
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lisa Adams | + 2471 word(s) | 2471 | 2021-04-26 11:24:12 | | | |
| 2 | Catherine Yang | Meta information modification | 2471 | 2021-05-11 03:28:00 | | | | |
| 3 | Conner Chen | Meta information modification | 2471 | 2021-09-22 03:29:34 | | |
Video Upload Options
This entry outlines recent preclinical and clinical advances in molecular imaging of abdominal aortic aneurysms (AAA) with a focus on molecular magnetic resonance imaging (MRI) of the extracellular matrix (ECM). In addition, developments in pharmacologic treatment of AAA targeting the ECM are reviewed and results from animal studies are contrasted with clinical trials. Abdominal aortic aneurysm (AAA) is an often fatal disease without non-invasive pharmacologic treatment options. The ECM, with collagen type I and elastin as major components, is the key structural component of the aortic wall and is recognized as a target tissue for both initiation and the progression of AAA. Molecular imaging allows in vivo measurement and characterization of biological processes at the cellular and molecular level and sets forth to visualize molecular abnormalities at an early stage of disease, facilitating novel diagnostic and therapeutic pathways. By providing surrogate criteria for the in vivo evaluation of the effects of pharmacological therapies, molecular imaging techniques targeting the ECM may facilitate the development of pharmacological drugs. In addition, molecular targets can also be used within theranostic approaches that have the potential for timely diagnosis and simultaneous medical therapy. Recent successes in preclinical studies suggest future opportunities for clinical translation. However, further clinical studies are needed to validate the most promising molecular targets for human application.
1. Introduction
Abdominal aortic aneurysm (AAA) is a fatal, but often asymptomatic disease with controversial treatment and insufficient prediction of complications [1][2]. The extracellular matrix (ECM), with collagen type I and elastin as main components, is the key structural component of the aortic wall and is recognized as a target tissue for both the onset and the progression of AAA. While large and fast-growing AAAs are indicated for surgery/vascular repair, management of medium-sized AAAs remains challenging [3]. Currently, there is no effective treatment, which can slow down or prevent AAA growth.
In clinical routine, morphological criteria for the determination of the aortic diameter on ultrasound, computed tomography (CT) and MRI have been used in accordance with guidelines as evaluation criteria for therapeutic intervention or conservative therapy. By contrast, molecular imaging enables an in vivo measurement and characterization of biological processes at cellular and molecular level and sets forth to visualize molecular abnormalities at an early stage of disease, facilitating novel diagnostic and also therapeutic pathways [4]. MRI is a non-ionizing modality well suited for imaging and characterizing the relatively thin arterial vessel wall as it allows imaging with high spatial resolution and excellent soft tissue contrast [5]. By combining molecular imaging with MR target-specific probes, pathological processes can be detected and characterized in vivo [6]. This information can provide new insights into the pathogenesis of diseases in vivo and help to develop new diagnostic targets and monitor potential therapeutic success. By providing surrogate criteria for the in vivo evaluation of the effects of pharmacological therapies, molecular imaging techniques targeting the ECM may facilitate the development of pharmacological drugs. In addition, they can help to improve the clinical efficacy of medical therapies by guiding the intensity of treatment based on the properties of the molecular tissue. For example, molecular imaging of the ECM could on the one hand enable non-invasive assessment of aneurysmal tissue changes and rupture risk, and on the other hand allow to monitor the levels of key ECM proteins in response to therapeutic intervention, such as elastin and collagen [7].
2. The Role of the Extracellular Matrix in AAA
In each organ, the composition of the ECM has a different three-dimensional structure and a constant pattern of remodeling to regulate tissue homeostasis [8]. Within the vascular system, the ECM is essential to resist the range of blood pressures and shear forces acting on the vessel walls. Development of AAA involves localized inflammatory response with proinflammatory cells (e.g., macrophages) and degradation/remodeling of the ECM. Disturbances in the synthesis and proteolytic degradation of the aortic structural ECM proteins, particularly collagen and elastin, have been shown to be critical to AAA pathogenesis [9]. Besides elastolysis and collagen lysis, which are reasonably well known, there is as yet little information on changes in other ECM proteins [10]. Prior proteomics analyses revealed thrombospondin 1 and 2, periostin, fibronectin und tenascin to be significantly altered proteins in the ECM of aneurysm tissue [10][11]. The protein thrombospondin 1 is a large glycosylated secretory protein with adhesive properties towards ECM components, including collagen, fibrinogen and fibronectin [11]. Periostin (fasciclin 1 family) is an ECM protein, which interacts with integrin molecules on cell surfaces, providing signals for tissue development and remodeling [12] and promoting the secretion of matrix metalloproteinases (MMPs) from cardiac cells [13]. In a mouse model of AAA, it was shown, that periostin was upregulated during the progression of AAA, particularly at times when active inflammation caused destruction of the ECM [14]. Periostin could thus function both in terms of a clinical biomarker of disease activity in AAA and in terms of a therapeutic target for patients with AAA. Fibronectin is another important glycoprotein of the aortic ECM and its expression has been reported to be increased in AAA of patients with tricuspid aortic valve stenosis or bicuspid aortic valve [15].
