1. The Role of Epithelial–Mesenchymal Transition through the Activation of Transcription FactorT-TFs in Cancer Invasion and Metastasis in Salivary Adenoid Cystic ACCarcinoma
EMT-TFs serve the purpose of maintaining stem cell characteristics, increasing the tumorigenicity of cells, and connecting to cancer stem cells. Furthermore, EMT-TFs create a phenotype that fosters cell survival by supporting DNA repair, evading immune recognition, resisting treatments, delaying aging, and avoiding cell death, which is advantageous under various stressful conditions. Taken together, the classical EMT functions and the varied, context-dependent, unique, and non-classical functions of EMT-TFs, which are also dynamically modulated by the tumor microenvironment, allow cancer cells to continuously adjust to changing conditions
[1][35].
1.1. TWIST Expression in SACC and Its Significance in Pathogenesis and Prognosis
TWIST is a crucial protein that drives the EMT process by decreasing the expression of E-cadherin and increasing the expression of mesenchymal markers
[2][13]. TWIST consists of two proteins, namely TWIST1 and TWIST2, that exhibit comparable levels of expression in tumors
[3][36]. Studies revealed TWIST to be the most highly expressed factor among EMT-TFs in SACC tumor tissue samples, with the solid variant being the most reactive
[4][5][37,39]. These results agree with a study performed by Shen et al., which showed significantly higher TWIST expression in SACC compared to pleomorphic adenomas (PAs) and normal parotid gland tissues
[6][38]. Additionally, TWIST expression was documented to be higher in cases with DM than in those without DM
[6][38].
The overexpression of TWIST has been observed to prompt an EMT-like transformation of metastatic cell lines of human SACC, leading to upgraded migration and invasion capabilities
[5][39]. Elevated TWIST expression has also been strongly tied to perineural invasion (PNI), local regional recurrence, and DM of SACC. A high, predominantly nuclear TWIST expression in SACC was recorded when compared to mucoepidermoid carcinoma (MEC), normal glands, and benign tumors
[7][40]. Kerche et al. similarly detected broad nuclear expression of the TWIST protein in SACC, although they did not find any link between TWIST expression and the absence of E-cadherin expression
[8][41]. These findings corroborate previous research conducted on various types of tumors, which demonstrated a possible association between the elevated nuclear expression of TWIST and the increased invasiveness of tumors
[9][10][42,43].
Recently, siRNA was used to reduce TWIST levels in SACC cells in order to investigate how TWIST is involved in the neurophilic invasion of these cells. Inhibiting TWIST resulted in decreased cancer cell migration, invasion, and PNI ability, as well as a reduction in the expression of genes associated with EMT and Schwann cell markers. When S100A4 was overexpressed using a plasmid, researchers noted a significant growth in the migratory, invasive, and PNI ability of SACC cells, along with a more fibroblast-like appearance and an increase in pseudopodia formation
[11][44].
1.2. SNAIL and SLUG: Key Transcription Factors Driving Epithelial–Mesenchymal Transition and Metastasis in Salivary Adenoid Cystic Carcinoma
1.2. SNAIL and SLUG: Key Transcription Factors Driving EMT and Metastasis in SACC
According to broad evidence, the SNAIL transcription factor suppresses the expression of E-cadherin, which results in the inhibition of cell–cell adhesion and augmented cancer invasion in different types of cancer
[12][13][46,47]. SNAIL has a significant role in triggering the process of EMT and its reactivity was shown to be heterogeneous, with a more distinct nuclear pattern in the solid variant and invasive tumor front of SACC
[4][14][37,48]. Remarkably, high levels of SNAIL expression are linked to PNI, local and regional recurrence, as well as DM of SACC
[14][48]. Zhao et al. pointed out that the increased expression of SNAIL and Integrin-linked kinase (ILK) strongly correlated with the solid pattern, advanced TNM stages, a high risk of recurrences, and DM
[15][52]. Of note, the authors found a correlation between the overexpression of SNAIL and N-cadherin and a neural invasive phenotype of SACC.
