire surface. In this process, one or more volatile precursors are reacted on the surface of a substrate, forming a thin solid film. This method is extremely sensitive to the applied pressure and temperature conditions [47]. An example of the CVD process is shown in Figure 1a. Generally, the CVD process is divided into six steps according to the temperature, pressure, and heat source. These steps include (a) heat transfer and reactant diffusion to the reaction zone, (b) chemical reaction in the gas phase, (c) transfer of reactants and products to the substrate, (d) chemical adsorption and diffusion of the products on the substrate, (e) formation of the coating on the substrate, and (f) heat transfer and removal of the by-products from the chamber (Figure 1b) [48].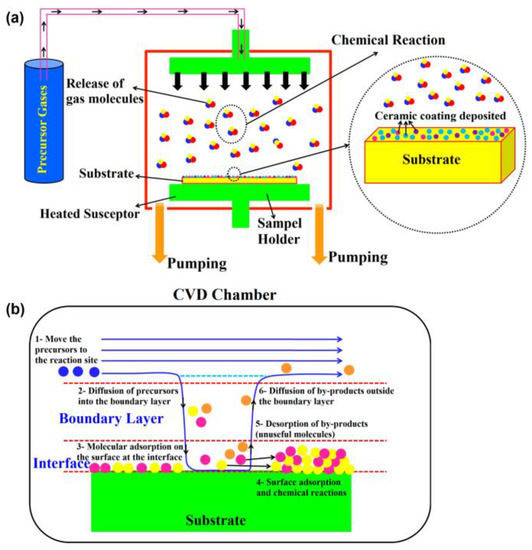
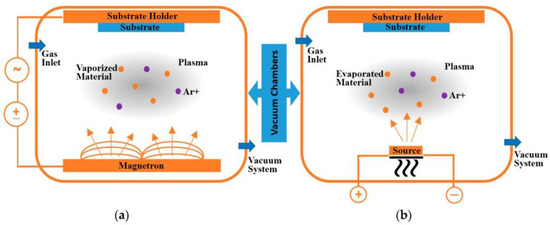
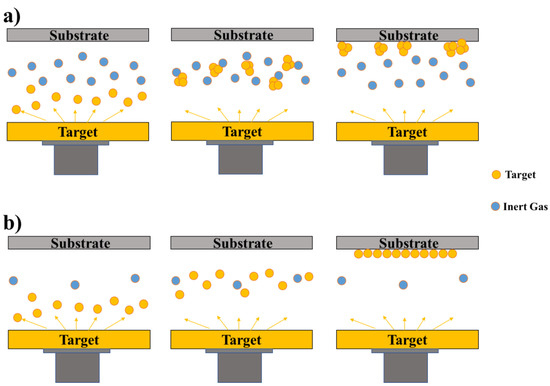
2.1.2. Physical Vapour Deposition (PVD) Process
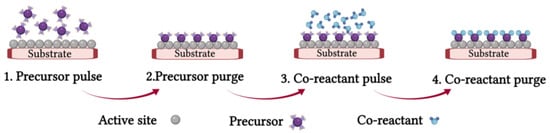
2.1.3. Atomic Layer Deposition (ALD) Process
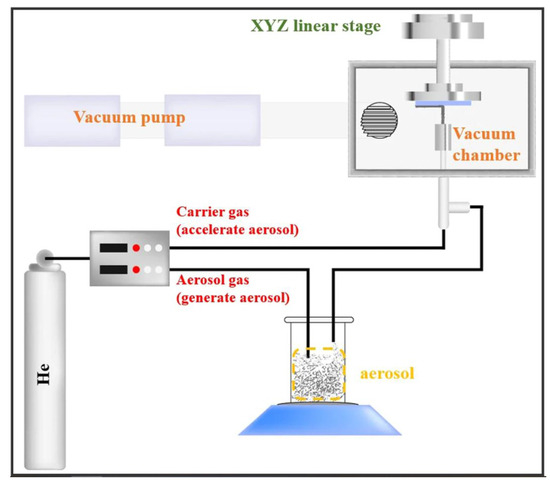
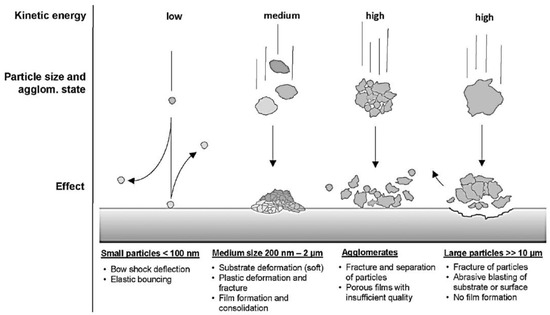
2.1.4. Aerosol Deposition (AD) Process
2.1.5. Dip Coating Process
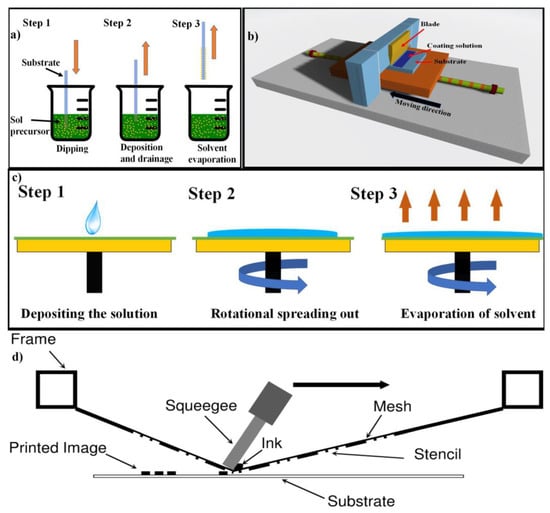
2.1.6. Tape Casting Process
2.1.7. Spin Coating Process
2.1.8. Screen Printing Process
2.2. Other Fabrication Methods
2.2.1. Solid-State Reaction
It is well known that different synthesis processes can greatly alter the microstructure of the products, resulting in changes in (a) the grain size and grain boundaries in the electrolyte, and (b) the durability and electrochemically active surface area of the electrodes [140][141]. In addition, the number of impurities observed in different SOFC components is also closely dependent on the synthesis and fabrication procedures used. Thus, the activation energy and electrical conductivity can be greatly affected by different procedures, as impurity levels significantly alter the characteristics of the grain boundary, grain, and density levels. The solid-state reaction is a well-known preparation method for its high yield, high selectivity, simplicity, and the absence of numerous solvents and side reactions. This method requires intimate mixing of the ceramic compounds in the form of oxides or other forms such as carbonates, where repetitive cycles of grinding and heating are commonly applied in order to reach a complete reaction between all reagents [142][143]. However, it requires a long process time and can lead to high contamination levels. In addition, due to the large number of uncertain factors involved in multiphase reactions, the final structure and composition of the samples are often inhomogeneous and nonstoichiometric [144]. When compared with data obtained with other methods, lower electrolyte conductivities, and much higher activation energies were reported for samples prepared using a solid-state reaction [145][146]. Zhan et al. [147] studied the contribution of grain boundary resistance to the total resistance measured for electrolytes produced through this method and reported an overwhelming contribution of more than 90%, resulting in low total conductivities. It was further reported that it is the presence of high levels of impurities in the grain boundaries of samples prepared through solid-state processing that blocked oxygen ion migration in the electrolyte structure. In addition, high temperatures required for the annealing processes during solid-state reactions commonly result in the formation of strongly agglomerated particles with large particle sizes [148]. Thus, very high sintering temperatures are often required for the full densification of the resulting electrolyte powder. Such high sintering temperatures can be extremely problematic when the electrolyte is co-fired with the electrode material [149]. As was mentioned in the previous section, it is often required for a thin electrolyte layer to be supported on an electrode substrate in order to improve cell performance by decreasing the ohmic resistance of the electrolyte layer [42]. In such configurations, the prepared electrode-electrolyte bilayers are co-fired to reach a dense electrolyte film while preserving the electrode porosity [150]. High sintering temperatures required for electrolyte powders prepared by the solid-state reaction can either lead to extreme interfacial reactions between the electrode and electrolyte materials or a loss in the electrode porosity [42]. Both of which are fatal for the fuel cell performance. Thus, alternative preparation methods, such as wet chemical methods, have been proposed in order to overcome such drawbacks.2.2.2. Sol–Gel Process
The sol–gel chemical synthesis method has also been used as a useful wet chemical route for laboratory synthesis of ceramic oxides [151]. Since, during a wet chemical reaction, the precursors are commonly dissolved and mixed in a solution, it is possible to reach a high level of homogeneity in the final product. Thus, by using the sol–gel method, it is possible to obtain stoichiometric ceramic powders at relatively low temperatures, avoiding problems aroused from solid-state reactions. However, numerous properties need to be controlled to reach superior properties for produced ceramics when compared to solid-state processing [152][153]. Sol–gel synthesis is an increasingly popular wet chemical synthesis method, first adopted for the synthesis of glass materials and novel compositions of ceramic oxides in the 1960s [154]. However, numerous properties need to be controlled to reach superior properties for produced ceramics when compared to solid-state processing [152][155][156]. The synthesis process includes the transition of monomers from a solution or colloidal liquid system (sol) into an integrated solid network (gel) [157]. The formed gel is basically a solid-state network in a liquid. In general, the synthesis process can be characterised by the following steps: (1) the formation of stable solutions of the solvated metal precursors (the sol); (2) gel formation resulting from the formation of an alcohol- or oxide-bridged network (the gel), commonly through polycondensation reactions. The initiation of such reactions can be observed in the form of a dramatic increase in the viscosity of the solution; (3) Gel ageing, where a solid mass is gradually formed. During this step, the continuous polycondensation reaction contracts the gel network, also expulsing the solvent from the gel pores; (4) drying the gel by removing water and other volatile liquids from the gel network; (5) decomposition and densification of the dried gel at high temperatures. Such processes would result in the formation of smaller particles with improved sinterability [158]. An important advantage of the sol–gel process or, in general, wet chemical processes is the possibility of incorporating different types of dopants at different stages of the process, where, due to the mixing of the precursors at the molecular level, an enhanced distribution of the dopant in the final solid solution can be achieved [159]. Thus, a greater grain interior conductivity can be reached for the electrolyte layer. Although the properties of materials synthesised through sol–gel processes are commonly superior to solid-state reactions, the relatively low powder yield observed for the sol–gel process limits its use in small-scale productions [21]. In addition, there have been cases where even lower conductivities and higher activation energies were reported for samples prepared using the sol–gel process in comparison to solid-state reactions [147][160][161]. Haung et al. [160] reported the synthesis of Ce0.2Sm0.8O1.9 using the sol–gel process and investigated the structure, thermal properties, and ionic conductivity of the solid electrolyte. The electrolyte powder revealed a lower sintering temperature (1400 °C) than powders prepared by solid-state processes (1650 °C). In the study, a considerably low conductivity (5.0 × 10−3 S·cm−1) and high activation energy (0.97 eV) were reported for the Ce0.2Sm0.8O1.9 sample prepared using the sol–gel process (at 600 °C). Similar observations were also reported for GDC powders prepared using the sol–gel process, where a conductivity and activation energy of 3.6 × 10−3 S·cm−1 and 1.08 eV, respectively, were reported for Gd0.1Ce0.9O1.95 [162]. Since such high activation energies and low conductivities are not expected for samples prepared using the sol–gel process, most studies have addressed this issue to the possible presence of impurities in the precursors and solvents used in the process. However, due to the considerable number of similar observations reported by different groups, especially for the synthesis of doped ceria, a more detailed investigation seems to be required.2.2.3. Coprecipitation Process