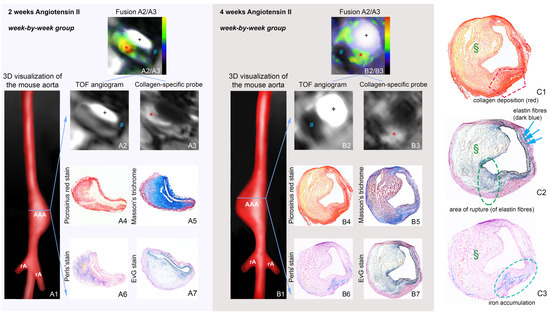
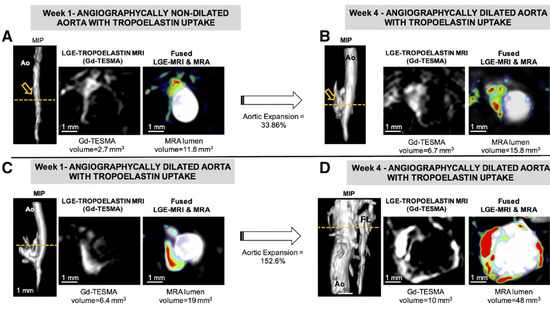
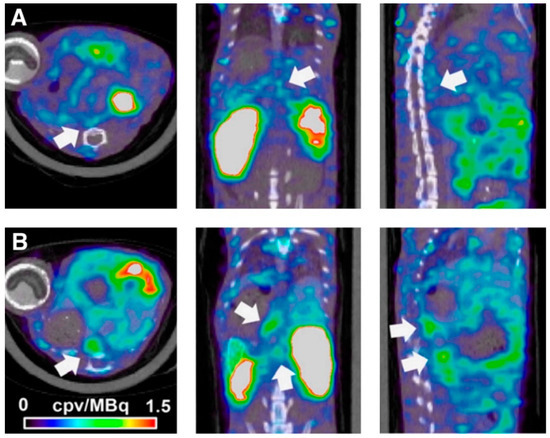
2.1. Molecular Magnetic Resonance Imaging for Identification of Potential Diagnostic Targets and Monitoring of Therapeutic Success
2.2. Pharmacologic Treatment Strategies Targeted to the ECM
Experimental targets for pharmaceutical AAA stabilization, that target the ECM, are thrombospondin inhibitors [36], cysteine protease inhibitors [37], serine protease inhibitors [38], protease inhibitors such as MMP inhibitors [39], and interleukins [40]. Other potential targets include inhibition of c-Jun N-terminal kinase as well as miR-29b (microRNA), both of which demonstrated a reduction of AAA via modulation of the ECM metabolism [41][42].
In humans, elevated thrombospondin-1 (TSP-1) was associated with MMP activitaion, ECM degradation as well as tissue infiltration [43]. Cysteine cathepsins (Cat) are a diverse group of proteases that are abundant in VSMCs, macrophages and endothelial cells of atherosclerotic plaques and aneurysmal lesions. Among the cysteine cathepsins, Cat S directly modulates inflammatory and immune responses and apoptosis of VSMCs, whereby elevated expression levels in the vessel wall and plasma of human AAA were recently confirmed [44]. In AAA, blockade of Cat S induced ECM degradation is still in the preclinical phase [45][46]. Recent preliminary preclinical studies in murine models showed promising results for two serine protease inhibitors, serpina3n (SA3N)—a potent inhibitor of granzyme B [38] and ulinastatin [47].
