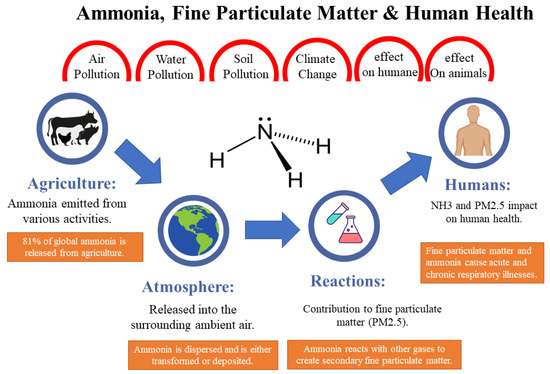
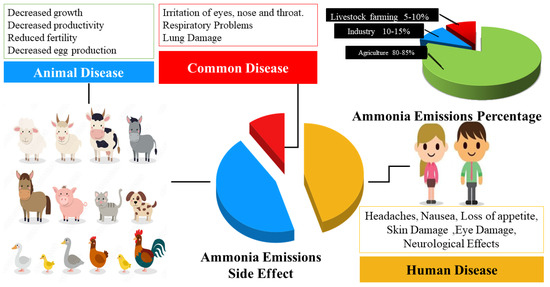
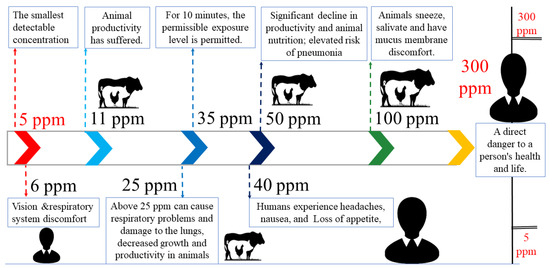
Ammonia (NH3) represents a perilous gas that poses a substantial hazard to both human well-being and the environment, particularly within agricultural regions. Agricultural activities constitute a primary source of ammonia emissions. Thus, effective monitoring and measurement of ammonia sources in agriculture are imperative for mitigating its adverse impact. However, not all existing ammonia detection methods are suitable for discerning the low concentrations typically encountered in agricultural ammonia volatilizing (ranging from 0.01 to 5 parts per million). Consequently, curtailing ammonia volatilization from farmland assumes paramount importance, with real-time monitoring serving as a crucial mechanism for assessing environmental contamination and minimizing agricultural ammonia losses. Deploying appropriate detection methodologies ensures that requisite measures are taken to safeguard human health and the environment from the deleterious repercussions of ammonia exposure.
- ammonia
- detection methods
- ammonia gas emissions
- E-nose
- concentrations
1. Introduction
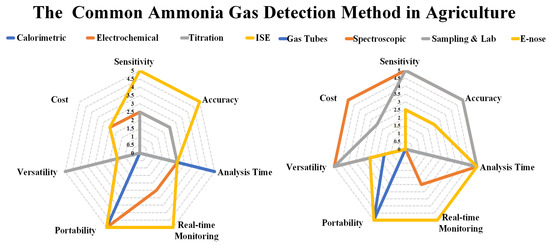
2. Performance Evaluation of Traditional Methods vs. E-Nose
The traditional methods have been used in agriculture for many years and are still widely used due to their simplicity, affordability, and ease of use. However, they also have limitations, such as the need for manual sampling, the potential for measurement errors, and time-consuming laboratory analysis. These methods can provide accurate and reliable results for ammonia detection, but their performance may vary depending on factors such as sensitivity, specificity, real-time monitoring capability, ease of use, and maintenance requirements. This has led to the emergence of alternative methods, such as E-nose, which offer potential advantages in terms of real-time, non-invasive, and automated ammonia detection in agricultural settings. When selecting a method for ammonia detection, it is crucial to consider both the specific application and the desired level of accuracy. The cost, speed, and accuracy of ammonia detection methods in agriculture can vary greatly depending on the method used and should be taken into account. According to [27], the most used methods in agriculture, one of the most commonly used methods for ammonia detection is the use of Electrochemical sensors. Electrochemical sensors are widely employed for their high sensitivity, good selectivity, and relatively fast response time. These sensors are capable of accurately measuring ammonia gas concentrations in the agricultural environment, making them suitable for applications such as monitoring livestock facilities, assessing fertilizer efficiency, and managing air quality in crop production. Electrochemical sensors offer several advantages for ammonia detection in agriculture. They are cost-effective, easy to use, and provide real-time measurements. Additionally, they can detect low concentrations of ammonia gas accurately, enabling farmers to monitor and adjust ammonia levels to optimize crop growth and minimize environmental impact. It is important to note that, while electrochemical sensors are commonly used, the choice of ammonia detection method can vary depending on specific agricultural practices, regulatory requirements, and the desired level of accuracy and monitoring capabilities. A deeper comparison shows that the cost of these methods can vary, with Calorimetric