The coprecipitation process is a well-known wet chemical method used in the earliest syntheses of nanoparticles, and it is based on the simultaneous precipitation of multiple species [163][164][165][166]. In this method, metal cations can be coprecipitated in different forms, e.g., carbonates, bicarbonates, and oxalates, followed by calcination and decomposition steps. The required calcination step will, however, result in particle agglomeration, which, at relatively high temperatures, could lead to aggregation and sintering. Fortunately, nanoparticulates of carbonate and oxalate have been shown to decompose at relatively low temperatures, minimising the level of agglomeration of the final powder [167][168][169][170]. Both carbonate and oxalate coprecipitation methods have been proven to produce highly sinteractive and homogeneous products and show improved performance over samples prepared using the solid-state reaction and sol–gel techniques [171][172], especially when used for the synthesis of doped ceria [168][170]. However, different problems, such as relatively low green density (for oxalate) and composition deviation from feed ratios (for carbonate), limit their application as an ideal synthesis method for SOFC applications [171]. In the oxalate coprecipitation synthesis process, oxalate acid (C2H2O4) is commonly used to react with the present metal cations in the solution, forming fine precipitates. The precipitates are then calcined to obtain the desired metal oxide composition. Generally, oxalate precipitates show good stability towards different treatment conditions, such as washing and drying, and possess a relatively high production yield [173]. In addition, the high homogeneity of the electrolyte powders produced by this method commonly results in the report of high bulk conductivities [170]. Such observations can be traced back to the early applications of this method for the production of doped ceria-based electrolyte powders [174]. Nevertheless, samples prepared using the oxalate coprecipitation method do require higher sintering temperatures when compared to other wet chemical methods, i.e., carbonate coprecipitation [175]. It has been seen that the oxalate coprecipitation method usually results in the formation of large rod-like particles, being the agglomerates of smaller irregular-shaped particles. Thus, electrolytes prepared using this method illustrate relatively low green densities and commonly require high sintering temperatures in order to reach full densification [176][177][178]. The carbonate synthesis route is similar to a coprecipitation method where ammonium carbonate is commonly used as the precipitant. Carbonates are known as precursors for highly sinterable oxides [168]. The non-gelatinous nature of carbonate precursors allows for low agglomeration of the resultant powders, resulting in well-distributed nano-sized particles with relatively low sintering temperatures [179]. Thus, most of the literature surrounding the carbonate coprecipitation method is mainly focused on the aspects of microstructure and sinterability, whereas a smaller number of studies address its effect on the electrical properties of the synthesised samples. Although the reported conductivity data for electrolytes prepared by carbonate precipitation show a slight inconsistency, especially for doped ceria, high conductivities and considerably low activation energies are often reported [180][181][182]. Tok et al. [183] reported a conductivity of 18.3 × 10−3 S·cm−1 (600 °C) for Gd0.1Ce0.9O1.95, while even higher conductivities of 22 × 10−3 S·cm−1 (600 °C) have been reported for Sm0.2Ce0.8O1.9 prepared using carbonate coprecipitation [184]. Both studies revealed activation energies lower than 0.7 eV. Nanosized spherical-shaped particles with high surface areas are usually reported for powders prepared by the carbonate method [182][185]. Such microstructural properties have been proven to facilitate the sinterability of the synthesised electrolyte powders, making it possible to reach high densification levels at relatively low temperatures [186]. In addition, low sintering temperatures offer the possibility of fabricating thin electrolyte layers with submicron grain sizes which can greatly enhance the grain boundary conductivity of the electrolyte layer, especially for doped ceria solid electrolytes.2.2.4. Glycine Nitrate Process (Combustion)
Recently, the development of novel combustion preparation processes has made it possible to produce ultrafine ceramic oxide powders at a surprisingly low reaction time and calcination temperatures with improved powder characteristics. The method includes an exothermic redox reaction between suitable oxidisers (e.g., metal nitrates) and an organic fuel (e.g., glycine, urea, and citric acid) [42][187][188][189]. In this synthesis process, important characteristics such as surface area, crystallite size, and the nature of the agglomerates can be controlled by the flame temperature and level of generated gaseous products, which in turn show great dependence on both the nature of the fuel and the oxidant-to-fuel ratio [190][191][192]. In general, all fuels used in this synthesis process serve two purposes: (1) acting as the H and C source, whereupon combustion forms H2O, CO2, and liberated heat; (2) forming complexes with the metal ions, improving the homogeneous mixing of metal cations in the final product [193]. When compared to conventional synthesis techniques, the combustion method provides attractive advantages. First, the relatively high heat generated from the redox reaction can greatly decrease the need for a rather energy-intensive high-temperature furnace, commonly used as an external energy source for conventional synthesis methods. Furthermore, this simple method is capable of producing ultrafine ceramic powders with very high purity [188][194]. The generated high temperature purges the powder of any possible volatile impurities present in the reactants. The combination of high-temperature gradients with rapid cooling rates in the generated combustion wave can result in the formation of unique microstructures, making it possible to produce powders with small average particle sizes and high porosity [188][190][194]. In addition, the rapid formation of large volumes of gaseous products generated in this method greatly dissipates the combustion heat, limiting the temperature rise and, therefore, preventing premature particle sintering between primary particles [195]. Furthermore, the gas evolution limits the formation of hard agglomerates by reducing the interparticle contacts, thus improving the sinterability of the final product [196]. Compared to solid-state and sol–gel processes, combustion reactions occur at high temperatures, typically in the range of several hundred to several thousand degrees Celsius. These high temperatures are necessary to initiate and sustain the exothermic reactions involved in combustion. While combustion processes typically involve the rapid oxidation of a fuel source, such as hydrocarbons, in the presence of oxygen, and the temperature required for combustion depends on the specific fuel and the nature of the combustion reaction, controlled reactions and phase transformation are the main characteristics of solid-state reactions [189][197]. On the other hand, the sol–gel process, in which hydrolysis and condensation take place at relatively lower temperatures ranging from room temperature to a few hundred degrees Celsius, enables controlling the composition, structure, and morphology of the product [158]. Regarding fuel cell application, combustion processes are not typically utilised directly in fuel cell applications or materials synthesis due to their high temperatures and rapid, uncontrolled nature. However, the heat generated from combustion reactions can be harnessed to generate high-temperature steam or thermal energy for fuel cell systems [198]. Solid-state processes are commonly employed in fuel cell materials syntheses, such as the fabrication of ceramic electrolytes or electrodes. The high temperatures used in solid-state processes enable the formation of dense, crystalline structures with optimised properties for fuel cell operation [146][199][200]. Sol–gel processes are particularly relevant in fuel cell applications for the production of thin films and nanoscale materials. They provide precise control over composition, morphology, and surface properties, allowing for the development of advanced fuel cell components with improved performance and stability [154][201][202]. Glycine (NH2CH2COOH), with an amino group at one end and a carboxylic acid group at the other end, has been commonly used as a complexing agent when reacted with different metal ions [192]. It is this zwitterionic characteristic of the molecule that allows for an efficient mixture of a variety of metal ions with different ionic sizes. Such characteristics can greatly inhibit selective precipitation among the reactive elements, greatly improving the homogeneity of the metal composites. In addition, glycine can act as a reliable fuel in the combustion reaction when oxidised by nitrate ions [203][204]. A great deal of research has reported the rapid and simple production of single- or multicomponent oxide ceramic powders using the glycine nitrate process [205]. The rapid and self-sustaining nature of glycine nitrate combustion reaction has made it possible to immediately produce homogeneous composite powders with relatively high surface area and very low carbon residue [206][207]. It is often claimed that a well-adjusted stoichiometry can result in an almost complete reaction, where the gaseous combustion products would mostly consist of CO2, N2, and H2O [208]. However, due to the fast reaction rates and, thus, the possible limitations imposed on the reaction kinetics, it is more likely that the combustion processes end up with incomplete reactions [209]. In fact, Pine et al. [210] reported a relatively high emission level of NOx and CO for a wide range of fuel-to-oxidant ratios used in this method. Despite such disadvantages, this synthesis technique has proven to be very successful for the laboratory-scale production of ceria-based SOFCs. Figure 8a,b illustrate a highly porous foam-like GDC particle using the glycine nitrate process before and after calcination. The powder was reported to possess an extremally porous structure with loosely agglomerated particles. Such properties have made it possible to prepare thin doped ceria films (<20 μm) using dry pressing, where the resulting powders were pelletised using hardened metal dies (Figure 7c,d) [197]. When compared with the carbonate precipitation method, lower conductivities have been reported for electrolyte material, especially doped ceria, prepared using the glycine nitrate process [197][211]. Despite the differences between the conductivity values reported by different groups, it can be generally said that electrolyte powders, especially doped ceria, prepared using the coprecipitation and glycine methods show higher conductivity and lower activation energy than samples prepared using the solid-state and sol–gel techniques.
