Trypanosomiases are a group of tropical diseases that have devastating health and socio-economic effects worldwide. In humans, these diseases are caused by the pathogenic kinetoplastids Trypanosoma brucei, causing African trypanosomiasis or sleeping sickness, and Trypanosoma cruzi, causing American trypanosomiasis or Chagas disease. Antimicrobial peptides (AMPs) are small peptides synthesized by both prokaryotes and (unicellular and multicellular) eukaryotes, where they fulfill functions related to competition strategy with other organisms and immune defense. These AMPs can bind and induce perturbation in cell membranes, leading to permeation of molecules, alteration of morphology, disruption of cellular homeostasis, and activation of cell death. These peptides have activity against various pathogenic microorganisms, including parasitic protists.
- trypanosomiases
- human sleeping sickness
- Chagas disease
- antimicrobial peptides
- anti-Trypanosoma activity
1. Introduction
2. Antimicrobial Peptides (AMPs)
AMPs are a class of small peptides synthesized by pro- and eukaryotic organisms, used as a strategy for competition and defense during invasion by foreign organisms. They are encoded by specific genes and expressed constitutively or in response to specific environmental stimuli [12][19]. In some insects, AMPs are key for vector–microorganism interaction and are effective against both quiescent and actively proliferating pathogenic organisms [13][14][15][20,21,22]. These peptides are synthesized through three pathways, which include classical ribosomal synthesis, non-ribosomal synthesis, and proteolytic digestion of proteins. Ribosomally synthesized AMPs (RS-AMPs) are those encoded by genes and produced by ribosomal translation of specific mRNAs into the biologically active amino acids sequences. These AMPs are widely distributed in nature, produced by various organisms (such vertebrates, insects, plants, and bacteria) [16][17][23,24]. Among the RS-AMPs are the mammalian defensins and amphibian dermaceptins [16][23]. Non-ribosomally synthesized AMPs (NR-AMPs) are produced by enzymes known as non-ribosomal peptide synthases (NRPSs), which incorporate non-proteinogenic amino acids into the sequence and are found mainly in filamentous fungi and bacteria [17][18][24,25]. So far, hundreds of AMPs synthesized in a NRPS-dependent manner have been described, among which are gramicidin S and isopenicillin [19][26]. Other AMPs are produced via the proteolytic digestion pathway (peptides also known as cryptides) by proteases-mediated cleavage of precursor proteins or larger proteins with other functions, to yield matured bioactive factors [17][20][24,27]. During these processes, various fragmented peptides are also produced that can vary in their biological activity [20][27]. Buforin II is one of the most studied cryptid peptides [21][28]. Although natural AMPs are molecules with considerable diversity in their structural properties, origins, and mechanisms of action, they have certain characteristics in common. Generally, they are short molecules (≈10–100 amino acids) of a cationic nature at neutral pH (generally ranging from +2 to +11), which facilitates their interaction with charged cell membranes through electrostatic interaction [22][23][29,30]. Additionally, most AMPs have a considerable proportion of hydrophobic residues (close to 50%) and an amphipathic structure [23][24][30,31]. This latter property is responsible for their structural