Another protein, the small heat-shock protein of 27 kDa (HSP27), has also been connected to cancer progression in SACC. The overexpression of HSP27 in SACC cells encouraged migration and invasion, induced EMT, and upregulated the expression of SNAIL and paired related homeobox protein 1 (PRRX1)
[16][53]. In contrast, HSP27 silencing reduced migration and invasion and contributed to the MET of SACC cells. Additionally, HSP27 downregulated the expression of E-cadherin indirectly via the activation of SNAIL and PRRX1 expressions, while the overexpression of SNAIL or PRRX1 restored migration and invasion in HSP27 knockdown cells. The results of the study align with the clinical outcome of patients with SACC, as HSP27 expression is related to radioresistance and the poor prognosis of SACC patients, as well as with the expression of PRRX1 or SNAIL in SACC tissues.
More recently, Du et al. identified protein kinase D1 (PKD1) as a key regulator of SACC progression
[17][55]. The inhibition of PKD1 resulted in the decreased proliferation, migration, invasion, and EMT of SACC cells. Conversely, the overexpression of kinase-active PKD1 evoked EMT and promoted cell migration in human SACC cells. The downregulation of PKD1 was found to control SNAIL via phosphorylation at Ser-11 on SNAIL protein and the promotion of proteasome-mediated degradation, which ultimately reduced lung metastasis in vivo.
Belulescu et al. reported that SLUG was the second most abundant EMT-TF detected in SACC, with reactivity observed in 78% of cases, mainly in the stroma at the level of cancer-associated fibroblasts (CAFs) and endothelial cells
[4][37]. SLUG expression was noted to be higher at the invasive front and in cases associated with DM and lymph node metastases
[4][37]. Similarly, Tang et al. stated that 72% of studied SACC exhibited reactivity to SLUG, and this correlated significantly with TNM stages, PNI, local regional recurrence, and DM
[18][56]. The mitogen-activated protein kinase (MAPK)–SLUG pathway has been shown to contribute to the metastatic formation of SACC
[19][58]. The high expression of SLUG and the extracellular matrix metalloproteinase inducer (EMMPRIN), along with low E-cadherin expression, were significantly linked with PNI and SACC
[20][59]. Silencing SLUG inhibited the EMT process by reducing EMMPRIN expression and subsequently increasing E-cadherin expression in SACC. Two studies also revealed that elevated levels of BTBD7, a spliced variant of BTBD containing binding sites for significant transcription factors, were significantly linked to SLUG expression in SACC
[21][22][60,61]. The reduction in BTBD7 considerably impeded the expression of SLUG in SACC cells. Furthermore, Yang et al. found a significant difference in SLUG expression between SACC and normal salivary gland cases, which was significantly linked with TNM stage, tissue typing, and DM.
Liu et al. displayed evidence that the epiregulin (EREG)–epidermal growth factor receptor (EGFR)–SNAIL/SLUG pathway propels EMT and metastatic characteristics in SACC cells, suggesting it could be an effective target for treating metastatic SACC
[23][62]. In accordance with these data, epidermal growth factor (EGF) stimulation resulted in EGRF activation, which stabilized SNAIL expression and provoked EMT
[24][63]. The suppression of SNAIL expression significantly inhibited EGF-induced EMT in these tumors. Jiang et al. further supported the aforementioned findings, as the authors displayed that the EMT process in SACC cell lines was reversed by pretreatment with Nimotuzumab, an anti-EGFR monoclonal antibody
[25][64]. Moreover, high levels of BMI-1, nuclear/cytoplasmic SNAIL and SLUG expression, and low membranous E-cadherin expression were significantly coupled with DM in SACC
[26][65].