References
- Ilbas, M.; Kumuk, B. Numerical Modelling of a Cathode-Supported Solid Oxide Fuel Cell (SOFC) in Comparison with an Electrolyte-Supported Model. J. Energy Inst. 2019, 92, 682–692.
- Li, R.; Peng, L.; Wang, X.; Yang, J.; Yan, D.; Pu, J.; Chi, B.; Li, J. Investigating the Performance of Glass/Al2O3 Composite Seals in Planar Solid Oxide Fuel Cells. Compos. Part B Eng. 2020, 192, 107984.
- Baldi, F.; Wang, L.; Pérez-fortes, M.; Maréchal, F. A Cogeneration System Based on Solid Oxide and Proton Exchange Membrane Fuel Cells With Hybrid Storage for Off-Grid Applications. Front. Energy Res. 2019, 6, 139.
- Vostakola, M.F.; Salamatinia, B.; Horri, B.A. A Review on Recent Progress in the Integrated Green Hydrogen Production Processes. Energies 2022, 15, 1209.
- Wang, J.; Zhang, X.; Li, Y.; Liu, P.; Chen, X.; Zhang, P.; Wang, Z.; Liu, X. Sweet Drinks as Fuels for an Alkaline Fuel Cell with Nonprecious Catalysts. Energies 2021, 14, 206.
- Abderezzak, B. Introduction to Hydrogen Technology. In Introduction to Transfer Phenomena in PEM Fuel Cell; ISTE Press-Elsevier: London, UK, 2018; pp. 1–51.
- Study, A.F.; Hu, H.; Zou, J.; Shan, L.; Jiang, X.; Ni, Y.; Li, X.; Qian, X. Conductivities in Yttrium-Doped Barium Zirconate: A First-Principles Study. Crystals 2023, 13, 401.
- Recirculation, G.; Bian, J.; Duan, L.; Yang, Y. Simulation and Economic Investigation of CO2 Separation from Gas Turbine Exhaust Gas by Molten Carbonate Fuel Cell with Exhaust Gas Recirculation and Selective Exhaust Gas Recirculation. Energies 2023, 16, 3511.
- Hooshyari, K.; Amini Horri, B.; Abdoli, H.; Fallah Vostakola, M.; Kakavand, P.; Salarizadeh, P. A Review of Recent Developments and Advanced Applications of High-Temperature Polymer Electrolyte Membranes for PEM Fuel Cells. Energies 2021, 14, 5440.
- Rabissi, C.; Zago, M.; Bresciani, F.; Gazdzicki, P.; Casalegno, A. A Novel Accelerated Stress Test for a Representative Enhancement of Cathode Degradation in Direct Methanol Fuel Cells. Energies 2023, 16, 3226.
- Weber, A. Fuel Flexibility of Solid Oxide Fuel Cells. Fuel Cells 2021, 21, 440–452.
- Ng, K.H.; Rahman, H.A.; Somalu, M.R. Review: Enhancement of Composite Anode Materials for Low-Temperature Solid Oxide Fuels. Int. J. Hydrogen Energy 2019, 44, 30692–30704.
- Das, S.; Dutta, K.; Nessim, G.D.; Kader, M.A. Introduction to Direct Methanol Fuel Cells. In Direct Methanol Fuel Cell Technology; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–12. ISBN 9780128191583.
- Ali, M.; Hassan, W.; El Haj, M.; Allagui, A.; Cha, S.W. On the Technical Challenges a Ff Ecting the Performance of Direct Internal Reforming Biogas Solid Oxide Fuel Cells. Renew. Sustain. Energy Rev. 2019, 101, 361–375.
- Taroco, H.A.; Santos, J.A.F.; Domingues, R.Z.; Matencio, T. Ceramic Materials for Solid Oxide Fuel Cells. In Advances in Ceramics-Synthesis and Characterization, Processing and Specific Applications; Sikalidis, C., Ed.; IntechOpen: Minas Gerais, Brazil, 2011; pp. 423–446.
- Pianko-Oprych, P.; Jaworski, Z.; Kendall, K. Cell, Stack and System Modelling. In High-temperature Solid Oxide Fuel Cells for the 21st Century; Academic Press: Cambridge, MA, USA, 2016; pp. 407–460. ISBN 9780124104532.
- Zhang, J.; Lenser, C.; Menzler, N.H.; Guillon, O. Comparison of Solid Oxide Fuel Cell (SOFC) Electrolyte Materials for Operation at 500 °C. Solid State Ionics 2020, 344, 115138.
- Wang, J. Barriers of Scaling-up Fuel Cells: Cost, Durability and Reliability. Energy 2015, 80, 509–521.
- Wang, J. System Integration, Durability and Reliability of Fuel Cells: Challenges and Solutions. Appl. Energy 2017, 189, 460–479.
- Ding, D.; Li, X.; Lai, Y.; Liu, M. Enhancing SOFC Cathode Performance by Surface Modification through Infiltratio. Energy Environ. Sci. 2014, 7, 552–575.
- Bi, L.; Traversa, E. Synthesis Strategies for Improving the Performance of Doped-BaZrO3 Materials in Solid Oxide Fuel Cell Applications. J. Mater. Res. 2014, 29, 1–15.
- Munira, S.; Hafiz, M.; Othman, D.; Rahman, M.A.; Jaafar, J.; Ismail, A.F.; Li, K. Recent Fabrication Techniques for Micro-Tubular Solid Oxide Fuel Cell Support: A Review. J. Eur. Ceram. Soc. 2015, 35, 1–22.
- Jiang, S.P. Development of Lanthanum Strontium Cobalt Ferrite Perovskite Electrodes of Solid Oxide Fuel Cells—A Review. Int. J. Hydrogen Energy 2019, 44, 7448–7493.
- Huang, N.; Han, B.; Wang, Y.; Li, Y.; Su, Y.; Guan, W.; Zhou, X.; Chai, M.; Singhal, S.C. On the Dependence of Interfacial Resistance on Contact Materials between Cathode and Interconnect in Solid Oxide Fuel Cells. Int. J. Hydrogen Energy 2021, 46, 20078–20086.
- He, S.; Jiang, S.P. Electrode/Electrolyte Interface and Interface Reactions of Solid Oxide Cells: Recent Development and Advances. Prog. Nat. Sci. Mater. Int. 2021, 31, 341–372.
- Santoni, F.; Silva Mosqueda, D.M.; Pumiglia, D.; Viceconti, E.; Conti, B.; Boigues Muñoz, C.; Bosio, B.; Ulgiati, S.; McPhail, S.J. In-Situ Study of the Gas-Phase Composition and Temperature of an Intermediate-Temperature Solid Oxide Fuel Cell Anode Surface Fed by Reformate Natural Gas. J. Power Sources 2017, 370, 36–44.
- Su, H.; Hu, Y.H. Progress in Low-Temperature Solid Oxide Fuel Cells with Hydrocarbon Fuels. Chem. Eng. J. 2020, 402, 126235.
- Liu, J.; Zhu, C.; Zhu, D.; Jia, X.; Zhang, Y.; Yu, J.; Li, X.; Yang, M. High Performance Low-Temperature Solid Oxide Fuel Cells Based on Nanostructured Ceria-Based Electrolyte. Nanomaterials 2021, 11, 2231.
- Zarabi, S.; Imran, M.; Lund, P.D. A Review on Solid Oxide Fuel Cell Durability: Latest Progress, Mechanisms, and Study Tools. Renew. Sustain. Energy Rev. 2022, 161, 112339.
- Prakash, B.S.; Pavitra, R.; Kumar, S.S.; Aruna, S.T. Electrolyte Bi-Layering Strategy to Improve the Performance of an Intermediate Temperature Solid Oxide Fuel Cell: A Review. J. Power Sources 2018, 381, 136–155.
- Afzal, M.; Raza, R.; Du, S.; Lima, R.B.; Zhu, B. Synthesis of Ba0.3Ca0.7Co0.8Fe0.2O3-δ Composite Material as Novel Catalytic Cathode for Ceria-Carbonate Electrolyte Fuel Cells. Electrochim. Acta 2015, 178, 385–391.
- Eric, D. Wachsman; Kang Taek Lee Lowering the Temperature of Solid Oxide Fuel Cells. Science 2011, 334, 928–935.
- Gao, Z.; Mogni, L.; Miller, E.C.; Railsback, J.; Barnett, S.A. A Perspective on Low-Temperature Solid Oxide Fuel Cells. Energy Environ. Sci. 2016, 9, 1602–1644.
- Coddet, P.; Liao, H.; Coddet, C. A Review on High Power SOFC Electrolyte Layer Manufacturing Using Thermal Spray and Physical Vapour Deposition Technologies. Adv. Manuf. 2014, 2, 212–221.
- Liang, F.; Yang, J.; Zhao, Y.; Zhou, Y.; Yan, Z.; He, J.; Yuan, Q.; Wu, J.; Liu, P.; Zhong, Z.; et al. A Review of Thin Film Electrolytes Fabricated by Physical Vapor Deposition for Solid Oxide Fuel Cells. Int. J. Hydrogen Energy 2022, 47, 36926–36952.