3. Discussion and Outlook
Over the past 30 years, a variety of promising gadolinium-based molecular tracers have been developed and their utility confirmed in proof-of-concept animal studies. Nevertheless, only non-targeted contrast agents have made it to clinical approval for use in humans. A challenge in clinical translation is that toxicological testing is time-consuming and costly, requiring substantial financial resources. This contradicts the expected revenues, which are lower for molecular tracers than for clinically proven nonspecific contrast agents due to their limited application in specific settings. Nonetheless, these markers play a prominent role especially in preclinical research and, if validated, can non-invasively image new therapeutic approaches, for example targeting ECM remodeling. In this context, the elastin-specific MRI probe Gd-ESMA and tropoelastin-specific MRI probe Gd-TESMA appear promising, as they can specifically detect the effect of therapeutic strategies affecting the elastin content in the aortic wall via their different pathways. Another novel approach are multitarget probes, which could allow for concurrent imaging of different pathophysiological processes at the molecular level. Molecular agents may also be used within theranostic approaches with the potential for timely diagnosis and concurrent medical therapy. Here, the use of miRNA-based targets is a novel and promising method.
Currently, there is no established pharmacologic therapy for the treatment of AAA. The apparent discordance between successes in preclinical studies and partly disappoint results in clinical trials suggests an incomplete understanding of the various pathological processes involved in the development of AAAs and points to possible inadequacies in animal models of the disease. To increase future therapeutic strategies, molecular imaging markers that can specifically visualize and quantify targets of pharmacological agents in ECM in a non-invasive manner hold promise for a better reflection of the pathophysiological processes behind the disease and during therapy. In addition, recent preclinical approaches in large animal models represent a promising option for evaluating therapeutic strategies and create better options for multiparametric functional imaging, which seems to be limited in parts by the low volume in small animal models. With the increasing availability of image-based molecular biomarkers and promising pharmacotherapies, as well as their evaluation in large animal models of AAA and initial human clinical trials, there is reason to hope that molecular therapeutic approaches targeting ECM will enter clinical research in the coming years.
References
- Toczek, J.; Meadows, J.L.; Sadeghi, M.M. Novel Molecular Imaging Approaches to Abdominal Aortic Aneurysm Risk Stratification. Circ. Cardiovasc. Imaging 2016, 9, e003023.
- Hallett, J.W. Management of Abdominal Aortic Aneurysms. Mayo Clin. Proc. 2000, 75, 395–399.
- Sakalihasan, N.; Limet, R.; Defawe, O. Abdominal aortic aneurysm. Lancet 2005, 365, 1577–1589.
- Weissleder, R.; Mahmood, U. Molecular Imaging. Radiology 2001, 219, 316–333.
- Makowski, M.R.; Botnar, R.M. MR Imaging of the Arterial Vessel Wall: Molecular Imaging from Bench to Bedside. Radiology 2013, 269, 34–51.
- Makowski, M.R.; Wiethoff, A.J.; Jansen, C.H.; Botnar, R.M. Molecular Imaging with Targeted Contrast Agents. Top. Magn. Reson. Imaging 2009, 20, 247–259.
- Okamura, H.; Pisani, L.J.; Dalal, A.R.; Emrich, F.; Dake, B.A.; Arakawa, M.; Onthank, D.C.; Cesati, R.R.; Robinson, S.P.; Milanesi, M.; et al. Assessment of Elastin Deficit in a Marfan Mouse Aneurysm Model Using an Elastin-Specific Magnetic Resonance Imaging Contrast Agent. Circ. Cardiovasc. Imaging 2014, 7, 690–696.
- Sonbol, H.S. Extracellular matrix remodeling in human disease. J. Microsc. Ultrastruct. 2018, 6, 123–128.
- Keeling, W.B.; Armstrong, P.A.; Stone, P.A.; Bandyk, D.F.; Shames, M.L. An Overview of Matrix Metalloproteinases in the Pathogenesis and Treatment of Abdominal Aortic Aneurysms. Vasc. Endovasc. Surg. 2005, 39, 457–464.
- Didangelos, A.; Yin, X.; Mandal, K.; Saje, A.; Smith, A.; Xu, Q.; Jahangiri, M.; Mayr, M. Extracellular matrix composition and remodeling in human abdominal aortic aneurysms: A proteomics approach. Mol. Cell Proteom. 2011, 10, M111.008128.
- Yagi, H.; Nishigori, M.; Murakami, Y.; Osaki, T.; Muto, S.; Iba, Y.; Minatoya, K.; Ikeda, Y.; Ishibashi-Ueda, H.; Morisaki, T. Discovery of novel biomarkers for atherosclerotic aortic aneurysm through proteomics-based assessment of disease progression. Sci. Rep. 2020, 10, 1–12.
