Lung transplantation is often the only viable treatment option for a patient with end-stage lung disease. Lung transplant results have improved substantially over time, but ischemia-reperfusion injury, primary graft dysfunction, acute rejection, and chronic lung allograft dysfunction (CLAD) continue to be significant problems. Mesenchymal stromal cells (MSC) are pluripotent cells that have anti-inflammatory and protective paracrine effects and may be beneficial in solid organ transplantation.
- mesenchymal stromal cell
- lung transplantation
- cell therapy
- gene therapy
- ex vivo lung perfusion
1. Introduction
2. Mesenchymal Stromal Cells
Mesenchymal stromal cells (MSC) and multipotent adult progenitor cells (MAPC) are mesenchymal, non-hematopoietic progenitor cells that are found in most tissues and are capable of self-renewal and differentiation into various types of stromal cells (Figure 1). Resident MSCs are also present in the lung, localized in perivascular spaces, where they act as important regulators of pulmonary homeostasis and the balance between lung injury and repair [15][18]. For therapeutic purposes, MSCs are commonly isolated from bone marrow, adipose tissue, the umbilical cord, and the placenta [6][7][6,7]. As considerable heterogeneity in cell sources, and isolation and expansion methods exist, the International Society for Cell Therapy has recommended minimal standard criteria to identify and characterize MSCs. Accordingly, MSCs should adhere to plastic in standard culture conditions, express typical stromal cell surface markers, lack expression of hematopoietic markers, and differentiate into osteoblasts, adipocytes, and chondroblasts in vitro under appropriate conditions [16][19]. While MAPCs resemble MCSs, they are biologically more primitive and have greater differentiation potential than classical MSCs [17][20].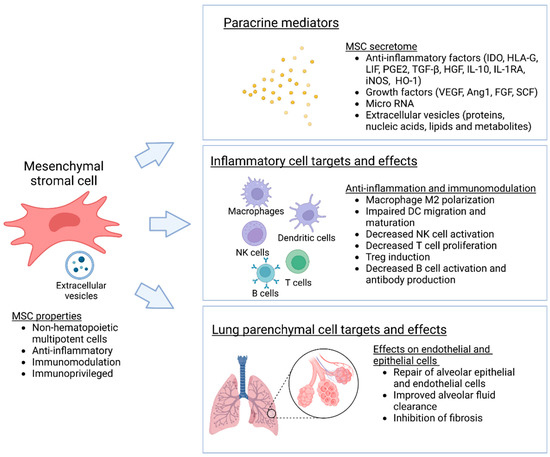
3. Lung Transplant MSC Therapy
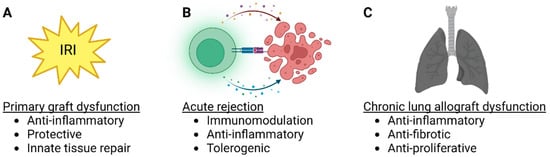
The protective anti-inflammatory and immunomodulatory properties make MSCs attractive therapeutic candidates for lung transplantation. MSCs have been delivered to the lungs through the airways or intravascularly and can be administered into the donor lung either before transplantation or to the recipient before or after transplantation (Figure 2). Most of the preclinical studies have used MSCs to inhibit IRI, while some experiments have targeted acute rejection, and some clinical trials have explored MSC therapy in patients with CLAD (Table 1). Importantly, there is significant interaction between IRI, acute rejection, and CLAD (Figure 3), as lung injury may trigger alloimmune responses, and various non-immunological and immunological factors participate in the development of CLAD [5][21][5,42].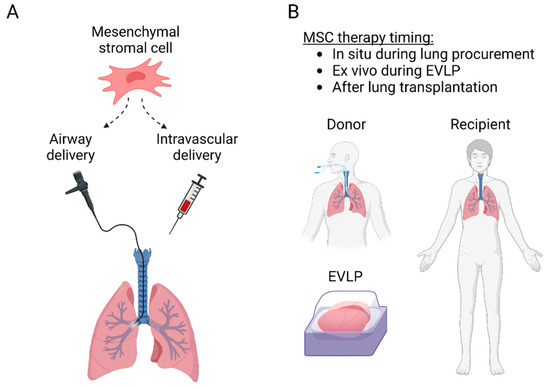
|
Target |
Species |
Model |
MSC |
Administration |
Effect |
Ref |
|---|---|---|---|---|---|---|
|
IRI |
Rat |
Lung hilar clamping |
Engineered BM-MSC (MSC-vIL-10) |
Intravenous |
Improved oxygenation, inflammation and permeability |
|
|
IRI |
Pig |
SLTx |
BM-MSC |
Pulmonary artery vs. endobronchial |
Endobronchial MSC delivery improved lung compliance |