- Horri, B.A.; Selomulya, C.; Wang, H. Electrochemical Characteristics and Performance of Anode-Supported SOFCs Fabricated Using Carbon Microspheres as a Pore-Former. Int. J. Hydrogen Energy 2012, 37, 19045–19054.
- Kharton, V.V.; Marques, F.M.B.; Atkinson, A. Transport Properties of Solid Oxide Electrolyte Ceramics: A Brief Review. Solid State Ionics 2004, 174, 135–149.
- Lamy, C.; Millet, P. A Critical Review on the Definitions Used to Calculate the Energy Efficiency Coefficients of Water Electrolysis Cells Working under near Ambient Temperature Conditions. J. Power Sources 2020, 447, 227350.
- Welaya, Y.M.A.; Mosleh, M.; Ammar, N.R. Thermodynamic Analysis of a Combined Gas Turbine Power Plant with a Solid Oxide Fuel Cell for Marine Applications. Int. J. Nav. Archit. Ocean Eng. 2013, 5, 529–545.
- Ouyang, T.; Zhao, Z.; Lu, J.; Su, Z.; Li, J. Waste Heat Cascade Utilisation of Solid Oxide Fuel Cell for Marine Applications. J. Clean. Prod. 2020, 275, 124133.
- Micoli, L.; Russo, R.; Coppola, T.; Pietra, A. Performance Assessment of the Heat Recovery System of a 12 MW SOFC-Based Generator on Board a Cruise Ship through a 0D Model. Energies 2023, 16, 3334.
- Fallah Vostakola, M.; Amini Horri, B. Progress in Material Development for Low-Temperature Solid Oxide Fuel Cells: A Review. Energies 2021, 14, 1280.
- Shina, J.W.; Go, D.; Kye, S.H.; Leea, S.; An, J. Review on Process-Microstructure-Performance Relationship in ALD- Engineered SOFCs. J. Phys. Energy 2019, 1, 042002.
- Frishberg, I. V Gas-Phase Method of Metal Powder Production. In Handbook of Non-Ferrous Metal Powders; Elsevier Ltd.: Yekaterinburg, Russia, 2019; Volume 1, pp. 187–200. ISBN 9780081005439.
- Sarangan, A. Nanofabrication. In Fundamentals and Applications of Nanophotonics; Woodhead Publishing: Dayton, OH, USA, 2016; pp. 149–184. ISBN 9781782424642.
- Igumenov, I.K.; Lukashov, V. V Modern Solutions for Functional Coatings in CVD Processes. Coatings 2022, 12, 1265.
- Carlsson, J.; Martin, P.M. Chemical Vapor Deposition. In Handbook of Deposition Technologies for Films and Coatings; William Andrew: Norwich, NY, USA, 2010; pp. 314–363.
- Sabzi, M.; Anijdan, S.H.M.; Shamsodin, M.; Farzam, M.; Hojjati-Najafabadi, A.; Feng, P.; Park, N.; Lee, U. A Review on Sustainable Manufacturing of Ceramic-Based Thin Films by Chemical Vapor Deposition (CVD): Reactions Kinetics and the Deposition Mechanisms. coatings 2023, 13, 188.
- Choi, H.R.; Neoh, K.C.; Choi, H.J.; Han, G.D.; Jang, D.Y.; Kim, D.; Shim, J.H. Nanoporous Silver Cathode Surface-Treated by Aerosol-Assisted Chemical Vapor Deposition of Gadolinia-Doped Ceria for Intermediate-Temperature Solid Oxide Fuel Cells. J. Power Sources 2018, 402, 246–251.
- Jang, D.Y.; Kim, M.; Kim, J.W.; Bae, K.; Son, J.; Schlupp, M.V.F.; Shim, J.H. High Performance Anode-Supported Solid Oxide Fuel Cells with Thin Film Yttria-Stabilized Zirconia Membrane Prepared by Aerosol-Assisted Chemical Vapor Deposition. J. Electrochem. Soc. 2017, 164, F484–F490.
- Sakai, T.; Kato, T.; Tanaka, Y.; Goto, T. Preparation of an Yttria-Stabilized Zirconia Electrolyte on a Porous Ni-Based Cermet Substrate by Laser Chemical Vapor Deposition. Mater. Today Commun. 2022, 33, 104169.
- Sawka, A.; Kwatera, A. Low Temperature Synthesis of Y2O3–Doped CeO2 Layers Using MOCVD. Mater. Sci. Eng. B 2022, 276, 115580.
- Sawka, A. Chemical Vapour Deposition of Scandia-Stabilised Zirconia Layers on Tubular Substrates at Low Temperatures. Materials 2022, 15, 2120.
- Sawka, A. MOCVD Growth of Gadolinium Oxide Layers on Tubes. Ceram. Int. J. 2023, 49, 23835–23843.
- Lee, Y.H.; Chang, I.; Cho, G.Y.; Park, J.; Yu, W.; Tanveer, W.H.; Cha, S.W. Thin Film Solid Oxide Fuel Cells Operating Below 600 ℃: A Review. Int. J. Precis. Eng. Manuf. Technol. 2018, 5, 441–453.
- Zhang, Y.; Knibbe, R.; Sunarso, J.; Zhong, Y.; Zhou, W.; Shao, Z. Recent Progress on Advanced Materials for Solid-Oxide Fuel Cells Operating Below 500 °C. Adv. Mater. 2017, 29, 1700132.
- Pinto, G. Sputtering Physical Vapour Deposition (PVD) Coatings: ACritical Review on Process Improvement AndMarket Trend Demands. Coatings 2018, 8, 402.
- Seshan, K.; Schepis, D. Handbook of Thin Film Deposition, 4th ed.; Seshan, K., Globalfoundries, D.S., Eds.; William Andrew: Norwich, NY, USA, 2018; ISBN 9780128123126.
- Infortuna, B.A.; Harvey, A.S.; Gauckler, L.J. Microstructures of CGO and YSZ Thin Films by Pulsed Laser Deposition. Adv. Funct. Mater. 2008, 18, 127–135.
- Coppola, N.; Polverino, P.; Carapella, G.; Ciancio, R.; Rajak, P.; Dario, M.; Martinelli, F.; Maritato, L.; Pianese, C. Large Area Deposition by Radio Frequency Sputtering of Gd0.1Ce0.9O1.95 Buffer Layers in Solid Oxide Fuel Cells: Structural, Morphological and Electrochemical Investigation. Materials (Basel) 2021, 14, 5826.
- Mattox, D.M. Handbook of Physical Vapor Deposition (PVD) Processing, 2nd ed.; William Andrew: Norwich, NY, USA, 2010; ISBN 9780815520375.
- Lamas, J.S.; Leroy, W.P.; Depla, D. Influence of Target–Substrate Distance and Composition on the Preferential Orientation of Yttria-Stabilized Zirconia Thin Films. Thin Solid Films 2012, 520, 4782–4785.
- Nédélec, R.; Uhlenbruck, S.; Sebold, D.; Haanappel, V.A.C.; Buchkremer, H.; Stöver, D. Dense Yttria-Stabilised Zirconia Electrolyte Layers for SOFC by Reactive Magnetron Sputtering. J. Power Sources 2012, 205, 157–163.
- Vafaeenezhad, S.; Hanifi, A.R.; Laguna-bercero, M.A.; Etsell, T.H. Microstructure and Long-Term Stability of Ni–YSZ Anode Supported Fuel Cells: A Review To. Mater. Futur. 2022, 1, 042101.
- Hanifi, A.R.; Shinbine, A.; Etsell, T.H. Development Of Monolithic YSZ Porous and Dense Layers through Multiple Slip Casting for Ceramic Fuel Cell Applications. Int. J. Appl. Ceram. Technol. 2012, 9, 1011–1021.
- Fonseca, F.C.; Uhlenbruck, S.; Nédélec, R.; Sebold, D.; Buchkremer, H.P. Temperature and Bias Effects on Sputtered Ceria Diffusion Barriers for Solid Oxide Fuel Cells. J. Electrochem. Soc. 2010, 157, B1515–B1519.
- Ostadi, A.; Hosseini, S.H.; Fordoei, M.E. The Effect of Temperature and Roughness of the Substrate Surface on the Microstructure and Adhesion Strength of EB-PVD ZrO2-%8wtY2O3 Coating. Ceram. Int. 2020, 46, 2287–2293.
- Tanhaei, M.; Mozammel, M. Yttria-Stabilized Zirconia Thin Film Electrolyte Deposited by EB-PVD on Porous Anode Support for SOFC Applications. Ceram. Int. 2017, 43, 3035–3042.
- Shim, J.H.; Han, G.D.; Choi, H.J.; Kim, Y.; Xu, S.; An, J.; Kim, Y.B.; Graf, T.; Schladt, T.D.; Gür, T.M.; et al. Atomic Layer Deposition for Surface Engineering of Solid Oxide Fuel Cell Electrodes. Int. J. Precis. Eng. Manuf.-Green Technol. 2019, 6, 629–646.
- Lee, S.; Lee, Y.; Park, J.; Yu, W.; Cho, G.Y.; Kim, Y.; Cha, S.W. Effect of Plasma-Enhanced Atomic Layer Deposited YSZ Inter-Layer on Cathode Interface of GDC Electrolyte in Thin Film Solid Oxide Fuel Cells. Renew. Energy 2019, 144, 123–128.
- Shin, J.W.; Oh, S.; Lee, S.; Yu, J.G.; Park, J.; Go, D.; Yang, B.C.; Kim, H.J.; An, J. Ultrathin Atomic Layer-Deposited CeO2 Overlayer for High-Performance Fuel Cell Electrodes. ACS Appl. Mater. Interfaces 2019, 11, 46651–46657.
- Ahn, C.H.; Kim, S.H.; Yun, M.G.; Cho, H.K. Design of Step Composition Gradient Thin Film Transistor Channel Layers Grown by Atomic Layer Deposition. Appl. Phys. Lett. 2014, 105, 223513.