Methods and Spectroscopic Methods generally being more expensive, while Electrochemical and Gas Sensitive Tube methods tend to be more affordable. In terms of speed, Electrochemical sensors and Gas Sensitive Tubes offer fast response times, while Sampling and Laboratory Analysis and Spectroscopic Methods can be slower due to the need for sample collection and analysis. Regarding accuracy, Calorimetric Methods [28], Electrochemical sensors [29], Ion Selective Electrode (ISE) [30], and Spectroscopic Methods [31] are known for providing high accuracy in ammonia detection. The measurement range can vary depending on the specific method and equipment used. Generally, Electrochemical sensors have a moderate to wide measurement range, while Calorimetric Methods and Ion Selective Electrode (ISE) have a narrower to moderate range. E-nose can also be used for spatial mapping of ammonia levels across large areas, providing valuable information for site-specific management strategies. Additionally, E-nose has the potential to detect other odor compounds or gases that may be indicative of specific agricultural practices or environmental conditions [23]. However, E-nose also has limitations. It may require calibration and validation against reference methods to ensure accuracy and reliability. The cost of initial investment and maintenance may be higher compared to some traditional methods. The complexity of data analysis and interpretation may also pose challenges, as E-nose generates large amounts of data that require advanced analytical techniques [26]. The comparative analysis of advantages and disadvantages pertaining to diverse domains concerning Electronic nose (E-nose) and conventional methodologies are elucidated in Table 1 presented hereunder.| Advantages | E-Nose | Traditional Methods |
|---|---|---|
| Sensitivity | High detection limits in the low ppm or even ppb range | Highly selective for ammonia |
| Precision and Accuracy | Precise and accurate measurements | Familiarity and wide adoption |
| Real-time Monitoring and Rapid Results | Provides real time monitoring and quick results | Easier to implement and validate |
| Portability and Ease of Use | Very portable and easy to use | High accuracy and precision in ammonia measurement |
| Potential Selectivity towards Specific Analytes | Including ammonia | Potentially reducing interference from other compounds in the sample |
| Disadvantages | E-Nose | Traditional Methods |
| Sensor Limitations | It has sensor limitations | Require complex sample preparation steps |
| Cost | E-nose devices can have varying costs depending on the complexity of the sensor array | Longer analysis times compared to E-nose |
| Standardization | Lack standardized methods or regulatory guidelines for ammonia measurement | Require specialized equipment, reagents, and skilled personnel for operation and maintenance |
| Application Range | Limited | Less portable compared to E-nose devices |
References
- Michaels, R.A. Emergency planning and the acute toxic potency of inhaled ammonia. Environ. Health Perspect. 1999, 107, 617–627.
- Jiang, B.; Xia, D. Ammonia control represents the key for PM2.5 elimination: Insights for global air pollution control interconnected from PM2.5 events in China. Clean Technol. Environ. Policy 2021, 23, 829–841.
- Simion, C.E.; Florea, O.G.; Mercioniu, I.; Dinu, I.V.; Stanoiu, A. Gas sensing mechanism involved in NH3 detection with NiO material. In Proceedings of the 2022 International Semiconductor Conference (CAS), Brasov, Romania, 12–14 October 2022; pp. 109–112.