1.3. ZEB1/ZEB2: A Key Regulator of Epithelial–Mesenchymal Transition in Salivary Adenoid Cystic Carcinoma
1.3. ZEB1/ZEB2: A Key Regulator of EMT in SACC
ZEB1/ZEB2 is a critical regulator in various tumor types and is downstream of nuclear factor kappa B (NF-κB)
[27][28][66,67]. However, the immunohistochemical (IHC) detection of ZEB1 in SACC tumor parenchyma did not depict any positive reaction, while positive staining was examined in the tumor stroma at the level of CAFs and endothelial blood cells in all cases
[4][37]. These results align with a study carried out by Kerche et al., that observed ZEB1 expression on both tumor cells and stromal cells
[8][41]. Moreover, NF-κB directly binds to the promoter region of ZEB1, leading to its transcriptional activation
[29][68]. Insulin-like growth factor-binding protein 2, which is significantly upregulated in SACC, plays a crucial role in the invasion and metastasis of this cancer type by modulating the NF-κB/ZEB1 signaling pathway. Interestingly, the knockdown of cancer cell-derived immunoglobulin G (cancer-IgG) in SACC suppressed EMT by upregulating E-cadherin and downregulating ZEB1/ZEB2, suggesting that cancer-IgG could be a useful prognostic marker for this disease
[30][69]. Additionally, cancer-IgG overexpression was significantly correlated with metastasis, recurrence, and invasion in SACC.
2. Expression of Major Epithelial–Mesenchymal TransitionT Markers in Salivary Adenoid Cystic ACCarcinoma
Epithelial cells are transdifferentiated towards a mesenchymal cell phenotype during EMT, as they lose epithelial markers such as E-cadherin or various types of cytokeratin (CK), while gaining mesenchymal markers such as vimentin, fibronectin or N-cadherin
[31][70].
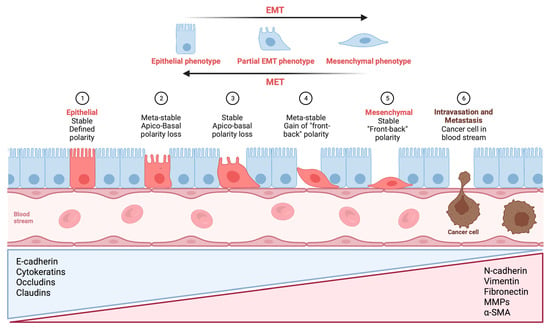
Figure 1 provides a visual illustration that outlines the various phases of EMT.
Figure 1. Visual illustration of a typical epithelial–mesenchymal transition (EMT) program. Epithelial cells exhibit apical–basal polarity and express specific molecules associated with the maintenance of cell polarity and the epithelial state, which are listed in the blue triangular box. As epithelial cells undergo a progressive loss of their distinctive characteristics, they acquire mesenchymal features while retaining certain epithelial traits. In certain situations, they may acquire a complete set of mesenchymal features. Mesenchymal cells display front-to-back polarity, with a highly reorganized cytoskeleton, and they express a distinct set of molecules that promote and preserve the mesenchymal state (listed in the red triangular box). As a result, cells undergoing EMT exhibit increased migration, invasion, and stem-like properties, which can result in treatment resistance, metastasis formation, and recurrences in patients, ultimately affecting their clinical outcomes. Metastatic colonization requires a reversal of EMT, referred to as mesenchymal-to-epithelial transition (MET), which facilitates the expansion of tumor cells, an essential prerequisite for metastatic growth. E-cadherin: epithelial cadherin; N-cadherin: neural cadherin; MMP: matrix metalloproteinase; α-SMA: alpha-smooth muscle actin (Figure created using BioRender, Toronto, ON, Canada).
2.1. Expression of CK in Salivary Adenoid Cystic Carcinoma
2.1. Expression of CK in SACC
CK is a group of intermediate filaments that offer structural reinforcement to epithelial cells. CK expression is dependent on the tissue or organ and the degree of cell differentiation. Due to the specific cytokeratin patterns associated with different cellular sources, CK can be employed as a diagnostic marker for tumors, particularly epithelial cancers
[32][33][71,72].