flexibility and solubility in aqueous environments. [23][30]. The overall positive net charge and amphipathicity are the two characteristics that contribute to the high affinity of AMPs for membranes [25][32]. Structurally, AMPs are commonly classified into four groups based on their secondary structure, which include linear α-helical peptides, β-sheet peptides (usually stabilized with one or more disulfide bonds), linear extension or loop (devoid of α- or β-elements) structure, and mixed (α-helical/β-sheet) peptides [22][23][25][29,30,32] (Figure 1). However, some peptides with cyclic structures and unusual complete topologies have also been documented [25][32]. Most studied among the groups of AMPs are the peptides with an α-helix structure [26][33].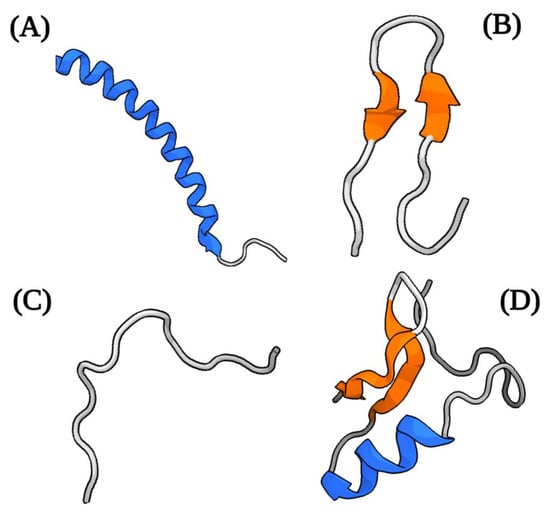
3. Current Treatment of Trypanosomiases
4. AMPs with Antiparasitic Activity
Several studies have shown the antiparasitic effect of some AMPs [43][44][45][35,107,108], including activity against parasites that cause important tropical diseases [46][109] (Figure 1). Many of these AMPs have been isolated from various vertebrate and invertebrate hosts of these parasites [44][47][48][107,110,111]. Plasmodium is the parasite on which most studies with AMPs have been carried out [49][112]. In this protist, many natural AMPs act primarily by disrupting the integrity of cell membranes [50][51][52][53][54][55][117,118,119,120,121,122]. However, some others can interfere with other important cellular processes of the parasite. In Plasmodium berghei, some fungal AMPs have an inhibitory effect on histone deacetylase (HDA), thus inducing histone hypermethylation and subsequent alteration of gene expression in the parasite [56][123]. Other AMPs derived from Gram-positive bacteria, such as epoxomicin and derivatives of the natural cyclic oligopeptide thiostrepton, have an inhibitory effect on protein synthesis and turnover, due to their binding to and inhibition of catalytic activity of proteasome β subunits (20S) [57][58][124,125]. Additionally, thiostrepton can inhibit mRNA translation in the apicoplast through its binding to the plasmodial organellar rRNA promoting structural alterations that prevent its function during protein synthesis [58][59][125,126]. Importantly, antimalarial activities have been attributed to some AMPs with semi-synthetic and synthetic origin. Synthetic AMPs inhibit the plasmodial cysteine protease falcipain and aspartic proteases plasmepsin I and plasmepsin II, involved in hemoglobin hydrolysis and hemozoin formation, thus interfering with parasite metabolism and growth [60][61][62][127,128,129]. Notably, some synthetic peptides have also shown an effect on some enzymes such as topoisomerase I, affecting the parasite’s DNA metabolism [63][130]. Several of these AMPs not only have antiplasmodial activity against different developmental stages of some Plasmodium species (P. falciparum, P. berghei, and P. yoelii nigeriensis) in vitro conditions [54][57][64][121,124,131], but are also effective at high parasitemia in an animal model [55][122]. In helminths, studies have focused primarily on Schistosoma and Brugia. In these parasites, AMPs have effects on motility, development, egg deposition, and the integumentary surface [65][66][67][68][69][136,137,138,139,140]. In Brugia pahangi, synthetic cecropins A/B, AMPs from insect hemolymph, attenuate microfilariae mobility and larval development in adult female Aedes aegypti [65][136]. In Schistosoma, dermaseptin, a