- Pigni, A.; Tugnolo, A.; Beghi, R.; Cocetta, G.; Finzi, A. Rapid and continuous monitoring of air ammonia concentration in dairy milking parlors. In Proceedings of the 2021 IEEE International Workshop on Metrology for Agriculture and Forestry (MetroAgriFor), Bolzano, Italy, 3–5 November 2021; pp. 167–171.
- Bulbul, A.; Kim, H. ppb level gas quantification by bubble chromatography. In Proceedings of the 2017 19th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS), Kaohsiung, Taiwan, 18–22 June 2017; pp. 660–663.
- Pauluhn, J. Acute inhalation toxicity of ammonia: Revisiting the importance of RD50 and LCT01/50 relationships for setting emergency response guideline values. Regul. Toxicol. Pharmacol. 2013, 66, 315–325.
- Andre, R.S.; Facure, M.H.; Mercante, L.A.; Correa, D.S. Electronic nose based on hybrid free-standing nanofibrous mats for meat spoilage monitoring. Sens. Actuators B Chem. 2022, 353, 131114.
- Guido, V.; Finzi, A.; Piazzi, P.; Ferrari, O.; Ricco, C.R.; Riva, E.; Provolo, G. Effect of mitigation techniques on ammonia emissions and nutrients recovery: The role of fertigation with digestate. In Proceedings of the 2020 IEEE International Workshop on Metrology for Agriculture and Forestry (MetroAgriFor), Trento, Italy, 4–6 November 2020; pp. 39–43.
- Liu, T.; Chen, Z.; Fu, Q.; Shi, B.; Yang, L. Acute toxicity test of landfill leachates using protozoan communities. In Proceedings of the 2010 4th International Conference on Bioinformatics and Biomedical Engineering, Chengdu, China, 18–20 June 2010; pp. 1–4.
- Zhang, B.; Wu, B.; Liu, J. PM2.5 pollution-related health effects and willingness to pay for improved air quality: Evidence from China’s prefecture-level cities. J. Clean. Prod. 2020, 273, 122876.
- Zheng, M.; Wang, Y.; Yuan, L.; Chen, N.; Kong, S. Ambient observations indicating an increasing effectiveness of ammonia control in wintertime PM2.5 reduction in Central China. Sci. Total Environ. 2022, 824, 153708.
- Yuanxi, W.; Zhang, W.; Jun, W. Research on Preparation and Performance of a New Solid Electrolyte Based Nitrogen Oxides Sensor. IEEE Sens. J. 2022, 22, 13908–13914.
- Singh, A.K.; Pandey, A.; Chakrabarti, P. Poly thiophene] organic polymer based-interdigitated channel enabled thin film transistor for detection of selective low ppm Ammonia sensing at 25 C. IEEE Sens. J. 2019, 20, 4047–4055.
- Insausti, M.; Timmis, R.; Kinnersley, R.; Rufino, M.C. Advances in sensing ammonia from agricultural sources. Sci. Total Environ. 2020, 706, 135124.
- Swotinsky, R.B.; Chase, K.H. Health effects of exposure to ammonia: Scant information. Am. J. Ind. Med. 1990, 17, 515–521.
- Conti, C.; Borgonovo, F.; Guarino, M. Ammonia concentration and recommended threshold values in pig farming: A review. In Proceedings of the 2021 IEEE International Workshop on Metrology for Agriculture and Forestry (MetroAgriFor), Bolzano, Italy, 3–5 November 2021; pp. 162–166.
- Liu, Q.X.; Zhou, Y.; Li, X.M.; Ma, D.D.; Xing, S.; Feng, J.H.; Zhang, M.H. Ammonia induce lung tissue injury in broilers by activating NLRP3 inflammasome via Escherichia/Shigella. Poult. Sci. 2020, 99, 3402–3410.
- Bouwman, A.F.; Lee, D.S.; Asman, W.A.; Dentener, F.J.; Van Der Hoek, K.W.; Olivier, J.G.J. A global high-resolution emission inventory for ammonia. Glob. Biogeochem. Cycles 1997, 11, 561–587.
- Zhou, Y.; Liu, Q.X.; Li, X.M.; Ma, D.D.; Xing, S.; Feng, J.H.; Zhang, M.H. Effects of ammonia exposure on growth performance and cytokines in the serum, trachea, and ileum of broilers. Poult. Sci. 2020, 99, 2485–2493.