- Kudo, A. Periostin in fibrillogenesis for tissue regeneration: Periostin actions inside and outside the cell. Cell. Mol. Life Sci. 2011, 68, 3201–3207.
- Hakuno, D.; Kimura, N.; Yoshioka, M.; Mukai, M.; Kimura, T.; Okada, Y.; Yozu, R.; Shukunami, C.; Hiraki, Y.; Kudo, A. Periostin advances atherosclerotic and rheumatic cardiac valve degeneration by inducing angiogenesis and MMP production in humans and rodents. J. Clin. Investig. 2010, 120, 2292–2306.
- Yamashita, O.; Yoshimura, K.; Nagasawa, A.; Ueda, K.; Morikage, N.; Ikeda, Y.; Hamano, K. Periostin Links Mechanical Strain to Inflammation in Abdominal Aortic Aneurysm. PLoS ONE 2013, 8, e79753.
- Della Corte, A.; Quarto, C.; Bancone, C.; Castaldo, C.; Di Meglio, F.; Nurzynska, D.; De Santo, L.S.; De Feo, M.; Scardone, M.; Montagnani, S.; et al. Spatiotemporal patterns of smooth muscle cell changes in ascending aortic dilatation with bicuspid and tricuspid aortic valve stenosis: Focus on cell–matrix signaling. J. Thorac. Cardiovasc. Surg. 2008, 135, 8–18.
- Brangsch, J.; Reimann, C.; Collettini, F.; Buchert, R.; Botnar, R.M.; Makowski, M.R. Molecular Imaging of Abdominal Aortic Aneurysms. Trends Mol. Med. 2017, 23, 150–164.
- Salarian, M.; Ibhagui, O.Y.; Yang, J.J. Molecular imaging of extracellular matrix proteins with targeted probes using magnetic resonance imaging. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1622.
- Brangsch, J.; Reimann, C.; Kaufmann, J.O.; Adams, L.C.; Onthank, D.C.; Thöne-Reineke, C.; Robinson, S.P.; Buchholz, R.; Karst, U.; Botnar, R.M.; et al. Concurrent Molecular Magnetic Resonance Imaging of Inflammatory Activity and Extracellular Matrix Degradation for the Prediction of Aneurysm Rupture. Circ. Cardiovasc. Imaging 2019, 12, e008707.
- Adams, L.C.; Brangsch, J.; Reimann, C.; Kaufmann, J.O.; Buchholz, R.; Karst, U.; Botnar, R.M.; Hamm, B.; Makowski, M.R. Simultaneous molecular MRI of extracellular matrix collagen and inflammatory activity to predict abdominal aortic aneurysm rupture. Sci. Rep. 2020, 10, 1–13.
- Botnar, R.M.; Brangsch, J.; Reimann, C.; Janssen, C.H.P.; Razavi, R.; Hamm, B.; Makowski, M.R. In Vivo Molecular Characterization of Abdominal Aortic Aneurysms Using Fibrin-Specific Magnetic Resonance Imaging. J. Am. Hear Assoc. 2018, 7, e007909.
- Klink, A.; Heynens, J.; Herranz, B.; Lobatto, M.E.; Arias, T.; Sanders, H.M.H.F.; Strijkers, G.J.; Merkx, M.; Nicolay, K.; Fuster, V.; et al. In Vivo Characterization of a New Abdominal Aortic Aneurysm Mouse Model With Conventional and Molecular Magnetic Resonance Imaging. J. Am. Coll. Cardiol. 2011, 58, 2522–2530.
- Botnar, R.M.; Wiethoff, A.J.; Ebersberger, U.; Lacerda, S.; Blume, U.; Warley, A.; Jansen, C.H.; Onthank, D.C.; Cesati, R.R.; Razavi, R.; et al. In Vivo Assessment of Aortic Aneurysm Wall Integrity Using Elastin-Specific Molecular Magnetic Resonance Imaging. Circ. Cardiovasc. Imaging 2014, 7, 679–689.
- Investigators, M.R.S. Aortic Wall Inflammation Predicts Abdominal Aortic Aneurysm Expansion, Rupture, and Need for Surgical Repair. Circulation 2017, 136, 787–797.