|
|
IRI |
Human |
EVLP |
MAPC |
Airways |
Decreased edema and inflammation |
|
|
IRI |
Human |
EVLP |
BM-MSC |
Intravascular |
Restored alveolar fluid clearance |
|
|
IRI |
Mouse |
SLTx |
BM-MSC |
Recipient intravenous |
Decreased IRI, MSC homing preferentially into the lung transplant |
|
|
IRI |
Pig |
EVLP |
UC-MSC |
Airway vs. intravascular, 3 different doses |
Intravascular delivery improved MSC lung retention, optimal dose 150 × 106 MSC decreased IL-8 and increased VEGF |
|
|
IRI |
Mouse |
SLTx |
BM-MSC |
Ex vivo pulmonary artery |
Decreased IRI |
|
|
IRI |
Pig |
SLTx |
BM-MSC |
Pulmonary artery vs. endobronchial |
No short-term differences detected |
|
|
IRI |
Mouse |
Lung hilar clamping and EVLP |
Human UC-MSC vs. MSC-EVs |
Intravascular |
MSCs and MSC-EVs attenuate IRI |
|
|
IRI |
Human |
EVLP |
MAPC |
Airways |
Decreased BAL neutrophilia, TNF-α, IL-1β and IFN-γ |
|
|
IRI |
Pig |
SLTx |
BM-MSC |
Intravenous or intrabronchial |
Heterogenous localization, in alveoli after endobronchial and in blood vessels after intravascular administration |
|
|
IRI |
Rat |
SLTx |
BM-MSC |
Intravenous |
Protection against IRI |
|
|
IRI |
Pig |
EVLP and SLTx |
UC-MSC |
Intravascular |
Decreased IRI during EVLP and after TX |
|
|
IRI |
Rat |
EVLP |
UC-MSC |
Intravascular |
Improved inflammation and IRI |
|
|
IRI |
Rat |
EVLP |
BM-MSC-EVs |
Intravascular |
Multiple influences on pulmonary energetics, tissue integrity and gene expression |
|
|
IRI |
Human |
EVLP |
Engineered UC-MSC (MSCIL−10) |
Intravascular |
Safe and feasible, results in rapid IL-10 elevation |
|
|
IRI |
Rat |
SLTx |
Donor vs. recipient adipose tissue MSC |
Intravenous |
MSCs, regardless of their origin, exert similar immunosuppressive effects |
|
|
IRI/ ARDS |
Human |
EVLP/endotoxin |
BM-MSC |
Airways |
Restored alveolar fluid clearance |
|
|
IRI/ ARDS |
Human |
EVLP/e.coli pneumonia |
BM-MSC |
Airways |
Restored alveolar fluid clearance, reduced inflammation and increased antimicrobial activity |
|
|
Acute rejection |
Rat |
SLTx |
BM-MSC |
1 vs. 2 recipient intravenous doses |
Protection from acute rejection, best result with 2 recipient doses |
|
|
Acute rejection/ CLAD |
Mouse |
Ortotopic tracheal Tx |
iPSC-MSC |
Intravascular |
Induces immune tolerance and supports long-term graft survival |
|
|
CLAD |
Mouse |
Heterotopic tracheal Tx |
MSC (various sources) |
Intravenous |
Prevents airway occlusion |
|
|
CLAD |
Mouse |
Ortotopic tracheal Tx |
BM-MSC |
Intravenous |
Prevents airway occlusion through macrophage cytokines |
|
|
CLAD |
Mouse |
Heterotopic tracheal Tx |
BM-MSC |
Local vs. systemic vs. combination |
Prevents airway occlusion through modulation of immune response, best effect with combination treatment |
|
|
CLAD |
Human |
Clinical Tx |
BM-MSC |
Intravenous twice weekly for 2 weeks |
Safe and feasible in patients with advanced CLAD |
[13] |
|
CLAD |
Human |
Clinical Tx |
BM-MSC |
Intravenous |
Safe and feasible in patients with moderate CLAD |
[12] |
|
CLAD |
Human |
Clinical Tx |
BM-MSC |
Intravenous |
Well tolerated in moderate-to-severe CLAD, low-dose may slow progression of CLAD in some patients |
[11] |