- Kristensen, H.H.; Wathes, C.M. Ammonia and poultry welfare: A review. World’s Poult. Sci. J. 2000, 56, 235–245.
- Lin, T.H.; Li, Y.T.; Hao, H.C.; Fang, I.C.; Yang, C.M.; Yao, D.J. Surface acoustic wave gas sensor for monitoring low concentration ammonia. In Proceedings of the 2011 16th International Solid-State Sensors, Actuators and Microsystems Conference, Beijing, China, 5–9 June 2011; pp. 1140–1143.
- Ansari, M.; Moravvej-Farshi, M.K. Ammonia Vapor Sensor based on Tapered Multimode Fiber Coated with Silica-Gel. In Proceedings of the 2020 28th Iranian Conference on Electrical Engineering (ICEE), Tabriz, Iran, 4–6 August 2020; pp. 1–4.
- Moshayedi, A.J.; Gharpure, D.C. Evaluation of bio inspired Mokhtar: Odor localization system. In Proceedings of the 2017 18th International Carpathian Control Conference (ICCC), Sinaia, Romania, 28–31 May 2017; pp. 527–532.
- Geng, K.; Ata, J.M.; Chen, J.; Hu, J.; Zhang, H. ENOSE Performance in Transient Time and Steady State Area of Gas Sensor Response for Ammonia Gas: Comparison and Study. In Proceedings of the 2023 2nd Asia Conference on Algorithms, Computing and Machine Learning, Shanghai, China, 17–19 March 2023; pp. 247–252.
- Chen, D.; Miao, Z.; Peng, M.; Xing, H.; Zhang, H.; Teng, X. The co-expression of circRNA and mRNA in the thymuses of chickens exposed to ammonia. Ecotoxicol. Environ. Saf. 2019, 176, 146–152.
- Moshayedi, A.J.; Khan, A.S.; Yang, S.; Geng, K.; Hu, J. ENose design and structures from statistical analysis to application in robotic: A compressive review. EAI Endorsed Trans. AI Robot. 2023, 1, e13.
- Hobbs, P.J.; Misselbrook, T.H.; Pain, B.F. Assessment of odours from livestock wastes by a photoionization detector, an Electronic nose, olfactometry and gas chromatography-mass spectrometry. J. Agric. Eng. Res. 1995, 60, 137–144.
- Ahn, C.K.; Han, K.; Lee, M.S.; Kim, J.Y.; Chun, H.D.; Kim, Y.; Park, J.M. Experimental studies of additives for suppression of ammonia vaporization in the ammonia based CO2 capture process. Energy Procedia 2013, 37, 7108–7116.
- Fang, C.S.; Oh, K.H.; Park, J.K.; Yang, H. Rapid and sensitive electrochemical detection of carbaryl based on enzyme inhibition and thiocholine oxidation mediated by a ruthenium (III) complex. Electroanalysis 2017, 29, 339–344.
- Kwak, D.; Lei, Y.; Maric, R. Ammonia gas sensors: A comprehensive review. Talanta 2019, 204, 713–730.
- Wang, G.; Lahib, A.; Duncianu, M.; Gou, Q.; Stevens, P.S.; Dusanter, S.; Tomas, A.; Sigrist, M.W.; Chen, W. Monitoring of peroxy radicals by chemical amplification enhanced photoacoustic spectroscopy. In Proceedings of the European Conference on Lasers and Electro-Optics, Munich, Germany, 21–25 June 2021.
- Pan, Y.; Gu, M.; Song, L.; Tian, S.; Wu, D.; Walters, W.W.; Yu, X.; Lü, X.; Ni, X.; Wang, Y.; et al. Systematic low bias of passive samplers in characterizing nitrogen isotopic composition of atmospheric ammonia. Atmos. Res. 2020, 243, 105018.
- Kashour, H.; Soubh, L. Comparative between Ammonia Ion selective electrode and dye binding method to study effect of processing methods on protein content of plain Yogurt. Res. J. Pharm. Technol. 2021, 14, 6257–6261.