- Conlisk, N.; Forsythe, R.O.; Hollis, L.; Doyle, B.J.; McBride, O.M.; Robson, J.M.; Wang, C.; Gray, C.D.; Semple, S.I.; MacGillivray, T.; et al. Exploring the Biological and Mechanical Properties of Abdominal Aortic Aneurysms Using USPIO MRI and Peak Tissue Stress: A Combined Clinical and Finite Element Study. J. Cardiovasc. Transl. Res. 2017, 10, 489–498.
- Makowski, M.R.; Wiethoff, A.; Ebersberger, U.; Blume, U.; Warley, A.; Jansen, C.; Onthank, D.C.; Cesati, R.R.; Razavi, R.; Marber, M.; et al. Molecular assessment of aortic aneurysm wall integrity using an elastin-specific MR imaging probe. J. Cardiovasc. Magn. Reson. 2013, 15, O4.
- Brangsch, J.; Reimann, C.; Kaufmann, J.O.; Adams, L.C.; Onthank, D.; Thöne-Reineke, C.; Robinson, S.; Wilke, M.; Weller, M.; Buchholz, R.; et al. Molecular MR-Imaging for Noninvasive Quantification of the Anti-Inflammatory Effect of Targeting Interleukin-1β in a Mouse Model of Aortic Aneurysm. Mol. Imaging 2020, 19.
- Lavin, B.; Lacerda, S.; Andia, M.E.; Lorrio, S.; Bakewell, R.; Smith, A.; Rashid, I.; Botnar, R.M.; Phinikaridou, A. Tropoelastin: An in vivo imaging marker of dysfunctional matrix turnover during abdominal aortic dilation. Cardiovasc. Res. 2019, 116, 995–1005.
- Bazeli, R.; Coutard, M.; Duport, B.D.; Lancelot, E.; Corot, C.; Laissy, J.-P.; Letourneur, D.; Michel, J.-B.; Serfaty, J.-M. In Vivo Evaluation of a New Magnetic Resonance Imaging Contrast Agent (P947) to Target Matrix Metalloproteinases in Expanding Experimental Abdominal Aortic Aneurysms. Investig. Radiol. 2010, 45, 662–668.
- Golestani, R.; Razavian, M.; Nie, L.; Zhang, J.; Jung, J.-J.; Ye, Y.; De Roo, M.; Hilgerink, K.; Liu, C.; Robinson, S.P.; et al. Imaging vessel wall biology to predict outcome in abdominal aortic aneurysm. Circ. Cardiovasc. Imaging 2015, 8, e002471.
- Toczek, J.; Ye, Y.; Gona, K.; Kim, H.-Y.; Han, J.; Razavian, M.; Golestani, R.; Zhang, J.; Wu, T.L.; Jung, J.-J.; et al. Preclinical Evaluation of RYM1, a Matrix Metalloproteinase–Targeted Tracer for Imaging Aneurysm. J. Nucl. Med. 2017, 58, 1318–1323.
- Yao, Y.; Cheng, K.; Cheng, Z. Evaluation of a smart activatable MRI nanoprobe to target matrix metalloproteinases in the early-stages of abdominal aortic aneurysms. Nanomed. Nanotechnol. Biol. Med. 2020, 26, 102177.
- Sadeghi, M.M.; Krassilnikova, S.; Zhang, J.; Gharaei, A.A.; Fassaei, H.R.; Esmailzadeh, L.; Kooshkabadi, A.; Edwards, S.; Yalamanchili, P.; Harris, T.D.; et al. Detection of injury-induced vascular remodeling by targeting activated alphavbeta3 integrin in vivo. Circulation 2004, 110, 84–90.
- Meoli, D.F.; Sadeghi, M.M.; Krassilnikova, S.; Bourke, B.N.; Giordano, F.J.; Dione, D.P.; Su, H.; Edwards, D.S.; Liu, S.; Harris, T.D.; et al. Noninvasive imaging of myocardial angiogenesis following experimental myocardial infarction. J. Clin. Investig. 2004, 113, 1684–1691.
- Razavian, M.; Marfatia, R.; Mongue-Din, H.; Tavakoli, S.; Sinusas, A.J.; Zhang, J.; Nie, L.; Sadeghi, M.M. Integrin-Targeted Imaging of Inflammation in Vascular Remodeling. Arter. Thromb. Vasc. Biol. 2011, 31, 2820–2826.
- English, S.J.; Sastriques, S.E.; Detering, L.; Sultan, D.; Luehmann, H.; Arif, B.; Heo, G.S.; Zhang, X.; Laforest, R.; Zheng, J.; et al. CCR2 Positron Emission Tomography for the Assessment of Abdominal Aortic Aneurysm Inflammation and Rupture Prediction. Circ. Cardiovasc. Imaging 2020, 13, e009889.