To characterize the CK profile of SACC, several studies have employed IHC examination of CK reactivity. In one study of 32 SACC cases, AE1/AE3 staining was found to be most intense in solid histological subtypes, followed by the tubular and cribriform subtypes
[34][73]. SACC cells demonstrated strong and widespread positivity for AE1/AE3, comparable to the residual parenchyma of the major or minor salivary gland
[35][74]. Furthermore, SACC exhibited positivity for CK7 but negativity for CK20, consistent with the results reported by Lee et al.
[36][75]. Ben Salha et al. observed that AE1/AE3 and CK7 showed widespread and positive reactivity in solid SACC, whereas CK5/6 and CAM5.2 were only reactive in some neoplastic cells
[37][76]. Notably, CK14-positive cells at the invasive front of SACC were reported to stimulate cancer cell invasion and may serve as a potential prognostic marker in these patients
[38][77].
2.2. The Role of E-Cadherin in Salivary Adenoid Cystic Carcinoma: Implications for Tumor Invasion, Metastasis, and Epithelial–Mesenchymal Transition
2.2. The Role of E-Cadherin in SACC: Implications for Tumor Invasion, Metastasis, and EMT
E-cadherin is a critical protein that plays a vital role in maintaining the structural integrity of epithelial tissue and is involved in regulating epithelial cell growth, differentiation, and survival. Multiple research projects have proven a reduction in E-cadherin expression in specimens of SACC compared to adjacent normal salivary parenchyma, leading to PNI, regional recurrence, and DM
[8][11][15][39][40][41][42][41,44,52,78,79,80,81]. These studies revealed that regardless of the histological subtype, SACC cells located at the invasive tumor front and near nerve fibers exhibit reduced E-cadherin expression, with the alteration of the subcellular pattern from membranous to predominantly cytoplasmic
[34][43][73,82]. Interestingly, the co-expression of E-cadherin with the gene cadherin 4 (CDH4), encoding retinal cadherin (R-cadherin), inhibits the growth and metastasis of SACC
[44][83]. Moreover, the overexpression of ILK via the dysregulation of E-cadherin/N-cadherin might trigger EMT and thus facilitate SACC invasion and metastasis. Notably, a reduction in E-cadherin expression has also been explored in the myoepithelial cells of SACC compared to normal gland tissue
[8][45][41,84], and the absence of E-cadherin expression correlated with PNI, vascular invasion, and DM in SACC patients.
In contrast to previous studies, two groups of researchers have observed E-cadherin expression in most of their SACC samples, suggesting that EMT in SACC could be related to the transformation of ductal epithelial cells into myoepithelial cells
[46][47][85,86]. However, van der Wal et al. reported a favorable prognosis of SACC patients with diminished E-cadherin expression
[48][87]. Wu et al. conducted a study on the invasion of neoplastic epithelial cells into adjacent tissue in SACC samples and noticed that the cells invaded as multicellular units and maintained their expression of E-cadherin
[49][88]. The study’s IHC results also showed that cancer cells at the invasive front did not exhibit a complete EMT signature. The three-dimensional (3D) spheroid invasion assay revealed that leader cells displayed an elongated phenotype while still retaining E-cadherin expression, representing a partial EMT phenotype. These findings are consistent with previous studies that have noted that leader cells lack a complete EMT signature and cannot be identified through canonical mesenchymal markers
[50][51][89,90]. EMT is not usually uniformly evident throughout the entire tumor tissue, and partial EMT is more common in cancer than complete EMT
[52][53][91,92].
2.3. Vimentin: A Key Intermediate Filament in Mesenchymal Cells and Its Role in Salivary Adenoid Cystic Carcinoma Metastasis
2.3. Vimentin: A Key Intermediate Filament in Mesenchymal Cells and Its Role in SACC Metastasis
Vimentin is a type of intermediate filament that is exclusively present in mesenchymal cells. It is a key structural protein that is widely distributed in different types of cells, including fibroblasts, vascular endothelial cells, smooth muscle cells, cartilage and bone cells, and nerve sheath cells, that form the connective tissue
[32][54][71,93].
