Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Mohammad Fouad Bayan and Version 2 by Rita Xu.
The nature of microorganisms and the efficiency of antimicrobials have witnessed a huge co-dependent change in their dynamics over the last few decades. On the other side, metals and metallic compounds have gained popularity owing to their effectiveness against various microbial strains.
- metals
- nanoparticles
- antimicrobial action
1. Introduction
Several kinds of microorganisms lead to the initiation and further development of microbial infections. Such infections primarily and solely manifest in many pathological conditions with variant degrees of severity. Their pathologies precipitate numerous mild symptoms (fever, fatigue, nausea, headache) to serious symptoms (cyanosis, tissue necrosis, lymphadenopathy, respiratory effects) [1][2][1,2]. These have also evolved to be one of the major secondary factors in various diseased conditions, while in some, they ultimately cause death [3][4][3,4]. Microbial infections and their manifestations interfere at every step of medical methodologies from concluding misleading and erroneous diagnoses to resulting in deleterious surgeries as well as unsuccessful and incomplete treatments [5]. The severity of the condition worsens when there is any kind of additional or major infection. All of this has made antimicrobials a very important and fundamental part of therapeutics and pharmacology [6][7][6,7].
The last century has seen excessive use of antibiotics for various kinds of infections. They work by targeting bacterial cell components and altering necessary processes like DNA replication, cell wall synthesis, etc. [8][9][10][8,9,10]. However, they do have certain drawbacks that make them insufficient and problematic in various ways:
Along with these deficiencies, increasing antibiotic resistance has hinted at the clinical need for newer antimicrobials to tackle microbial growth and biofilm production effectively. Continuous mutations, irrational use of antibiotics, and the production of enzymes that inactivate the bacterial cells have contributed to this increasing resistance to the agents [16][17][16,17].
Barring antibiotics, metal compounds were largely in use and practice before the 1920s, after which antibiotics took over [18][19][20][18,19,20]. The potential of metals to conveniently restrict biofilm production make them the best possible alternative in present times [21][22][21,22]. Antimicrobial properties of metals have been used since ancient times for disinfecting food and water, managing plant diseases in agriculture, and in medical areas as well [23][24][23,24]. Certain metals are necessary for cell functioning and cell membrane formation but their presence in excess amounts can be lethal, whereas specific other metals like non-essential groups, such as mercury, silver, etc., are found to be microbicidal even at very low concentrations [25].
2. Metals as Antimicrobials
Metals are abundant in the earth’s crust and ecosphere. The Great Oxidation Event (GOE), which took place 2.3–2.4 billion years ago, exposed bacteria to a wide range of metal ions. The earth’s crust contains a variety of oxidized forms of metal compounds as a result of the atmosphere’s rising oxygen level. Enzymes used metals like copper, iron, and zinc for their redox reactions. Metals are necessary for the process of life but are toxic at high intracellular concentrations, and, thus, cells need a homeostasis mechanism to keep the intracellular concentration constant. Zinc and copper share a similar pathogen-killing mechanism in eukaryotes, where oxidative stress is used to destroy the encapsulated bacterium. Metals like gold, silver, and mercury are extremely poisonous to microorganisms at low concentrations [26][29]. Metals were once utilized as antibacterial agents, but their industrial usage can harm the ecological system, although they do have a medical use. Infections were treated with arsenic, mercury, silver, copper, zinc, and other elements. Antimony and arsenic are employed as fungicides, rodenticides, insecticides, and to treat protozoal illnesses. While zinc salts can be used to treat diarrhea, copper salts are used to make the Bordeaux and Burgundy mixture, which is used to prevent bacterial and fungal problems in plants and to promote animal growth. Burns can be relieved with silver. Organic mercury compounds are utilized to keep eye drops in good condition. Mercury was utilized as a disinfectant and a syphilis infection treatment. In dental restorations, mercury is combined with copper, silver, and tin [27][30].2.1. Metal-Based Nanoparticles as Antimicrobials