peptide isolated from frogs, can synergistically interact with other natural compounds and contribute to parasite killing and infection control. In combination with piplartine, an amide alkaloid of Piper longum L. (long piper), dermaseptin not only exerts activity against the Schistosoma mansoni (S. mansoni) stages (schistosomula and adult) and affects the reproductive fitness of adult worms, but also induces structural alterations of the tegument and extensive destruction of the tubercles [66][67][137,138]. Although the anthelmintic mechanism of AMPs has not been elucidated, it has been proposed that disruption of cell structure by pore formation by direct interaction with the lipid bilayer seems to be the most likely [65][67][70][71][136,138,141,142]. It should be noted that the integument is essential for the survival of the helminth parasites, since it is involved in nutrient absorption and in the interaction with the host [72][73][74][75][143,144,145,146]. In both Brugia and Schistosoma, divalent metal transporter 1 (DMT1) molecules are present in the integument and are essential for the absorption of iron, an essential ion for the development and reproduction of these parasites [73][75][76][144,146,147]. In this sense, directing AMPs against the tegument of these parasites could be a good anthelmintic strategy. Unlike in apicomplexan parasites, the AMPs tested so far on helminths exert their antiparasitic action at micromolar concentrations [65][66][67][136,137,138]. Some AMPs with antimicrobial properties have been discovered in helminths [71][77][78][142,148,149]. In S. mansoni, an AMP called schistocins has been obtained from the protein SmKI-1, a key protein for the survival of this nematode, which has activity against Schistosoma itself [71][142]. Likewise, putative neuropeptides derived from this parasite alter the behavior of the cercariae stage, therefore their use has been proposed as strategy for the control of the infection [69][140]. In trypanosomes such as Trypanosoma evansi and Trypanosoma equiperdum, causing surra and dourine in animals, some AMPs have been shown to exert an trypanocidal effect; hence, they have been proposed for use in new treatment strategies of trypanosomiasis in animals [79][80][150,151]. Furthermore, AMPs isolated from triatomine hemolymph have been shown to have trypanolytic activity against different strains of Trypanosoma rangeli, an infectious but non-pathogenic human parasite [81][152]. In these trypanosomes, AMPs exert their action through different mechanisms, including plasma membrane permeabilization, mitochondrial alteration, and parasite lysis [79][80][81][150,151,152].5. Antimicrobial Peptides against Kinetoplastids Causing Neglected Tropical Diseases
5.1. AMPs against T. brucei
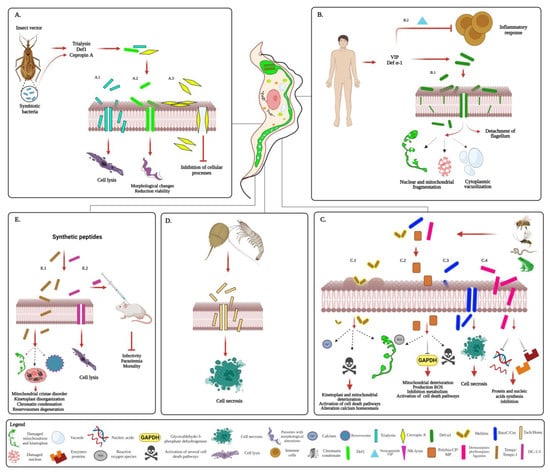
Many of the AMPs that are active against T. brucei are produced by a wide variety of organisms, including mammals and the insect vector [82][83][16,153]. These can carry out their action extracellularly, by plasma membrane disturbance, or intracellularly, by altering the function of some intracellular compartments [82][82][83][84][16,16,153,154] (Figure 23).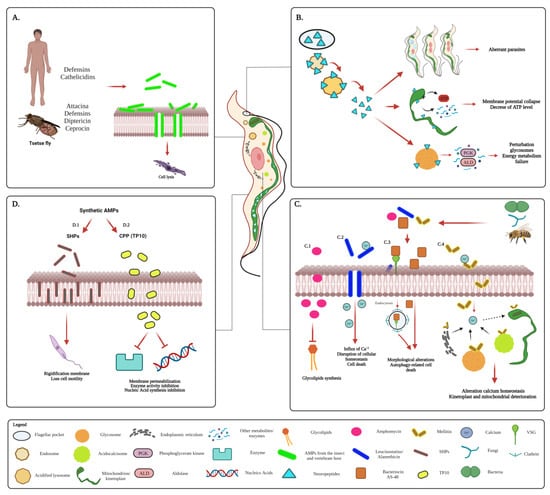
5.2. AMPs against T. cruzi
The antiparasitic activity of AMPs has also been evaluated on T. cruzi, using some peptides obtained from a variety of natural sources and others synthetically prepared [15][85][86][87][88][89][90][17,22,103,106,178,184,185] (Figure 34).