Belulescu et al. reported that the solid variant of SACC exhibited a more prominent EMT process, likely due to the more obvious vimentin expression perceived in these cases
[34][73]. Furthermore, mesenchymal features were more pronounced in neoplastic cells located at the invasive tumor front and those associated with PNI, as demonstrated by Zhang et al., who found enlarged vimentin expression in areas of nerve invasion
[11][44]. In accordance with these results, various authors declared positive vimentin staining in SACC together with the co-expression of keratin in these cells
[55][56][94,95].
2.4. The Complex Role of Fibronectin in Salivary Adenoid Cystic Carcinoma: Promoting Perineural Invasion, Inhibiting Invasion, and Potential Prognostic Marker
2.4. The Complex Role of Fibronectin in SACC: Promoting PNI, Inhibiting Invasion, and Potential Prognostic Marker
Fibronectin is a type of glycoprotein that creates an extracellular matrix. It is a polypeptide that combines to form a dimer with a molecular weight of approximately 250 kDa. Its primary function is to facilitate the attachment of various cell types such as fibroblasts, hepatocytes, and nerve cells. The cell surface-specific receptor known as Integrin plays a significant role in processes such as cell adhesion, migration, and phagocytosis, particularly in the context of tissue damage
[32][57][71,96].
Similar to the results regarding vimentin expression, fibronectin seems to act as a key player in the process of SACC invasiveness. In solid subtypes, fibronectin expression was observed to diminish in the outer region of invasive tumor clusters, while strong expression was detected in neoplastic cells that surround and infiltrate the nerve fibers, suggesting its potential dual role in promoting PNI, while potentially inhibiting invasion at the invasive tumor front
[34][58][59][60][73,97,98,99]. In contrast, other studies have found lower fibronectin expression in the solid SACC variant compared to the tubular and cribriform subtypes
[61][62][100,101]. Interestingly, the authors found a significant association between fibronectin expression, low TNM stages, and non-metastatic SACC cases
[61][100]. Moreover, they postulated a potential inverse correlation between BTBD7 and fibronectin, indicating BTBD7′s potential prognostic role in predicting the invasion and metastasis of SACC.
2.5. N-Cadherin Expression in Salivary Adenoid Cystic Carcinoma: Association with Perineural Invasion and Epithelial–Mesenchymal Transition
2.5. N-Cadherin Expression in SACC: Association with PNI and EMT
N-cadherin is a transmembrane glycoprotein that belongs to the classic cadherin superfamily and has a molecular weight of 130-kDa. Its expression has been detected in a range of cell types, such as neurons, endothelial cells, and cardiomyocytes
[32][63][71,102].
Zhao et al. found that N-cadherin was expressed in 68.1% of the investigated SACC cases, with a membranous and/or cytoplasmic reaction pattern, and the highest expression levels were found in cases with PNI
[15][52]. These findings are supported by Zhang et al., who also reported heightened N-cadherin expression in the area of nerve invasion in SACC
[11][44]. In contrast, Belulescu et al. observed N-cadherin reactivity in only 35% of SACC cases, with low intensity and no significant difference between the three histological subtypes
[34][73]. The discrepancy in results may be due to differences in the sample size, technique, or antibodies used for N-cadherin detection. Additionally, N-cadherin expression was found to be negatively aligned with E-cadherin expression and positively associated with the expression of SNAIL and ILK in SACC, implying that ILK overexpression might induce EMT by dysregulating the expression of these cadherins, leading to SACC invasion and metastasis
[15][52]. Jiang et al. reported that thioredoxin 1 (TXN) and thioredoxin reductase 1 were overexpressed in SACC cases with metastasis and were partnered with high N-cadherin expression and low E-cadherin expression, proposing that TXN incites an EMT-like phenotype that promotes SACC migration and invasion
[64][103]. These findings highlight the potential of N-cadherin, SNAIL, ILK, and TXN as prognostic markers for SACC.