Metallic NPs of sizes ranging from 1 nm to 100 nm can be synthesized by two approaches, i.e., top-down and bottom-up. The top-down approach involves beginning with the material in bulk, which is then broken down into the size of a nanoscopic scale via ball milling or attrition etc. It is an easy method to employ, but increased accommodated impurities and non-uniform sizes of particles limit its use [28][29][31,32]. On the other hand, the bottom-up nanofabrication approach includes variant techniques such as the colloidal synthesis, the sol-gel method, the chemical vapor decomposition process, and the atomic layer deposition among others. The process, though time consuming and tedious, has the benefit of uniform-sized and uniform-shaped smaller particles bearing the least number of defects and controlled surface properties [29][30][31][32,33,34]. The use of metal-based nanoparticles as components in the creation of antibacterial agents has been made possible by nanotechnologies. Metal-based nanoparticles (NPs) demonstrate an effective role in locating and eliminating bacteria through a variety of mechanisms, including attraction to the surface of the bacteria, disruption of the cell wall and membrane, and induction of a toxic mechanism mediated by an increase in oxidative stress (e.g., the production of reactive oxygen species (ROS)) [32][33][34][35][35,36,37,38]. The creation of oxidative stress is a valuable and effective antibacterial method to combat MDR bacteria, given the absence of new antimicrobial medicines with unique mechanisms of action. Therefore, it is important to identify and properly characterize whether NPs might cause oxidative stress in these bacteria [36][37][38][39,40,41]. Metal-based NPs physically interact with bacterial cell surfaces, disrupt their membrane, and, ultimately, restrict the formation of biofilms [39][42]. The formation of biofilms also leads to the development of resistance against antimicrobial agents, so their hindrance ultimately restricts the modulation of resistant mutants, too [40][41][42][43,44,45]. The shape of metal-based NPs along with their ultra-small, compliantly controllable size, and resultant greater surface area to mass ratio all contribute to the prevention of biofilm formation [43][44][45][46,47,48]. The target microorganisms and their mechanisms of action for a few metal-based nanoparticles are provided in Table 1.Table 1. Antimicrobial activity mechanisms of different metal-based NPs.
| S. No. | Metal-Based Nanoparticles | Microorganism | Mechanism of Action | Reference | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. | Smaller silica nanoparticles | E. coli | bacteria | Cell wall rupturing | [46] | [49] | |||||
| 2. | AgNPs | K. pneumoniae | Damage to bacterial cell wall Reactive Oxygen Species (ROS) generation |
[47] | [50] | ||||||
| 3. | CuNPs | F. oxysporum | Structural and functional changes in fungi cell, affects DNA and its replication, and protein synthesis | [48] | [51] | ||||||
| 4. | AuNPs | B. subtilis | Bacterial membrane damage | [49] | [52] | ||||||
| 5. | Iron nanoparticles | P. aeruginosa | , | E. coli | , | S. aureus | and | B. subtilis | Bacterial cell membrane rupture ultimately led to bacteria death | [50] | [53] |
| 6. | Gallium based nanoparticles | P. aeruginosa | ROS-mediated bacterial cell wall damage | [51] | [54] |
2.2. Mechanisms Involved in Antimicrobial Activity of Metal and Metal-Based NPs
Metal and metal NPs interfere with bacteria’s hemostasis in 3 major ways: disruption of the membrane, oxidative stress, and interaction with proteins and enzymes (Figure 1).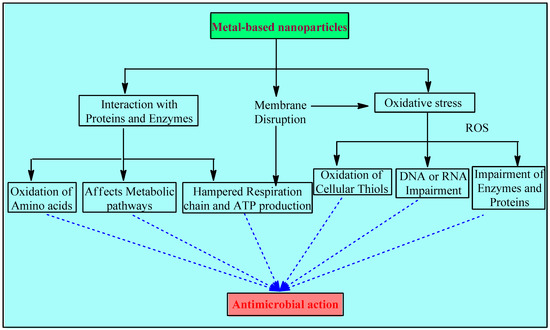
Figure 1. Mechanisms involved in the antimicrobial activity of metals and metal-based NPs.