- Knoops, H.C.M.; Potts, S.E.; Bol, A.A.; Kessels, W.M.M. Atomic Layer Deposition. In Handbook of Crystal Growth: Thin Films and Epitaxy, 2nd ed.; Elsevier B.V.: Eindhoven, The Netherlands, 2015; pp. 1101–1134. ISBN 9780444633057.
- Shahmohammadi, M.; Mukherjee, R.; Sukotjo, C.; Diwekar, U.M.; Takoudis, C.G. Recent Advances in Theoretical Development of Thermal Atomic Layer Deposition: A Review. Nanomaterials 2022, 12, 831.
- Shim, J.H.; Choi, H.J.; Kim, Y.; Torgersen, J.; An, J.; Lee, M.H.; Prinz, F.B. Process-Property Relationship in High-K ALD SrTiO3 and BaTiO3: A Review. J. Mater. Chem. C 2017, 5, 8000–8013.
- Detavernier, C.; Dendooven, J.; Sree, S.P.; Ludwigc, K.F.; Martens, J.A. Tailoring Nanoporous Materials by Atomic Layer Deposition. Chem. Soc. Rev. 2011, 40, 5242–5253.
- Koponen, S.E.; Gordon, P.G.; Barry, S.T. Principles of Precursor Design for Vapour Deposition Methods. Polyhedron 2016, 108, 59–66.
- Putkonen, B.M.; Sajavaara, T.; Johansson, L.; Niinistö, L. Low-Temperature ALE Deposition of Y2O3 Thin Films from β-Diketonate Precursors**. Chem. Vap. Depos. 2001, 7, 44–50.
- Hausmann, D.M.; Kim, E.; Becker, J.; Gordon, R.G. Atomic Layer Deposition of Hafnium and Zirconium Oxides Using Metal Amide Precursors. Chem. Mater. 2002, 14, 4350–4358.
- Cassir, M.; Putkonen, M.; Niinisto, L. Electrical Properties of Thin Yttria-Stabilized Zirconia Overlayers Produced by Atomic Layer Deposition for Solid Oxide Fuel Cell Applications. Appl. Surf. Sci. 2007, 253, 3962–3968.
- Kerman, K.; Lai, B.-K.; Ramanathan, S. Nanoscale Compositionally Graded Thin-Film Electrolyte Membranes for Low-Temperature Solid Oxide Fuel Cells. Adv. Energy Mater. 2012, 2, 656–661.
- Baek, J.D.; Yu, C.; Su, P. A Silicon-Based Nanothin Film Solid Oxide Fuel Cell Array with Edge Reinforced Support for Enhanced Thermal Mechanical Stability. Nano Lett. 2016, 16, 2413–2417.
- Shim, J.H.; Chao, C.-C.; Huang, H.; Prinz, F.B. Atomic Layer Deposition of Yttria-Stabilized Zirconia for Solid Oxide Fuel Cells. Chem. Mater. 2007, 19, 3850–3854.
- Lee, C.; Cho, M.-Y.; Kim, M.; Jang, J.; Oh, Y.; Oh, K.; Kim, S.; Park, B.; Kim, B.; Koo, S.-M.; et al. Applicability of Aerosol Deposition Process for Flexible Electronic Device and Determining the Film Formation Mechanism with Cushioning Effects. Sci. Rep. 2019, 9, 2166.
- Erilin, I.S.; Agarkov, D.A.; Burmistrov, I.N.; Pukha, V.E.; Yalovenko, D.V.; Lyskov, N.V.; Levin, M.N.; Bredikhin, S.I. Aerosol Deposition of Thin-Film Solid Electrolyte Membranes for Anode-Supported Solid Oxide Fuel Cells. Mater. Lett. 2020, 266, 127439.
- Bredikhin, S.I.; Agarkov, D.A.; Agarkova, E.A.; Burmistrov, I.N.; Cherkasov, A.M.; Pukha, V.E.; Yalovenko, D.V.; Lyskov, N.V. Aerosol Deposition as a Promising Technique to Fabricating a Thin-Film Solid Electrolyte of Solid Oxide Fuel Cells. ECS Trans. 2019, 91, 403–413.
- Hanft, D.; Exner, J.; Schubert, M.; Stöcker, T.; Fuierer, P.; Moos, R. An Overview of the Aerosol Deposition Method: Process Fundamentals and New Trends in Materials Applications. J. Ceram. Sci. Technol. 2015, 182, 147–182.
- Akedo, J.; Ichiki, M.; Kikuchi, K.; Maeda, R. Jet Molding System for Realization of Three-Dimensional Micro-Structures. Sens. Actuators A 1998, 69, 106–112.
- Akedo, J.; Lebedev, M. Microstructure and Electrical Properties of Lead Zirconate Titanate (Pb(Zr52/Ti48)O3) Thick Films Deposited by Aerosol Deposition Method. Jpn. J. Appl. Phys. 1999, 38, 5397.
- Akedo, J. Room Temperature Impact Consolidation (RTIC) of Fine Ceramic Powder by Aerosol Deposition Method and Applications to Microdevices. J. Therm. Spray Technol. 2008, 17, 181–198.
- Furuya, Y.; Tanaka, M.; Kitaoka, S.; Hasegawa, M. Method for Determining Particle Velocity during Aerosol Deposition by Impact Pressure of the Particles. Surf. Coat. Technol. 2022, 443, 128616.
- Furuya, Y.; Konuma, S.; Hasegawa, M. Deposition Mechanism of Alumina Particles in Aerosol Deposition Based on the Kinetic Energy of Particles. Surf. Coat. Technol. 2023, 458, 129362.
- Kuroyanagi, S.; Shinoda, K.; Yumoto, A.; Akedo, J. Size-Dependent Quasi Brittle–Ductile Transition of Single Crystalline Alpha-Alumina Particles during Microcompression Tests. Acta Mater. J. 2020, 195, 588–596.
- Yeganeh, A.Z.; Jadidi, M.; Moreau, C.; Dolatabadi, A. Surface & Coatings Technology Numerical Modeling of Aerosol Deposition Process. Surf. Coat. Technol. 2019, 370, 269–287.
- Matsubayashi, Y.; Akedo, J. Aerosol Deposition of Alumina Films on Microstructured Silicon Substrates. Ceram. Int. 2023, 49, 21570–21576.
- Schubert, M.; Exner, J.; Moos, R. Influence of Carrier Gas Composition on the Stress of Al2O3 Coatings Prepared by the Aerosol Deposition Method. Materials 2014, 7, 5633–5642.
- Seto, N.; Endo, K.; Sakamoto, N.; Hirose, S.; Akedo, J. Hard A-Al2O3 Film Coating on Industrial Roller Using Aerosol Deposition Method. J. Therm. Spray Technol. 2014, 23, 1373–1381.
- Lee, W.; Kim, H.; Lee, D.; Jeong, M.; Lim, D.; Nam, S. Al2O3-Nanodiamond Composite Coatings with High Durability and Hydrophobicity Prepared by Aerosol Deposition. Surf. Coat. Technol. 2012, 206, 4679–4684.
- Xiao, Y.; Wang, M.; Bao, D.; Wang, Z.; Jin, F.; Wang, Y.; He, T. Performance of Fuel Electrode-Supported Tubular Protonic Ceramic Cells Prepared through Slip Casting and Dip-Coating Methods. Catalysts 2023, 13, 182.
- Winter, R.L.; Singh, P.; King, M.K., Jr.; Mahapatra, M.K.; Sampathkumaran, U. Protective Ceramic Coatings for Solid Oxide Fuel Cell (SOFC) Balance-of-Plant Components. Adv. Mater. Sci. Eng. 2018, 2018, 9121462.
- Lei, L.; Bai, Y.; Liu, Y. An Investigation on Dip-Coating Technique for Fabricating Anode-Supported Solid Oxide Fuel Cells. Int. J. Appl. Ceram. Technol. 2015, 12, 351–357.
- Sahu, S.K.; Panthi, D.; Soliman, I.; Feng, H.; Du, Y. Fabrication and Performance of Micro-Tubular Solid Oxide Cells. Energies 2022, 15, 3536.
- Exploration, C.A.B.; Butt, M.A. Thin-Film Coating Methods: A Successful Marriage of High-Quality and Cost-Effectiveness—A Brief Exploration. Coatings 2022, 12, 1115.
- Panthi, D.; Tsutsumi, A. A Novel Multistep Dip-Coating Method for the Fabrication of Anode-Supported Microtubular Solid Oxide Fuel Cells. J. Solid State Electrochem. 2014, 18, 1899–1905.
- Niazmand, M.; Maghsoudipour, A.; Alizadeh, M.; Khakpour, Z.; Kariminejad, A. Effect of Dip Coating Parameters on Microstructure and Thickness of 8YSZ Electrolyte Coated on NiO-YSZ by Sol-Gel Process for SOFCs Applications. Ceram. Int. 2022, 48, 16091–16098.
- Delory, M.; Chorein, B.; Brulez, A.; Mees, L.; Grosjean, N.; Fiorani, T.; Benayoun, S. Role of Surface-Tension Additive in Coating Framing Effect Topography Mathilde. Colloids Surf. A Physicochem. Eng. Asp. 2023, 658, 130554.
- Phillips, C.O.; Beynon, D.G.; Hamblyn, S.M.; Davies, G.R.; Gethin, D.T.; Claypole, T.C. A Study of the Abrasion of Squeegees Used in Screen Printing and Its Effect on Performance with Application in Printed Electronics. Coatings 2014, 4, 356–379.
- Howatt, G.N.; Breckenridge, R.G.; Brownlow, J.M. Fabrication of Thin Ceramic Sheets for Capacitors. J. Am. Ceram. Soc. 1947, 30, 237–242.