- Satoh, M.; Nasu, T.; Osaki, T.; Hitomi, S. Thrombospondin-1 contributes to slower aortic aneurysm growth by inhibiting maladaptive remodeling of extracellular matrix. Clin. Sci. 2017, 131, 1283–1285.
- Qin, Y.; Cao, X.; Guo, J.; Zhang, Y.; Pan, L.; Zhang, H.; Li, H.; Tang, C.; Du, J.; Shi, G.-P. Deficiency of cathepsin S attenuates angiotensin II-induced abdominal aortic aneurysm formation in apolipoprotein E-deficient mice. Cardiovasc. Res. 2012, 96, 401–410.
- Ang, L.; Boivin, W.; Williams, S.; Zhao, H.; Abraham, T.; Carmine-Simmen, K.; McManus, B.; Bleackley, R.; Granville, D. Serpina3n attenuates granzyme B-mediated decorin cleavage and rupture in a murine model of aortic aneurysm. Cell Death Dis. 2011, 2, e209.
- Ennis, T.; Jin, J.; Bartlett, S.; Arif, B.; Grapperhaus, K.; Curci, J.A. Effect of Novel Limited-Spectrum MMP Inhibitor XL784 in Abdominal Aortic Aneurysms. J. Cardiovasc. Pharmacol. Ther. 2012, 17, 417–426.
- Ju, X.; Ijaz, T.; Sun, H.; LeJeune, W.; Vargas, G.; Shilagard, T.; Recinos III, A.; Milewicz, D.M.; Brasier, A.R.; Tilton, R.G. IL-6 Regulates Extracellular Matrix Remodeling Associated With Aortic Dilation in a Fibrillin-1 Hypomorphic mgR/mgR Mouse Model of Severe M arfan Syndrome. J. Am. Heart Assoc. 2014, 3, e000476.
- Yoshimura, K.; Aoki, H.; Ikeda, Y.; Fujii, K.; Akiyama, N.; Furutani, A.; Hoshii, Y.; Tanaka, N.; Ricci, R.; Ishihara, T.; et al. Regression of abdominal aortic aneurysm by inhibition of c-Jun N-terminal kinase. Nat. Med. 2005, 11, 1330–1338.
- Zampetaki, A.; Attia, R.; Mayr, U.; Gomes, R.S.; Phinikaridou, A.; Yin, X.; Langley, S.R.; Willeit, P.; Lu, R.; Fanshawe, B.; et al. Role of miR-195 in Aortic Aneurysmal Disease. Circ. Res. 2014, 115, 857–866.
- Yang, H.; Zhou, T.; Sorenson, C.M.; Sheibani, N.; Liu, B. Myeloid-Derived TSP1 (Thrombospondin-1) Contributes to Abdominal Aortic Aneurysm Through Suppressing Tissue Inhibitor of Metalloproteinases-1. Arter. Thromb. Vasc. Biol. 2020, 40, 350.
- Klaus, V.; Schmies, F.; Reeps, C.; Trenner, M.; Geisbüsch, S.; Lohoefer, F.; Eckstein, H.-H.; Pelisek, J. Cathepsin S is associated with degradation of collagen I in abdominal aortic aneurysm. Vasa 2018, 47, 285–293.
- Jadhav, P.K.; Schiffler, M.A.; Gavardinas, K.; Kim, E.J.; Matthews, D.P.; Staszak, M.A.; Coffey, D.S.; Shaw, B.W.; Cassidy, K.C.; Brier, R.A.; et al. Discovery of Cathepsin S Inhibitor LY3000328 for the Treatment of Abdominal Aortic Aneurysm. ACS Med. Chem. Lett. 2014, 5, 1138–1142.
- Lai, C.H.; Chang, J.Y.; Wang, K.C.; Lee, F.T.; Wu, H.L.; Cheng, T.L. Pharmacological Inhibition of Cathepsin S Suppresses Abdominal Aortic Aneurysm in Mice. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 990–999.
- Li, W.; Qiu, X.; Jiang, H.; Zhi, Y.; Fu, J.; Liu, J. Ulinastatin inhibits the inflammation of LPS-induced acute lung injury in mice via regulation of AMPK/NF-κB pathway. Int Immunopharmacol 2015, 29, 560–567.