- Nishihora, R.K.; Rachadel, P.L.; Quadri, M.G.N.; Hotza, D. Manufacturing Porous Ceramic Materials by Tape Casting—A Review. J. Eur. Ceram. Soc. 2018, 38, 988–1001.
- Nie, L.; Sun, Q.; Liu, Z.; Liu, M. Graded Porous Solid Oxide Fuel Cells Fabricated by Multilayer Tape Casting and Co-Firing Progress. Int. J. Hydrogen Energy 2015, 40, 16503–16508.
- Lee, S.; Lee, K.; Jang, Y.; Bae, J. Fabrication of Solid Oxide Fuel Cells (SOFCs) by Solvent-Controlled Co-Tape Casting Technique. Int. J. Hydrogen Energy 2017, 42, 1648–1660.
- Snowdon, A.L.; Steinberger-wilckens, R. Five-Layer Reverse Tape Casting of IT-SOFC. Int. J. Appl. Ceram. Technol. 2022, 19, 289–298.
- Transactions, E.C.S.; Society, T.E. Tubular Solid Oxide Fuel Cells Fabricated by Tape-Casting and Dip-Coating Methods. ECS Trans. 2019, 91, 329–337.
- Bulatova, R.; Jabbari, M.; Kaiser, A.; Negra, M.D.; Andersen, K.B.; Gurauskis, J.; Bahl, C.R.H. Thickness Control and Interface Quality as Functions of Slurry Formulation and Casting Speed in Side-by-Side Tape Casting. J. Eur. Ceram. Soc. 2014, 34, 4285–4295.
- Aishwarya, K.P.; Darve, R.; Gadde, J.R.; Kale, H. Optimisation of Slurry Compositions for Improving the Mechanical Properties of Low Temperature Co-Fired Ceramic (LTCC) Tapes To. IOP Conf. Ser. 2022, 1248, 012096.
- Varghese, J.; Joseph, N.; Jantunen, H. Multilayer Glass–Ceramic/Ceramic Composite Substrates. In Encyclopedia of Materials: Technical Ceramics and Glasses; Elsevier: Oulu, Finland, 2021; Volume 3, pp. 437–451. ISBN 9780128185421.
- Myung, J.; Jun, H.; Park, H.; Hwan, M.; Hyun, S. Fabrication and Characterization of Planar-Type SOFC Unit Cells Using the Tape-Casting/Lamination/Co-Firing Method. Int. J. Hydrogen Energy 2011, 37, 498–504.
- Santos, T.H.; Grilo, J.P.F.; Loureiro, F.J.A.; Fagg, D.P.; Fonseca, F.C.; Macedo, D.A. Structure, Densification and Electrical Properties of Gd3+ and Cu2+ Co- Doped Ceria Solid Electrolytes for SOFC Applications: Effects of Gd2O3 Content. Ceram. Int. 2018, 44, 2745–2751.
- Jabbari, M.; Bulatova, R.; Tok, A.I.Y.; Bahl, C.R.H.; Mitsoulis, E.; Hattel, J.H. Ceramic Tape Casting: A Review of Current Methods and Trends with Emphasis on Rheological Behaviour and Flow Analysis. Mater. Sci. Eng. B 2016, 212, 39–61.
- Coduri, M.; Checchia, S.; Longhi, M.; Ceresoli, D. Rare Earth Doped Ceria: The Complex Connection Between Structure and Properties. Front. Chem. 2018, 6, 526.
- Mahmud, L.S.; Muchtar, A. Challenges in Fabricating Planar Solid Oxide Fuel Cells: A Review. Renew. Sustain. Energy Rev. 2017, 72, 105–116.
- Mishra, A.; Bhatt, N.; Bajpai, A.K. Nanostructured Superhydrophobic Coatings for Solar Panel Applications. In Nanomaterials-Based Coatings; Elsevier Inc.: Jabalpur, India, 2019; pp. 397–424. ISBN 9780128158845.
- Yilbas, B.S.; Al-Sharafi, A.; Ali, H. Surfaces for Self-Cleaning. In Self-Cleaning of Surfaces and Water Droplet Mobility; Elsevier: Dhahran, Saudi Arabia, 2019; pp. 45–98. ISBN 9780128147764.
- Cui, Z.; Liao, L. Coating and Printing Processes. In Solution Processed Metal Oxide Thin Films for Electronic Applications; Elsevier Inc.: Changsha, China, 2020; pp. 83–97. ISBN 9780128149300.
- Zhang, J.X.J.; Hoshino, K. Fundamentals of Nano/ Microfabrication and Scale Effect. In Molecular Sensors and Nanodevices Principles, Designs and Applications in Biomedical Engineering; Academic Press: Cambridge, MA, USA, 2019; pp. 43–111. ISBN 9780128148624.
- Xu, X.; Xia, C.; Huang, S.; Peng, D. YSZ Thin Films Deposited by Spin-Coating for IT-SOFCs. Ceram. Int. 2005, 31, 1061–1064.
- Wang, J.; Lu, Z.; Chen, K.; Huang, X.; Ai, N.; Hu, J.; Zhang, Y.; Su, W. Study of Slurry Spin Coating Technique Parameters for the Fabrication of Anode-Supported YSZ Films for SOFCs Jiaming. J. Power Sources 2007, 164, 17–23.
- Chen, M.; Hee, B.; Xu, Q.; Guk, B.; Ping, D. Fabrication and Performance of Anode-Supported Solid Oxide Fuel Cells via Slurry Spin Coating. J. Memb. Sci. 2010, 360, 461–468.
- Yang, Q.; Wang, Y.; Tian, D.; Wu, H.; Ding, Y.; Lu, X.; Chen, Y.; Lin, B. Enhancing Performance and Stability of Symmetrical Solid Oxide Fuel Cells via Quasi-Symmetrical Ceria-Based Buffer Layers. Ceram. Int. 2022, 48, 27509–27515.
- Ramasamy, D.; Nasani, N.; Pukazhselvan, D.; Fagg, D.P. Increased Performance by Use of a Mixed Conducting Buffer Layer, Terbia-Doped Ceria, for Nd2NiO4þd SOFC/SOEC Oxygen Electrodes. Int. J. Hydrogen Energy 2019, 44, 31466–31474.
- Wu, Y.; Sang, J.; Liu, Z.; Fan, H.; Cao, B.; Wang, Q.; Yang, J.; Guan, W.; Liu, X.; Wang, J. Enhancing the Performance and Stability of Solid Oxide Fuel Cells by Adopting Samarium-Doped Ceria Buffer Layer. Ceram. Int. 2023, 49, 20290–20297.
- Feng, Z.; Zhang, X.; Luo, D. Evolution and Effect on Electrolysis Performance of Pores in YSZ Electrolyte Films Prepared by Screen-Printing. Ceram. Int. 2023, 49, 10731–10737.
- Baharuddin, N.A. Fabrication of High-Quality Electrode Films for Solid Oxide Fuel Cell by Screen Printing: A Review on Important Processing Parameters. Int. J. Energy Res. 2020, 44, 8296–8313.
- Graca, V.C.D.; Holz, L.I.V.; Loureiro, F.J.A.; Mikhalev, S.M.; Fagg, D.P. Ba2NiMoO6-δ as a Potential Electrode for Protonic Ceramic Electrochemical Cells. Int. J. Hydrogen Energy 2023.
- Tomašegovi, T. Properties and Colorimetric Performance of Screen-Printed Thermochromic/UV-Visible Fluorescent Hybrid Ink Systems. Appl. Sci. 2021, 11, 11414.
- Somalu, M.R.; Yufit, V.; Brandon, N.P. The Effect of Solids Loading on the Screen-Printing and Properties of Nickel/Scandia-Stabilized-Zirconia Anodes for Solid Oxide Fuel Cells. Int. J. Hydrogen Energy 2013, 38, 9500–9510.
- Mücke, R.; Büchler, O.; Menzler, N.H.; Lindl, B.; Vaßen, R.; Buchkremer, H.P. High-Precision Green Densities of Thick Films and Their Correlation with Powder, Ink, and Film Properties. J. Eur. Ceram. Soc. 2014, 34, 3897–3916.
- Von Dollen, P.; Barnett, S. A Study of Screen Printed Yttria-Stabilized Zirconia Layers for Solid Oxide Fuel Cells. J. Am. Ceram. Soc. 2005, 3368, 3361–3368.
- Potts, S.; Phillips, C.; Jewell, E.; Clifford, B.; Lau, Y.C.; Claypole, T. High-Speed Imaging the Effect of Snap-off Distance and Squeegee Speed on the Ink Transfer Mechanism of Screen-Printed Carbon Pastes. J. Coat. Technol. Res. 2020, 17, 447–459.
- Na, S.; Tio, B.; Chaou, F.; Bendahhou, A.; Chourti, K.; Jalafi, I.; Yahakoub, E.H.; El Barkany, S.; Abou-salama, M. Optimisation of Electrical Conductivity and Dielectric Properties of Sn4+-Doped (Na0.5Bi0.5)TiO3 Perovskite. Ceram. Int. 2023, 49, 17940–17952.
- Kaur, P.; Singh, K. Structural, Morphological and Conducting Properties of Copper and Nickel-Substituted Strontium Manganite. Mater. Today Commun. 2023, 35, 106056.
- Kim, D.; Taek, K. Effect of Lanthanide (Ln=La, Nd, and Pr) Doping on Electrochemical Performance of Ln2NiO4+δ−YSZ Composite Cathodes for Solid Oxide Fuel Cells. Ceram. Int. J. 2021, 47, 2493–2498.
- Chandrasekaran, R.E.G. Structural, Optical and Magnetic Characterization of Cu-Doped ZnO Nanoparticles Synthesized Using Solid State Reaction Method. J. Mater. Sci. Mater. Electron. 2010, 21, 1168–1173.
- Rafique, M.; Nawaz, H.; Rafique, M.S.; Tahir, M.B.; Nabi, G.; Khalid, N.R. Material and Method Selection for Efficient Solid Oxide Fuel Cell Anode: Recent Advancements and Reviews. Int. J. Energy Res. 2019, 43, 2423–2446.
- Yang, H.; Wu, N. Ionic Conductivity and Ion Transport Mechanisms Ofsolid-state Lithium-ion Battery Electrolytes: A Review. Energy Sci. Eng. 2022, 10, 1643–1671.
- Jiang, S.P. Solid-State Electrochemistry and Solid Oxide Fuel Cells: Status and Future Prospects. Electrochem. Energy Rev. 2022, 5, 5–21.
- Zhan, Z.; Wen, T.; Tu, H.; Lu, Z. AC Impedance Investigation of Samarium-Doped Ceria. J. Electrochem. Soc. 2001, 148, 427–432.
- Martinelli, H.; Lamas, D.G.; Leyva, A.G.; Sacanell, J. Influence of Particle Size and Agglomeration in Solid Oxide Fuel Cell Cathodes Using Manganite Nanoparticles. Mater. Res. Express 2018, 5, 075013.
- Tolchard, J.R.; Grande, T. Chemical Compatibility of Candidate Oxide Cathodes for BaZrO3 Electrolytes. Solid State Ionics 2007, 178, 593–599.
- Mehranjani, A.S.; Cumming, D.J.; Sinclair, D.C.; Rothman, R.H. Journal of the European Ceramic Society Low-Temperature Co-Sintering for Fabrication of Zirconia / Ceria Bi-Layer Electrolyte via Tape Casting Using a Fe2O3 Sintering Aid. J. Eur. Ceram. Soc. 2017, 37, 3981–3993.
- Mah, J.C.W.; Aznam, I.; Muchtar, A.; Somalu, M.R.; Raharjo, J. Synthesis of (Cu,Mn,Co)3O4 Spinel: Effects of Citrate-to-Nitrate Ratio on Its Homogeneity and Electrical Properties. Energies 2023, 16, 1382.
- Lian, Z.; Chen, F.; Song, X.; Liao, D.; Peng, K. Electrochemical Performance of Li13.9Sr0.1Zn(GeO4)4 Prepared by Sol-Gel for Solid Oxide Fuel Cell Electrolyte. J. Alloys Compd. 2021, 882, 160696.
- Rivai, A.K.; Shabrina, N.; Setiawan, A.R. Synthesis and Characterization of 8YSZ/5GDC Composite and Dissimiliar-Bulk by Sonochemistry Assisted Sol-Gel Method. J. Phys. Conf. Ser. 2017, 817, 012001.
- Cushing, B.L.; Kolesnichenko, V.L.; Connor, C.J.O. Recent Advances in the Liquid-Phase Syntheses of Inorganic Nanoparticles. Chem. Rev. 2004, 104, 3893–3946.
- Vostakola, M.F.; Yekta, B.E.; Mirkazemi, S.M. The Effects of Vanadium Pentoxide to Oxalic Acid Ratio and Different Atmospheres on the Formation of VO2 Nanopowders Synthesized via Sol–Gel Method. J. Electron. Mater. 2017, 46, 6689–6697.
- Shabani, S.; Naghizadeh, R.; Fallah Vostakola, M.; Golestanifard, F. The Effect of MgO Addition on the Properties of Alumina-Based Ceramic Cores Prepared via Sol–Gel Process. J. Sol-Gel Sci. Technol. 2020, 96, 539–549.
- Medina-Ramirez, I.; Hernandez-Ramirez, A.; Maya-Trevino, M.L. Synthesis Methods for Photocatalytic Materials. In Photocatalytic Semiconductors; Springer: Cham, Switzerland, 2015; pp. 69–102. ISBN 9783319109992.
- Hossain, S.; Abdalla, A.M.; Noorazean, S.; Jamain, B.; Hj, J. A Review on Proton Conducting Electrolytes for Clean Energy and Intermediate Temperature-Solid Oxide Fuel Cells. Renew. Sustain. Energy Rev. 2017, 79, 750–764.
- Navas, D.; Fuentes, S.; Castro-alvarez, A.; Chavez-Angel, E. Review on Sol-Gel Synthesis of Perovskite and Oxide Nanomaterials. Gels 2021, 7, 275.
- Huang, W.; Shuk, P.; Greenblatt, M. Properties of Sol-Gel Prepared Ce1-XSmxO2-x/2 Solid Electrolytes. Solid State Ionics 1997, 100, 23–27.
- Piñol, S.; Najib, M.; Bastidas, D.M.; Calleja, A.; Capdevila, X.G.; Segarra, M.; Espiell, F.; Ruiz-Morales, J.C.; Marrero-López, D.; Nuñez, P. Microstructure–Conductivity Relationship in Gd- and Sm-Doped Ceria-Based Electrolytes Prepared by the Acrylamide Sol–Gel-Related Method. J. Solid State Electrochem. 2004, 8, 650–654.
- Huang, K.; Feng, M.; Goodenough, J.B. Synthesis and Electrical Properties of Dense Ce0.9Gd0.1O1.95 Ceramics Keqin. J. Am. Ceram. Soc. 1998, 62, 357–362.
- Choolaei, M.; Jakubczyk, E.; Amini, B. Synthesis and Characterisation of a Ceria-Based Cobalt-Zinc Anode Nanocomposite for Low-Temperature Solid Oxide Fuel Cells (LT-SOFCs). Electrochim. Acta 2023, 445, 142057.
- Bellardita, M.; Di Paola, A.; Yurdakal, S.; Palmisano, L. Preparation of Catalysts and Photocatalysts Used for Similar Processes. In Heterogeneous Photocatalysis; Elsevier B.V.: Amsterdam, The Netherlands, 2019; pp. 25–56. ISBN 9780444640154.
- Lesani, P.; Babaei, A.; Ataie, A.; Mostafavi, E. Nanostructured MnCo2O4 Synthesized via Co-Precipitation Method for SOFC Interconnect Application. Int. J. Hydrogen Energy 2016, 41, 20640–20649.
- Teng, Y.; Zuhairi, A.; Amini, B.; Salamatinia, B. Synthesis and Characterisation of Y2O3 Using Ammonia Oxalate as a Precipitant in Distillate Pack Co-Precipitation Process. Ceram. Int. 2018, 44, 18693–18702.
- Liu, S.; Ma, C.; Ma, M.; Xu, F. Magnetic Nanocomposite Adsorbents. In Composite Nanoadsorbents; Elsevier Inc.: Shihezi, China, 2019; pp. 295–316. ISBN 9780128141328.
- Agli, G.D.; Spiridigliozzi, L.; Pansini, M.; Accardo, G.; Yoon, S.P.; Frattini, D. Effect of the Carbonate Environment on Morphology and Sintering Behaviour of Variously Co-Doped (Ca, Sr, Er, Pr) Samarium-Doped Ceria in Co- Precipitation/Hydrothermal Synthesis. Ceram. Int. 2018, 44, 17935–17944.
- Hui, Y.; Liu, B.; Li, J.; Jun, Y. Improved Energy Storage Performance of Ba0.4Sr0.6TiO3 Nanocrystalline Ceramics Prepared by Using Oxalate Co-Precipitation and Spark Plasma Sintering. Mater. Res. Bull. 2019, 113, 141–145.
- Teng, Y.; Zuhairi, A.; Amini, B.; Salamatinia, B. Optimised Co-Precipitation Synthesis Condition for Oxalate-Derived Zirconia Nanoparticles. Ceram. Int. 2019, 45, 22930–22939.
- Dong, H.; Koenig, G.M., Jr. A Review on Synthesis and Engineering of Crystal Precursors Produced Via Coprecipitation for Multicomponent Lithium-Ion Battery Cathode Materials. CrystEngComm 2020, 22, 1514–1530.
- Pelosato, R.; Cristiani, C.; Dotelli, G.; Mariani, M.; Donazzi, A.; Natali, I.; Leonardo, P. Co-Precipitation Synthesis of SOFC Electrode Materials. Int. J. Hydrogen Energy 2012, 38, 480–491.
- Choolaei, M.; Bull, T.; Reina, T.R.; Horri, B.A. Synthesis and Characterisation of Nanocrystalline CuO–Fe2O3/GDC Anode Powders for Solid Oxide Fuel Cells. Ceram. Int. 2020, 46, 14776–14786.
- Dokiya, M.; Horita, T.; Kawada, T.; Sakai, N.; Yokokawa, H. Low Temperature Fabrication of (Y,Gd,Sm)-Doped Ceria Electrolyte. Solid State Ionics 1996, 86–88, 1255–1258.
- Zha, S.; Xia, C.; Meng, G. Effect of Gd (Sm) Doping on Properties of Ceria Electrolyte for Solid Oxide Fuel Cells. J. Power Sources 2003, 115, 44–48.
- Jongen, N.; Bowen, P.; Lemaitre, J.; Valmalette, J.-C.; Hofmann, H. Precipitation of Self-Organized Copper Oxalate Polycrystalline Particles in the Presence of Hydroxypropylmethylcellulose (HPMC): Control of Morphology. J. Colloid Interface Sci. 2000, 226, 189–198.
- Huang, J.; Mao, Z.; Liu, Z.; Wang, C. Development of Novel Low-Temperature SOFCs with Co-Ionic Conducting SDC-Carbonate Composite Electrolytes. Electrochem. Commun. 2007, 9, 2601–2605.
- Huang, J.; Mao, Z.; Liu, Z.; Wang, C. Performance of Fuel Cells with Proton-Conducting Ceria-Based Composite Electrolyte and Nickel-Based Electrodes. J. Power Sources 2008, 175, 238–243.
- Rafique, M.; Safdar, N.; Irshad, M.; Usman, M.; Akhtar, M.; Saleem, M.; Abbas, M.M.; Ashour, A.; Soudagar, M.E. Influence of Low Sintering Temperature on BaCe0.2Zr0.6Y0.2O3−δ IT-SOFC Perovskite Electrolyte Synthesized by Co-Precipitation Method. Materials 2022, 15, 3585.
- Fergus, J.; Hui, R.; Xianguo, L.; Wilkinson, D.; Zhang, J. Solid Oxide Fuel Cells: Materials Properties and Performance, 1st ed.; CRC Press: Boca Raton, FL, USA, 2008; ISBN 978-1-4200-8883-0.
- Filonova, E.; Medvedev, D. Recent Progress in the Design, Characterisation and Application of LaAlO3- and LaGaO3-Based Solid Oxide Fuel Cell Electrolytes. Nanomaterials 2022, 12, 1991.
- Konwar, D.; Nguyen, N.T.Q.; Yoon, H.H. Evaluation of BaZr0.1Ce0.7Y0.2O3-d Electrolyte Prepared by Carbonate Precipitation for a Mixed Ion-Conducting SOFC. Int. J. Hydrogen Energy 2015, 40, 11651–11658.
- Tok, A.I.Y.; Luo, L.H.; Boey, F.Y.C. Consolidation and Properties of Gd0.1Ce0.9O1.95 Nanoparticles for Solid-Oxide Fuel Cell Electrolytes. J. Mater. Res. 2006, 21, 119–124.
- Ding, D.; Liu, B.; Zhu, Z.; Zhou, S.; Xia, C. High Reactive Ce0.8Sm0.2O1.9 Powders via a Carbonate Co-Precipitation Method as Electrolytes for Low-Temperature Solid Oxide Fuel Cells. Solid State Ionics 2008, 179, 896–899.
- Spiridigliozzi, L.; Dell’Agli, G.; Marocco, A.; Accardo, G.; Pansini, M.; Yoon, S.P.; Ham, H.C.; Frattini, D. Engineered Co-Precipitation Chemistry with Ammonium Carbonate for Scalable Synthesis and Sintering of Improved Sm0.2Ce0.8O1.90 and Gd0.16Pr0.04Ce0.8O1.90 Electrolytes for IT-SOFCs. J. Ind. Eng. Chem. 2018, 59, 17–27.
- Roy, N.K.; Foong, C.S.; Cullinan, M.A. Effect of Size, Morphology, and Synthesis Method on the Thermal and Sintering Properties of Copper Nanoparticles for Use in Microscale Additive Manufacturing Processes. Addit. Manuf. 2018, 21, 17–29.
- Chen, Y.-M.; Lin, T.-N.; Liao, M.-W.; Kuo, H.-Y.; Yeh, C.-Y.; Kao, W.-X.; Yang, S.-F.; Wu, K.-T.; Ishihara, T. Applications of the Glycine Nitrate Combustion Method for Powder Synthesis on the LSGM-Based Electrolyte-Supported Solid Oxide Fuel Cells. ECS Trans. 2017, 78, 773–781.
- Jais, A.A.; Ali, S.A.M.; Anwar, M.; Somalu, M.R.; Muchtar, A.; Isahak, W.N.R.W.; Baharudin, N.A.; Lim, K.L.; Brandon, N.P. Performance of Ni/10Sc1CeSZ Anode Synthesized by Glycine Nitrate Process Assisted by Microwave Heating in a Solid Oxide Fuel Cell Fueled with Hydrogen or Methane. J. Solid State Electrochem. 2020, 24, 711–722.
- Minakshi, M.; Barmi, M.; Mitchell, D.R.G.; Barlow, A.J.; Fichtner, M. Effect of Oxidizer in the Synthesis of NiO Anchored Nanostructure Nickel Molybdate for Sodium-Ion Battery. Mater. Today Energy J. 2018, 10, 1–14.
- Jamale, A.P.; Natoli, A.; Jadhav, L.D. Synthesis of Active Electrocatalysts Using Glycine–Nitrate Chemistry. J. Phys. Chem. Solids 2021, 148, 109723.
- Wain-martin, A.; Morán-ruiz, A.; Vidal, K.; Larrañaga, A.; Laguna-bercero, M.A. Scalable Synthetic Method for SOFC Compounds. Solid State Ionics 2017, 313, 52–57.
- Komova, O.V.; Mukha, S.A.; Ozerova, A.M.; Odegova, G.V.; Simagina, V.I.; Bulavchenko, O.A.; Ishchenko, A.V.; Netskina, O. V The Formation of Perovskite during the Combustion of an Energy-Rich Glycine–Nitrate Precursor. Materials 2020, 13, 5091.
- Patil, K.C.; Aruna, S.T.; Mimani, T. Combustion Synthesis: An Update. Curr. Opin. Solid State Mater. Sci. 2002, 6, 507–512.
- Kulkarni, S.; Duttagupta, S.; Phatak, G. Study of Glycine Nitrate Precursor Method for the Synthesis of Gadolinium Doped Ceria (Ce0.8Gd0.2O1.90) as an Electrolyte for Intermediate Temperature Solid Oxide Fuel Cells. RSC Adv. 2014, 4, 46602–46612.
- Chen, W.; Li, F.; Yu, J.; Liu, L. A Facile and Novel Route to High Surface Area Ceria-Based Nanopowders by Salt-Assisted Solution Combustion Synthesis. Mater. Sci. Eng. B 2006, 133, 151–156.
- Zhang, X.; Jiang, W.; Song, D.; Sun, H.; Sun, Z.; Li, F. Salt-Assisted Combustion Synthesis of Highly Dispersed Superparamagnetic CoFe2O4 Nanoparticles. J. Alloys Compd. 2009, 475, 34–37.
- Park, S.-Y.; Na, C.W.; Ahn, J.H.; Song, R.-H.; Lee, J.-H. Preparation of Highly Porous NiO–Gadolinium- Doped Ceria Nano-Composite Powders by One- Pot Glycine Nitrate Process for Anode-Supported Tubular Solid Oxide Fuel Cells. J. Asian Ceram. Soc. 2018, 2, 339–346.
- Dong, H.; Wang, M.; Liu, Y.; Han, Z. Optimized Solid-State Synthesis of Sr2Fe1.5Mo0.5O6−δ Perovskite: Implications for Efficient Synthesis of Mo-Containing SOFC Electrodes. Crystals 2022, 12, 1533.
- Fan, L. Solid-State Electrolytes for SOFC. In Solid Oxide Fuel Cells: From Electrolyte-Based to Electrolyte-Free Devices; Zhu, B., Raza, R., Fan, L., Sun, C., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2020; pp. 35–78. ISBN 9783527812790.
- Yang, S.H.; Kim, K.H.; Yoon, H.H.; Kim, W.J.; Choi, H.W. Comparison of Combustion and Solid-State Reaction Methods for the Fabrication of SOFC LSM Cathodes. Mol. Cryst. Liq. Cryst. 2011, 539, 50–390.
- Kane, N.; Zhou, Y.; Zhang, W.; Ding, Y.; Luo, Z.; Hua, X.; Liu, M. Precision Surface Modification of Solid Oxide Fuel Cells via Layer-by-Layer Surface Sol-Gel Deposition. J. Mater. Chem. A 2022, 10, 8798–8806.
- Liu, Y.; Qin, H.; Li, M.; Cheng, J.; Tang, C.; Xiao, J.; Xie, Y. Direct Synthesis of Ce0.8Sm0.2−xZnxO2−δ Electrolyte by Sol–Gel for IT-SOFC. Ionics 2022, 28, 4675–4684.
- Tripathi, R.; Dur, L.; Ricardo, P.; Tudela, D.; Christoph, H.; Marx, D. Unveiling Zwitterionization of Glycine in the Microhydration Limit. ACS Omega 2021, 6, 12676–12683.
- Aruna, S.T.; Mukasyan, A.S. Combustion Synthesis and Nanomaterials. Curr. Opin. Solid State Mater. Sci. 2008, 12, 44–50.
- Birol, H.; Rambo, C.R.; Guiotokuc, M.; Hotza, D. Preparation of Ceramic Nanoparticles via Cellulose- Assisted Glycine Nitrate Process: A Review. RSC Adv. 2013, 3, 2873–2884.
- Yu, C.; Sakthinathan, S.; Lai, G.; Lin, C.; Chiu, T.-W.; Liu, M.-C. ZnO–ZnCr2O4 Composite Prepared by a Glycine Nitrate Process Method and Applied for Hydrogen Production by Steam Reforming of Methanol. RSC Adv. 2022, 12, 22097–22107.
- Cheng, H.-H.; Chen, S.-S.; Liu, H.-M.; Jang, L.-W.; Chang, S.-Y. Glycine–Nitrate Combustion Synthesis of Cu-Based Nanoparticles for NP9EO Degradation Applications. Catalysts 2020, 10, 1061.
- Varma, A.; Mukasyan, A.S.; Rogachev, A.S.; Manukyan, K. V Solution Combustion Synthesis of Nanoscale Materials. Chem. Rev. 2016, 116, 14493–14586.
- Stojanovic, B.D.; Dzunuzovic, A.S.; Ilic, N.I. Review of Methods for the Preparation of Magnetic Metal Oxides. In Magnetic, Ferroelectric, and Multiferroic Metal Oxides; Elsevier Inc.: Belgrade, Serbia, 2018; pp. 333–359. ISBN 9780128111802.
- Pine, T.; Lu, X.; Mumm, D.R.; Samuelsen, G.S.; Brouwer, J. Emission of Pollutants from Glycine–Nitrate Combustion Synthesis Processes. J. Am. Ceram. Soc. 2007, 90, 3735–3740.
- Park, S.; Woong, C.; Hyun, J.; Yun, U.; Lim, T.; Song, R.; Shin, D.; Lee, J. Intermediate-Temperature Nickeleyttria Stabilized Zirconia Supported Tubular Solid Oxide Fuel Cells Using Gadolinia-Doped Ceria Electrolyte. J. Power Sources 2012, 218, 119–127.
